Submitted:
27 February 2026
Posted:
02 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Material and Methods
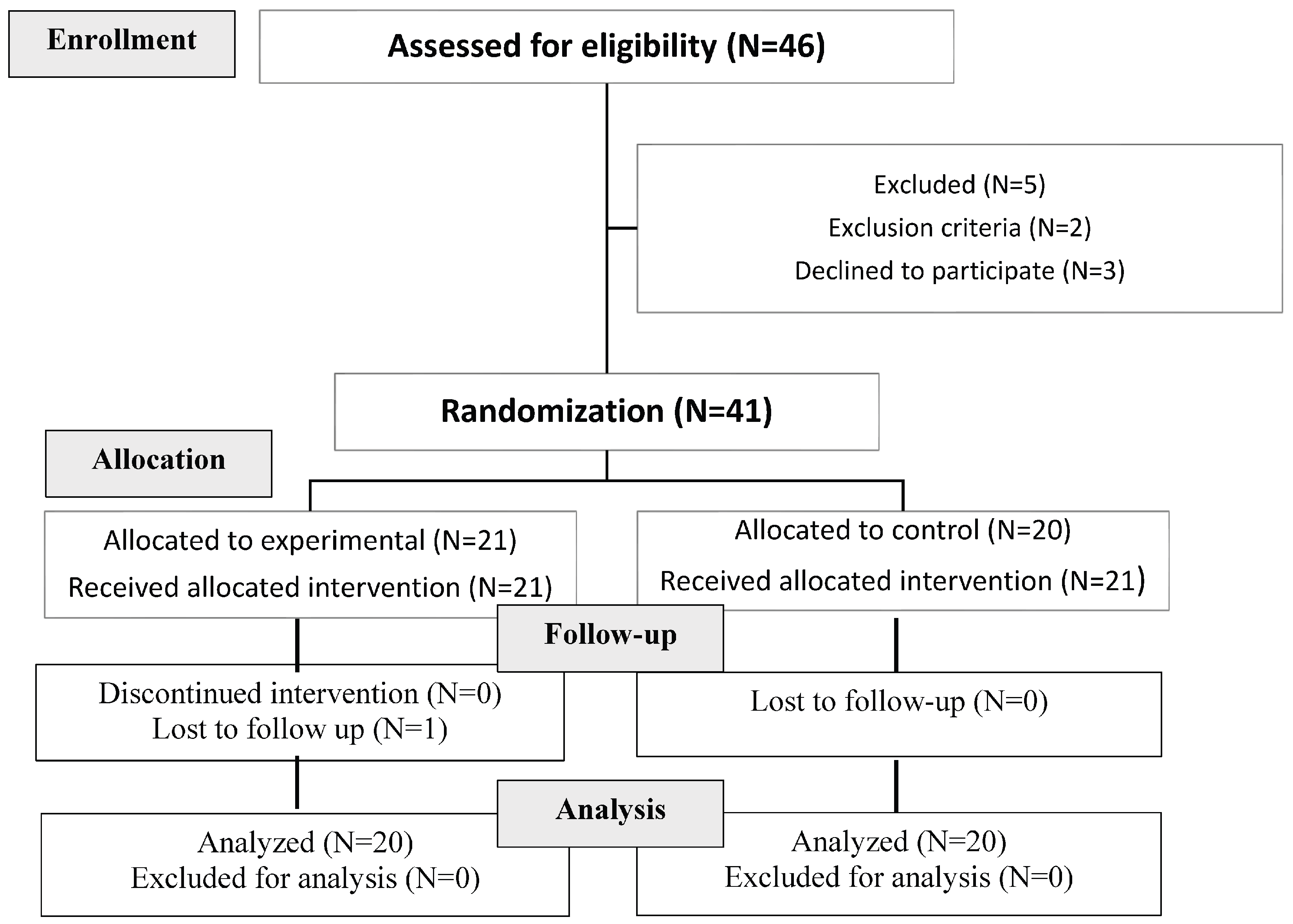
2.1. Study Design and Population
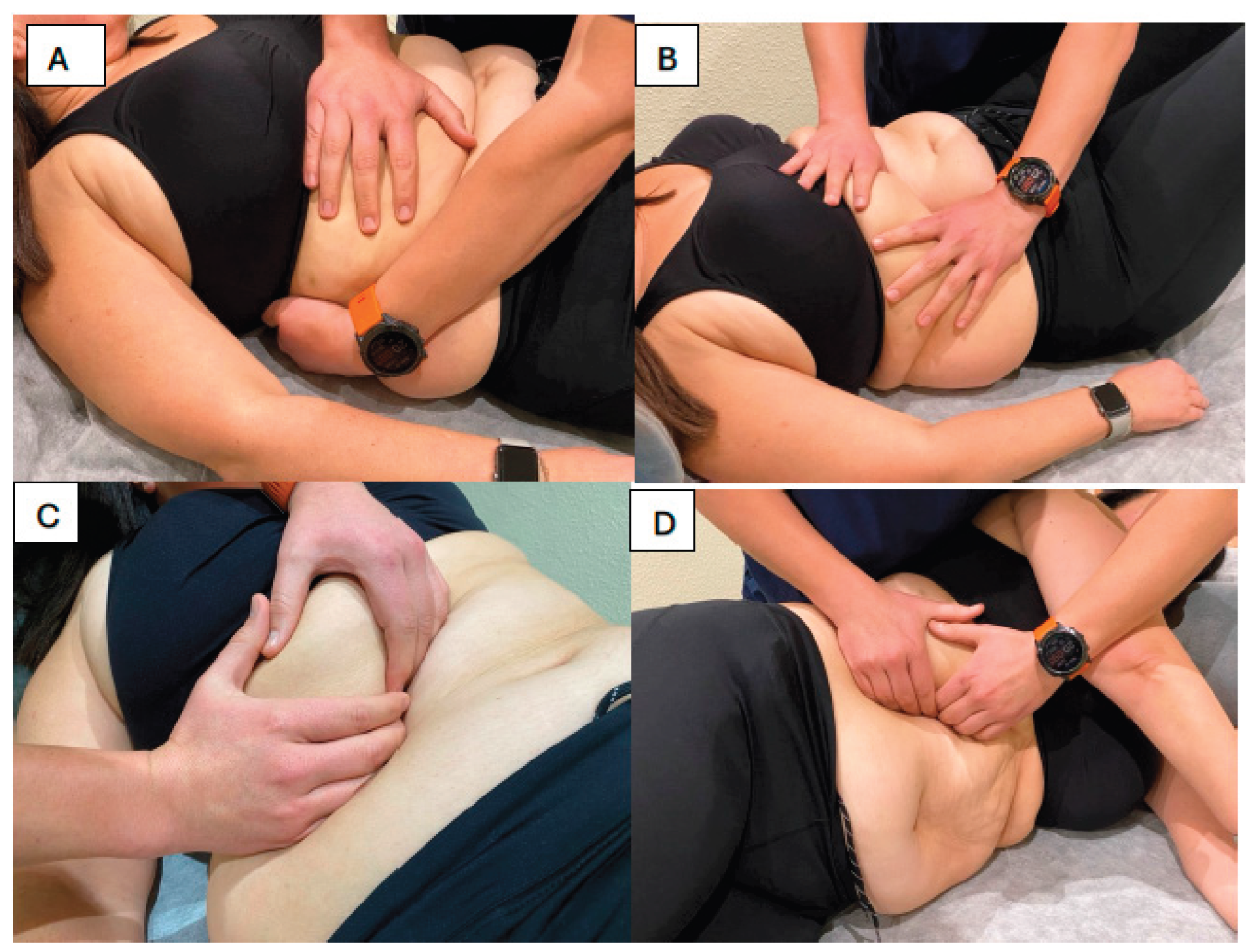
2.2. Study Protocol
2.3. End Points and Assessments
2.4. Data Analysis
3. Results
3.1. Patient Characteristics and Randomization
3.2. Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Appendix

- La participación en el estudio es totalmente voluntaria.
- Usted puede retirarse del estudio cuando así lo manifieste, sin dar explicaciones y sin que esto repercuta en sus cuidados médicos.
- Todos los datos carácter personal, obtenidos en este estudio son confidenciales y se tratarán conforme a la Ley Orgánica 03/2018 de Protección de Datos de Carácter Personal.
- La información obtenida se utilizará exclusivamente para los fines específicos de este estudio.
- He leído el documento informativo que acompaña a este consentimiento (Información al Paciente)
- He podido hacer preguntas sobre el estudio
- He recibido suficiente información sobre el estudio
- He hablado con el profesional sanitario informador:
- Comprendo que mi participación es voluntaria y soy libre de participar o no en el estudio.
- Se me ha informado que todos los datos obtenidos en este estudio serán confidenciales y se tratarán conforme establece la Ley Orgánica 03/2018 de Protección de Datos de Carácter Personal.
- Se me ha informado de que la información obtenida sólo se utilizará para los fines específicos del estudio.
- Deseo ser informado/a de mis datos médicos y otros de carácter personal que se obtengan en el curso de la investigación, incluidos los descubrimientos inesperados que se puedan producir, siempre que esta información sea necesaria para evitar un grave perjuicio para mi salud o la de mis familiares biológicos.
- Cuando quiera
- Sin tener que dar explicaciones
- Sin que esto repercuta en mis cuidados médicos
| Firma del paciente | Firma del profesional |
| (o representante legal en su caso) | sanitario informador |
| Nombre y apellidos:……………. | Nombre y apellidos: …………….... |
| DNI: | |
| Fecha: ……………………………… | Fecha: ………………………. |
- Revocar el consentimiento otorgado para el tratamiento de mis datos de carácter personal, de conformidad con lo establecido en la normativa vigente sobre protección de datos.
- Cese en la participación del proyecto
| Firma del paciente | Firma del profesional |
| (o representante legal en su caso) | sanitario informador |
| Nombre y apellidos:………………. | Nombre y apellidos: …………….... |
| DNI: | |
| Fecha: ……………………………… | Fecha: ………………………. |
References
- Rinella, M.E.; Lazarus, J.V.; Ratziu, V.; et al. A Multisociety Delphi Consensus Statement on New Fatty Liver Disease Nomenclature. Hepatology 2023, 78, 1966–1986. [CrossRef]
- Song, S.J.; Lai, J.C.T.; Wong, G.L.H.; et al. Can We Use Old NAFLD Data under the New MASLD Definition? J. Hepatol. 2024, 80, e54–e56. [CrossRef]
- Clayton-Chubb, D.; Scott, R.A.; Roberts, S.K.; et al. Metabolic Dysfunction-Associated Steatotic Liver Disease in Community-Dwelling Older Adults: Prevalence, Risk Factors and Association with Frailty and Functional Outcomes. J. Gerontol. A Biol. Sci. Med. Sci. 2024, 79, 1543–1552. [CrossRef]
- Huang, S.; Hu, W.; Fang, T. The Global Impact of Non-Alcoholic Fatty Liver Disease (Including Cirrhosis) in the Elderly from 1990 to 2021 and Future Projections of Disease Burden. PLoS ONE 2025, 20, e0325961. [CrossRef]
- Bjornsson, E.S. The Importance of Age for Liver-Related Mortality in Patients with Metabolic Dysfunction-Associated Steatotic Liver Disease. Hepatobiliary Surg. Nutr. 2024, 13, 124–127. [CrossRef]
- Mitten, E.K.; Baffy, G. Mechanotransduction in the Pathogenesis of Non-Alcoholic Fatty Liver Disease. J. Hepatol. 2022, 77, 1642–1656. [CrossRef]
- Kang, N. Mechanotransduction in Liver Diseases. Semin. Liver Dis. 2020, 40, 84–90. [CrossRef]
- Bercík, P.; Bouley, L.; Dutoit, P.; et al. Quantitative Analysis of Intestinal Motor Patterns: Spatiotemporal Organization of Nonneural Pacemaker Sites in the Rat Ileum. Gastroenterology 2000, 119, 386–394. [CrossRef]
- Bercík, P.; Wang, L.; Verdú, E.F.; et al. Visceral Hyperalgesia and Intestinal Dysmotility in a Mouse Model of Postinfective Gut Dysfunction. Gastroenterology 2004, 127, 179–187. [CrossRef]
- Lee, J.H.; Kim, D.; Kim, H.J.; et al. Hepatic Steatosis Index: A Simple Screening Tool Reflecting Nonalcoholic Fatty Liver Disease. Dig. Liver Dis. 2010, 42, 503–508. [CrossRef]
- Wen, B.; Zhang, G.; Zhan, C.; et al. The 2024 Revision of the Declaration of Helsinki: A Modern Ethical Framework for Medical Research. Postgrad. Med. J. 2024. [CrossRef]
- Ricard, F. Tratado de Osteopatía Visceral y Medicina Interna. Tomo II: Sistema Digestivo; MEDOS: Madrid, Spain, 2016.
- Gaggini, M.; Morelli, M.; Buzzigoli, E.; et al. Non-Alcoholic Fatty Liver Disease (NAFLD) and Its Connection with Insulin Resistance, Dyslipidemia, Atherosclerosis and Coronary Heart Disease. Nutrients 2013, 5, 1544–1560. [CrossRef]
- Dongiovanni, P.; Stender, S.; Pietrelli, A.; et al. Causal Relationship of Hepatic Fat with Liver Damage and Insulin Resistance in Nonalcoholic Fatty Liver. J. Intern. Med. 2018, 283, 356–370. [CrossRef]
- Stefan, N.; Staiger, H.; Häring, H.U. Dissociation between Fatty Liver and Insulin Resistance: The Role of Adipose Triacylglycerol Lipase. Diabetologia 2011, 54, 7–9. [CrossRef]
- Bende, F.; Szabó, B.; Sándor, J.; et al. Association between Visceral Adiposity and the Prediction of Hepatic Steatosis and Fibrosis in Patients with Metabolic Dysfunction-Associated Steatotic Liver Disease. J. Clin. Med. 2025, 14, 3405. [CrossRef]
- Patel, P. Treatment Landscape of Metabolic-Dysfunction-Associated Steatotic Liver Disease. J. Clin. Med. 2025, 14, 6060. [CrossRef]
- Lake-Bakaar, G. Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD): New Perspectives on an Evolving Epidemic. J. Clin. Med. 2025, 14, 8872. [CrossRef]
- Tacke, F.; Horn, P.; Wong, V.W.; et al. EASL-EASD-EASO Clinical Practice Guidelines on the Management of Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD). J. Hepatol. 2024, 81, 492–542. [CrossRef]
- Hickman, I.J.; Jonsson, J.R.; Prins, J.B.; et al. Modest Weight Loss and Physical Activity in Overweight Patients with Chronic Liver Disease Results in Sustained Improvements in Alanine Aminotransferase, Fasting Insulin, and Quality of Life. Gut 2004, 53, 413–419. [CrossRef]
- Li, N.; Zhang, X.; Zhou, J.; et al. Multiscale Biomechanics and Mechanotransduction from Liver Fibrosis to Cancer. Adv. Drug Deliv. Rev. 2022, 188, 114448. [CrossRef]
- Yosri, M.M.; Hamada, H.A.; Yousef, A.M. Effect of Visceral Manipulation on Menstrual Complaints in Women with Polycystic Ovarian Syndrome. J. Osteopath. Med. 2022, 122, 411–422. [CrossRef]
- Zakaryaei, S.A.; Ravanbakhsh, M.; Javaherizadeh, H.; et al. Effect of Visceral Manipulation on Children with Refractory Chronic Functional Constipation: A Randomized Controlled Trial. Arq. Gastroenterol. 2024, 61. [CrossRef]
- Eguaras, N.; Rodríguez-López, E.S.; Lopez-Dicastillo, O.; et al. Effects of Osteopathic Visceral Treatment in Patients with Gastroesophageal Reflux: A Randomized Controlled Trial. J. Clin. Med. 2019, 8, 1738. [CrossRef]
- Carretero Gómez, J.; Ena, J.; Seguí Ripoll, J.M.; et al. Effect of Newer Antihyperglycemic Drugs on Liver Steatosis Indices in Patients with Diabetes and Obesity. Curr. Med. Res. Opin. 2021, 37, 1867–1873. [CrossRef]
- Carretero-Gómez, J.; Carrasco-Sánchez, F.J.; Fernández-Rodríguez, J.M.; et al. Effect of Semaglutide on Fatty Liver Disease Biomarkers in Patients with Diabetes and Obesity. Rev. Clin. Esp. 2023, 223, 134–143. [CrossRef]
- Newsome, P.N.; Buchholtz, K.; Cusi, K.; et al. A Placebo-Controlled Trial of Subcutaneous Semaglutide in Nonalcoholic Steatohepatitis. N. Engl. J. Med. 2021, 384, 1113–1124. [CrossRef]
- Loomba, R.; Hartman, M.L.; Lawitz, E.J.; et al. Tirzepatide for Metabolic Dysfunction–Associated Steatohepatitis with Liver Fibrosis. N. Engl. J. Med. 2024, 390, 299–310. [CrossRef]
- Jastreboff, A.M.; Kaplan, L.M.; Frías, J.P.; et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity: A Phase 2 Trial. N. Engl. J. Med. 2023, 389, 514–526. [CrossRef]
- Sanyal, A.J.; Bedossa, P.; Fraessdorf, M.; et al. A Phase 2 Randomized Trial of Survodutide in MASH and Fibrosis. N. Engl. J. Med. 2024, 391, 185–197. [CrossRef]
| Visceral Manual Therapy (N=20) |
Control Group (N=20) |
P value | |
| Male, n (%) | 10 (50) | 16 (80) | 0.047 |
| Age, mean (SD) | 52 (13,39) | 54,5 (11,83) | 0.264 |
| BMI, mean (SD) | 33,4 (4,68) | 31,6 (3,06) | 0.076 |
| Smoking, n (%) | 3 (15) | 3 (15) | 1 |
| Systolic BP, n (%) | 136.8 (20.22) | 141.65 (22.60) | 0.24 |
| Diastolic BP, n (%) | 89.65 (11.82) | 85,1 (11.67) | 0.11 |
| Comorbidities, n (%) | |||
| Obesity (BMI >30) | 15 (75) | 14 (70) | 0.723 |
| Type 2 Diabetes, n (%) | 10 (50) | 7 (35) | 0,337 |
| Hypercholesterolemia, n (%) | 16 (80) | 16 (80) | 1 |
| Hypertriglyceridemia, n (%) | 16 (80) | 8 (40) | 0.010 |
| Peripheral arterial disease, n (%) | 4 (20) | 2(10) | 0.376 |
| Chronic Kidney disease, n (%) | 3 (15) | 3 (15) | 1 |
| Medical Treatment, n (%) | |||
| Statins | 16 (80) | 13 (65) | 0.288 |
| Fibrates | 6 (30) | 2 (10) | 0.114 |
| Ezetimibe | 12 (60) | 9 (45) | 0.342 |
| Omega-3 | 6 (30) | 1 (5) | 0.037 |
| Metformin | 15 (75) | 7 (35) | 0.011 |
| SGLT2 inhibitors | 11 (55) | 5 (25) | 0.053 |
| GLP1 analogues | 12 (60) | 6 (30) | 0.057 |
| Insulins | 3 (15) | 1 (5) | 0.292 |
| ACE inhibitors/ARB | 15 (75) | 14 (70) | 0.723 |
| Betablockers | 5 (25) | 3 (15) | 0.429 |
| Diuretics | 12 (60) | 9 (45) | 0.342 |
| Calcio Antagonist | 9 (45) | 10 (50) | 0.752 |
| Hemoglobin | 13.95 (1.53) | 14.21 (1.57) | 0.299 |
| Plaquettes | 267,550 (7,3010.80) | 262,400 (51,657.17) | 0.399 |
| Glucose | 125 (46.47) | 102.15 (13.08) | 0.020 |
| Urea | 41.29 (17.48) | 39.05 (17.77) | 0.323 |
| Creatinine | 0.86 (0.25) | 0.97 (0.19) | 0.059 |
| Sodium | 140.4 (2.74) | 141.20 (2.69) | 0.179 |
| Potassium | 4.69 (0.40) | 4.48 (0.46) | 0.065 |
| Albumin | 4.58 (0.36) | 4.63 (0.26) | 0.325 |
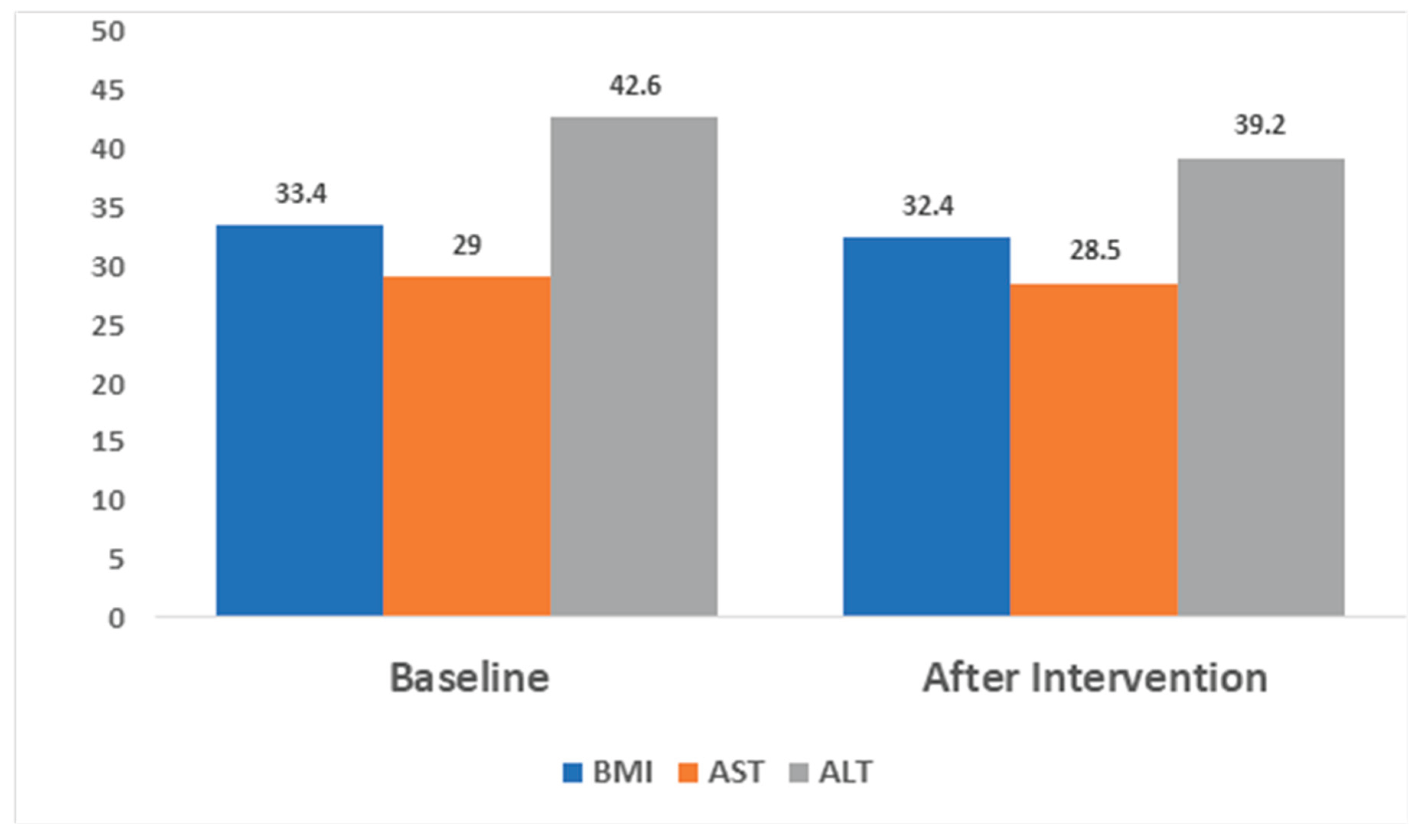
| AST | 29 (15.71) | 22.55 (6.99) | 0.051 |
| ALT | 42.6 (25.71) | 28.05 (12.15) | 0.014 |
| GGT | 73.15 (66.54) | 54.85 (60) | 0.183 |
| Bilirubin | 0.44 (0.19) | 0.53 (0.23) | 0.098 |
| LDH | 173.15 (39.74) | 178.30 (25.58) | 0.314 |
| Total Cholesterol | 155.5 (33.93) | 164.8 (44.82) | 0.232 |
| LDL-Cholesterol | 71.84 (34.36) | 94.5 (42.79) | 0.036 |
| HDL-Cholesterol | 43.54 (11.40) | 50.35 (11.36) | 0.033 |
| No-HDL Cholesterol | 111.85 (31.43) | 113.5 (46.87) | 0.448 |
| Triglycerides | 214.55 (115.38) | 130.35 (63.01) | 0.004 |
| Apolipoprotein B | 85.80 (21.26) | 83.75 (25.07) | 0.391 |
| Ferritin, median (25th-75th) | 133 (50.9-174.9) | 169 (56.9-376) | 0.327 |
| C-protein | 5.02 (5.2) | 2.75 (2.57) | 0.044 |
| HbA1c for T2D | 6.66 (1.3) | 5.81 (0.79) | 0.009 |
| UACR, median (25th-75th) | 7 (2.7-35.1) | 4.9 (2.5-8.1) | 0.192 |
|
Visceral Manual Therapy Group (N=20) |
Control Group (N=20) |
P value | |
| HOMA (mean, SD) | 7.22 (4.77) | 4.17 (2.2) | 0.008 |
| HIS (mean, SD) | 47.40 (6.17) | 42.60 (4.53) | 0.004 |
| NAFLD score (median, 25th-75th) | 1.42 (0.17-2.62) | 1.43 (-0.4-1.82) | 0.672 |
| HAIR (median, 25th-75th) | 2 (1-2) | 1 (1-2) | 0.127 |
| FIB-4 (mean, SD) | 1.03 (0.75) | 0.95 (0.39) | 0.356 |
| APRI (mean, SD) | 0.35 (0.24) | 0.252 (0.08) | 0.054 |
| NAFLD fibrosis (mean, SD) | -91.44 (12.9) | -92.55 (12.32) | 0.784 |
| Visceral Manual Therapy (N=20) | |||
| Basal Score | After proceedings | P value | |
| HOMA (mean, SD) | 7.22 (4.77) | 5.5 (3.20) | 0.018 |
| HIS (mean, SD) | 47.40 (6.17) | 45.55 (4.8) | 0.036 |
| NAFLD score (median, 25th-75th) | 1.42 (0.17-2.62) | 1.32 (0.35-2.63) | <0.001 |
| HAIR (median, 25th-75th) | 2 (1-2) | 1 (1-2) | 0.527 |
| FIB-4 (mean, SD) | 1.03 (0.75) | 1.07 (0.67) | 0.600 |
| APRI (mean, SD) | 0.35 (0.24) | 0.34(0.21) | 0.950 |
| NAFLD fibrosis (mean, SD) | -91.44 (12.9) | -90.35(13.0) | 0.480 |
| Triglycerides (mean, SD) | 214 (115) | 188 (145) | 0.290 |
| Body Mass Index (mean, SD) | 33.43 (8.6) | 32.40 (4.2) | <0.001 |
| Control Group (N=20) | |||
| Basal Score | After proceedings | P value | |
| HOMA (mean, SD) | 4.17 (2.2) | 4.7 (2.22) | 0.266 |
| HIS (mean, SD) | 42.60 (4.53) | 41.9 (3.7) | 0.228 |
| NAFLD score (median, 25th-75th) | 1.43 (-0.4-1.82) | 1.41(0.26-2.1) | 0.125 |
| HAIR (median, 25th-75th) | 1 (1-2) | 1(1-1.75) | 0.257 |
| FIB-4 (mean, SD) | 0.95 (0.39) | 0.99 (0.48) | 0.533 |
| APRI (mean, SD) | 0.252 (0.08) | 0.247 (0.08) | 0.302 |
| NAFLD fibrosis (mean, SD) | -92.55 (12.32) | -92.30 | 0.535 |
| Triglycerides (mean, SD) | 130.8 (63) | 156.4 (136) | 0.352 |
| Body Mass Index (mean, SD) | 31.6 (3.0) | 31.3 (2.6) | 0.865 |
| Diabetes Mellitus (N=17) | |||
| Basal Score | After proceedings | P value | |
| HOMA (mean, SD) | 5.59 (4.8) | 5.25 (3.3) | 0.708 |
| HIS (mean, SD) | 44.53 (5.1) | 43.32 (4.5) | 0.074 |
| GLP1 agonist (N=18) | |||
| Basal Score | After proceedings | P value | |
| HOMA (mean, SD) | 6.47 (5.03) | 5.58 (3.40) | 0.275 |
| HIS (mean, SD) | 46.62 (5.7) | 44.94 (5.03) | 0.028 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




