Submitted:
25 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MPS | Mucopolysaccharidosis |
| ARSB | Arylsulfatase B |
| HGMD | Human Gene Mutation Database |
| ENT | Ear, Nose and Throat |
| GAG | Glycos Amino Glycans |
| ERT | Enzyme Replacement Therapy |
| BMI | Body Mass Index |
| FVC | Forced Vital Capacity |
| FEV1 | Forced Expiratory Volume in 1 second |
| CT | Computerised Tomography |
| 3D | Three Dimensions |
| LMA | Laryngeal Mask Airway |
| CPAP | Continuous Positive Airway Pressure |
References
- Mehta, A.B.; Winchester, B. Lysosomal storage disorders: a practical guide; Wiley-Blackwell Chicester, 2012. [Google Scholar]
- Muenzer, J. Overview of the mucopolysaccharidoses. Rheumatology 2011, 50, v4–v12. [Google Scholar] [CrossRef] [PubMed]
- Braunlin, E.A.; Harmatz, P.R.; Scarpa, M.; Furlanetto, B.; Kampmann, C.; Loehr, J.P.; Ponder, K.P.; Roberts, W.C.; Rosenfeld, H.M.; Giugliani, R. Cardiac disease in patients with mucopolysaccharidosis: presentation, diagnosis and management. Journal of inherited metabolic disease 2011, 34, 1183–1197. [Google Scholar] [CrossRef] [PubMed]
- Neufeld, E.; Muenzer, J.; Scriver, CR; Beaudet, AL; Sly, WS; Valle, D; Childs, R; Kinzler, KW. The mucopolysaccharidoses, In: The Metabolic and Molecular Bases of Inherited Diseases, 8 ed.; McGraw-Hill: New York, 2001; pp. 3421–3452. [Google Scholar]
- Valayannopoulos, V.; Nicely, H.; Harmatz, P.; Turbeville, S. Mucopolysaccharidosis vi. Orphanet journal of rare diseases 2010, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Tulebayeva, A.; Mukhambetova, G.; Sharipova, M.; Tylki-Szymanska, A. The birth prevalence of mucopolysaccharidosis types I, II, III, IVA, VI, and VII in the Republic of Kazakhstan between 1984 and 2023. Diagnostics 2025, 15, 679. [Google Scholar] [CrossRef] [PubMed]
- D’Avanzo, F.; Zanetti, A.; De Filippis, C.; Tomanin, R. Mucopolysaccharidosis type VI, an updated overview of the disease. International Journal of Molecular Sciences 2021, 22, 13456. [Google Scholar] [CrossRef] [PubMed]
- Harmatz, P.; Shediac, R. Mucopolysaccharidosis VI: pathophysiology, diagnosis and treatment. Front Biosci (Landmark Ed) 2017, 22, 385–406. [Google Scholar] [CrossRef] [PubMed]
- Gadepalli, C.; Stepien, K.M.; Sharma, R.; Jovanovic, A.; Tol, G.; Bentley, A. Airway Abnormalities in Adult Mucopolysaccharidosis and Development of Salford Mucopolysaccharidosis Airway Score. Journal of Clinical Medicine 2021, 10, 3275. [Google Scholar] [CrossRef] [PubMed]
- Karageorgos, L.; Brooks, D.A.; Pollard, A.; Melville, E.L.; Hein, L.K.; Clements, P.R.; Ketteridge, D.; Swiedler, S.J.; Beck, M.; Giugliani, R. Mutational analysis of 105 mucopolysaccharidosis type VI patients. Human mutation 2007, 28, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, I.; El-Boghdadly, K.; Iliff, H.; Dua, G.; Higgs, A.; Huntington, M.; Mir, F.; Nouraei, S.R.; O’Sullivan, E.P.; Patel, A. Difficult Airway Society 2025 guidelines for management of unanticipated difficult tracheal intubation in adults. British Journal of Anaesthesia 2025. [Google Scholar] [CrossRef] [PubMed]
- Akyol, M.U.; Alden, T.D.; Amartino, H.; Ashworth, J.; Belani, K.; Berger, K.I.; Borgo, A.; Braunlin, E.; Eto, Y.; Gold, J.I. Recommendations for the management of MPS IVA: systematic evidence-and consensus-based guidance. Orphanet journal of rare diseases 2019, 14, 137. [Google Scholar] [CrossRef] [PubMed]
- Nicolas-Jilwan, M.; AlSayed, M. Mucopolysaccharidoses: overview of neuroimaging manifestations. Pediatric Radiology 2018, 48, 1503–1520. [Google Scholar] [CrossRef] [PubMed]
- Garcia, P.; Phillips, D.; Johnson, J.; Martin, K.; Randolph, L.M.; Rosenfeld, H.; Harmatz, P. Long-term outcomes of patients with mucopolysaccharidosis VI treated with galsulfase enzyme replacement therapy since infancy. Molecular Genetics and Metabolism 2021, 133, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Demis, A.A.; Oikonomidou, S.; Daglis, F.; Polymenakos, S.; Panagiotou, M. Double valve replacement in a patient with Maroteaux - Lamy syndrome as an ultimate team challenge. Journal of cardiothoracic surgery 2021, 16, 141–141. [Google Scholar] [CrossRef] [PubMed]
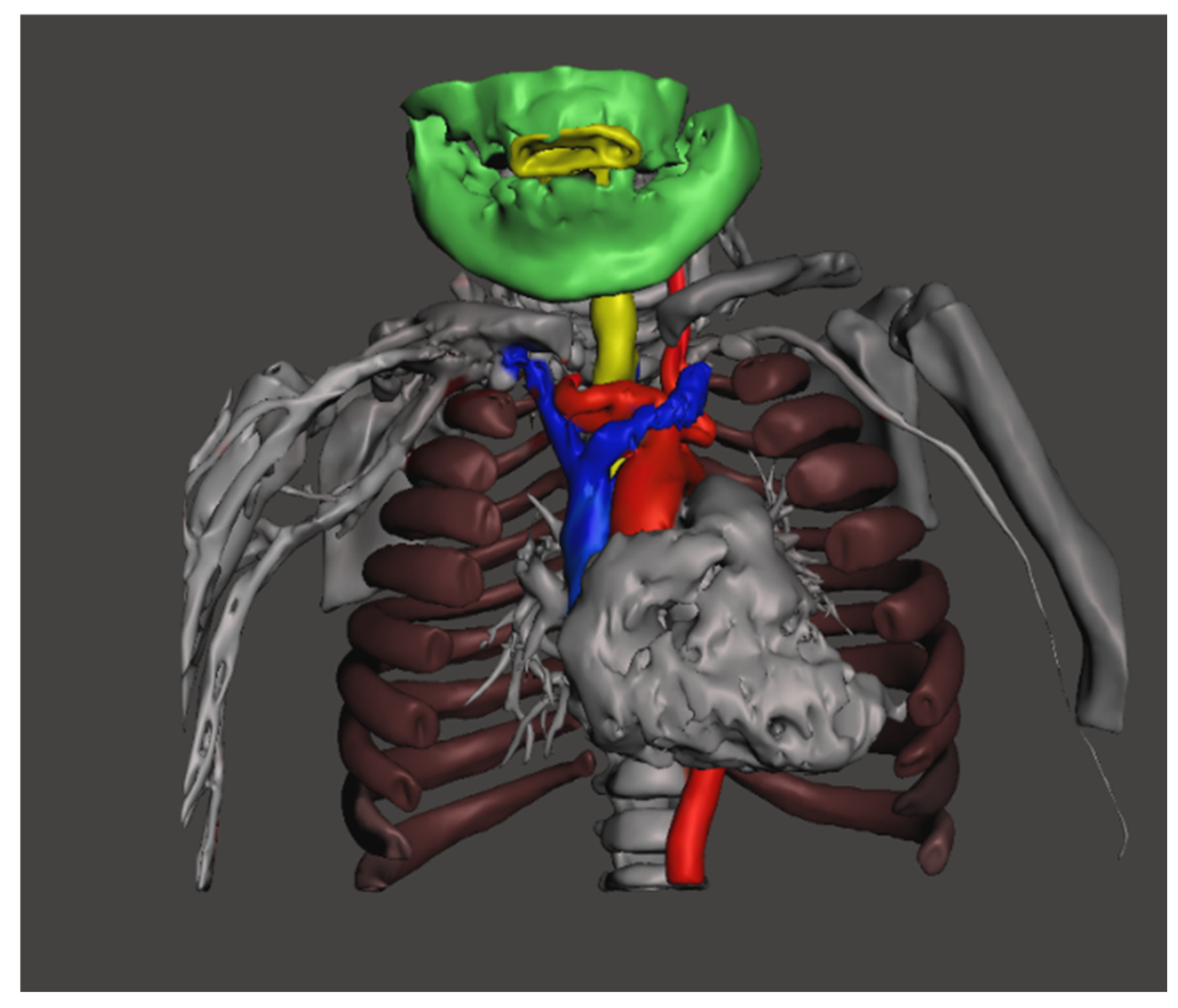
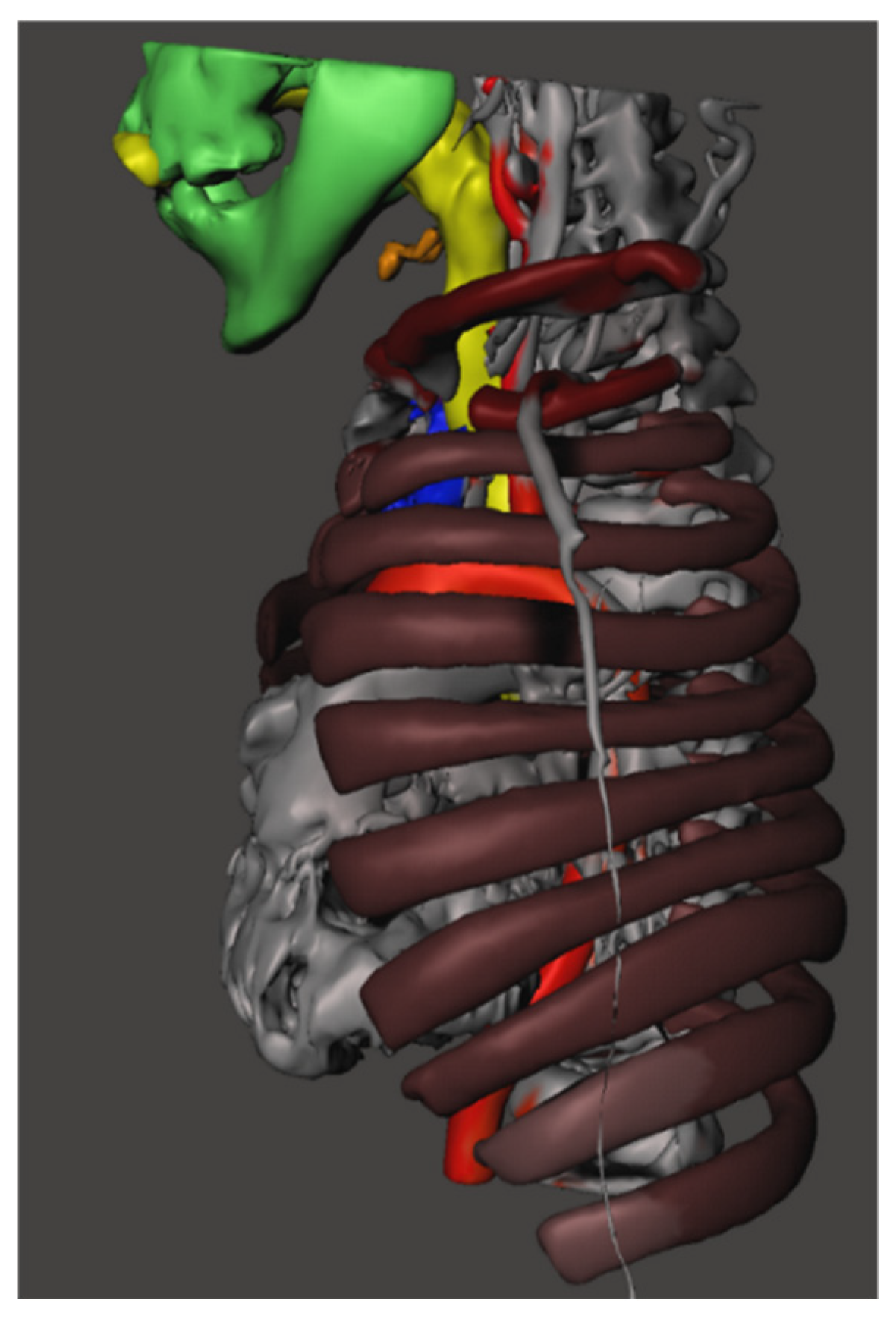
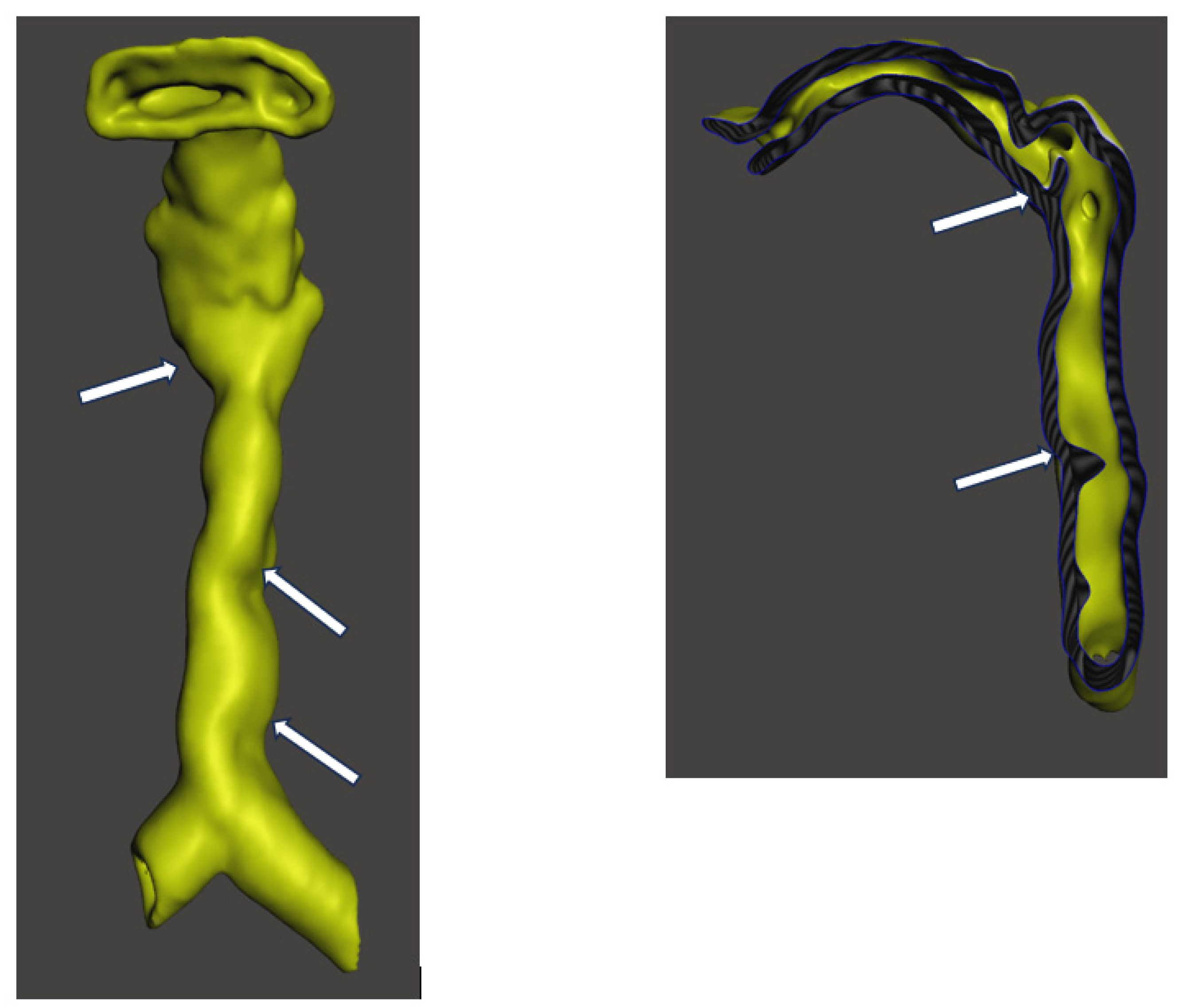
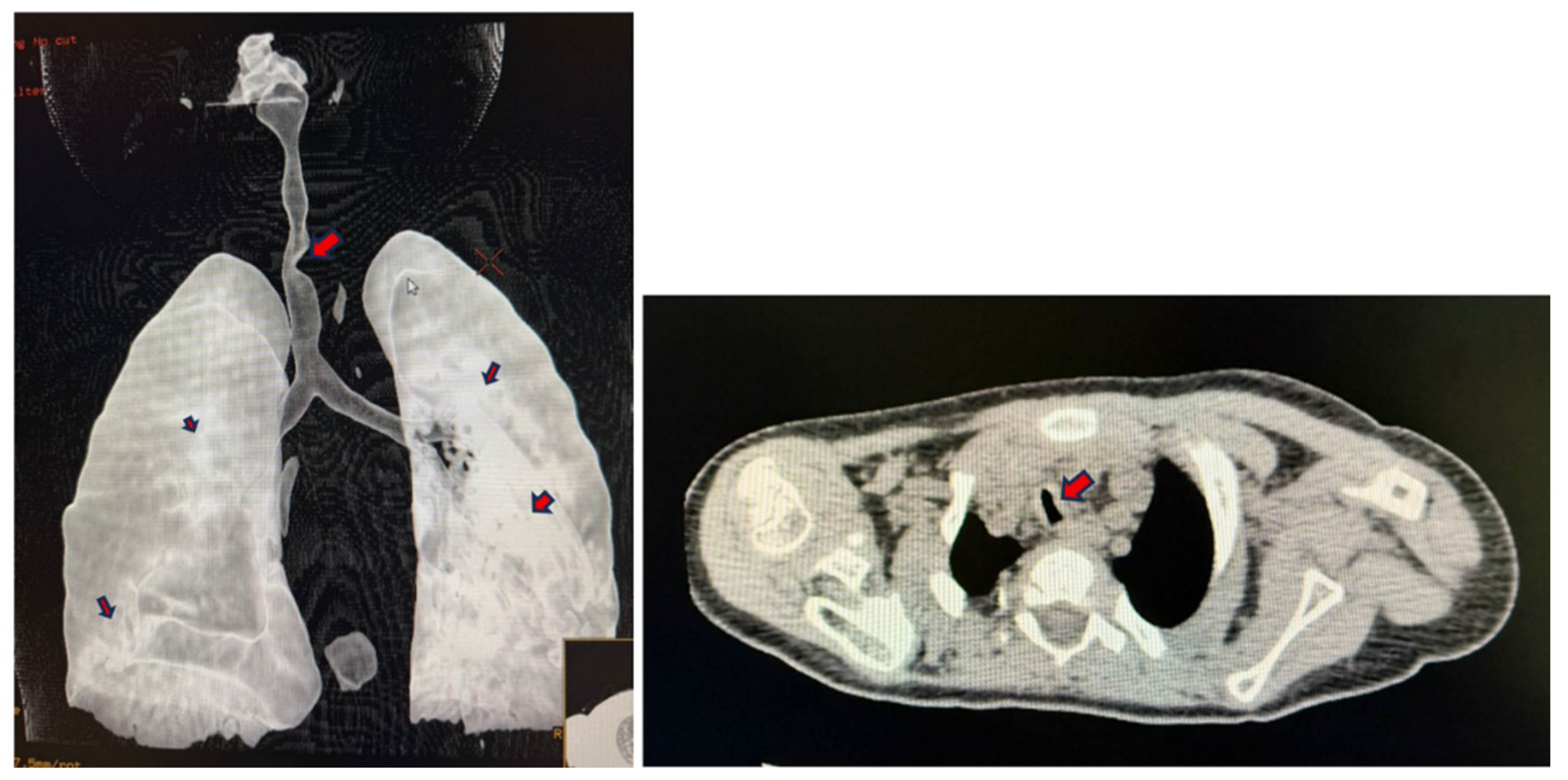
| MPS type (eponym) | Incidence per 105 live births; inheritance pattern | Typical age at diagnosis | Typical life expectancy if untreated | Enzyme deficiency | GAG |
|---|---|---|---|---|---|
| MPS I Hurler (H) MPS I Hurler-Scheie (H-S) MPS I Scheie (S) | 0.11-1.67; AR | H: < 1 year H-S: 3–8 years S: 10–20 years | H: death in childhood H-S: death in teens or early adulthood S: normal to slightly reduced lifespan | α-L-iduronidase | DS, HS |
| MPS II (Hunter) | 0.1-1.07; XR | 1-2 years when rapidly progressing | rapidly progressing: death < 15 years slowly progressing: death in adulthood | iduronate-2-sulfatase | DS, HS |
| MPS III (Sanfilippo) A-B-C-D | 0.39-1.89; AR | 4-6 years | death in puberty or early adulthood | heparan sulfamidase (A) N-acetyl-α-D-glucosaminidase (B) acetyl-CoA-α-glucosaminidase N-acetyltransferase (C) N-acetylglucosamine-6-sulfatase (D) | HS |
| MPS IV (Morquio) A-B |
0.15-0.47; AR | 1-3 years | death in childhood- middle age | N-acetylgalactosamine-6-sulfatase (A) β-galactosidase (B) | CS, KS (A) KS (B) |
| MPS VI (Maroteaux-Lamy) |
0-0.38; AR | rapidly progressing: 1–9 years slowly progressing: > 5 years | rapidly progressing: death in 2nd-3rd decade slowly progressing: death in 4-5th decade | N-acetylgalactosamine-4-sulfatase | DS |
| MPS VII (Sly) | 0-0.29; AR | neonatal to adulthood | death in infancy- 4th decade** | β-D-glucuronidase | CS, DS, HS |
| MPS IX (Natowicz)* | unknown | adolescence | unknown | hyaluronidase | CS |
| MPS X*** | unknown AR | childhood-onset | unknown | Arylsulfatase K | DS |
| Airway plan | Challenges | Consequence | Methods to mitigate |
|---|---|---|---|
| Plan A Endo tracheal intubation |
Reduced mouth opening, large tongue, high anterior larynx, large tongue, limited neck extension. Tortuous trachea with multiple narrowing’s can make passage of the endo tracheal tube difficult. | Access to the larynx and passage of the endo tracheal tube into the trachea will be difficult | Using small endo tracheal tube 1.Nasal intubation 2.Oral intubation with video laryngoscope or Hopkins rod telescope 3.Awake nasal or oral fiberoptic using an airway conduit |
| Plan B Laryngeal Mask airway (LMA) |
Limited mouth opening, large tongue, high anterior larynx, bulky supraglottic | Inserting the LMA and securing a seal will be difficult | Using re-enforced LMA, which are more flexible to reach anterior larynx |
| Plan C Bag and mask ventilation |
Limited mouth opening, large tongue which can fall posteriorly occluding the airway, bulky oropharynx, bulky supra glottis | Inability to pass oxygen beyond the oropharynx due to obstruction | 1.Guedel’s airway to by-pass the tongue base 2.Nasopharyngeal airway to bypass the epiglottis |
|
Plan D Front of neck access |
Short neck, limited extension, large head, small torso Large vessels in the thoracic inlet |
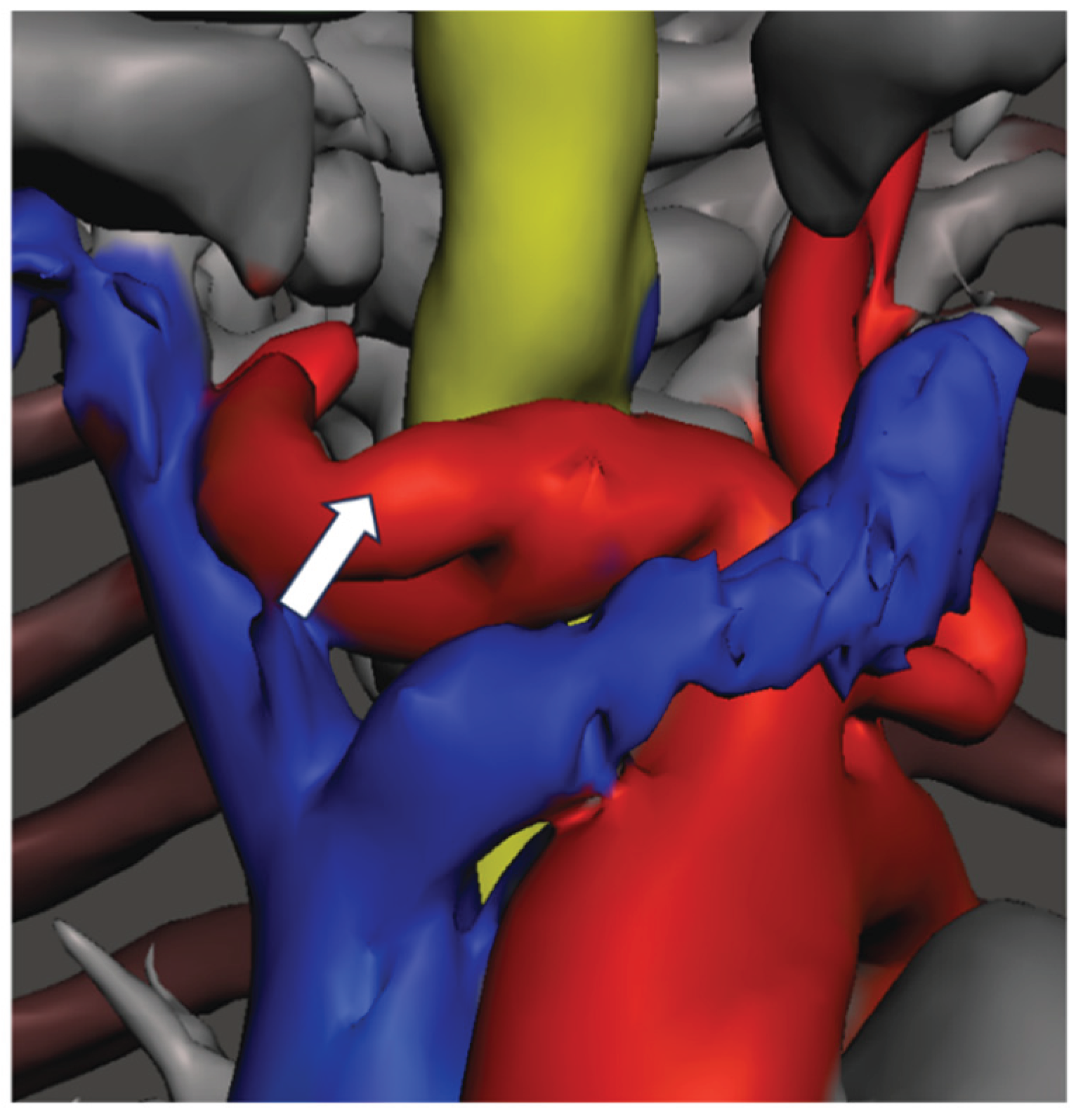
Accessing the cervical trachea will be difficult, large vessel catastrophic haemorrhage, inserting the right sized tracheostomy tube | Avoid tracheostomy, if attempted perform high tracheostomy. Planning the right tracheostomy tube before the surgery |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




