Submitted:
26 February 2026
Posted:
28 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Determination of the Median Lethal Dose (LD50) of Carvacrol and P-Cymene in Rats
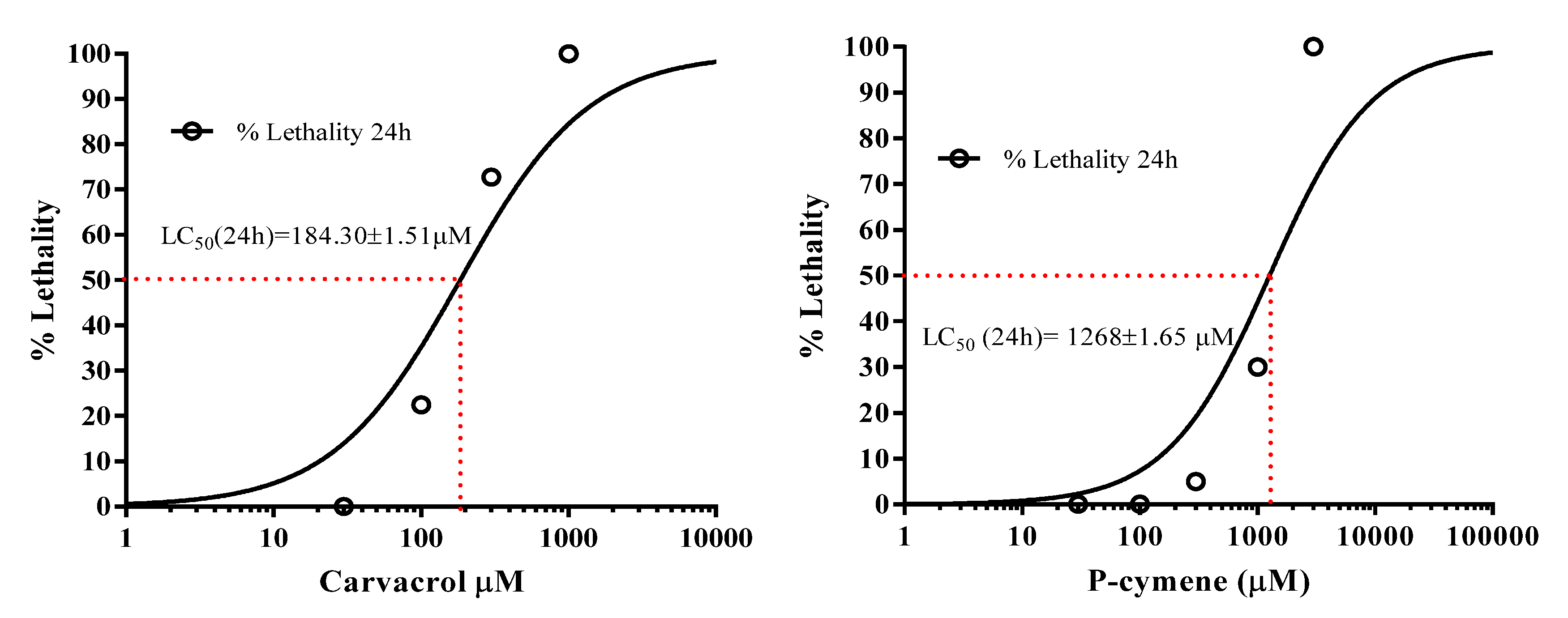
2.2. Determination of the Median Lethal Concentration (LC50) of P-Cymene and Carvacrol in C. elegans
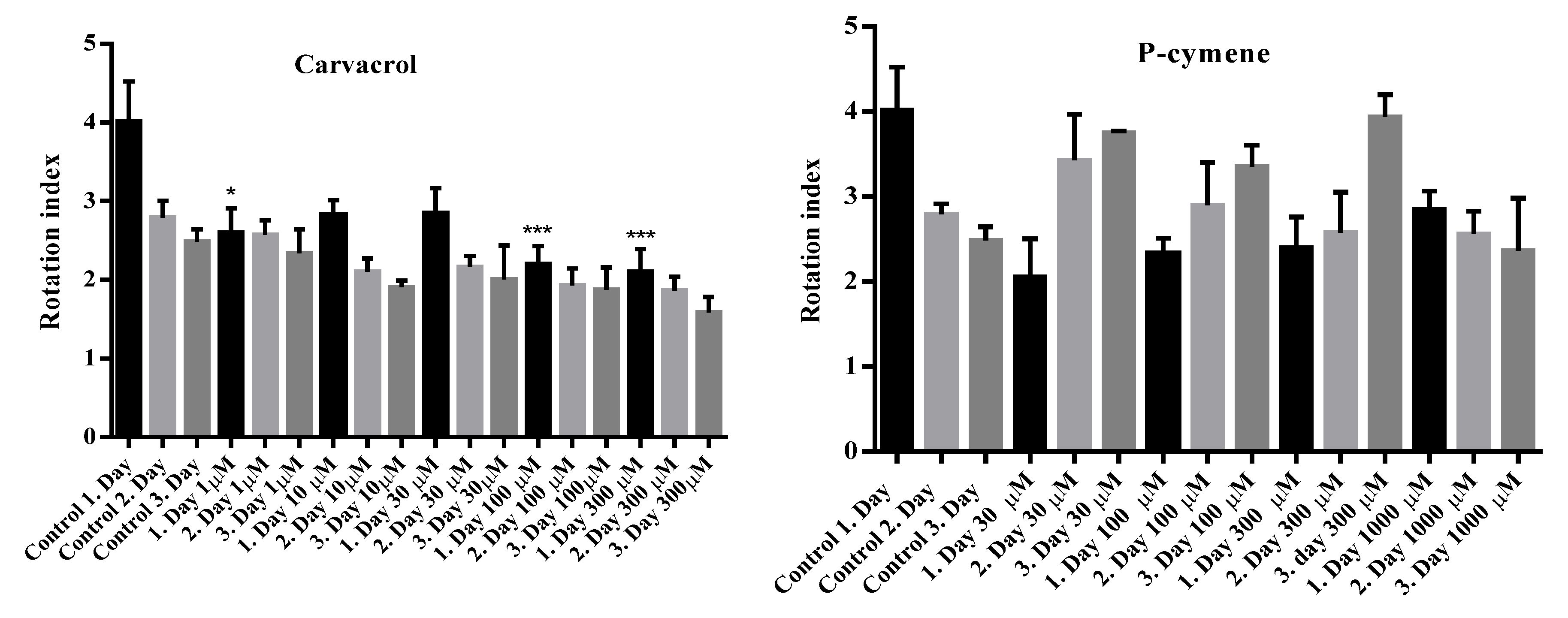
2.3. Rota-Rod Performance
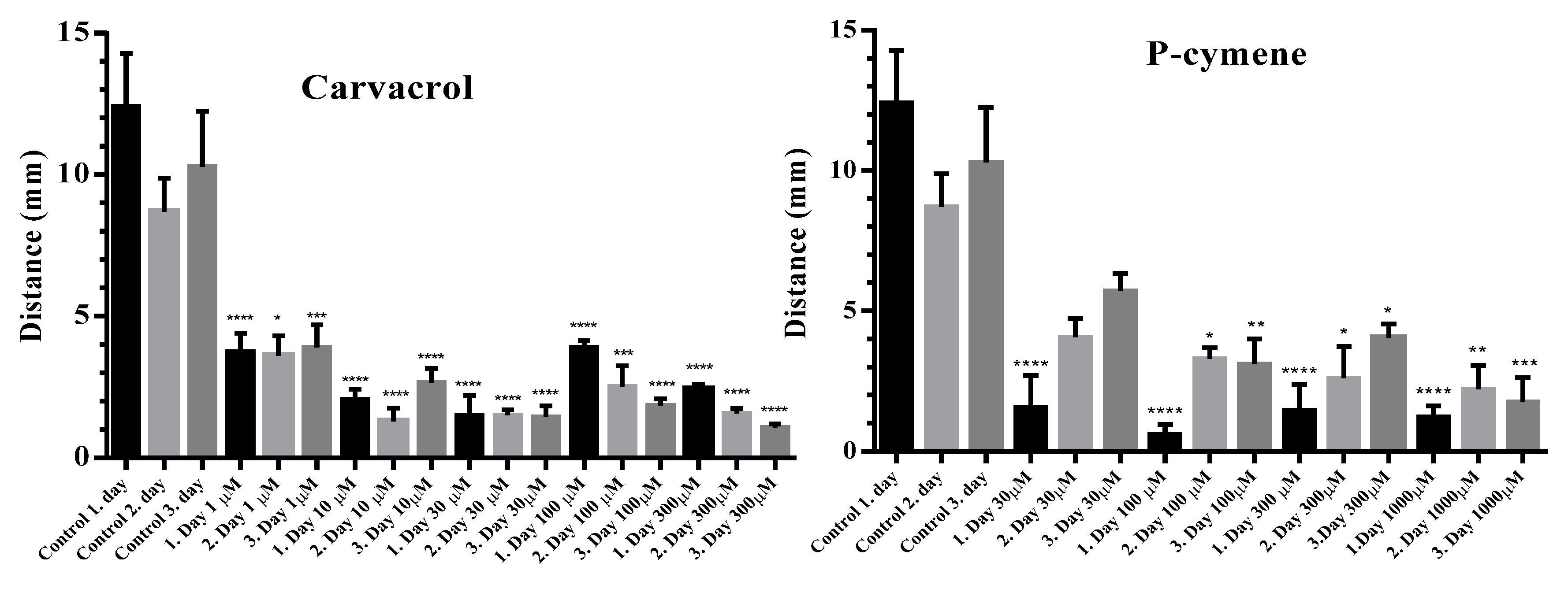
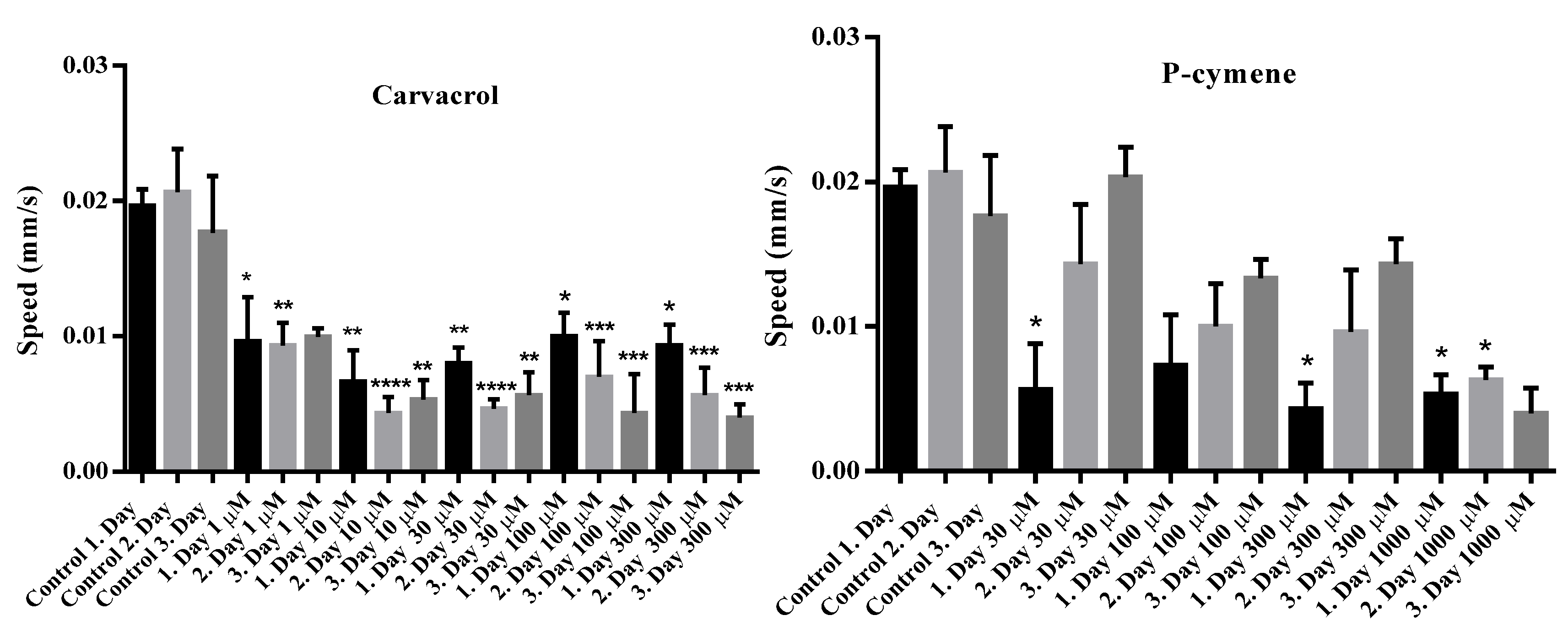
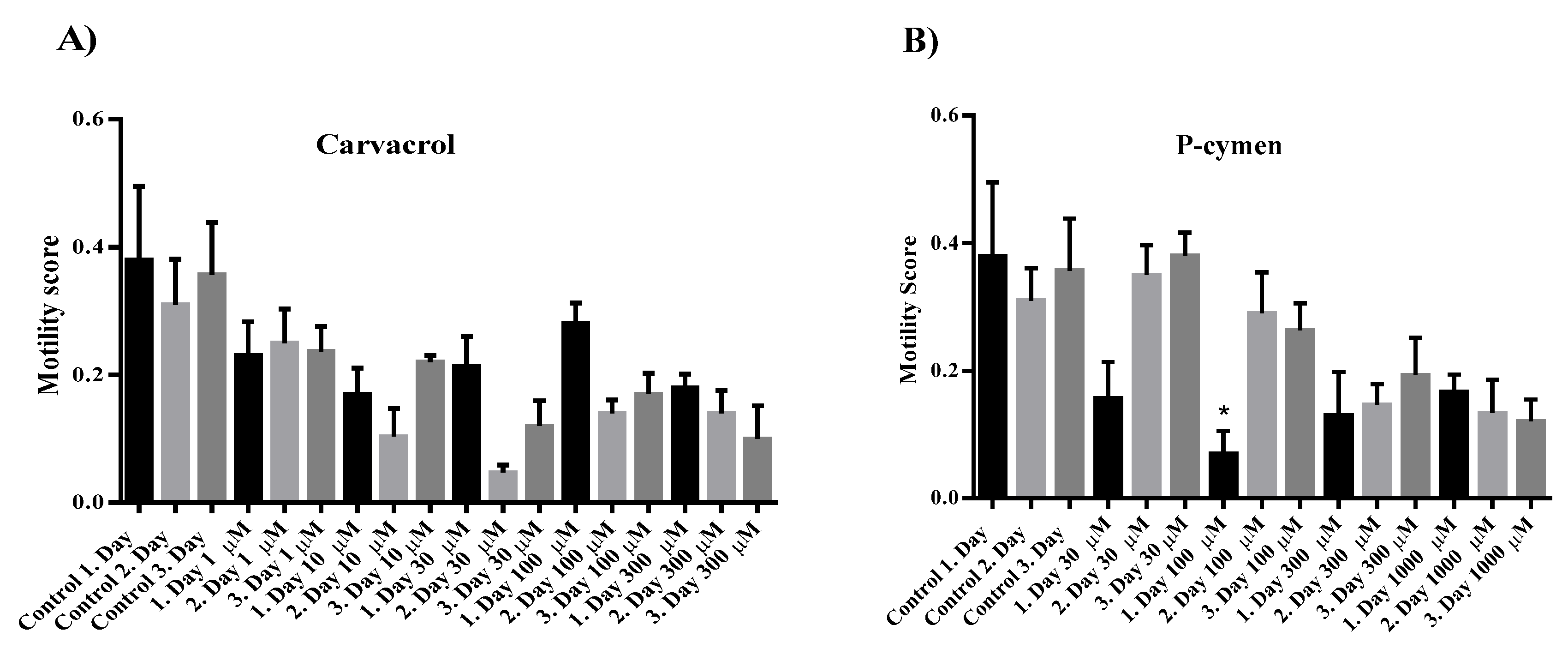
2.4. Investigation of the Influence of Carvacrol and P-Cymene on the Motility of Adult C. elegans
3. Discussion
4. Materials and Methods
4.1. Animals
4.1.1. Rats
4.2. Chemicals and Method of Administration
4.3. Procedures
4.3.1. Determination of the Median Lethal Dose (LD50) of carvacrol and p-cymene in rats
4.3.2. Determination of the Median Lethal Concentration (LC50) of p-cymene and carvacrol in C. elegans
4.3.3. Rota-Rod Performance
4.3.4. Investigation of the Influence of Carvacrol and P-Cymene on the Motility of Adult C. elegans.
4.4. Statistics
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Imran, M.; Aslam, M.; Alsagaby, S.A.; Saeed, F.; Ahmad, I.; Afzaal, M.; Arshad, M.U.; Abdelgawad, M.A.; El-Ghorab, A.H.; Khames, A.; Shariati, M.A.; Ahmad, A.; Hussain, M.; Imran, A.; Islam, S. Therapeutic application of carvacrol: A comprehensive review. Food Sci Nutr. 2022, 10, 3544–3561. [Google Scholar] [CrossRef]
- Balahbib, A.; El Omari, N.; Hachlafi, N.E.; Lakhdar, F.; El Menyiy, N.; Salhi, N; Mrabtiž, H.N.; Bakrim, S.; Zengin, G.; Bouyahya, A. Health beneficial and pharmacological properties of p-cymene. Food Chem Toxicol. 2021, 153, 112259. [Google Scholar] [CrossRef] [PubMed]
- Hyldgaard, M.; Mygind, T.; Meyer, R.L. Essential oils in food preservation: mode of action, synergies, and interactions with food matrix components. Front Microbiol. 2012, 3, 12. [Google Scholar] [CrossRef]
- Philis, J.G. The S1 <-- S0 spectrum of jet-cooled p-cymene. Spectrochim Acta A Mol Biomol Spectrosc. 2005, 61, 1239–41. [Google Scholar] [CrossRef] [PubMed]
- de Santana, M.F.; Guimarães, A.G.; Chaves, D.O.; Silva, J.C.; Bonjardim, L.R.; de Lucca Júnior, W.; Ferro, J.N.; Barreto, Ede. O.; dos Santos, F.E.; Soares, M.B.; Villarreal, C.F.; Quintans; Jde, S.; Quintans-Júnior, LJ. The anti-hyperalgesic and anti-inflammatory profiles of p-cymene: Evidence for the involvement of opioid system and cytokines. Pharm Biol. 2015, 53, 1583–90. [Google Scholar] [CrossRef]
- Milovanović, M.; Marjanović, Đ.; Trailović, S.; Stojanović, D. Antinociceptive effect of p-cymene and cinnamaldehyde: their relationship with the L-arginine-nitric oxide pathway in rats. Acta Veterinaria 2025, 75, 193–211. [Google Scholar] [CrossRef]
- Suntres, Z.E.; Coccimiglio, J.; Alipour, M. The bioactivity and toxicological actions of carvacrol. Crit Rev Food Sci Nutr. 2015, 55, 304–18. [Google Scholar] [CrossRef]
- Marjanović, D.S.; Zdravković, N.; Milovanović, M.; Trailović, J.N.; Robertson, A.P.; Todorović, Z.; Trailović, S.M. Carvacrol acts as a potent selective antagonist of different types of nicotinic acetylcholine receptors and enhances the effect of monepantel in the parasitic nematode Ascaris suum. Veterinary parasitology 2020, 278, 109031. [Google Scholar] [CrossRef]
- Trailovic, S.M.; Rajkovic, M.; Marjanovic, D.S.; Neveu, C.; Charvet, C.L. Action of Carvacrol on Parascaris sp. and Antagonistic Effect on Nicotinic Acetylcholine Receptors. Pharmaceuticals (Basel) 2021, 14, 505. [Google Scholar] [CrossRef]
- Marjanović, D.S.; Trailović, S.M.; Milovanović, M. Interaction of agonists of a different subtype of the nAChR and carvacrol with GABA in Ascaris suum somatic muscle contractions. J Nematol. 2021, 53, e2021-22. [Google Scholar] [CrossRef]
- Baser, K.H. Biological and pharmacological activities of carvacrol and carvacrol bearing essential oils. Curr Pharm Des. 2008, 14, 3106–19. [Google Scholar] [CrossRef] [PubMed]
- European Chemicals Agency (ECHA), p-cymene, 2014.
- Stojković, M.; Todorović, Z.; Protic, D.; Stevanovic, S.; Medić, D.; Charvet, C.L.; Courtot, E.; Marjanović, D.S.; Nedeljković Trailović, J.; Trailović, S.M. Corrigendum: Pharmacological effects of monoterpene carveol on the neuromuscular system of nematodes and mammals. Front Pharmacol. 2024, 15, 1466575. [Google Scholar] [CrossRef]
- Fuentes, C.; Verdú, S.; Fuentes, A.; Ruiz, M.J.; Barat, J.M. Effects of essential oil components exposure on biological parameters of Caenorhabditis elegans. Food Chem Toxicol. 2022, 159, 112763. [Google Scholar] [CrossRef] [PubMed]
- Lei, J.; Leser, M.; Enan, E. Nematicidal activity of two monoterpenoids and SER-2 tyramine receptor of Caenorhabditis elegans. Biochem Pharmacol. 2010, 79, 1062–71. [Google Scholar] [CrossRef] [PubMed]
- Santana, M.F.; Lucindo, J.; Quintans-Júnior, S.C.H.; Cavalcanti, M.G.B.; Oliveira, A. G.; Guimarães, E.S.; Cunha, M.S.; Melo, M.R.V.; Santos, A.A.S.; Araújo, L.R.B. p-Cymene reduces orofacial nociceptive response in mice. Revista Brasileira de Farmacognosia 2011, 21, 1138–1143. [Google Scholar] [CrossRef]
- Austgulen, L.T.; Solheim, E.; Scheline, R.R. Metabolism in rats of p-cymene derivatives: carvacrol and thymol. Pharmacol Toxicol. 1987, 61, 98–102. [Google Scholar] [CrossRef]
- Walde, A.; Ve, B.; Scheline, R.R.; Monge, P. p-Cymene metabolism in rats and guinea-pigs. Xenobiotica 1983, 13, 503–12. [Google Scholar] [CrossRef]
- Braendle, C.; Paaby, A. Life history in Caenorhabditis elegans: from molecular genetics to evolutionary ecology. Genetics 2024, 228, iyae151. [Google Scholar] [CrossRef]
- Stojković, M.; Marjanović, D.S.; Medić, D.; Charvet, C.L.; Trailović, S.M. Neuromuscular System of Nematodes Is a Target of Synergistic Pharmacological Effects of Carvacrol and Geraniol. Pharmaceuticals (Basel) 2025, 18, 1232. [Google Scholar] [CrossRef]
- Higgins, B.J.; Hirsh, D. Roller mutants of the nematode Caenorhabditis elegans. Mol Gen Genet. 1977, 150, 63–72. [Google Scholar] [CrossRef]
- Charvet, C.L.; Guégnard, F.; Courtot, E.; Cortet, J.; Neveu, C. Nicotine-sensitive acetylcholine receptors are relevant pharmacological targets for the control of multidrug resistant parasitic nematodes. Int J Parasitol Drugs Drug Resist. 2018, 8, 540–549. [Google Scholar] [CrossRef]
- Jones, B.J.; Roberts, D.J. The quantiative measurement of motor inco-ordination in naive mice using an acelerating rotarod. J Pharm Pharmacol. 1968, 20, 302–4. [Google Scholar] [CrossRef]
- Trailovic, S.M.; Ivanovic, S.R.; Varagić, V.M. Ivermectin effects on motor coordination and contractions of isolated rat diaphragm. Res Vet Sci. 2011, 91, 426–33. [Google Scholar] [CrossRef]
- Home Office, Code of Practice for the Humane Killing of Animals under Schedule 1 to the Animals (Scientific Procedures) Act 1986, 1997.
| Control | Carvacrol 1mM | Carvacrol 10⚜M | Carvacrol 30⚜M | Carvacrol 100⚜M | Carvacrol 300⚜M | |
|---|---|---|---|---|---|---|
| Day 1 | 20.00±4.16 | 14.33±7.84 | 19.00±11.79 | 20.00±8.33 | 8.33±6.33 | 10.00±2.65 |
| Day 2 | 27.67±6.38 | 18.33±10.34 | 21.67±7.22 | 24.00±8.00 | 11.67±4.67 | 8.00±1.73 |
| Day 3 | 32.33±2.03 | 25.66±3.38 | 25.67±3.38 | 24.67±7.88 | 14.33±5.49 | 5.33±1.33 |
| Control | P-cymene 30mM | P-cymene 100⚜M | P-cymene 300⚜M | P-cymene 1000⚜M | |
|---|---|---|---|---|---|
| Day 1 | 20.00±4.16 | 22.00±10.26 | 29.00±10.44 | 12.00±2.52 | 32.67±2.40 |
| Day 2 | 27.67±6.38 | 25.33±8.51 | 28.00±4.93 | 11.00±2.65 | 27.67±11.15 |
| Day 3 | 32.33±2.03 | 40.00±4.00 | 30.33±2.90 | 26.00±0.58 | 16.00±3.21 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




