Submitted:
28 February 2026
Posted:
02 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
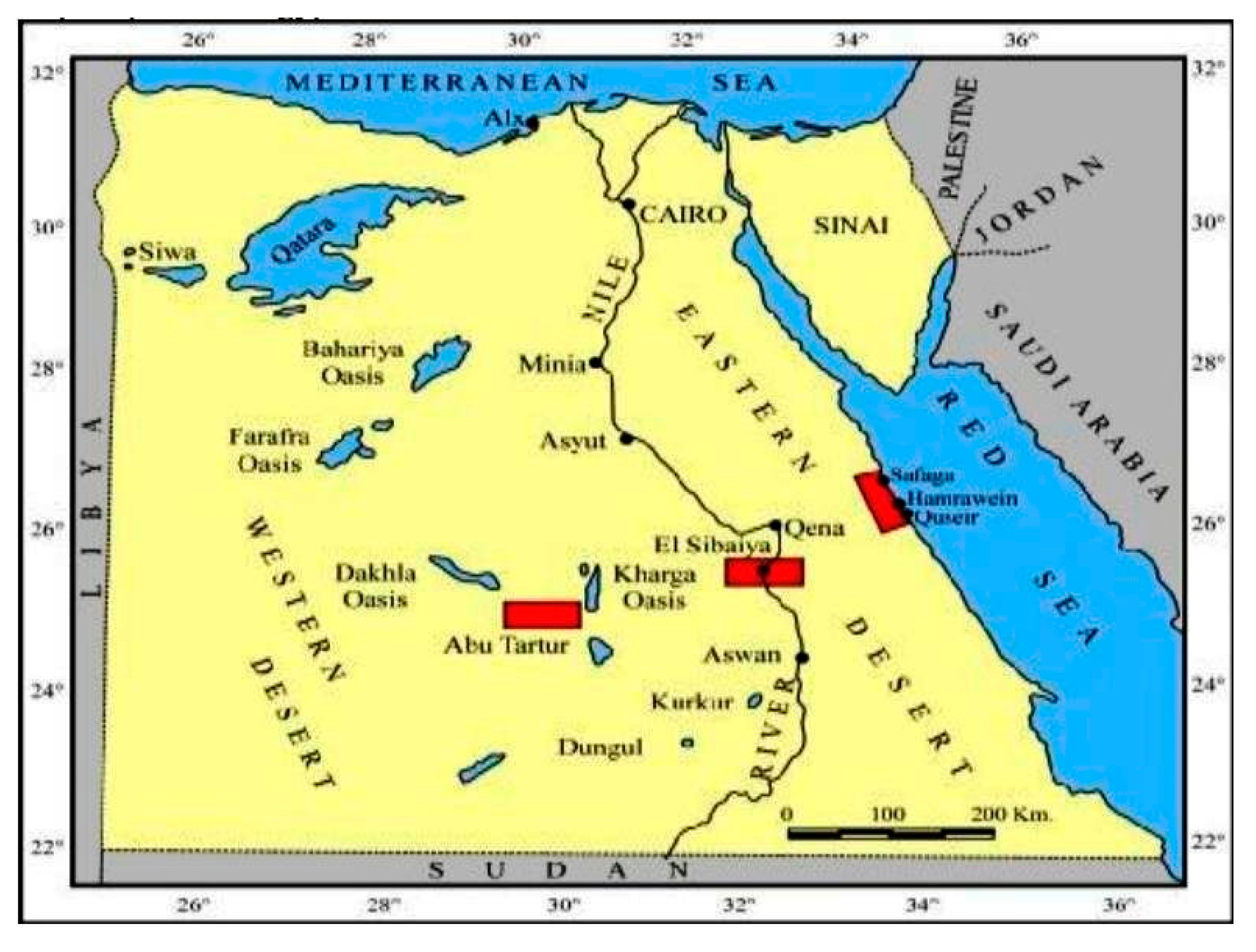
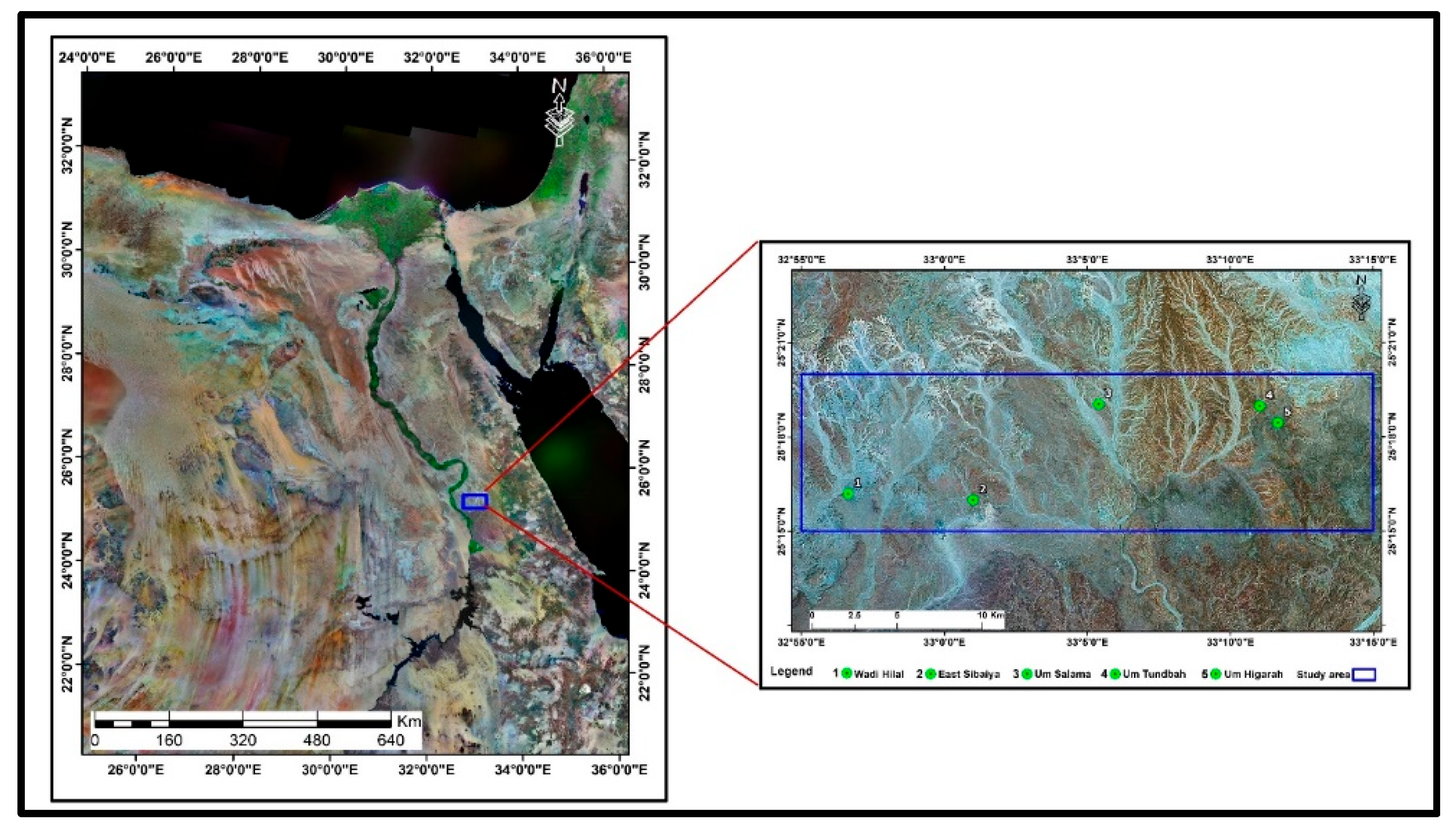
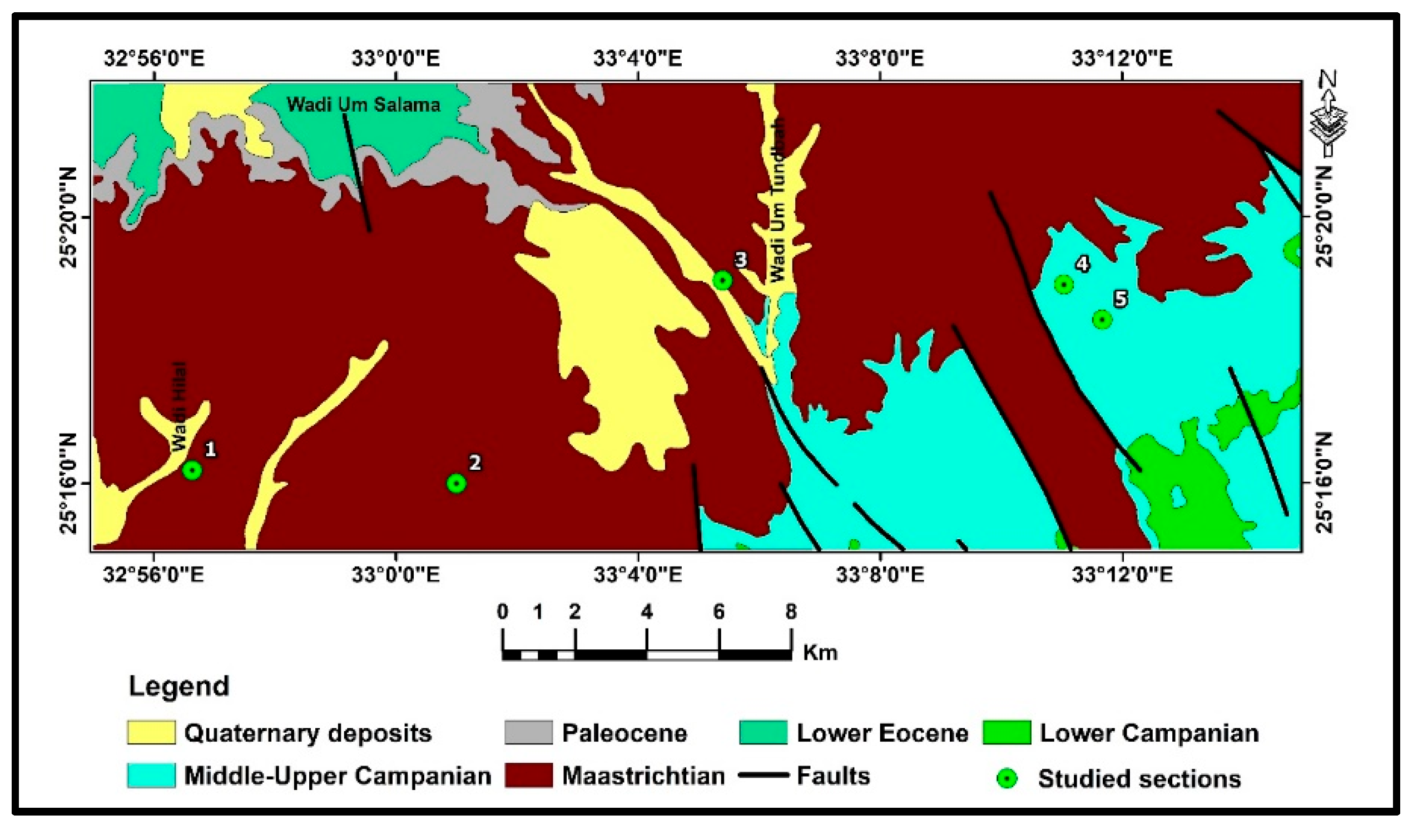
3. Geological Setting
4. Results
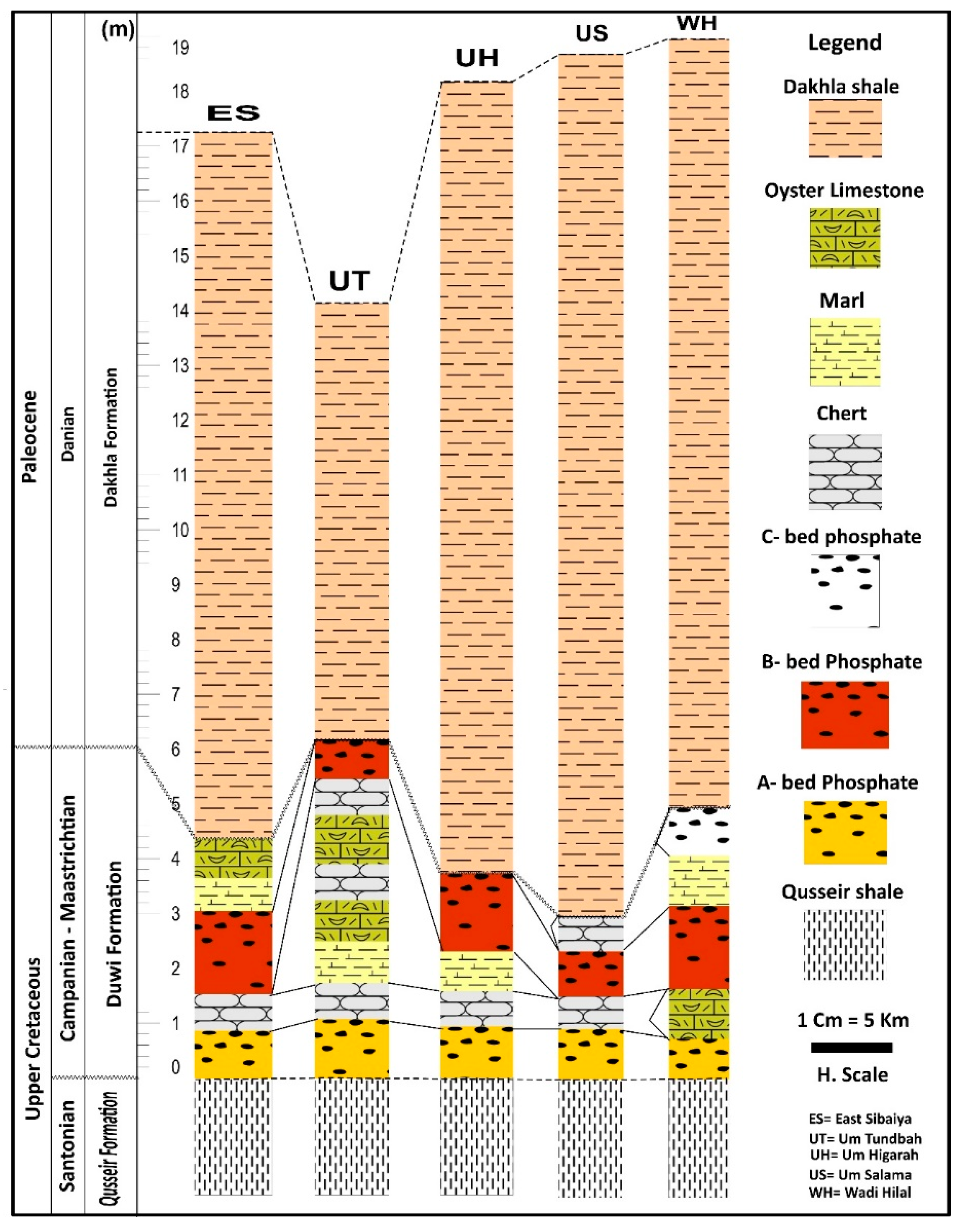
4.1. Field Description
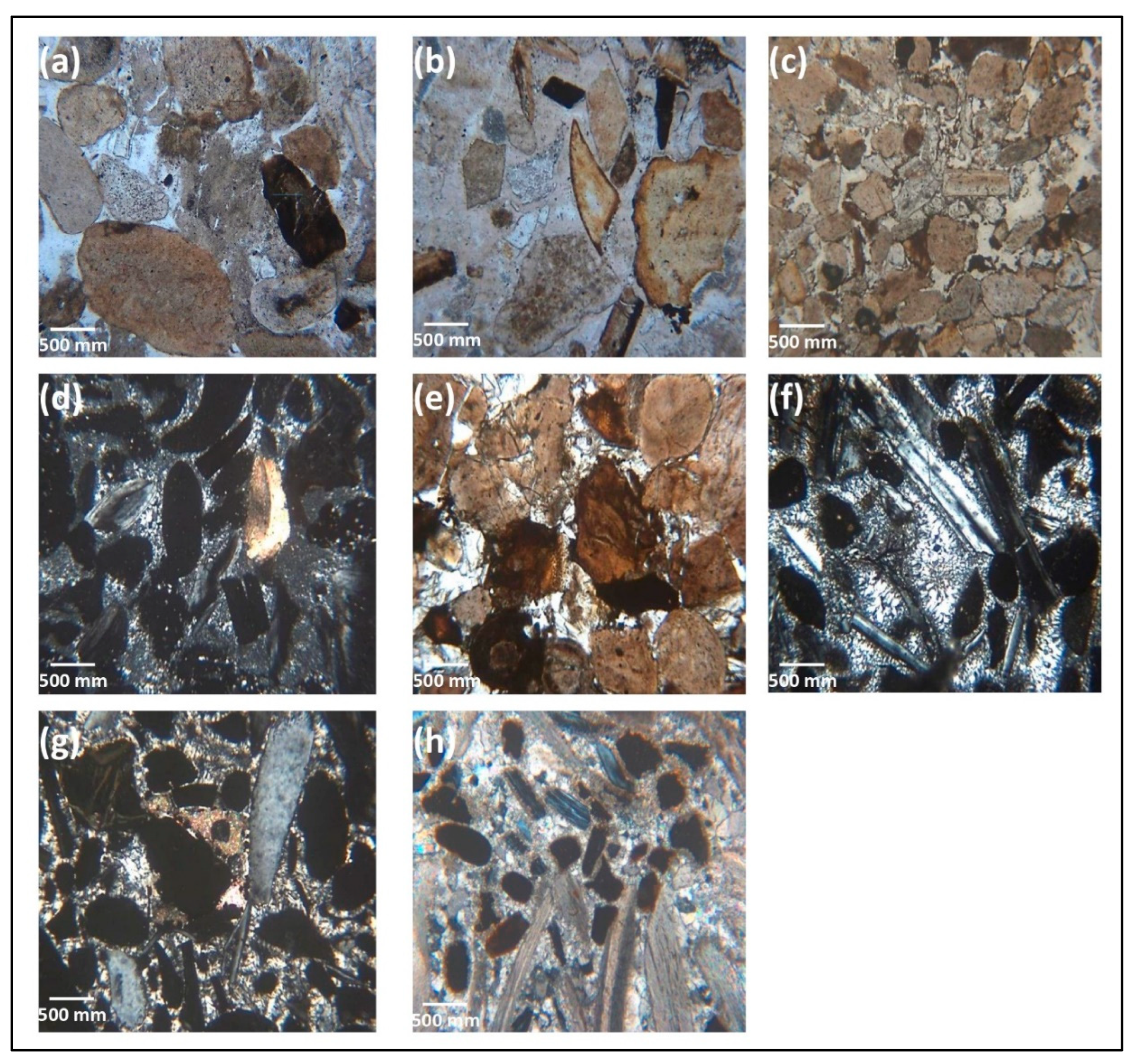
4.2. Mineralogical Composition
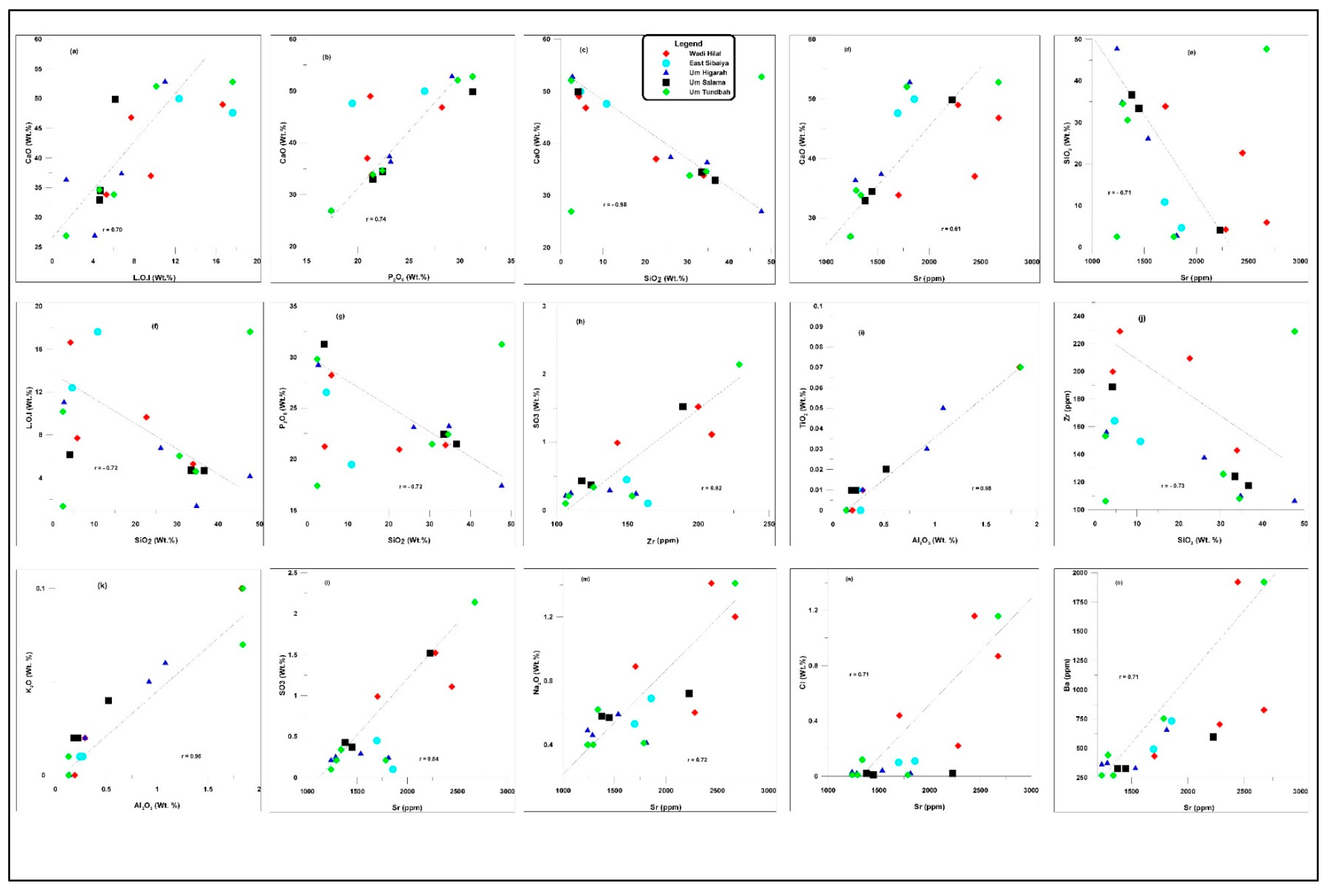
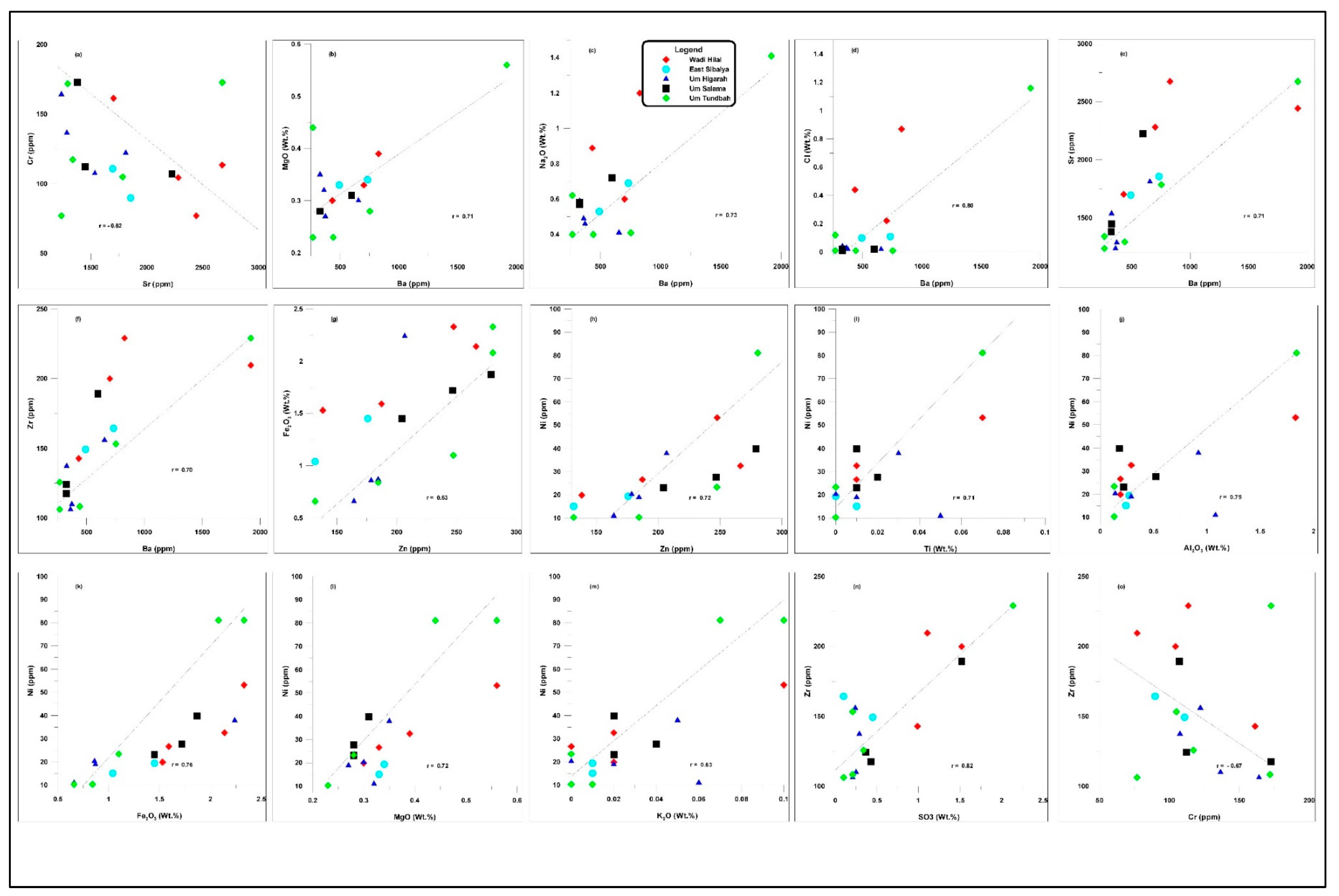
4.3. Whole-Rock Geochemistry
4.3.1. Major Elements
4.3.2. Trace and Radioactive Elements
5. Discussion
5.1. Lithostratigraphic Positions
5.2. Petrographic Characteristics
5.3. Geochemical Implications
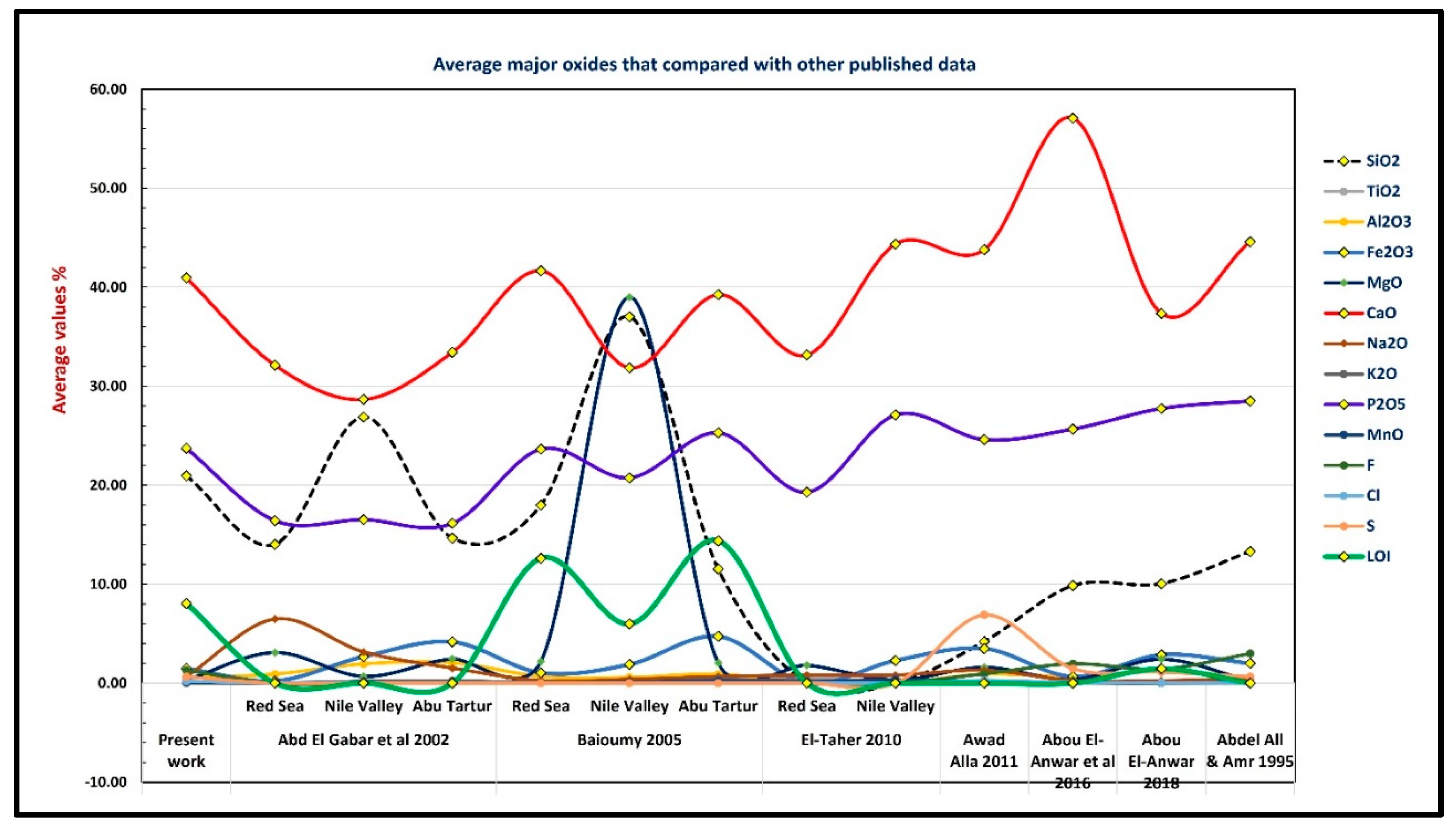
5.4. Comparative Study
5.4.1. Average Major Oxides and Trace Elements
5.4.2. Radioactive Elements
5.4.3. Rare Earth Elements
6. Conclusions
- The stratigraphic and petrographic characteristics of the studied succession collectively reflect a progressive transition from continental siliciclastic settings to fully marine environments during the Late Cretaceous–early Paleocene.
- The Qusseir variegated shale marks the initial phase of this transition, recording low-energy coastal to delta-plain conditions influenced by alternating oxidation states and episodic clastic influxes. This unit represents the onset of marine encroachment over the Nubia Sandstone.
- The overlying Duwi Formation preserves the key stages of phosphorite formation on a shallow epicontinental shelf affected by Late Cretaceous global sea-level rise. Phosphogenesis was driven by high marine productivity, redox fluctuations, upwelling activity, and low sedimentation rates.
- The Dakhla shale records the maximum transgression and marks the shift from shallow-shelf phosphatic deposition to deeper, low-energy offshore sedimentation. Its fine-grained, organic-rich character indicates dysoxic bottom-water conditions conducive to enhanced preservation of organic matter.
- Petrographic evidence, such as abundant pellets, bioclasts, and reworked phosphatic grains, indicates oscillation between low and moderate energy conditions, allowing condensed phosphate accumulation. Associated lithofacies, including oyster limestone, marl, and chert, further confirm deposition under nutrient-rich, intermittently oxygen-deficient waters.
- Geochemical data support these interpretations. Silica is mostly detrital, with minor diagenetic microcrystalline forms. Variations in Fe₂O₃ reflect diagenetic oxidation of pyrite, whereas low MgO contents indicate minimal dolomitization. Trace-element relationships highlight the roles of lattice substitution and diagenetic redistribution, with Sr content linked to calcite and Zn enrichment related to its partial substitution for Ca in the apatite lattice.
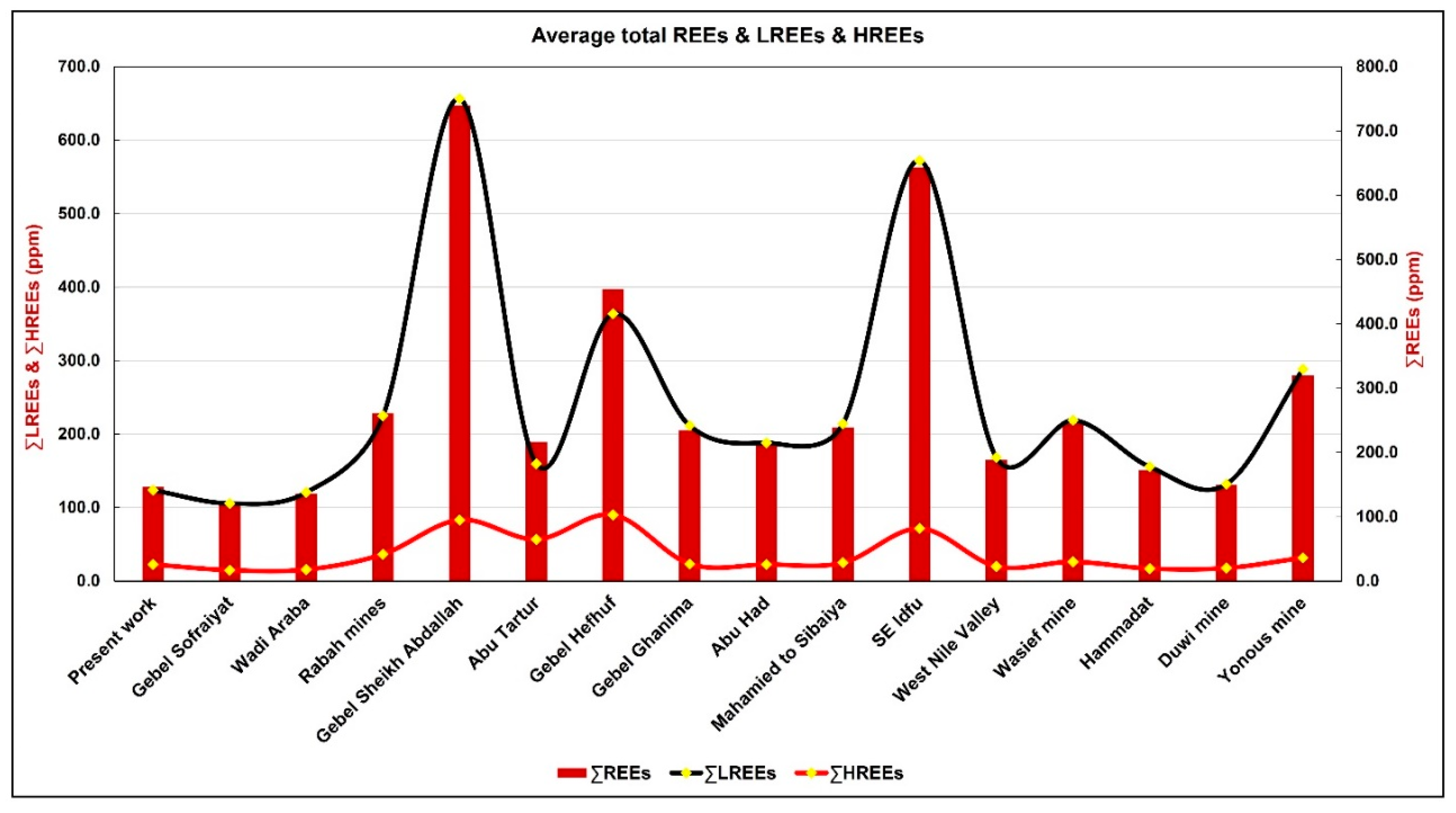
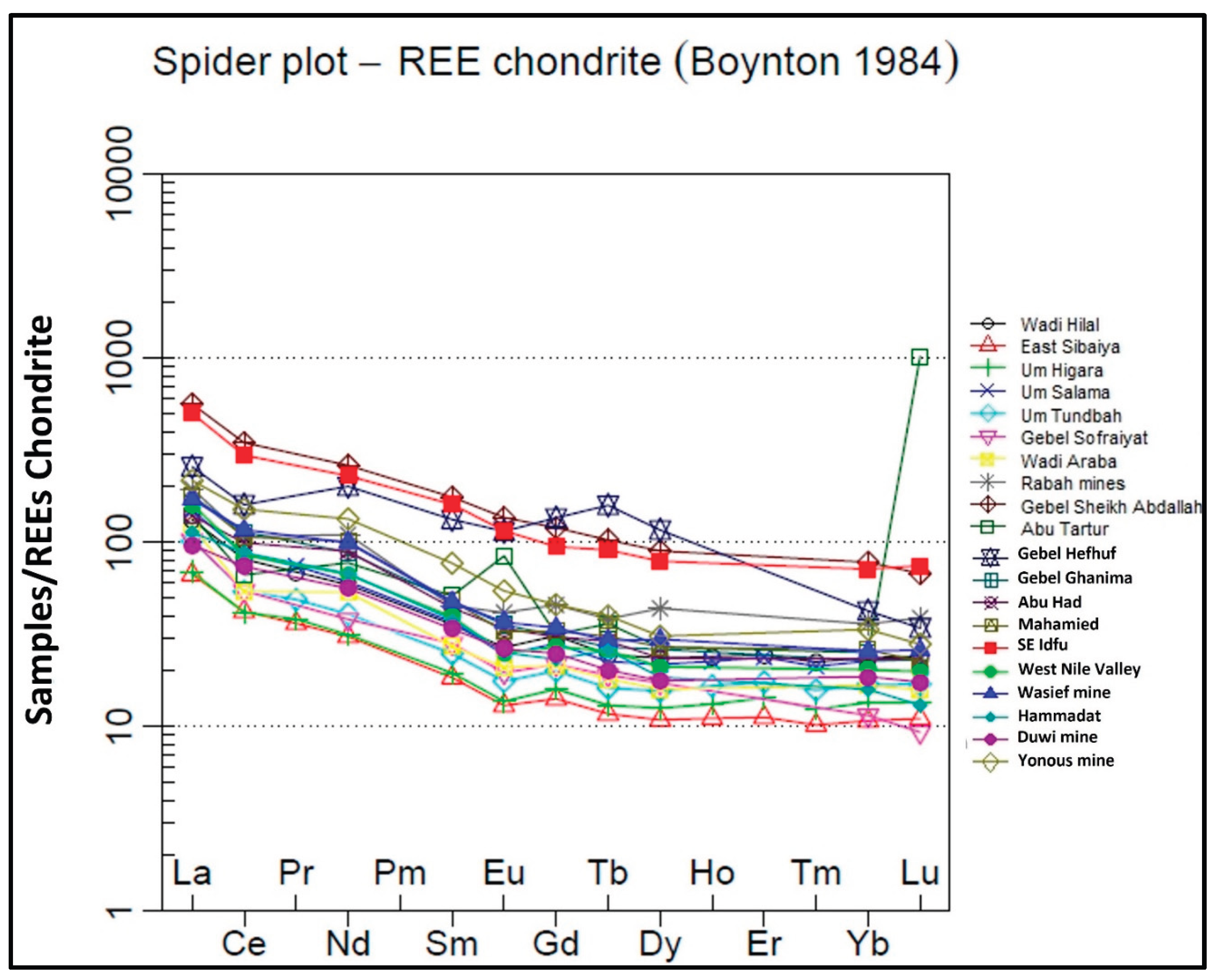
- Light rare earth element patterns (LREE) enrichment and slight negative Ce and Eu anomalies indicate marine deposition under predominantly reducing conditions and subsequent post-depositional modification.
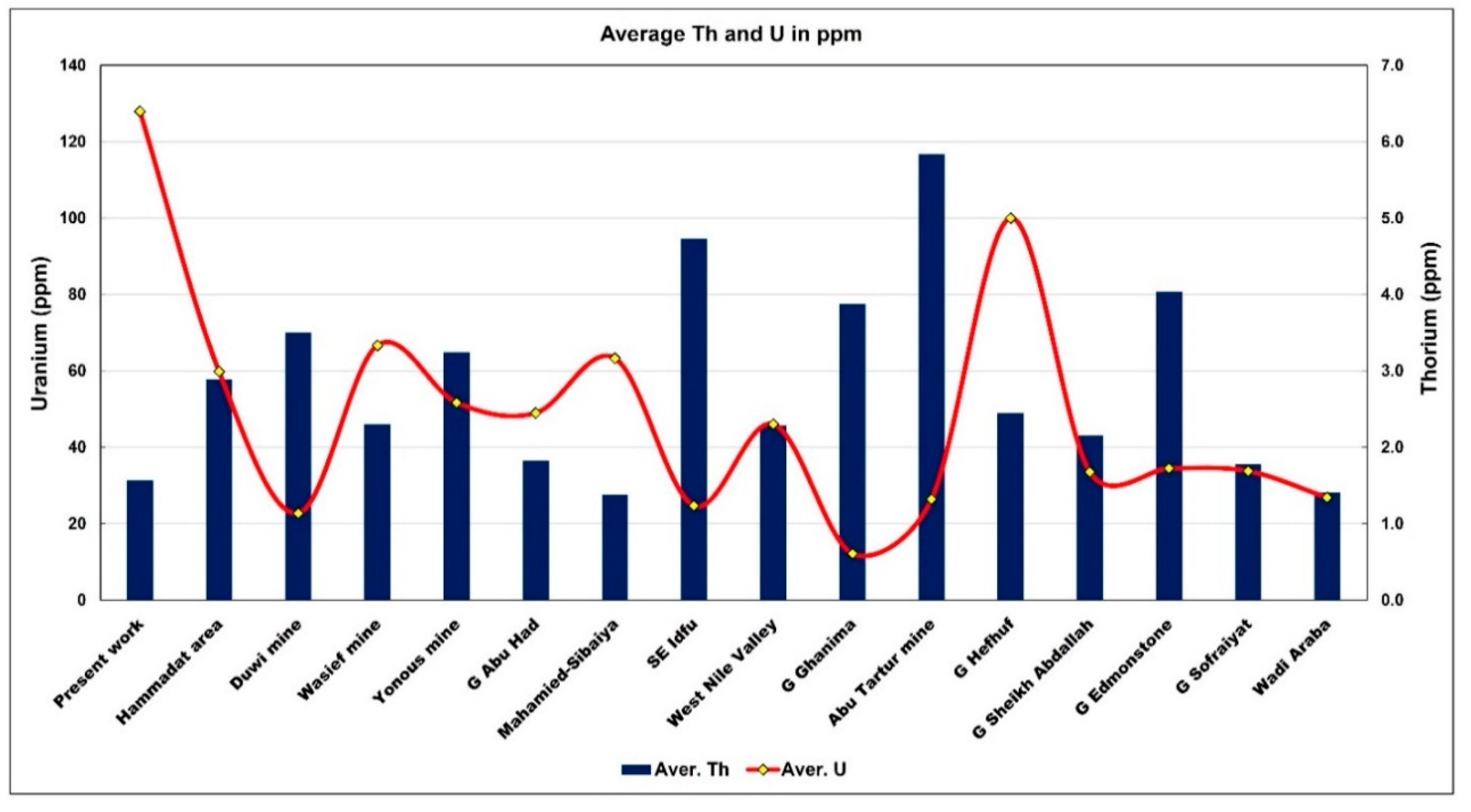
- The thermal activity related to the Oligo-Miocene age seems to be responsible for the high uranium and REEs contents.
- In general, the geological, petrographic, and geochemical evidence demonstrates that the Duwi phosphorites formed under conditions highly favorable for economically significant phosphate accumulation, shaped by upwelling-driven productivity, fluctuating redox conditions, and episodic sediment reworking across a shallow epicontinental shelf.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| REEs | Rare Earth Elements |
| HREEs | Heavy Rare Earth Elements |
| LREEs | Light Rare Earth Elements |
| XRF | X-Ray Fluorescence |
| ICP-MS | Inductively Coupled Plasma-Mass Spectrometry |
| LOI | Loss on Ignition |
References
- Hermina, M.H., 1972, Review of the phosphate deposits of Egypt. Proceedings of the 2nd Arab Conference of Mineral Resources, Conf. papers, pp.109-149.
- Notholt, A.J.G., 1985, Phosphorite resources in the Mediterranean (Tethyan) phosphatic province: a progress report. Sciences Gʹeologiques, Mʹemoire 77, pp.9-17.
- Youssef, M. I., 1954, Stratigraphy of Gabel Oweina section near Esna, Upper Egypt, Bull. Inst. Desert Egypt, v. 4/ 2, pp. 83-93.
- Youssef, M.I., 1957, Upper Cretaceous rocks in the Qusseir area. Bull. Inst., Desert Egypt, 7, pp.35-54.
- Awad, G. H., and Ghobrial, M. G., 1966, Zonal stratigraphy of Kharga Oasis, Annals of the Geological Survey of Egypt, v.34, pp.1-77.
- El-Naggar, Z. R., 1966, Stratigraphy and planktonic foraminifer of the Upper Cretaceous-Lower-Tertiary succession in the Esna-Idfu region, Nile Valley, Egypt. Bulletin of the British Museum (Natural History), Geology Suppl., 2, 291p.
- El-Naggar, Z. R., 1970, The genus Rugoglobigerina in the Maastrichtian Sharawna shale, Egypt. Proceedings II Planktonic Conference, Roma, I, pp.477-537.
- Issawi, B., 1972, Review of Upper Cretaceous-Lower Tertiary stratigraphy in central and southern Egypt, Ann. Assoc. Petrol. Geol. Bull., 56/ 8, pp.1448-1463.
- Issawi, B., Abdallah, A.M., and Said, M.M., 1978, Geology of Wadi El Mashash Area, Eastern Desert-Egypt. Ann. Geol. Surv. Egypt, 8, pp.163-185.
- Issawi, B., El-Hinnawi, M., Francis, M., and Mazhar, A., 1999, The Phanerozoic geology of Egypt: A geodynamic approach. Special Publication No. 76. Geol. Survey, Cairo.
- Klitzsch, E., and Wycisk, P., 1987, Geology of sedimentary basins of Northern Sudan and bordering areas. Ber. Gewiss. Abh, 75 (A), 1, pp. 97-136.
- Klitzsch, E., Groeschke, M., and Hermann-Degen, W., 1990, Wadi Qena: Paleozoic and Precambrian Cretaceous strata. In R. Said (ed.), The Geology of Egypt, Rotterdam, Balkema, pp. 321-327.
- Baioumy, H.M., Tada, R., 2005, Origin of Upper Cretaceous phosphorites in Egypt. Cretac. Res., 26, pp.261-275. [CrossRef]
- Abou El-Anwar, E.A., Mekky, H.S., Abd El Rahim, S.H., Aita, S.K., 2017, Mineralogical, geochemical characteristics, and origin of Late Cretaceous phosphorite in Duwi Formation (Gebel Duwi Mine), Red Sea region, Egypt. Egyptian J Petrol, 26, pp.157 ̶ 169. [CrossRef]
- Abou El-Anwar, E.A., 2019a, Lithologic characterization of the phosphorite-bearing Duwi Formation (Campanian), South Esna, West Nile Valley, Egypt. Carbonates and Evaporates 34, pp.793-805.
- Abou El-Anwar, E.A., and Abd El Rahim, S.H., 2022, Mineralogy, geochemistry, and origin of the phosphorites at Um El-Huwtat mine, Qusseir, Central Eastern Desert, Egypt. Carbonates and Evaporites, 37:16, 18p.
- Youssef, M. I., 1949, Stratigraphical studies in Qusseir area. Ph. D. Thesis, Alex. Univ., Egypt, pp.35-54.
- Ghorab, M. A., 1956, A summary of proposed rock stratigraphic classification for the Upper Cretaceous rocks in Egypt. Geol. Soc. Egypt. April 9th.
- Said, R., 1962, The geology of Egypt. Elsevier, Amsterdam, 337p.
- El-Kammar, A., 1974, Comparative geochemical and mineralogical studies on Egyptian phosphorites. Ph.D. Dissertation, Geology Department, Faculty of Science, Cairo University, Giza, Egypt, 169p.
- Hassan, F., and El-Kammar, A.M., 1975, Environmental conditions affecting the accumulation of uranium and rare earths in Egyptian phosphorites. Egyptian Journal of Geology, v.19 (2), pp.169-178.
- Said, R., 1990, Geology of Egypt, A. A. Balkeina, Brockfield Rotterdam, Rotterdam, 722p.
- El-Kammar, A., and El-Kammar, M., 2002, on the trace elements composition of the Egyptian phosphorites: A new approach. 6th international conference on the geology of the Arab World, Cairo University, Feb., pp.227-244.
- El-Kammar, A.M., Darwish, M., Philip, G., and El-Kamaar, M.M., 1990, Composition and origin of black shales from Qusseir area, Red Sea. Egypt Journal of the University of Kuwait-Science, 17, pp.177-1989.
- Cook, P.J., and McElhinny, M.W., 1979, A reevaluation of the spatial and temporal distribution of sedimentary phosphate deposits in the light of plate tectonics. Economic Geology 74, pp.315 -330. [CrossRef]
- Glenn, C.R., and Arthur, M.A., 1990, Anatomy and origin of a Cretaceous phosphorite-greensand giant, Egypt. Sedimentology, 37, pp.123 ̶ 154.
- Lucas, J., Chaabani, F., and Prevot, L., 1979, Phosphorites et evaporates: deux formations de mileux sedimentaires voisins dans la coup Paleogene de Foum Selja (Metlaoui, Tunisie). Sci. Geol. Bull., v.32, pp.7-20. [CrossRef]
- McHargue, T.R., and Price, B., 1982, Dolomite from clay in argillaceous or shale-associated marine carbonates. J Sed. Petro., v.52, pp.873-886.
- El-Kammar, A., and El-Kammar, M., 1996, potentially of chemical weathering under arid conditions of black shales from Egypt. J Arid Environments, v.33, pp.179-199. [CrossRef]
- Tucker, M.E., 1984, Introduction to sedimentary petrology, Geoscience Texts, 3, London and Tonbridge, 252p.
- Cathcart, J., and Gulbradsen, R., 1973, Phosphate deposits, U.S. Geol. Surv., Prof. Paper, pp.820-515.
- Zidan, I.H., 2014, Evaluation of phosphorite and Uranium in Lower phosphorite Member, Duwi Formation at Kummer area, South Esna, West Nile Valley, Egypt. Sedimentology of Egypt, 21, pp.143-154.
- De Silva, E.F.D., Ammar, M., Celso, G., Fernando, N., Abdelkrim, C., Cristina, S., Valdemar, E., Ana, R., and Marques, F., 2010, Heavy elements in the phosphorite from Kalaat Khasba mine (North-western Tunisia): potential implications on the environment and human health. J Hazard Mater., 182, pp.232–245. [CrossRef]
- McConnell, D., 1973, Apatite, its crystal chemistry, mineralogy, utilization, and geologic and biologic occurrences, New York.
- Blatt, D., Middleton, G., and Murray, R., 1972, Origin of sedimentary rocks. Prentice-Hall, New Jersey.
- Dabous, A. A., 1981, Mineralogy, geochemistry, and radioactivity of some Egyptian phosphate deposits. Ph.D. Thesis. Florida State Univ. College of Arts and Sciences. 201p.
- Nemliher, J. G., Baturin, G. N., Kallaste, T. E., and Murdmaa, I. O., 2004, Transformation of hydroxyapatite of bone phosphate from the ocean bottom during fossilization. Lithology Mineral Resources. 39, pp.468-479. [CrossRef]
- El-Kammar, A.M., Zayed, M.A., Amer, S.A., 1979, Rare earths of the Nile Valley phosphorites, Upper Egypt. Chem Geol., 24, pp.69-81. [CrossRef]
- Zidan, I.H., 2002, Geological and Geochemical studies of Abu-Tartur phosphate, Western Desert, Egypt, Ph. Fac. Sci., Al-Azhar Univ., 235p.
- Altschuler, Z.S., 1980, The geochemistry of trace elements in marine phosphorites, part I: characteristic abundances and enrichment. In: Bentor YK (ed) Marine phosphorites, Spec Publication. 29, pp.19-30.
- Basta, E., and El Kammar, A. M., 1976, Mineralogy and geochemistry of the phosphorites of Abu Tartur, WesternDesert, Egypt. The 25th International Geological Congress, Sydney, Australia, 2, Section 14, 555p.
- Weinberg, J.M., and Cowart, J.B., 2001, Hydrogeologic implications of uranium-rich phosphate in northeastern Lee County. In: Missimer, T.M. and Thomas, M.S. (Eds.), Geology and Hydrogeology of Lee County, Florida. Florida Geological Survey Special Publication No. 49, pp.151-165.
- Abd El-Gabar, M., El-Arabia, I., and Khalifa, H., 2002, Application of multivariate statistical analyses in the interpretation of geochemical behavior of uranium in phosphatic rocks in the Red Sea, Nile Valley and Western Desert, Egypt. J. Environ. Radioact., 6, pp.169-190.
- Baioumy, H.M., 2005, Preliminary data on cadmium and arsenic geochemistry for some phosphorites in Egypt. Journal of African Earth Sciences, 41, pp.266-274. [CrossRef]
- El-Taher, A., 2010, Elemental analysis of two Egyptian phosphate rock mines by instrumental neutron activation analysis and atomic absorption spectrometry. App Radiat Isot., 68, pp.511-515.
- Awadalla, G.S., 2011, Geochemistry and microprobe investigations of Abu Tartur REE-bearing phosphorite, Western Desert, Egypt. Journal of African Earth Sciences, 57, pp.431-443. [CrossRef]
- Abou El-Anwar, E.A., Mekky, H.S., Abd El Rahim, S.H., Aita, S.K., 2017, Mineralogical, geochemical characteristics, and origin of Late Cretaceous phosphorite in Duwi Formation (Geble Duwi Mine), Red Sea Region, Egypt. Egyptian J Pet., 26, pp.157–169. [CrossRef]
- Abou El-Anwar, E.A., 2019, Lithologic characterization of the phosphorite-bearing Duwi Formation (Campanian), South Esna, West Nile Valley, Egypt. Carbonates and Evaporites, 34, pp.793–805.
- Abdel-All, E.A., and Amer, A.M., 1995, Evaluation of Sibaiya-West phosphate concentrate for nitrophosphate fertilizer production. Miner. Eng., 8(10), pp.1221–1230. [CrossRef]
- Ismael, I.S., 2002, Rare Earth Elements in Egyptian Phosphorites. Chinese Journal of Geochemistry, v. 21 (1), 10p. [CrossRef]
- Meneisy, M.Y., and Kreuzer, H., 1974, Potassium-Argon ages of Egyptian basaltic rocks. Geol. Jb., D9, pp.21-31.
- Germann, K., Bock, W.D., and Schroter, T., 1984, Facies development of Upper Cretaceous phosphorites in Egypt: sedimentological and geochemical aspects. Berliner Geowiss. Abh., 50, pp.345-361.
- McArthur, J.M., and Walsh, J.N., 1984, Rare-earth element geochemistry of phosphorites. Chem. Geol., 47, pp.191-220.
- Altschuler, Z.S., Berman, S., Cuttita, F., 1967, Rare earths in phosphorites: Geochemistry and potential recovery. U.S. Geological Survey Professional Papers 525b, pp.1-9.
- Kolodny, Y., 1981, Phosphorites. In: Emiliani, C. (Ed.), The Sea, J. Wiley & Sons, New York, 7, pp. 981-1023.
- Baioumy, H.M., 2011, Rare earth elements and sulfur and strontium isotopes of upper Cretaceous phosphorites in Egypt. Cretaceous Research, v. 32(3), pp.368-377. [CrossRef]
- Mazumdar, A., Banerjee, D., Schidlowski, M., and Balaram, V., 1999, Rare earth elements and stable isotope geochemistry of Early Cambrian chert-phosphorite assemblages from the Lower Tal Formation of the Krol Belt, Lesser Himalaya, India. Chem. Geol., 156, pp. 275-297. [CrossRef]
- Boynton, W.V., 1984, Geochemistry of the REE: meteorite studies. In: Henderson, P. (Ed.), Rare Earth Element Geochemistry. Elsevier, pp.63 ̶ 114.
- Egyptian Geological Survey and Mining Authority, 1968, Geological map of Idfu-Qena area, scale 1:200.000, phosphate project group between Soviet and EGSMA, unpublished – internal report.

| Area | Wadi Hilal | East Sibaiya | Um Higara | Um Salama | Um Tundbah | Total average | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | 1 | 3 | 4 | 6 | Aver. | 1 | 3 | Aver. | 1 | 2 | 5 | 6 | Aver. | 1 | 2 | 4 | Aver. | 1 | 2 | 6 | Aver. | |
| SiO2 | 4.25 | 5.93 | 33.89 | 22.63 | 16.68 | 4.66 | 10.85 | 7.76 | 2.71 | 34.77 | 47.67 | 26.1 | 27.81 | 4.11 | 36.63 | 33.42 | 24.72 | 2.48 | 34.53 | 30.6 | 22.54 | 20.95 |
| TiO2 | 0.01 | 0.01 | 0.01 | 0.07 | 0.02 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.05 | 0.03 | 0.02 | 0.01 | 0.01 | 0.02 | 0.01 | 0.01 | 00.1 | 0.07 | 0.02 | 0.02 |
| Al2O3 | 0.19 | 0.29 | 0.19 | 1.83 | 0.63 | 0.27 | 0.24 | 0.26 | 0.14 | 0.29 | 1.08 | 0.92 | 0.61 | 0.18 | 0.22 | 0.52 | 0.31 | 0.13 | 0.13 | 1.84 | 0.70 | 0.53 |
| Fe2O3 | 1.59 | 2.14 | 1.53 | 2.33 | 1.90 | 1.45 | 1.04 | 1.25 | 0.86 | 0.87 | 0.66 | 2.24 | 1.16 | 1.87 | 1.45 | 1.72 | 1.68 | 1.1 | 0.84 | 2.08 | 1.34 | 1.49 |
| MgO | 0.33 | 0.39 | 0.3 | 0.56 | 0.40 | 0.34 | 0.33 | 0.34 | 0.3 | 0.27 | 0.32 | 0.35 | 0.31 | 0.31 | 0.28 | 0.28 | 0.29 | 0.28 | 0.23 | 0.44 | 0.32 | 0.33 |
| CaO | 48.98 | 46.82 | 33.84 | 37.02 | 41.67 | 49.96 | 47.6 | 48.78 | 52.78 | 36.33 | 26.91 | 37.35 | 38.34 | 49.87 | 32.94 | 34.49 | 39.10 | 52.07 | 34.65 | 33.85 | 40.19 | 40.97 |
| Na2O | 0.6 | 1.2 | 0.89 | 1.41 | 1.03 | 0.69 | 0.53 | 0.61 | 0.41 | 0.46 | 0.49 | 0.59 | 0.49 | 0.72 | 0.58 | 0.57 | 0.62 | 0.41 | 0.4 | 0.62 | 0.48 | 0.66 |
| K2O | 0.01 | 0.02 | 0.02 | 0.1 | 0.04 | 0.01 | 0.01 | 0.01 | 0.01 | 0.02 | 0.06 | 0.05 | 0.03 | 0.02 | 0.02 | 0.04 | 0.03 | 0.01 | 0.01 | 0.07 | 0.03 | 0.03 |
| P2O5 | 21.22 | 28.23 | 21.36 | 20.92 | 22.93 | 26.54 | 19.46 | 23.00 | 29.22 | 23.22 | 17.4 | 23.11 | 23.24 | 31.24 | 21.47 | 22.43 | 25.05 | 29.79 | 22.41 | 21.47 | 24.56 | 23.72 |
| MnO | 0.11 | 0.1 | 0.1 | 0.09 | 0.10 | 0.07 | 0.06 | 0.07 | 0.06 | 0.09 | 0.05 | 0.09 | 0.07 | 0.07 | 0.09 | 0.06 | 0.07 | 0.07 | 0.07 | 0.07 | 0.07 | 0.08 |
| F | 1.22 | 1.36 | 1.46 | 0.98 | 1.26 | 0.14 | 1.16 | 0.65 | 1.53 | 1.48 | 1.25 | 1.31 | 1.39 | 1.55 | 1.46 | 1.46 | 1.49 | 1.73 | 1.55 | 1.14 | 1.47 | 1.30 |
| Cl | 0.22 | 0.87 | 0.44 | 1.16 | 0.67 | 0.11 | 0.10 | 0.11 | 0.02 | 0.02 | 0.03 | 0.04 | 0.03 | 0.02 | 0.02 | 0.01 | 0.02 | 0.01 | 0.01 | 0.12 | 0.05 | 0.20 |
| S | 1.52 | 2.14 | 0.99 | 1.11 | 1.44 | 0.10 | 0.45 | 0.28 | 0.24 | 0.25 | 0.21 | 0.29 | 0.25 | 1.52 | 0.43 | 0.37 | 0.77 | 0.21 | 0.21 | 0.34 | 0.25 | 0.65 |
| LOI | 16.62 | 7.71 | 5.29 | 9.64 | 9.82 | 12.39 | 17.61 | 15.00 | 11.02 | 1.37 | 4.16 | 6.77 | 5.83 | 6.17 | 4.65 | 4.71 | 5.18 | 10.17 | 4.6 | 6.04 | 6.94 | 8.06 |
| Cr | 104.43 | 113.43 | 161.37 | 76.93 | 114.04 | 89.75 | 110.83 | 100.29 | 122.15 | 136.56 | 164.09 | 107.55 | 132.59 | 107.18 | 172.74 | 112.19 | 130.70 | 104.98 | 171.93 | 117.29 | 131.40 | 123.34 |
| Ni | 26.65 | 32.54 | 19.9 | 53.12 | 33.05 | 19.4 | 15.09 | 17.25 | 20.33 | 18.89 | 10.99 | 37.86 | 22.02 | 39.82 | 23.15 | 27.72 | 30.23 | 23.37 | 10.27 | 81.1 | 38.25 | 28.76 |
| Cu | 15.82 | 13.91 | 17.48 | 15.12 | 15.58 | 12.62 | 17.28 | 14.95 | 15.82 | 13.23 | 11.72 | 12.81 | 13.40 | 20.45 | 13.62 | 14.39 | 16.15 | 11.7 | 12.86 | 22.23 | 15.60 | 15.07 |
| Zn | 187.33 | 266.45 | 138.21 | 247.76 | 209.94 | 175.81 | 131.79 | 153.80 | 178.68 | 184.55 | 164.23 | 206.82 | 183.57 | 279.14 | 204.44 | 246.91 | 243.50 | 247.53 | 184.54 | 280.49 | 237.52 | 207.79 |
| Sr | 2281.44 | 2675.3 | 1703.01 | 2442.22 | 2275.49 | 1855.98 | 1695.39 | 1775.69 | 1811.86 | 1285.7 | 1237.11 | 1535.19 | 1467.47 | 2225.98 | 1378.25 | 1447.27 | 1683.83 | 1784.85 | 1292.11 | 1338.57 | 1471.84 | 1749.39 |
| Zr | 199.8 | 228.96 | 142.79 | 209.43 | 195.25 | 164.26 | 149.31 | 156.79 | 155.86 | 109.94 | 106.21 | 137.43 | 127.36 | 189.09 | 117.56 | 124.17 | 143.61 | 153.27 | 108.31 | 125.82 | 129.13 | 151.39 |
| Ba | 702.05 | 827.9 | 433.4 | 1920.91 | 971.07 | 733.35 | 491.73 | 612.54 | 655.38 | 372.97 | 361.54 | 328.22 | 429.53 | 597.64 | 326.33 | 327.21 | 417.06 | 754.07 | 441.89 | 268.6 | 488.19 | 596.45 |
| V | 90.89 | 156.65 | 172.78 | 82.11 | 125.61 | 112.43 | 68.16 | 90.30 | 199.01 | 281.43 | 0.01 | 0.01 | 120.11 | 208.32 | 254.19 | 286.66 | 249.72 | 322.82 | 462.7 | 121.06 | 302.19 | 176.20 |
| Pb | 5.96 | 6.29 | 6.11 | 4.64 | 5.75 | 3.59 | 2.58 | 3.09 | 3.6 | 4.86 | 4.49 | 4.49 | 4.36 | 6.37 | 9.36 | 12.26 | 9.33 | 4.9 | 5.62 | 6.19 | 5.57 | 5.71 |
| Th | 1.73 | 1.92 | 1.23 | 1.86 | 1.69 | 1.11 | 0.77 | 0.94 | 0.8 | 1.07 | 1.36 | 2.25 | 1.37 | 1.05 | 1.25 | 4.07 | 2.12 | 0.61 | 0.74 | 3.3 | 1.55 | 1.57 |
| U | 68.89 | 111.85 | 123.37 | 50.62 | 88.68 | 89.28 | 40.59 | 64.94 | 144.46 | 173.63 | 181.11 | 63.94 | 140.79 | 136.64 | 166.48 | 204.73 | 169.28 | 181 | 245.84 | 64.61 | 163.82 | 127.94 |
| Cao/P2O5 | 2.31 | 1.66 | 1.58 | 1.77 | 1.82 | 1.88 | 2.45 | 2.12 | 1.81 | 1.56 | 1.55 | 1.62 | 1.65 | 1.60 | 1.53 | 1.54 | 1.56 | 1.75 | 1.55 | 1.58 | 1.64 | 1.76 |
| Th/U | 0.03 | 0.02 | 0.01 | 0.04 | 0.02 | 0.01 | 0.02 | 0.01 | 0.01 | 0.01 | 0.01 | 0.04 | 0.01 | 0.01 | 0.01 | 0.02 | 0.01 | 0.00 | 0.00 | 0.05 | 0.01 | 0.02 |
| SiO2 | TiO2 | Al2O3 | Fe2O3 | MgO | CaO | Na2O | K2O | P2O5 | MnO | F | Cl | S | LOI | Cr | Ni | Cu | Zn | Sr | Zr | Ba | V | Pb | Th | U | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | 1.00 | ||||||||||||||||||||||||
| TiO2 | 0.40 | 1.00 | |||||||||||||||||||||||
| Al2O3 | 0.37 | 0.98 | 1.00 | ||||||||||||||||||||||
| Fe2O3 | -0.16 | 0.44 | 0.49 | 1.00 | |||||||||||||||||||||
| MgO | -0.11 | 0.77 | 0.80 | 0.69 | 1.00 | ||||||||||||||||||||
| CaO | -0.98 | -0.51 | -0.49 | 0.00 | -0.04 | 1.00 | |||||||||||||||||||
| Na2O | -0.12 | 0.38 | 0.40 | 0.71 | 0.77 | -0.03 | 1.00 | ||||||||||||||||||
| K2O | 0.49 | 0.94 | 0.95 | 0.52 | 0.75 | -0.61 | 0.51 | 1.00 | |||||||||||||||||
| P2O5 | -0.72 | -0.48 | -0.43 | 0.11 | -0.16 | 0.74 | 0.00 | -0.46 | 1.00 | ||||||||||||||||
| MnO | -0.09 | -0.10 | -0.08 | 0.48 | 0.22 | 0.02 | 0.49 | -0.05 | -0.03 | 1.00 | |||||||||||||||
| F | 0.16 | -0.21 | -0.28 | -0.21 | -0.44 | -0.10 | -0.29 | -0.21 | 0.17 | 0.03 | 1.00 | ||||||||||||||
| Cl | -0.12 | 0.37 | 0.40 | 0.57 | 0.77 | -0.01 | 0.95 | 0.47 | -0.06 | 0.50 | -0.21 | 1.00 | |||||||||||||
| S | -0.38 | -0.02 | -0.07 | 0.54 | 0.35 | 0.27 | 0.69 | 0.00 | 0.23 | 0.60 | 0.10 | 0.64 | 1.00 | ||||||||||||
| LOI | -0.72 | -0.17 | -0.16 | 0.02 | 0.22 | 0.70 | 0.05 | -0.31 | 0.06 | 0.03 | -0.38 | 0.13 | 0.17 | 1.00 | |||||||||||
| Cr | 0.67 | -0.22 | -0.27 | -0.54 | -0.59 | -0.60 | -0.39 | -0.19 | -0.36 | -0.06 | 0.44 | -0.36 | -0.25 | -0.56 | 1.00 | ||||||||||
| Ni | -0.04 | 0.71 | 0.75 | 0.76 | 0.72 | -0.09 | 0.41 | 0.63 | 0.04 | 0.15 | -0.13 | 0.33 | 0.23 | -0.07 | -0.45 | 1.00 | |||||||||
| Cu | -0.15 | 0.27 | 0.26 | 0.37 | 0.31 | 0.10 | 0.16 | 0.16 | 0.05 | 0.00 | 0.04 | 0.06 | 0.32 | 0.12 | -0.17 | 0.64 | 1.00 | ||||||||
| Zn | -0.19 | 0.38 | 0.37 | 0.63 | 0.40 | 0.10 | 0.34 | 0.36 | 0.46 | 0.05 | 0.17 | 0.24 | 0.36 | -0.24 | -0.41 | 0.72 | 0.29 | 1.00 | |||||||
| Sr | -0.71 | -0.05 | -0.05 | 0.53 | 0.51 | 0.61 | 0.72 | -0.03 | 0.45 | 0.46 | -0.16 | 0.71 | 0.84 | 0.49 | -0.62 | 0.21 | 0.16 | 0.35 | 1.00 | ||||||
| Zr | -0.73 | -0.01 | 0.00 | 0.56 | 0.55 | 0.62 | 0.71 | -0.01 | 0.43 | 0.45 | -0.21 | 0.70 | 0.82 | 0.52 | -0.67 | 0.27 | 0.19 | 0.37 | 1.00 | 1.00 | |||||
| Ba | -0.37 | 0.30 | 0.33 | 0.37 | 0.71 | 0.27 | 0.73 | 0.36 | 0.13 | 0.24 | -0.28 | 0.80 | 0.39 | 0.34 | -0.59 | 0.23 | -0.07 | 0.24 | 0.71 | 0.70 | 1.00 | ||||
| V | 0.06 | -0.50 | -0.48 | -0.35 | -0.58 | 0.02 | -0.32 | -0.44 | 0.34 | -0.08 | 0.50 | -0.27 | -0.17 | -0.37 | 0.37 | -0.27 | -0.15 | 0.15 | -0.24 | -0.29 | -0.17 | 1.00 | |||
| Pb | 0.35 | 0.01 | -0.04 | 0.27 | -0.20 | -0.37 | 0.03 | 0.09 | -0.06 | 0.10 | 0.32 | -0.08 | 0.13 | -0.45 | 0.22 | 0.13 | 0.05 | 0.41 | -0.14 | -0.16 | -0.26 | 0.39 | 1.00 | ||
| Th | 0.30 | 0.54 | 0.54 | 0.59 | 0.34 | -0.39 | 0.21 | 0.55 | -0.26 | 0.02 | -0.08 | 0.12 | 0.07 | -0.23 | -0.24 | 0.59 | 0.26 | 0.49 | -0.08 | -0.04 | -0.12 | -0.18 | 0.63 | 1.00 | |
| U | 0.38 | -0.38 | -0.41 | -0.57 | -0.71 | -0.27 | -0.47 | -0.29 | 0.14 | -0.32 | 0.53 | -0.43 | -0.31 | -0.61 | 0.62 | -0.51 | -0.43 | 0.00 | -0.47 | -0.53 | -0.36 | 0.77 | 0.43 | -0.14 | 1.00 |
| Samples No. | WH 1 | WH 3 | WH 4 | WH 6 | Aver. | ES 1 | ES 3 | Aver. | UH 1 | UH 2 | UH 5 | UH 6 | Aver. | US 1 | US 2 | US 4 | Aver. | UT 1 | UT 2 | UT 6 | Aver. | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| LREEs | La | 42.53 | 52.49 | 38.55 | 30.71 | 41.07 | 27.04 | 14.37 | 20.71 | 18.29 | 21.76 | 11.62 | 33.66 | 21.33 | 33.68 | 43.59 | 61 | 46.09 | 21.34 | 25.54 | 47.46 | 31.45 |
| Ce | 70.97 | 82.77 | 56.62 | 50.94 | 65.33 | 42.88 | 25.2 | 34.04 | 25.8 | 35.29 | 14.52 | 58.77 | 33.6 | 47.45 | 64.09 | 97.93 | 69.82 | 24.32 | 30.02 | 76.68 | 43.67 | |
| Pr | 9.11 | 10.8 | 7.53 | 6.18 | 8.405 | 5.56 | 3.29 | 4.425 | 3.75 | 4.99 | 2.24 | 7.65 | 4.658 | 6.23 | 8.42 | 12.5 | 9.05 | 3.56 | 4.39 | 9.93 | 5.96 | |
| Nd | 37.87 | 45.11 | 31.54 | 26.36 | 35.22 | 23.36 | 13.57 | 18.47 | 15.58 | 20.18 | 8.81 | 30.89 | 18.87 | 25.82 | 33.9 | 49.98 | 36.57 | 14.79 | 18.35 | 40.12 | 24.42 | |
| Sm | 7.34 | 9.08 | 6.35 | 5.17 | 6.985 | 4.49 | 2.7 | 3.595 | 3.07 | 4.04 | 1.75 | 6.36 | 3.805 | 5.12 | 6.64 | 9.84 | 7.20 | 2.94 | 3.55 | 8.04 | 4.843 | |
| Eu | 2.1 | 2.47 | 1.77 | 1.55 | 1.973 | 1.22 | 0.69 | 0.955 | 0.81 | 1.06 | 0.47 | 1.68 | 1.005 | 1.28 | 1.69 | 2.44 | 1.80 | 0.79 | 0.94 | 2.15 | 1.293 | |
| HREEs | Gd | 8.2 | 10.2 | 7.41 | 6.38 | 8.048 | 5.06 | 2.26 | 3.66 | 3.73 | 4.53 | 1.75 | 6.52 | 4.133 | 5.28 | 6.74 | 9.86 | 7.29 | 3.4 | 4.02 | 8.15 | 5.19 |
| Tb | 1.24 | 1.45 | 1.04 | 0.92 | 1.163 | 0.74 | 0.37 | 0.555 | 0.55 | 0.68 | 0.3 | 0.94 | 0.618 | 0.79 | 1.02 | 1.39 | 1.07 | 0.51 | 0.6 | 1.19 | 0.767 | |
| Dy | 7.86 | 9.78 | 6.91 | 5.78 | 7.583 | 4.91 | 2.08 | 3.495 | 3.7 | 4.45 | 2.04 | 6.01 | 4.05 | 5.18 | 6.62 | 9.22 | 7.01 | 3.32 | 4.17 | 7.55 | 5.013 | |
| Ho | 1.77 | 2.21 | 1.59 | 1.29 | 1.715 | 1.15 | 0.44 | 0.795 | 0.85 | 1.07 | 0.54 | 1.34 | 0.95 | 1.23 | 1.52 | 2.11 | 1.62 | 0.82 | 1.04 | 1.72 | 1.193 | |
| Er | 5.3 | 6.69 | 4.83 | 3.77 | 5.148 | 3.51 | 1.2 | 2.355 | 2.73 | 3.41 | 1.71 | 4.13 | 2.995 | 3.8 | 4.8 | 6.38 | 4.99 | 2.5 | 3.3 | 5.17 | 3.657 | |
| Tm | 0.74 | 0.94 | 0.66 | 0.56 | 0.725 | 0.49 | 0.17 | 0.33 | 0.37 | 0.46 | 0.23 | 0.55 | 0.403 | 0.52 | 0.67 | 0.87 | 0.69 | 0.34 | 0.47 | 0.72 | 0.51 | |
| Yb | 5.12 | 6.25 | 4.62 | 3.75 | 4.935 | 3.31 | 1.18 | 2.245 | 2.62 | 3.17 | 1.6 | 3.87 | 2.815 | 3.63 | 4.67 | 5.92 | 4.74 | 2.44 | 3.15 | 4.99 | 3.527 | |
| Lu | 0.79 | 1 | 0.73 | 0.58 | 0.775 | 0.54 | 0.17 | 0.355 | 0.41 | 0.48 | 0.25 | 0.6 | 0.435 | 0.58 | 0.75 | 0.92 | 0.75 | 0.39 | 0.5 | 0.75 | 0.547 | |
| ∑LREEs | 169.9 | 202.7 | 142.4 | 120.9 | 159 | 104.6 | 59.82 | 82.19 | 67.3 | 87.32 | 39.41 | 139 | 83.26 | 119.6 | 158.3 | 233.69 | 170.53 | 67.74 | 82.79 | 184.4 | 111.6 | |
| ∑HREEs | 31.02 | 38.52 | 27.79 | 23.03 | 30.09 | 19.71 | 7.87 | 13.79 | 14.96 | 18.25 | 8.42 | 23.96 | 16.4 | 21.01 | 26.79 | 36.67 | 28.157 | 13.72 | 17.25 | 30.24 | 20.4 | |
| ∑REEs | 200.9 | 241.2 | 170.2 | 143.9 | 189.1 | 124.3 | 67.69 | 95.98 | 82.26 | 105.6 | 47.83 | 163 | 99.66 | 140.6 | 185.1 | 270.36 | 198.69 | 81.46 | 100.04 | 214.6 | 132 | |
| Ce/La | 1.669 | 1.577 | 1.469 | 1.659 | 1.591 | 1.586 | 1.754 | 1.644 | 1.411 | 1.622 | 1.25 | 1.746 | 1.575 | 1.409 | 1.47 | 1.6054 | 1.5149 | 1.1396 | 1.1754 | 1.616 | 1.389 | |
| Eu/Eu* | 0.8 | 0.81 | 0.78 | 0.76 | 0.79 | |||||||||||||||||
| Elements | Present work | Abd El Gabar et al 2002 | Baioumy 2005 | El-Taher 2010 | Awadalla 2011 | Abou El Anwar et al 2016 | Abou El Anwar 2019 | Abdel-All & Amer 1995 |
|||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Red Sea | Nile Valley | Abu Tartur | Red Sea | Nile Valley | Abu Tartur | Red Sea | Nile Valley | ||||||
| SiO2 | 20.95 | 14.04 | 26.90 | 14.66 | 17.99 | 37.01 | 11.54 | ND | ND | 4.20 | 9.85 | 10.06 | 13.30 |
| TiO2 | 0.02 | 0.01 | 0.09 | 0.05 | 0.03 | 0.04 | 0.05 | 0.67 | ND | 0.10 | 0.04 | 0.03 | ND |
| Al2O3 | 0.53 | 0.92 | 1.94 | 2.06 | 0.70 | 0.58 | 0.91 | ND | ND | 0.90 | 0.70 | 1.24 | 0.62 |
| Fe2O3 | 1.49 | 0.23 | 2.68 | 4.17 | 1.05 | 1.90 | 4.73 | ND | 2.29 | 3.50 | 0.66 | 2.87 | 2.00 |
| MgO | 0.33 | 3.09 | 0.68 | 2.45 | 2.19 | 39.00 | 2.05 | 1.82 | 0.33 | 1.60 | 0.37 | 2.41 | 0.23 |
| CaO | 40.97 | 32.12 | 28.67 | 33.42 | 41.66 | 31.86 | 39.25 | 33.16 | 44.36 | 43.80 | 57.09 | 37.35 | 44.60 |
| Na2O | 0.66 | 6.48 | 3.15 | 1.52 | 0.26 | 0.39 | 0.66 | 0.81 | 0.81 | 1.30 | 0.26 | 0.24 | 0.50 |
| K2O | 0.03 | 0.02 | 0.20 | 0.24 | 0.08 | 0.09 | 0.40 | 0.29 | 0.10 | 0.20 | 0.05 | 0.03 | 0.15 |
| P2O5 | 23.72 | 16.43 | 16.53 | 16.15 | 23.65 | 20.75 | 25.29 | 19.30 | 27.10 | 24.60 | 25.67 | 27.75 | 28.50 |
| MnO | 0.08 | 0.04 | 0.06 | 0.06 | 0.03 | 0.12 | 0.28 | 0.13 | 0.39 | 0.10 | 0.06 | ND | ND |
| F | 1.30 | ND | ND | ND | ND | ND | ND | ND | ND | 0.95 | 1.96 | 1.43 | 2.99 |
| Cl | 0.20 | ND | ND | ND | ND | ND | ND | ND | ND | 0.30 | 0.07 | 0.04 | 0.02 |
| S | 0.65 | ND | ND | ND | ND | ND | ND | ND | ND | 6.90 | 1.48 | 1.12 | 0.65 |
| LOI | 8.06 | ND | ND | ND | 12.62 | 5.98 | 14.38 | ND | ND | ND | ND | 1.46 | ND |
| Cr | 121.80 | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | 113.25 | ND |
| Ni | 28.16 | ND | ND | ND | ND | ND | ND | 469 | 22 | ND | 77 | 35.25 | ND |
| Cu | 15.14 | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | 18.25 | ND |
| Zn | 205.66 | 119.7 | 86.3 | 106.2 | ND | ND | ND | 360 | 162 | 133 | 149 | 68.25 | 171 |
| Sr | 1734.86 | 1385 | 1584 | 391 | ND | ND | ND | 520 | 1032 | 1001 | 1705 | ND | 1350 |
| Zr | 150.4 | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| Ba | 583.68 | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| V | 177.59 | ND | ND | ND | ND | ND | ND | ND | 220 | 30 | 246 | ND | ND |
| Pb | 5.62 | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| CaO/P2O5 | 1.76 | 1.96 | 1.73 | 2.07 | 1.76 | 1.54 | 1.55 | 1.72 | 1.64 | 1.78 | 2.22 | 1.35 | 1.56 |
| Area | Aver. Th | Aver. U | Th/U |
|---|---|---|---|
| Present work | 1.57 | 127.94 | 0.01 |
| Hammadat area | 2.90 | 59.87 | 0.05 |
| Duwi mine | 3.50 | 22.77 | 0.15 |
| Wasief mine | 2.30 | 66.70 | 0.03 |
| Yonous mine | 3.25 | 51.75 | 0.06 |
| G Abu Had | 1.83 | 49.00 | 0.04 |
| Mahamied-Sibaiya | 1.38 | 63.33 | 0.02 |
| SE Idfu | 4.74 | 24.75 | 0.19 |
| West Nile Valley | 2.29 | 46.07 | 0.05 |
| G Ghanima | 3.88 | 12.20 | 0.32 |
| Abu Tartur mine | 5.84 | 26.38 | 0.22 |
| G Hefhuf | 2.45 | 100.00 | 0.02 |
| G Sheikh Abdallah | 2.16 | 33.60 | 0.06 |
| G Edmonstone | 4.04 | 34.50 | 0.12 |
| G Sofraiyat | 1.78 | 33.78 | 0.05 |
| Wadi Araba | 1.41 | 26.86 | 0.05 |
| MIN | 1.38 | 12.20 | 0.01 |
| MAX | 5.84 | 127.94 | 0.32 |
| Area | La | Ce | Pr | Nd | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ∑LREEs | ∑HREEs | ∑REEs | Ce/La | Eu/Eu* |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Present work | 32.7 | 50.3 | 6.6 | 27.3 | 5.4 | 1.4 | 5.8 | 0.9 | 5.6 | 1.3 | 4.0 | 0.5 | 3.8 | 0.6 | 123.7 | 22.5 | 146.2 | 1.5 | 0.79 |
| Gebel Sofraiyat | 31.8 | 44.0 | nd | 22.9 | 5.5 | 1.4 | 5.6 | 0.9 | 5.5 | nd | nd | nd | 2.4 | 0.3 | 105.7 | 14.8 | 120.4 | 1.4 | 0.79 |
| Wadi Araba | 38.1 | 43.7 | nd | 32.2 | 5.3 | 1.5 | 5.6 | 0.9 | 5.1 | nd | nd | nd | 3.5 | 0.5 | 120.8 | 15.6 | 136.4 | 1.1 | 0.86 |
| Rabah mines | 60.5 | 86.5 | nd | 66.0 | 8.8 | 3.1 | 11.8 | 1.8 | 14.1 | nd | nd | nd | 7.5 | 1.3 | 224.9 | 36.5 | 261.4 | 1.4 | 0.91 |
| Gebel Sheikh Abdallah | 174.6 | 280.4 | nd | 156.9 | 34.3 | 10.0 | 31.0 | 4.9 | 28.9 | nd | nd | nd | 16.3 | 2.2 | 656.1 | 83.2 | 739.3 | 1.6 | 0.94 |
| Abu Tartur | 43.3 | 53.7 | nd | 46.4 | 10.0 | 6.2 | 8.2 | 1.7 | 8.7 | nd | nd | nd | 5.3 | 32.9 | 159.6 | 56.7 | 216.3 | 1.2 | 2.09 |
| Gebel Hefhuf | 79.6 | 129.0 | nd | 120.6 | 25.9 | 8.4 | 35.1 | 7.5 | 37.5 | nd | nd | nd | 8.9 | 1.1 | 363.5 | 90.1 | 453.6 | 1.6 | 0.85 |
| Gebel Ghanima | 55.0 | 91.8 | nd | 52.8 | 9.3 | 2.6 | 8.1 | 1.3 | 8.4 | nd | nd | nd | 4.8 | 0.7 | 211.6 | 23.3 | 234.8 | 1.7 | 0.92 |
| Abu Had | 43.2 | 80.2 | nd | 53.7 | 8.6 | 2.5 | 7.9 | 1.4 | 7.6 | nd | nd | nd | 4.9 | 0.8 | 188.1 | 22.5 | 210.5 | 1.9 | 0.93 |
| Mahamied to Sibaiya | 55.5 | 86.4 | nd | 60.4 | 9.4 | 2.4 | 8.5 | 1.5 | 8.7 | nd | nd | nd | 5.4 | 0.7 | 214.1 | 24.9 | 239.0 | 1.6 | 0.82 |
| SE Idfu | 155.3 | 239.5 | nd | 137.5 | 31.3 | 8.5 | 24.5 | 4.3 | 25.4 | nd | nd | nd | 14.9 | 2.4 | 572.0 | 71.5 | 643.5 | 1.5 | 0.94 |
| West Nile Valley | 49.9 | 68.9 | nd | 40.1 | 7.7 | 1.8 | 7.0 | 1.2 | 6.8 | nd | nd | nd | 4.3 | 0.6 | 168.3 | 19.9 | 188.2 | 1.4 | 0.77 |
| Wasief mine | 52.9 | 94.4 | nd | 59.6 | 9.2 | 2.7 | 8.9 | 1.4 | 9.6 | nd | nd | nd | 5.4 | 0.8 | 218.8 | 26.0 | 244.9 | 1.8 | 0.91 |
| Hammadat | 35.0 | 70.8 | nd | 40.5 | 7.4 | 1.9 | 6.0 | 1.2 | 6.0 | nd | nd | nd | 3.3 | 0.4 | 155.6 | 16.9 | 172.5 | 2.0 | 0.85 |
| Duwi mine | 30.0 | 59.7 | nd | 33.7 | 6.6 | 1.9 | 6.5 | 1.0 | 5.7 | nd | nd | nd | 3.9 | 0.6 | 131.9 | 17.6 | 149.4 | 2.0 | 0.90 |
| Yonous mine | 67.0 | 122.0 | nd | 80.3 | 15.0 | 4.0 | 11.9 | 1.9 | 10.0 | nd | nd | nd | 7.0 | 0.9 | 288.2 | 31.6 | 319.9 | 1.8 | 0.92 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




