Submitted:
28 February 2026
Posted:
02 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Study Design and Population
2.2. Study Protocol
2.3. Study Outcomes
2.4. Statistical Analysis
3. Results
3.1. Clinical Characteristics of the Study Population
3.2. Trajectories of Clinical and Subclinical Congestion
3.3. Correlation Between Clinical and Subclinical Congestion
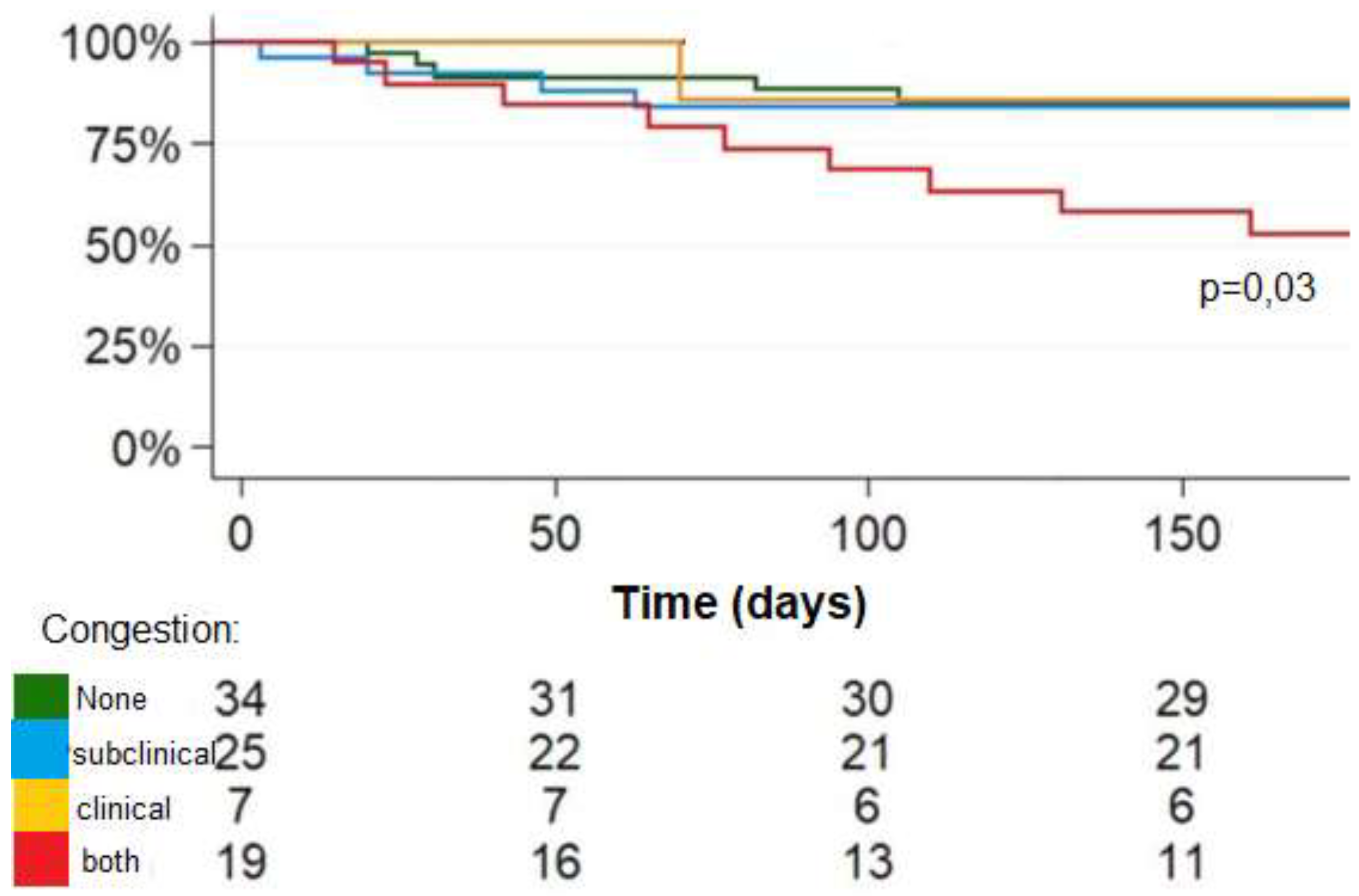
3.4. Prognostic Impact
4. Discussion
4.1. Prognostic Relevance of Congestion
4.2. Prognostic Value of Pulmonary Ultrasound (B-Lines)
4.3. Prognostic Value of ReDS
4.4. Prognostic value of Systemic Venous Congestion (VExUS)
4.5. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Farré, N; Vela, E; Clèries, M; et al. Real-world heart failure epidemiology and outcome: A population-based analysis of 88,195 patients. PLoS One 2017, 12(2), e0172745. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, CM; Miller, AB; Blair, JE; et al. Efficacy of Vasopressin Antagonism in heart Failure Outcome Study with Tolvaptan (EVEREST) investigators. Causes of death and rehospitalization in patients hospitalized with worsening heart failure and reduced left ventricular ejection fraction: results from Efficacy of Vasopressin Antagonism in Heart Failure Outcome Study with Tolvaptan (EVEREST) program. Am Heart J 2010, 159(5), 841–849. [Google Scholar] [PubMed]
- Nohria, A; Tsang, SW; Fang, JC; et al. Clinical assessment identifies hemodynamic profiles that predict outcomes in patients admitted with heart failure. J Am Coll Cardiol. 2003, 41(10), 1797–804. [Google Scholar] [CrossRef] [PubMed]
- Mullens, W; Damman, K; Harjola, VP; et al. The use of diuretics in heart failure with congestion — a position statement from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail 2019, 21(2), 137–55. [Google Scholar] [CrossRef]
- Araiza-Garaygordobil, D; Gopar-Nieto, R; Martinez-Amezcua, P; et al. A randomized controlled trial of lung ultrasound-guided therapy in heart failure (CLUSTER-HF study). Am Heart J 2020, 227, 31–9. [Google Scholar] [CrossRef]
- Marini, C; Fragasso, G; Italia, L; et al. Lung ultrasound-guided therapy reduces acute decompensation events in chronic heart failure. Heart 2020, 106(24), 1934–9. [Google Scholar] [CrossRef]
- Beaubien-Souligny, W; Rola, P; Korbin, H; et al. Quantifying systemic congestion with Point-Of-Care ultrasound: development of the venous excess ultrasound grading system. Ultrasound J 2020, 12(16). [Google Scholar] [CrossRef]
- Sattar, Y; Zghouzi, M; Suleiman, ARM; et al. Efficacy of remote dielectric sensing (ReDS) in the prevention of heart failure rehospitalizations: a meta-analysis. J Community Hosp Intern Med Perspect. 2021, 11(5), 646–52. [Google Scholar] [CrossRef]
- McDonagh, TA; Metra, M; Adamo, M; et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 2021, 42(36), 3599–726. [Google Scholar] [CrossRef]
- Rivas-Lasarte, M; Álvarez-García, J; Fernández-Martínez, J; et al. Lung ultrasound- guided treatment in ambulatory patients with heart failure: a randomized controlled clinical trial (LUS-HF study). Eur J Heart Fail 2019, 21(12), 1605–13. [Google Scholar] [CrossRef]
- Platz, E; Lewis, EF; Uno, H; et al. Detection and prognostic value of pulmonary congestion by lung ultrasound in ambulatory heart failure patients. Eur Heart J 2016, 37(15), 1244–51. [Google Scholar] [CrossRef]
- Álvarez García, J; Lala, A; Rivas-Lasarte, M; et al. Remote Dielectric Sensing before and after discharge in patients with ADHF: The ReDS-SAFE HF trial. JACC Heart Fail 2024, 12(4), 695–706. [Google Scholar] [CrossRef]
- Beaubien-Souligny, W; Benkreira, A; Robillard, P; et al. Alterations in portal vein flow and intrarenal venous flow are associated with acute kidney injury after cardiac surgery: a prospective observational cohort study. J Am Heart Assoc. 2018, 7(19), e009961. [Google Scholar] [CrossRef] [PubMed]
- Longino, A; Martin, K; Leyba, K; et al. Prospective Evaluation of Venous Excess Ultrasound for Estimation of Venous Congestion. CHEST 2024, 165(3), 590–600. [Google Scholar] [CrossRef] [PubMed]
- Islas-Rodríguez, JP; Miranda-Aquino, T; Romero-González, G; et al. Effect on.
- Kidney Function Recovery Guiding Decongestion with VExUS in Patients with Cardiorenal Syndrome: A Randomized Control Trial. Cardiorenal Med 2024, 14(1), 1–11.
- Aslaner, MA; Helvacı, Ö; Haycock, K; et al. Diagnostic accuracy of venous system ultrasound for subtypes of acute kidney injury. Emerg Med J 2024, 41(5), 304–10. [Google Scholar] [CrossRef] [PubMed]
- Sovetova, S; Charaya, K; Erdniev, T; et al. Venous Excess Ultrasound Score is.
- associated with Worsening Renal Function and reduced natriuretic response in patients with Acute Heart Failure. J Clin Med. 2024, 13(20), 6272. [CrossRef]
- Kanitkar, S; Soni, K; Vaishnav, B; et al. Venous Excess Ultrasound for fluid.
- assessment in complex cardiac patients with acute kidney injury. Cureus 2024, 16(8), e66003. [CrossRef] [PubMed]
| Variable | N (%) |
|---|---|
| Sex, male | 64 (68.0) |
| Comorbidities: | |
| Hypertension | 65 (69.1) |
| Diabetes mellitus | 35 (37.2) |
| Dyslipidaemia | 49 (52.1) |
| History of smoking | 39 (41.5) |
| Alcohol abuse | 18 (19.1) |
| Atrial fibrillation | |
| – None | 25 (26.7) |
| – Paroxysmal | 21 (22.3) |
| – Persistent | 13 (13.8) |
| – Permanent | 35 (37.2) |
| Chronic kidney disease (glomerular filtration <60 ml/min/m²) | 37 (39.4) |
| Chronic obstructive pulmonary disease | 16 (17.0) |
| Sleep apnoea-hypopnea syndrome | 10 (10.9) |
| Stroke | 9 (9.9) |
| Peripheral vascular disease | 5 (5.4) |
| HF profile: | |
| First episode of HF | 8 (8.5) |
| NYHA functional class | |
| – I | 2 (2.1) |
| – II | 58 (61.7) |
| – III | 31 (33.0) |
| – IV | 3 (3.2) |
| Aetiology | |
| – Valvular | 25 (26.7) |
| – Ischemic | 20 (21.6) |
| – Idiopathic | 15 (16.0) |
| – Hypertensive | 14 (14.9) |
| – Restrictive | 10 (10.9) |
| – Other | 9 (9.9) |
| LVEF category | |
| – Preserved (≥50%) | 55 (58.5) |
| – Intermediate (41-49%) | 20 (21.3) |
| – Reduced (<40%) | 19 (20.2) |
| Variable | Admission (n=94) | 72 hours (n=93)* | Pre-Discharge (n=89)* | HF clinic visit after discharge (n=79) | P value |
|---|---|---|---|---|---|
| Orthopnoea |
<0.001 |
||||
| - None | 28 (29.8) | 67 (72.0) | 86 (96.6) | 68 (90.6) | |
| - Sometimes | 14 (14.9) | 22 (23.6) | 3 (3.4) | 4 (5.5) | |
| - Always | 18 (19.1) | 1 (1.1) | 0 | 1 (1.3) | |
| - Paroxysmal nocturnal dyspnoea | 34 (36.2) | 3 (3.3) | 0 | 2 (2.6) | |
| Crackles |
<0.001 |
||||
| - None | 20 (21.3) | 69 (74.2) | 86 (96.6) | 67 (89.3) | |
| - Basal | 64 (68.1) | 23 (24.7) | 3 (3.4) | 8 (10.7) | |
| - Mid | 10 (10.6) | 1 (1.1) | 0 | 0 | |
| Pleural effusion | 26 (27.6) | 8 (8.6) | 3 (3.4) | 6 (8.1) | <0.001 |
| Weight, kg | 75.8 ±18.1 | 73.5 ±16.9 | 71.7 ±17.2 | 72.6 ±15.6 | <0.001 |
| Oedema |
<0.001 |
||||
| - None | 18 (19.1) | 54 (58.1) | 77 (86.5) | 54 (72.0) | |
| - Malleolar | 36 (38.3) | 27 (29.0) | 12 (13.5) | 16 (21.3) | |
| - Knee | 17 (18.1) | 8 (8.6) | 0 | 3 (4.0) | |
| - Above knee | 23 (24.5) | 4 (4.3) | 0 | 2 (2.7) | |
| Hepatomegaly |
<0.001 |
||||
| - None | 46 (48.9) | 69 (74.2) | 81 (91.0) | 52 (69.6) | |
| - 1–2 fingerbreadths | 28 (29.8) | 21 (22.6) | 7 (7.9) | 19 (24.0) | |
| - >2 fingerbreadths | 20 (21.3) | 3 (3.2) | 1 (1.1) | 4 (6.4) | |
| Jugular venous distension |
<0.001 |
||||
| - None | 39 (41.5) | 62 (66.7) | 72 (80.9) | 47 (62.7) | |
| - < SCM | 33 (35.1) | 27 (29.0) | 17 (19.1) | 24 (32.0) | |
| - SCM | 14 (14.9) | 3 (3.2) | 0 | 3 (4.0) | |
| - Mandible | 8 (8.5) | 1 (1.1) | 0 | 1 (1.3) | |
| Hepatojugular reflux | 37 (39.4) | 19 (20.7) | 7 (7.9) | 14 (18.9) | <0.001 |
| Clinical congestion score | 4.0 ±2.1 | 1.5 ±1.6 | 0.3 ±0.6 | 0.8 ±1.3 | <0.001 |
| Variable | Admission (n=94) | 72 hours (n=93) |
Pre-Discharge (n=89) | HF clinic visit after discharge (n=79) | P value |
|---|---|---|---|---|---|
| NT-ProBNP, pg/mL | 6,180 (3,749–11,162) | 3,406 (1,655–7,085) | 2,713 (1,046–5,423) | 3,414 (818–6,642) | 0.003 |
| CA125, U/mL | 37.5 (21–62) | 48 (12–86) | 40 (11–40) | Not available | <0.001 |
| Creatinine, mg/dL | 1.4 (0.7) | 1.5 (0.8) | 1.4 (0.7) | 1.6 (0.7) | 0.472 |
| Presence of B-lines, n (%) |
97% | 82% | 52% | 54% | <0.001 |
| No. of zones with ≥3 B-lines | 4.8 ± 2.0 | 2.7 ± 2.1 | 1.3 ± 1.8 | 1.8 ± 2.2 | <0.001 |
| ReDS, % | 34.0 ± 7.6 | 32.3 ± 6.1 | 29.9 ± 6.5 | 29.2 ± 5.9 | <0.001 |
| IVC diameter, mm | 22.6 ± 5.1 | 19.7 ± 4.9 | 18.2 ± 5.3 | 18.7 ± 6.1 | <0.001 |
| Suprahepatic veins |
<0.001 |
||||
| - S>D | 28 (33.7) | 37 (43.5) | 41 (51.2) | 35 (54.7) | |
| - D>S | 21 (25.3) | 20 (23.5) | 13 (16.2) | 6 (25.0) | |
| - Inversion | 34 (41.0) | 28 (33.0) | 26 (32.6) | 23 (20.3) | |
| Portal vein pulsatility |
<0.001 |
||||
| - <30% | 31 (39.7) | 47 (58.8) | 53 (61.7) | 35 (54.7) | |
| - 30–50% | 29 (37.2) | 22 (27.5) | 16 (25.0) | 13 (20.3) | |
| - >50% | 18 (23.1) | 11 (13.7) | 13 (20.3) | 8 (10.1) | |
| Renal Doppler |
<0.001 |
||||
| - Continuous | 26 (32.1) | 43 (50.6) | 48 (59.3) | 30 (46.9) | |
| - Biphasic | 41 (50.6) | 37 (42.0) | 26 (32.1) | 22 (34.4) | |
| - Monophasic | 14 (17.3) | 6 (7.4) | 7 (8.6) | 12 (18.7) | |
| VExUS score, mean ± SD | 1.3 ± 1.0 | 0.95 ± 1.0 | 0.64 ± 1.0 | 0.97 ± 1.3 | |
| VExUS points, n (%) | <0.001 | ||||
| - 0 | 22 (25.6) | 45 (51.1) | 60 (69.8) | 39 (57.4) | |
| - 1 | 30 (34.9) | 21 (23.9) | 8 (9.3) | 9 (13.2) | |
| - 2 | 20 (23.2) | 14 (15.9) | 11 (12.8) | 5 (7.4) | |
| - 3 | 14 (16.3) | 8 (9.1) | 7 (8.1) | 15 (22.0) | |
| Variable | VExUS | B-lines | NT-proBNP | Creatinine | ReDS | Congestion | Crackles | Hepatomegaly | HJR |
|---|---|---|---|---|---|---|---|---|---|
| B-lines | 0.36* | ||||||||
| NT-proBNP | 0.26* | 0.29* | |||||||
| Creatinine | 0.11* | -0.02 | 0.25* | ||||||
| ReDS | 0.23* | 0.38* | 0.09 | 0.11 | |||||
| Congestion | 0.38* | 0.53* | 0.30* | 0.02 | 0.23* | ||||
| Crackles | 0.17* | 0.54* | 0.18* | -0.03 | 0.26* | 0.57* | |||
| Hep | 0.40* | 0.38* | 0.29* | 0.10 | 0.03 | 0.63* | 0.25* | ||
| HJR | 0.29* | 0.21* | 0.19* | 0.09 | 0.08 | 0.53* | 0.23* | 0.51* | |
| Oedema | 0.31* | 0.41* | 0.21* | -0.03 | 0.11 | 0.83* | 0.46* | 0.53* | 0.41* |
| Variable | HR | 95% CI | P value |
|---|---|---|---|
| Presence of B-lines | 3.50 | 1.41–8.72 | 0.007 |
| NT-ProBNP | 1.00 | 0.99–1.00 | 0.09 |
| ReDS >35% | 2.05 | 0.69–6.13 | 0.20 |
| Clinical congestion | 2.67 | 1.13–6.30 | 0.02 |
| VExUS ≥1 | 1.84 | 0.73–4.68 | 0.20 |
| IVC diameter | 1.07 | 1.01–1.13 | 0.01 |
| Portal vein pulsatility | 1.92 | 1.01–3.66 | 0.05 |
| SH | 1.38 | 0.82–2.32 | 0.22 |
| Renal Doppler | 1.44 | 0.77–2.68 | 0.25 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




