Submitted:
27 February 2026
Posted:
02 March 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Material and Methods
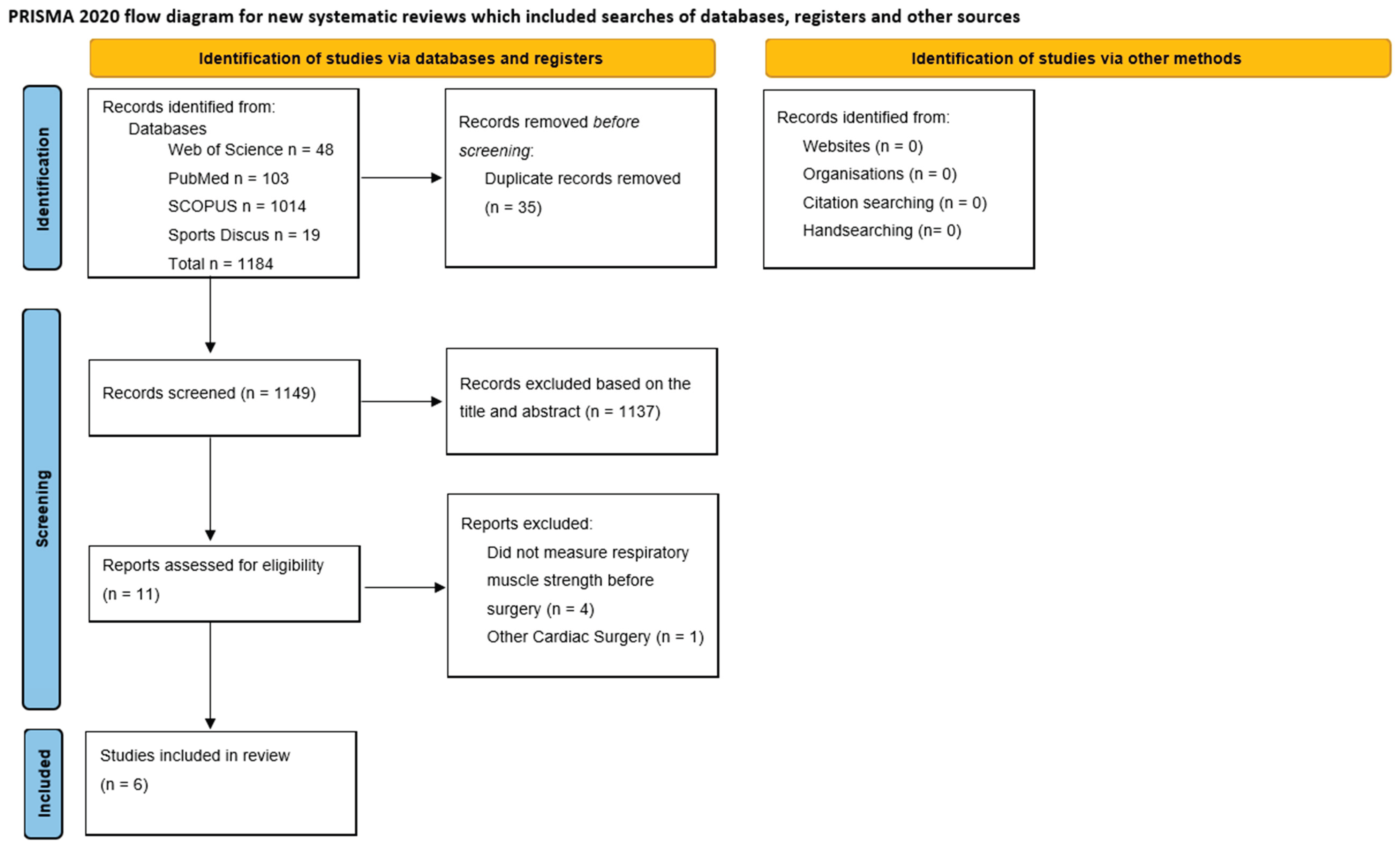
Eligibility criteria
Search Strategy
Selection Process and Data Extraction
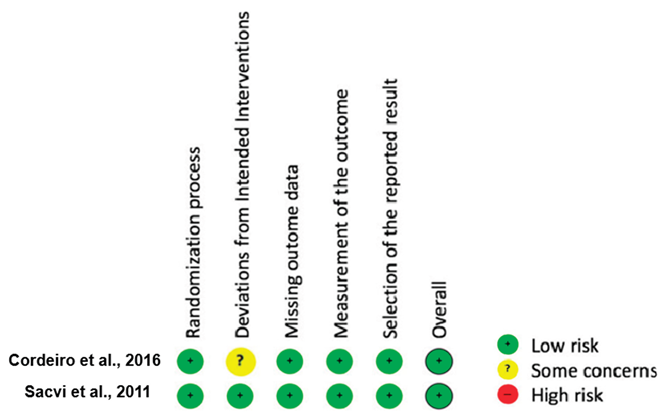
Study Risk of Bias Assessment
Results
Discussion
Limitations
Conclusions
References
- Fonseca, C; Bras, D; Araujo, I; Ceia, F. Heart failure in numbers: Estimates for the 21st century in Portugal. Rev Port Cardiol (Engl Ed) 2018, 37, 97–104. [Google Scholar] [CrossRef]
- Song, Y; Ren, C; Liu, P; Tao, L; Zhao, W; Gao, W. Effect of Smartphone-Based Telemonitored Exercise Rehabilitation among Patients with Coronary Heart Disease. J Cardiovasc Transl Res. 2020, 13, 659–67. [Google Scholar] [CrossRef]
- Cordeiro, AL; de Melo, TA; Neves, D; Luna, J; Esquivel, MS; Guimaraes, AR; et al. Inspiratory Muscle Training and Functional Capacity in Patients Undergoing Cardiac Surgery. Braz J Cardiovasc Surg. 2016, 31, 140–4. [Google Scholar] [PubMed]
- Pantoni, CBF; Thommazo-Luporini, LD; Mendes, RG; Caruso, FCR; Castello-Simoes, V; Mezzalira, D; et al. Effect of continuous positive airway pressure associated to exercise on the breathing pattern and heart rate variability of patients undergoing coronary artery bypass grafting surgery: a randomized controlled trial. Braz J Med Biol Res. 2021, 54, e10974. [Google Scholar] [CrossRef]
- Hermes, BM; Cardoso, DM; Gomes, TJ; Santos, TD; Vicente, MS; Pereira, SN; et al. Short-term inspiratory muscle training potentiates the benefits of aerobic and resistance training in patients undergoing CABG in phase II cardiac rehabilitation program. Rev Bras Cir Cardiovasc. 2015, 30, 474–81. [Google Scholar]
- Patel, DK; Duncan, MS; Shah, AS; Lindman, BR; Greevy, RA, Jr.; Savage, PD; et al. Association of Cardiac Rehabilitation With Decreased Hospitalization and Mortality Risk After Cardiac Valve Surgery. JAMA Cardiol. 2019, 4, 1250–9. [Google Scholar] [CrossRef]
- Brown, TM; Pack, QR; Aberegg, E; Brewer, LC; Ford, YR; Forman, DE; et al. Core Components of Cardiac Rehabilitation Programs: 2024 Update: A Scientific Statement From the American Heart Association and the American Association of Cardiovascular and Pulmonary Rehabilitation. Circulation 2024, 150, e328–e347. [Google Scholar] [CrossRef]
- Dos Santos, TD; Pereira, SN; Portela, LOC; Cardoso, DM; Lago, PD; Dos Santos Guarda, N; et al. Moderate-to-high intensity inspiratory muscle training improves the effects of combined training on exercise capacity in patients after coronary artery bypass graft surgery: A randomized clinical trial. Int J Cardiol. 2019, 279, 40–6. [Google Scholar] [CrossRef]
- Naseer, BA; Al-Shenqiti, AM; Ali, ARH; Aljeraisi, T. Effect of cardiac surgery on respiratory muscle strength. J Taibah Univ Med Sci. 2019, 14, 337–42. [Google Scholar] [CrossRef] [PubMed]
- Urell, C; Emtner, M; Hedenstrom, H; Westerdahl, E. Respiratory muscle strength is not decreased in patients undergoing cardiac surgery. J Cardiothorac Surg. 2016, 11, 41. [Google Scholar] [CrossRef] [PubMed]
- Morgan, RL; Whaley, P; Thayer, KA; Schunemann, HJ. Identifying the PECO: A framework for formulating good questions to explore the association of environmental and other exposures with health outcomes. Environ Int. 2018, 121, 1027–31. [Google Scholar] [CrossRef]
- Ardern, CL; Buttner, F; Andrade, R; Weir, A; Ashe, MC; Holden, S; et al. Implementing the 27 PRISMA 2020 Statement items for systematic reviews in the sport and exercise medicine, musculoskeletal rehabilitation and sports science fields: the PERSiST (implementing Prisma in Exercise, Rehabilitation, Sport medicine and SporTs science) guidance. Br J Sports Med. 2022, 56, 175–95. [Google Scholar] [PubMed]
- Higgins, JPT; Morgan, RL; Rooney, AA; Taylor, KW; Thayer, KA; Silva, RA; et al. A tool to assess risk of bias in non-randomized follow-up studies of exposure effects (ROBINS-E). Environ Int. 2024, 186, 108602. [Google Scholar] [CrossRef] [PubMed]
- Sterne, JAC; Savovic, J; Page, MJ; Elbers, RG; Blencowe, NS; Boutron, I; et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019, 366, l4898. [Google Scholar] [CrossRef]
- Johnson, D; Hurst, T; Thomson, D; Mycyk, T; Burbridge, B; To, T; et al. Respiratory function after cardiac surgery. J Cardiothorac Vasc Anesth. 1996, 10, 571–7. [Google Scholar] [CrossRef] [PubMed]
- Riedi, C; Mora, CTR; Driessen, T; Coutinho, MDG; Mayer, DM; Moro, FL; et al. Relation between respiratory muscle strength with respiratory complication on the heart surgery. Revista Brasileira De Cirurgia Cardiovascular 2010, 25, 500–5. [Google Scholar]
- Savci, S; Degirmenci, B; Saglam, M; Arikan, H; Inal-Ince, D; Turan, HN; et al. Short-term effects of inspiratory muscle training in coronary artery bypass graft surgery: a randomized controlled trial. Scand Cardiovasc J. 2011, 45, 286–93. [Google Scholar] [CrossRef]
- Silva, CR; Andrade, LB; Maux, DA; Bezerra, AL; Duarte, MC. Effectiveness of prophylactic non-invasive ventilation on respiratory function in the postoperative phase of pediatric cardiac surgery: a randomized controlled trial. Braz J Phys Ther. 2016, 20, 494–501. [Google Scholar] [CrossRef]
- Cargnin, C; Karsten, M; Guaragna, J; Dal Lago, P. Inspiratory Muscle Training After Heart Valve Replacement Surgery Improves Inspiratory Muscle Strength, Lung Function, and Functional Capacity: A RANDOMIZED CONTROLLED TRIAL. J Cardiopulm Rehabil Prev. 2019, 39, E1–E7. [Google Scholar] [CrossRef]
- Supinski, GS; Morris, PE; Dhar, S; Callahan, LA. Diaphragm Dysfunction in Critical Illness. Chest 2018, 153, 1040–51. [Google Scholar] [CrossRef]
- Jung, B; Moury, PH; Mahul, M; de Jong, A; Galia, F; Prades, A; et al. Diaphragmatic dysfunction in patients with ICU-acquired weakness and its impact on extubation failure. Intensive Care Med. 2016, 42, 853–61. [Google Scholar] [CrossRef] [PubMed]
- Girgin, Z; Cigerci, Y; Yaman, F. The Effect of Pulmonary Rehabilitation on Respiratory Functions, and the Quality of Life, following Coronary Artery Bypass Grafting: A Randomised Controlled Study. Biomed Res Int. 2021, 2021, 6811373. [Google Scholar] [CrossRef] [PubMed]
| PECOS | Inclusion criteria | Exclusion criteria | Search term | |
|---|---|---|---|---|
| Population | Individuals with more than 18 years of age submitted to coronary artery bypass grafting | Individuals with less than 18 years of age or submitted to other types of cardiac surgery | Adult | |
|
Exposure Comparison |
Individuals submitted to coronary artery bypass grafting | Individuals submitted to other types of cardiac surgery | Cardiac rehabilitation Heart surgery Coronary heart disease Cardiac surgery Coronary artery bypass grafting GABG |
|
| Pre-post surgery respiratory function comparison | ||||
| Outcome | Respiratory function | No respiratory function assessment or other outcomes evaluated | Respiratory function Respiratory muscle strength Muscle strength Inspiratory muscle training |
|
| Study Design | Original research articles | Systematic reviews, meta-analysis and editorials | ||
| Authors | Sample size and sex | Age | Study Design | Timing of Assessment | Respiratory Function Outcome | Main Conclusions |
|---|---|---|---|---|---|---|
| Cordeiro et al., 2016 | Control group (25) M - 16; F - 9 |
Control Group: 57 ± 14.7 | Randomized controlled trial | Preoperative and at discharge | Maximal inspiratory pressure | Significant reduction in maximal inspiratory pressure in both groups after surgery, more significant in the control group (97.5 ± 18.2 to 69.5 ± 14.9 cmH2O in the control group, p = 0.00001, and 94.2 ± 16.2 to 83.1 ± 19.1 cmH2O in the intervention group, p < 0.01). |
| Intervention group (25) M - 11; F - 14 |
Intervention: 56.4 ± 13 | |||||
| Johnson et al., 1996 | 90 patients | CABG: 65 ± 8.8 | Observational study | Preoperative, at discharge and 8 weeks after discharge | Forced vital capacity Maximal expiratory volume in one second Maximal expiratory pressure |
Postoperative changes in respiratory muscle strength persisted up to at least 8 weeks after surgery (FVC 3.51± 0.82 to 2 91± 0.72 cmH2O, p < 0.01; FEV₁ 2.74 ± 0.74 to 22.2 ± 0.63 cmH2O, p < 0.01; MEP 93 ± 28 to 89 ± 26 cmH2O, p > 0.01). |
| Naseer et al., 2019 | 28 patients | 65 ± 7 | Observational study | Preoperative and 8 weeks after surgery | Forced expiratory volume one second Maximal inspiratory pressure Maximal expiratory pressure |
Significantly decreased in all outcomes observed 8 weeks after surgery, showing a relationship between a reduction in inspiratory muscle strength and lung function (FEV₁ 2.87 ± 0.45 to 2.5 ± 0.68 cmH2O, p = 0.0001; MIP 81.75 ± 22.04 to 74.56 ± 18.86 cmH2O, p = 0.146; MEP 98.55 ± 22.24 to 88.86 ± 18.14 cmH2O, p = 0.019). |
| Riedi et al., 2010 | 34 patients | 54.05 ± 13.6 | Observational study | Preoperative and 5 days after surgery | Maximal inspiratory pressure Maximal expiratory pressure |
Significantly decreased in respiratory muscle strength in the postoperative period (MEP 89.18 ± 30.18 to 66.8 ± 22.11 cmH2O, p > 0.05, and MIP 106.2 ± 49.42 to 91.5 ± 52.2 cmH2O, p < 0.05) |
| Sacvi et al., 2011 | Control group (21) M - 19; F - 2 |
Control group: 57.48 ± 11.48 | Randomized controlled trial | Preoperative and 5 days after surgery | Forced expiratory volume one second Forced vital capacity Maximum inspiratory pressure Maximum expiratory pressure |
Significantly decreased in all outcomes five days after cardiac surgery in both groups, with a reduction in lung function (FVC 85.00 ± 13.71 to 66.43 ± 14.42 %, p < 0.05, in the control group, and 88.00 ± 16.36 to 64.00 ± 14.94 %, p < 0.05, in the intervention group; FEV₁ 77.24 ± 14.59 to 64.29 ± 14.90 %, p < 0.05 in the control group and 85.95 ± 16.75 to 63.73 ± 15.06 %, p < 0.05, in the intervention group); MEP 101.71 ± 22.22 to 73.43 ± 25.52 cmH2O, p < 0.05, in the control group, and 106.55 ± 33.27 to 69.82 ± 14.60 cmH2O, p < 0.05, in the intervention group; ); MIP 84.62 ± 17.26 to 57.24 ± 19.48 cmH2O, p < 0.05, in the control group and 82.64 ± 29.31 to 95.45 ± 30.32 cmH2O, p < 0.05, in the intervention group) |
| Intervention group (22) M – 19; F - 3 |
Intervention group: 62.82 ± 8.69 | |||||
| Urell et al., 2016 | 16 patients | 67 ± 10 | Observational study | Preoperative and 2 months after surgery | Maximal inspiratory pressure Maximal expiratory pressure Forced expiratory volume one second |
Differences in respiratory muscle strength two months after surgery, although an association between decreased inspiratory muscle strength and impaired lung function was shown (MIP 78 ± 24 to 73 ± 22 cmH2O, p = 0.19; MEP 122 ± 33 to 115 ± 38 cmH2O, p = 0.018; FEV₁ 3.0 ± 0.8 to 2.8 ± 0.7 cmH2O, p = 0.001) |
| Confounding | Selection | Measurement of exposure | Departure from exposure | Missing data | Measurement of outcomes | Reported results | Overall bias | |
| Johnson et al., 1996 | M | M | L | M | L | L | M | M |
| Naseer et al., 2019 | M | M | L | M | L | M | M | M |
| Riedi et al., 2010 | M | M | L | M | M | M | M | M |
| Urell et al., 2016 | M | M | L | M | L | M | M | M |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




