Submitted:
03 March 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Search Strategy
3. Theoretical Models of Quantum Consciousness
4. The Technological Landscape for Investigating Quantum Brain Processes
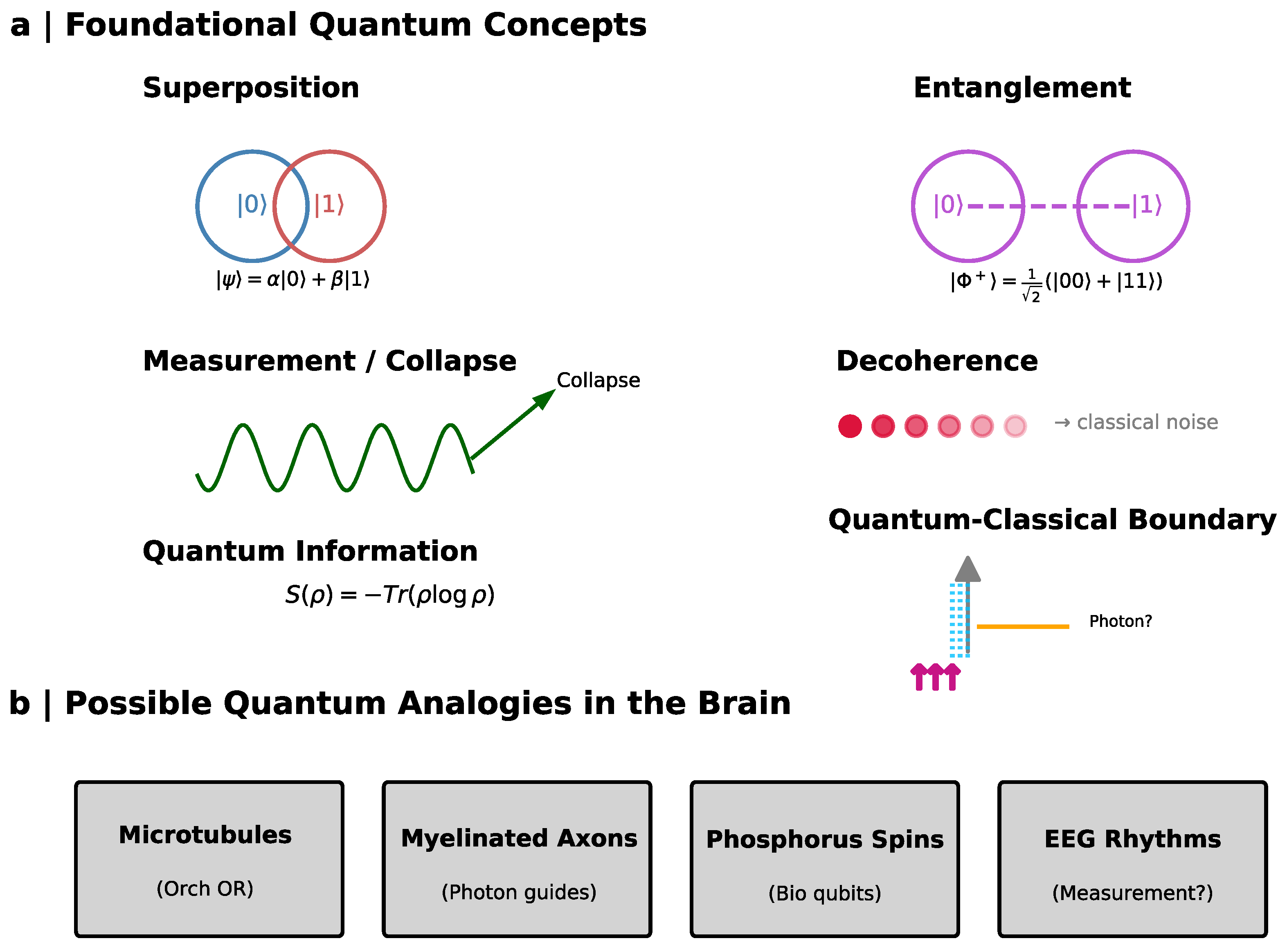
5. Basic Principles of Quantum Mechanics for Consciousness
5.1. Superposition: Structured Ambiguity and Parallel Representations
5.2. Entanglement: Beyond Classical Correlation
5.3. Measurement and Collapse: From Potentiality to Experience
5.4. Decoherence: The Thermodynamic Objection and the Biological Counterexample
5.5. Quantum Information: Entropy, Uncertainty, and Cognitive Transitions
5.6. The Quantum–Classical Boundary: Proposed Neural Substrates
6. Insights From Quantum Computing and Neural Networks
6.1. Quantum Neural Networks: Computation in a Higher Algebra
6.2. Quantum Tensor Networks: Hierarchies, Abstraction, and the Binding Problem
6.3. The Neurobiological Challenge: Connecting Formalism to Physiology
6.4. Emerging Empirical Signals
6.5. Speculative Neurobiological Substrates
6.6. A Foundational Question: Mechanism or Metaphor?
7. Experimental Evidence and Challenges
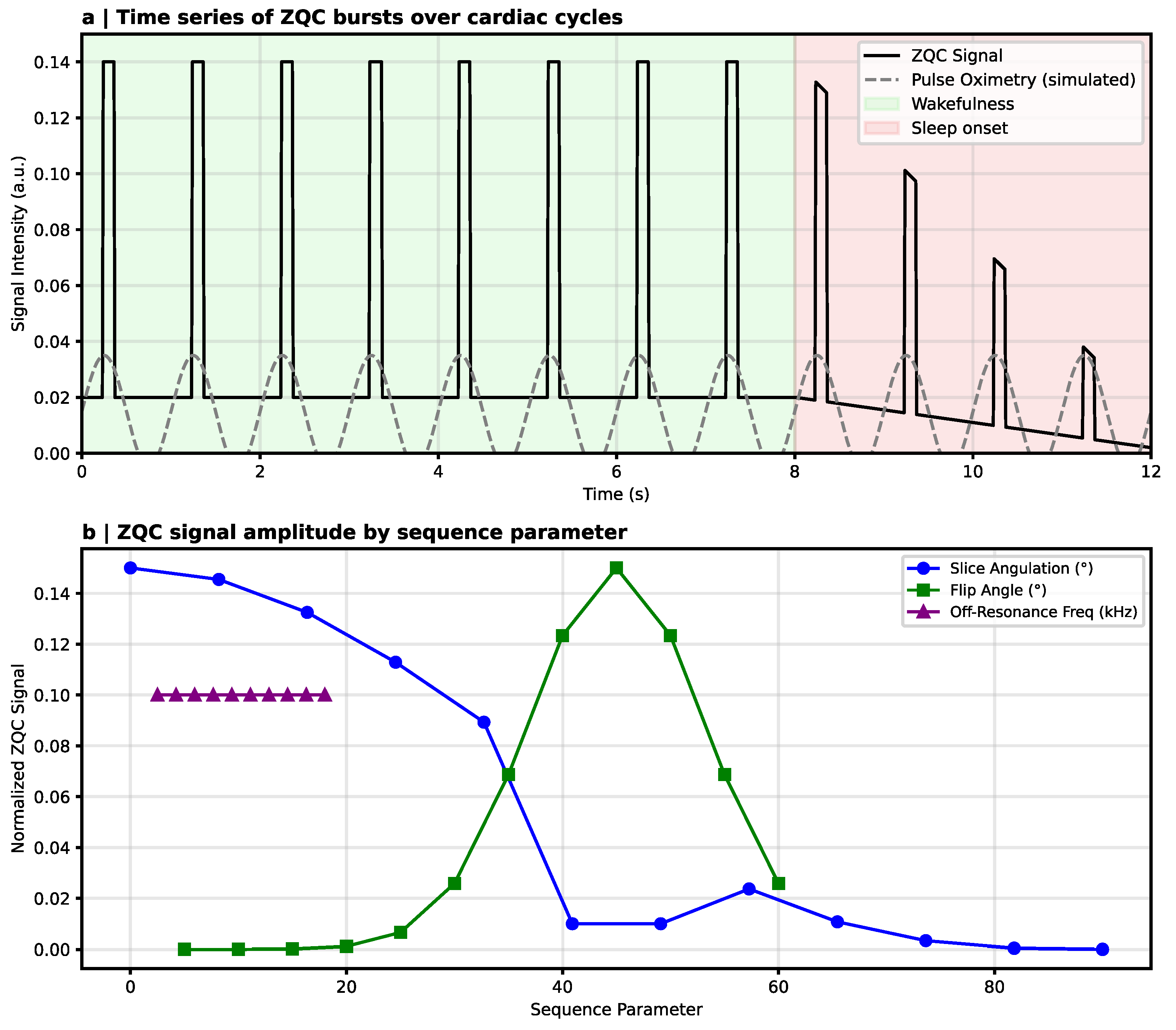
7.1. MRI-Based Indicators of Non-Classical Dynamics
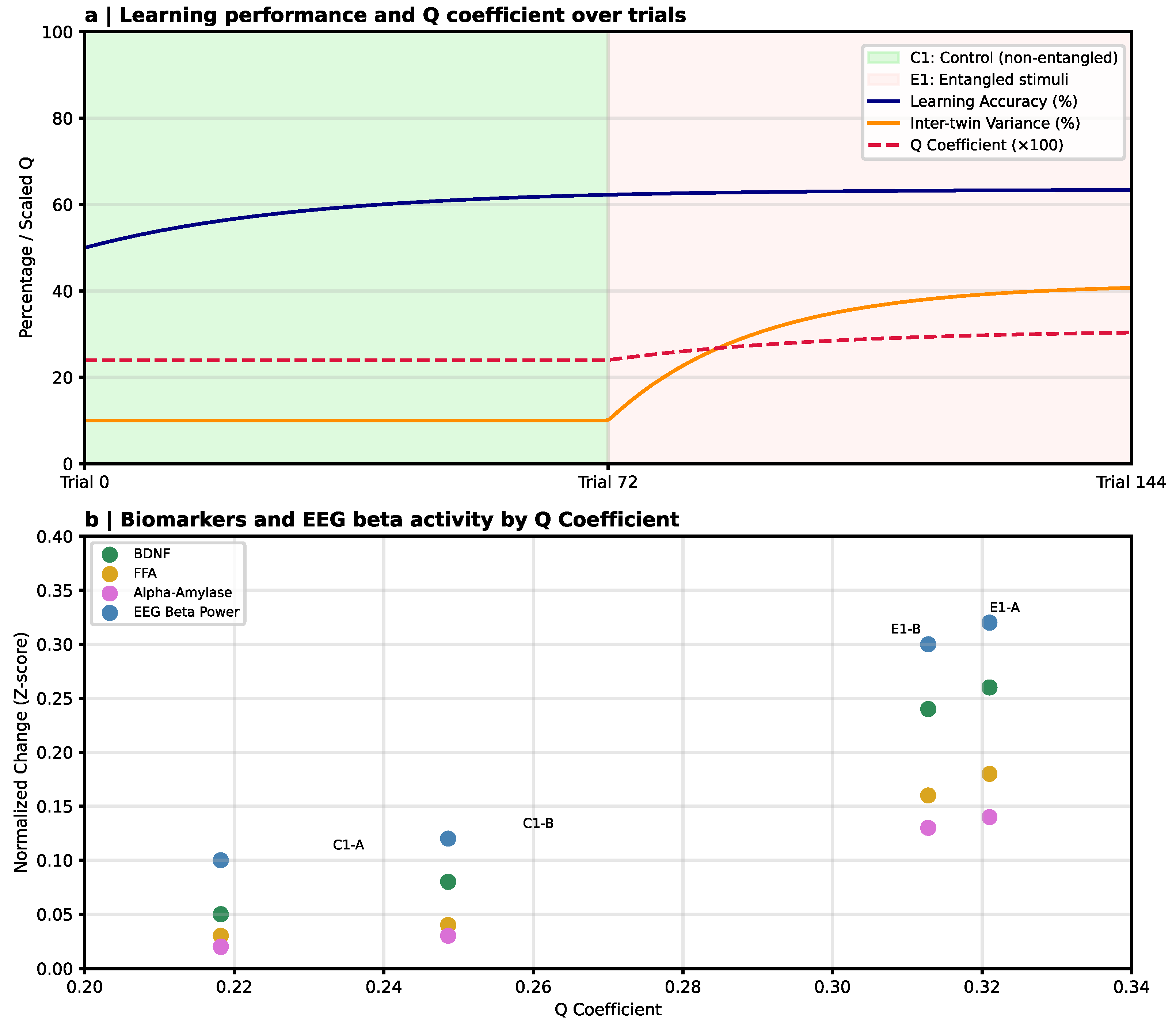
7.2. Entanglement-Structured Learning and Neuroplasticity Markers
7.3. Quantum-Like Implicit Learning and Anomalous Information Anticipation
7.4. Microtubule Coherence and Orchestrated Objective Reduction
7.5. Contextuality, Interference, and Classical Explanations of Quantum-Like Cognition
7.6. Stochastic Reduction Models and the Physics of Collapse
7.7. Cross-Cutting Challenges in Evaluating Quantum Hypotheses
8. Discussion
8.1. Limitations and Alternative Explanations
8.2. Challenges and Opportunities
8.3. Future Directions and Interrogations
- Quantitative modeling of calcium-mediated signal integration under realistic thermal conditions;
- Direct measurement of coherence lifetimes in neural microenvironments;
- Experimental tests capable of distinguishing structured ionic energy transfer from diffusion-based classical propagation;
- Adversarial null-model comparisons between nonlinear classical field dynamics and proposed non-classical effects.
9. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACT-R | Adaptive Control of Thought–Rationale |
| A-C | Access Consciousness |
| AIA | Anomalous Information Anticipation |
| ANN(s) | Artificial Neural Network(s) |
| BDNF | Brain-Derived Neurotrophic Factor |
| CRQA | Cross-Recurrence Quantification Analysis |
| EEG | Electroencephalography |
| FFA | Free Fatty Acids |
| fMRI | Functional Magnetic Resonance Imaging |
| GPT | Generalized Probability Theory |
| MEG | Magnetoencephalography |
| MF-DFA | Multifractal Detrended Fluctuation Analysis |
| MRI | Magnetic Resonance Imaging |
| MTL | Medial Temporal Lobe |
| NV | Nitrogen-Vacancy (diamond magnetometry) |
| Orch OR | Orchestrated Objective Reduction |
| P-C | Phenomenal Consciousness |
| Q (coefficient) | Quantum-Multilinear Integrated Coefficient |
| QAOA | Quantum Approximate Optimization Algorithm |
| QNN(s) | Quantum Neural Network(s) |
| SQC | Single Quantum Coherence |
| SP-RP | Stated Preference–Revealed Preference |
| SSE | Stochastic Schrödinger Equation |
| UtPBM | Unilateral Transcranial Photobiomodulation |
| ZQC | Zero Quantum Coherence |
References
- Chalmers, D.J. Facing up to the problem of consciousness. Journal of consciousness studies 1995, 2, 200–219. [Google Scholar]
- Keppler, J. Macroscopic quantum effects in the brain: new insights into the fundamental principle underlying conscious processes. Front. Hum. Neurosci. 2025, 19. [Google Scholar] [CrossRef] [PubMed]
- Theise, N.D.; Tuszynski, J.A. Non-linearity, complexity, and quantization concepts in biology. Front. Hum. Neurosci. 2026, 19. [Google Scholar] [CrossRef]
- Brody, D.C. Quantum formalism for the dynamics of cognitive psychology. Sci. Rep. 2023, 13, 16104. [Google Scholar] [CrossRef]
- Gunji, Y.-P.; Sonoda, K.; Basios, V. Quantum cognition based on an ambiguous representation derived from a rough set approximation. Biosystems 2016, 141, 55–66. [Google Scholar] [CrossRef]
- Khrennikov, A.; Ozawa, M.; Benninger, F.; Shor, O. Coupling quantum-like cognition with the neuronal networks within generalized probability theory. J. Math. Psychol. 2025, 125, 102923. [Google Scholar] [CrossRef]
- Acacio de Barros, J.; Suppes, P. Quantum mechanics, interference, and the brain. J. Math. Psychol. 2009, 53, 306–313. [Google Scholar] [CrossRef]
- Bruza, P.D.; Wang, Z.; Busemeyer, J.R. Quantum cognition: a new theoretical approach to psychology. Trends in Cognitive Sciences 2015, 19, 383–393. [Google Scholar] [CrossRef]
- Wang, Z.; Busemeyer, J.R. A Quantum Question Order Model Supported by Empirical Tests of an A Priori and Precise Prediction. Topics in Cognitive Science 2013, 5, 689–710. [Google Scholar] [CrossRef]
- Tegmark, M. Importance of quantum decoherence in brain processes. Physical Review E 2000, 61, 4194–4206. [Google Scholar] [CrossRef]
- Rosa, L.P.; Faber, J. Quantum models of the mind: Are they compatible with environment decoherence? Physical Review E 2004, 70, 031902. [Google Scholar] [CrossRef]
- Lambert, N.; Chen, Y.-N.; Cheng, Y.-C.; Li, C.-M.; Chen, G.-Y.; Nori, F. Quantum biology. Nature Physics 2013, 9, 10–18. [Google Scholar] [CrossRef]
- Hameroff, S.; Penrose, R. Consciousness in the universe: A review of the ‘Orch OR’ theory. Physics of Life Reviews 2014, 11, 39–78. [Google Scholar] [CrossRef]
- Liu, Z.; Chen, Y.-C.; Ao, P. Entangled biphoton generation in the myelin sheath. Physical Review E 2024, 110, 024402. [Google Scholar] [CrossRef] [PubMed]
- Zarkeshian, P.; Kergan, T.; Ghobadi, R.; Nicola, W.; Simon, C. Photons guided by axons may enable backpropagation-based learning in the brain. Sci. Rep. 2022, 12, 20720. [Google Scholar] [CrossRef] [PubMed]
- Neven, H.; Zalcman, A.; Read, P.; Kosik, K.S.; van der Molen, T.; Bouwmeester, D.; Bodnia, E.; Turin, L.; Koch, C. Testing the Conjecture That Quantum Processes Create Conscious Experience. Entropy 2024, 26, 460. [Google Scholar] [CrossRef] [PubMed]
- Kerskens, C.M.; López Pérez, D. Experimental indications of non-classical brain functions. Journal of Physics Communications 2022, 6, 105001. [Google Scholar] [CrossRef]
- Jedlicka, P. Revisiting the Quantum Brain Hypothesis: Toward Quantum (Neuro)biology? Front. Mol. Neurosci. 2017, 10–2017. [Google Scholar] [CrossRef]
- Escolà-Gascón, Á. Our brains sense the future through a new quantum-like implicit learning mechanism. Brain Res. Bull. 2024, 216, 111048. [Google Scholar] [CrossRef]
- Escolà-Gascón, Á. Evidence of quantum-entangled higher states of consciousness. Computational and Structural Biotechnology Journal 2025, 30, 21–40. [Google Scholar] [CrossRef]
- Escolà-Gascón, Á.; Benito-León, J. Mathematical proof of the Fisher-Escolà Q statistical distribution in quantum consciousness modeling. Computational and Structural Biotechnology Journal 2025, 30, 41–58. [Google Scholar] [CrossRef]
- Gassab, L.; Pusuluk, O.; Cattaneo, M.; Müstecaplıoğlu, Ö.E. Quantum Models of Consciousness from a Quantum Information Science Perspective. Entropy 2025, Vol. 27, 243. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.-G.; Liu, Z.-P.; Yin, H.-L.; Li, C.-L.; Xu, T.-K.; Chen, Z.-B. Quantum Neural Network for Quantum Neural Computing. Research 6 0134. [CrossRef] [PubMed]
- Silberstein, R.B.; Bigelow, F.J. Brain functional connectivity correlates of anomalous interaction between sensorily isolated monozygotic twins. Front. Hum. Neurosci. 2024, 18. [Google Scholar] [CrossRef]
- Abramov, D.M.; Quintanilha, D.d.F.; Lima, H.S.; Costa, R.P.; Kamil-Leite, C.; Lazarev, V.V.; Tsallis, C. Neurophysiological correlates to the human brain complexity through q-statistical analysis of electroencephalogram. Sci. Rep. 2025, 15, 36502. [Google Scholar] [CrossRef]
- Georgiev, D.D. Quantum information theoretic approach to the hard problem of consciousness. Biosystems 2025, 251, 105458. [Google Scholar] [CrossRef]
- Schiffer, F. A four-field quantum model of life, subjectivity, consciousness, and memory: integrating dual-brain psychology and biophoton quantum interactions. Med. Hypotheses 2025, 202, 111738. [Google Scholar] [CrossRef]
- Marshall, P. The role of quantum mechanics in cognition-based evolution. Prog. Biophys. Mol. Biol. 2023, 180-181, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.G.; Jayakrishnan, R. A quantum cognition model for bridging stated and revealed preference. Transportation Research Part B: Methodological 2018, 118, 263–280. [Google Scholar] [CrossRef]
- Pathak, P.; Innan, N.; Marchisio, A.; Shafique, M. Quantum-enhanced decision-making in ACT-R: Cognitive architectures, decision-making, and quantum optimization. In Quantum Computational AI; Cheng, L., Saurabh, N., Mao, Y., Eds.; Morgan Kaufmann, 2026; pp. 215–249. [Google Scholar] [CrossRef]
- Swan, M.; dos Santos, R.P.; Witte, F. Quantum Neurobiology. Quantum Reports 2022, Vol. 4, pp 107–126. [Google Scholar] [CrossRef]
- Swan, M.; dos Santos, R.P.; Lebedev, M.; Witte, F. Quantum Computing for the Brain; WORLD SCIENTIFIC (EUROPE), 2022; Volume 3, p. 552. [Google Scholar]
- Aerts, D.; Broekaert, J.; Gabora, L. A case for applying an abstracted quantum formalism to cognition. New Ideas in Psychology 2011, 29, 136–146. [Google Scholar] [CrossRef]
- Levine, Y.; Sharir, O.; Cohen, N.; Shashua, A. Quantum Entanglement in Deep Learning Architectures. Phys. Rev. Lett. 2019, 122, 065301. [Google Scholar] [CrossRef] [PubMed]
- Kyriazos, T.; Poga, M. Quantum concepts in Psychology: Exploring the interplay of physics and the human psyche. Biosystems 2024, 235, 105070. [Google Scholar] [CrossRef] [PubMed]
- Gunji, Y.-P.; Shinohara, S.; Haruna, T.; Basios, V. Inverse Bayesian inference as a key of consciousness featuring a macroscopic quantum logical structure. Biosystems 2017, 152, 44–65. [Google Scholar] [CrossRef]
- Eeles, E.; Pourzinal, D.; Baland, J.; Ray, J. Schrödinger’s cat and mouse: An adapted thought experiment for the context of consciousness. Behav. Brain Res. 2025, 483, 115459. [Google Scholar] [CrossRef]
- Wiest, M.C.; Puniani, A.S. Conscious active inference I: A quantum model naturally implements the path integral needed for real-time planning and control. Computational and Structural Biotechnology Journal 2025, 30, 108–121. [Google Scholar] [CrossRef]
- Wiest, M.C.; Puniani, A.S. Conscious active inference II: Quantum orchestrated objective reduction among intraneuronal microtubules naturally accounts for discrete perceptual cycles. Computational and Structural Biotechnology Journal 2025, 30, 94–107. [Google Scholar] [CrossRef]
- Brody, D.C.; Hughston, L.P. Quantum noise and stochastic reduction. Journal of Physics A: Mathematical and General 2006, 39, 833. [Google Scholar] [CrossRef]
- Waddup, O.J.; Yearsley, J.M.; Blasiak, P.; Pothos, E.M. Temporal Bell inequalities in cognition. Psychonomic Bulletin & Review 2023, 30, 1946–1953. [Google Scholar] [CrossRef]
- Hameroff, S.; Penrose, R. Orchestrated reduction of quantum coherence in brain microtubules: A model for consciousness. Mathematics and Computers in Simulation 1996, 40, 453–480. [Google Scholar] [CrossRef]
- Vitas, M. Towards a Possible Definition of Consciousness. Biosystems 2025, 254, 105526. [Google Scholar] [CrossRef] [PubMed]
- Baars, B.J.; Edelman, D.B. Consciousness, biology and quantum hypotheses. Physics of Life Reviews 2012, 9, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Loboguerrero, D.A. The collapse of the wave function as the mediator of free will in prime neurons. Front. Neurosci. 2025, 19. [Google Scholar] [CrossRef]
- Wiest, M.C. A quantum microtubule substrate of consciousness is experimentally supported and solves the binding and epiphenomenalism problems. Neuroscience of Consciousness 2025, 2025, niaf011. [Google Scholar] [CrossRef]
- Freitas da Rocha, A.; Pereira, A.; Bezerra Coutinho, F.A. N-methyl-d-aspartate channel and consciousness: from signal coincidence detection to quantum computing. Prog. Neurobiol. 2001, 64, 555–573. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A. Classical-quantum interfaces in living neural tissue supporting conscious functions. In Advances in Quantum Chemistry; Poznański, R.R., Brändas, E.J., Eds.; Academic Press, 2020; Vol. 82, pp. 213–252. [Google Scholar]
| Domain | Proposed Quantum Feature | Candidate Neural Mechanism | Representative Indicators | Key Challenges |
|---|---|---|---|---|
| Non-classical MRI dynamics | Non-classical spin correlations → ZQC-like coherence | Mesoscopic proton–spin networks | Heartbeat-linked ZQC bursts; awareness-dependent signal modulation | Artefact exclusion; entanglement not directly tested; replication needed |
| Entanglement-structured cognition | Entangled relational structure → variance amplification → enhanced learning | Network-level sensitivity to non-local task structure | Increased Q-coefficient; boosted learning variance; biomarker shifts | Task entanglement ≠, neural entanglement, expectancy and twin-based confounds |
| Quantum-like implicit learning (AIA) | Non-local prediction structure → implicit anticipation | Distributed implicit-learning circuits (occipital–parietal–MTL) | Above-chance AIA performance; trial-dependent learning acceleration; EEG correlates | Alternative classical models; need for blinded controls; no physical entanglement measurement |
| Microtubule quantum coherence (Orch OR) | Tubulin coherence → objective reduction → discrete conscious moments | Cytoskeletal microtubule lattices | Theoretical coherence thresholds; anesthetic isotope sensitivity predictions | Decoherence at physiological temperature; lack of in-situ microtubule coherence evidence |
| Contextuality-based explanations | Contextuality → interference-like cognitive states | Classical oscillatory or field-based interactions | Violations of classical probability; interference patterns in decisions | Not uniquely quantum; classical contextuality is sufficient |
| Stochastic reduction frameworks | Noise-driven collapse → measurement-like behavior | Hypothetical micro-scale collapse processes | Energy-based stochastic collapse models | No biological demonstration; unclear neural relevance |
| Domain | Proposed Quantum Feature | Candidate Neural Mechanism | Representative Indicators | Key Challenges |
| Study / Model | Method | Key Findings | Interpretation | Limitations / Challenges |
|---|---|---|---|---|
| ZQC–MRI [17] | 3T MRI → ZQC-sensitive sequences | Heartbeat-locked cortical signals → disappear in sleep/anesthesia | Awareness-dependent coherence-like activity | Vascular/diffusion artefacts possible → replication required |
| Twin EEG with entangled stimuli [20] | EEG → entangled vs. non-entangled stimuli → biomarker assays | ↑ Accuracy, ↑ twin variance, ↑ plasticity biomarkers | Entangled task structure → behavioral & neuroplastic modulation | Q-coefficient novel → expectancy & twin confounds → reproducibility pending |
| Anomalous information anticipation (AIA) [19] | Continuous flash suppression → 3D EEG → 144-trial protocol | Predictive accuracy 25–48% → posterior activation | Quantum-like implicit anticipation → non-classical learning profile | No entanglement measure → small sample → need for blinded designs |
| Quantum interference models [7] | Quantum probability → cognitive modelling | Captures contextual decisions → classical rule violations | Cognition may follow interference-like principles | Theoretical; no identified neural substrate |
| Stochastic Schrödinger models [40] | Energy-based stochastic Schrödinger equation | Predicts spontaneous collapse → conserved mean energy | Framework linking micro-indeterminacy → macroscopic dynamics | Mathematical only; biological relevance undemonstrated |
| Quantum biology analogues (photosynthesis; magnetoreception; axonal photon models) | Spectroscopy → radical pair dynamics → photonic modelling | Long-lived coherence → entangled radical pairs → theoretical biphoton guidance | Demonstrates the feasibility of biological coherence | Neural evidence speculative; no direct verification in the brain |
| Analytical approaches (CRQA, MF-DFA) | Nonlinear EEG/MRI analyses | Recurrence & coherence-like patterns | Could reflect deterministic chaos → alternative to quantum mechanisms | Susceptible to false positives → requires robust null models |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




