Submitted:
16 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
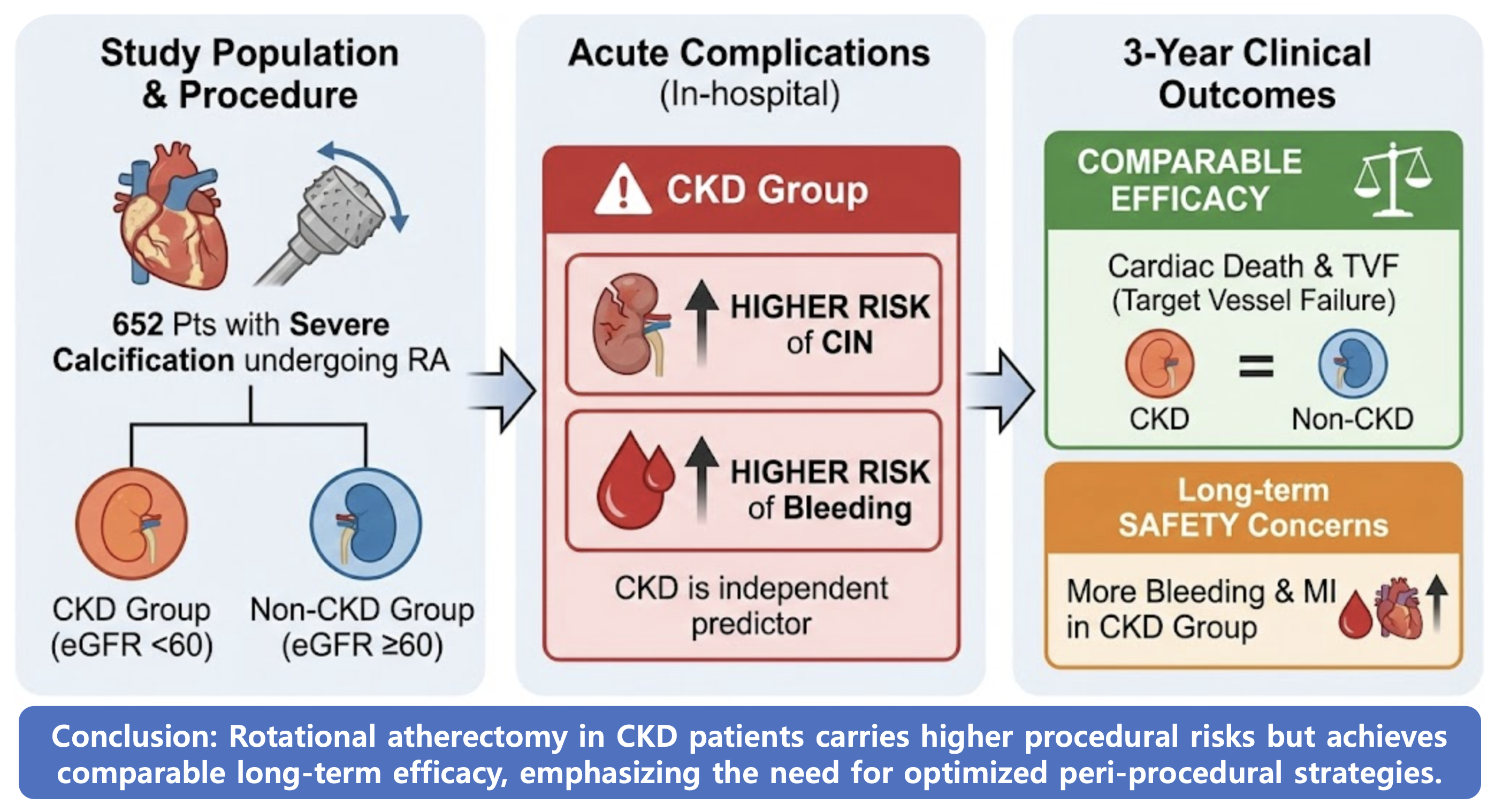
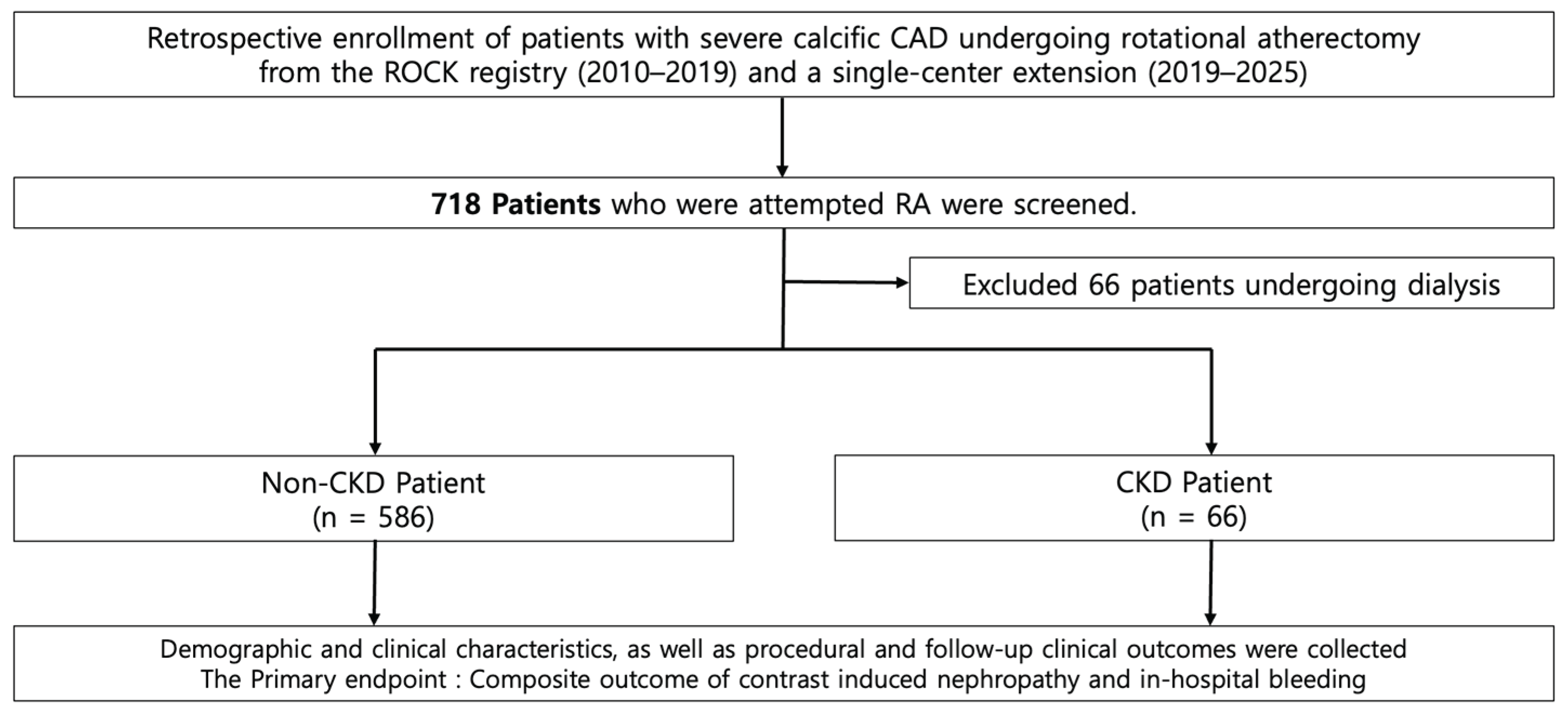
2.1. Study Design and Population
2.2. RA Procedure
2.3. Study Outcomes
2.4. Statistical Analyses
3. Results
3.1. Baseline Characteristics
3.2. Primary Outcomes
3.3. Secondary Outcomes
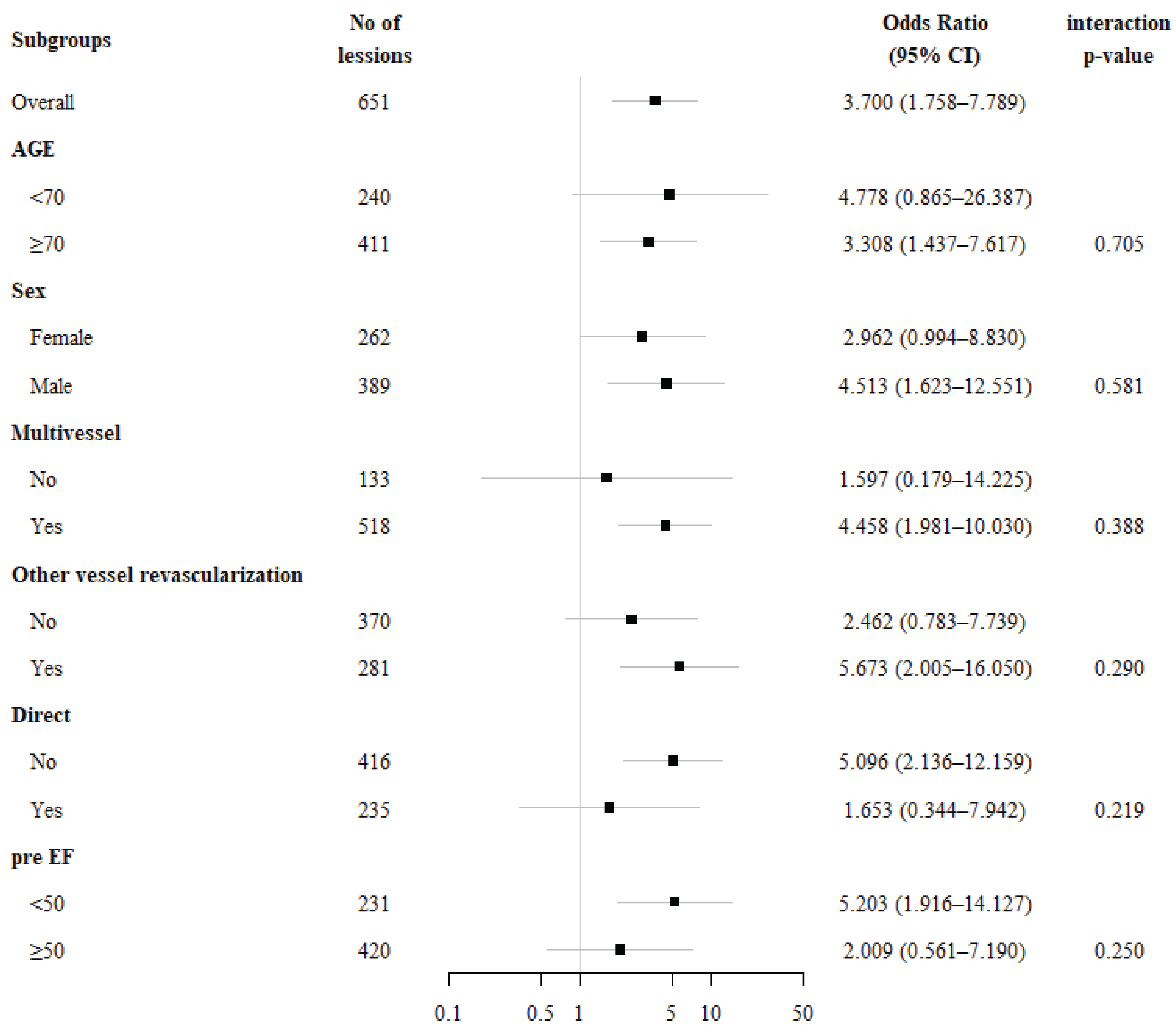
3.4. Subgroup Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CABG | coronary artery bypass graft |
| CD | cardiac death |
| CIN | contrast induced nephropathy |
| CKD | chronic kidney disease |
| CVA | cerebrovascular accident |
| LVEF | left ventricle ejection fraction |
| MVD | multivessel disease |
| PCI | percutaneous coronary intervention |
| RA | rotational atherectomy |
| ST | stent thrombosis |
| RR | repeat revascularization |
| TVF | target vessel failure |
| TVM | target-vessel spontaneous myocardial infarction |
| TVR | target-vessel revascularization |
References
- Russo, D; Palmiero, G; De Blasio, AP; Balletta, MM; Andreucci, VE. Coronary artery calcification in patients with CRF not undergoing dialysis. Am J Kidney Dis. 2004, 44(6), 1024–30. [Google Scholar] [CrossRef]
- Nitta, K; Akiba, T; Suzuki, K; Uchida, K; Ogawa, T; Majima, K; et al. Assessment of coronary artery calcification in hemodialysis patients using multi-detector spiral CT scan. Hypertens Res. 2004, 27(8), 527–33. [Google Scholar] [CrossRef] [PubMed]
- Wang, XR; Zhang, JJ; Xu, XX; Wu, YG. Prevalence of coronary artery calcification and its association with mortality, cardiovascular events in patients with chronic kidney disease: a systematic review and meta-analysis. Ren Fail. 2019, 41(1), 244–56. [Google Scholar] [CrossRef] [PubMed]
- Guedeney, P; Claessen, BE; Mehran, R; Mintz, GS; Liu, M; Sorrentino, S; et al. Coronary Calcification and Long-Term Outcomes According to Drug-Eluting Stent Generation. JACC Cardiovasc Interv. 2020, 13(12), 1417–28. [Google Scholar] [CrossRef] [PubMed]
- Généreux, P; Madhavan, MV; Mintz, GS; Maehara, A; Palmerini, T; Lasalle, L; et al. Ischemic outcomes after coronary intervention of calcified vessels in acute coronary syndromes. Pooled analysis from the HORIZONS-AMI (Harmonizing Outcomes With Revascularization and Stents in Acute Myocardial Infarction) and ACUITY (Acute Catheterization and Urgent Intervention Triage Strategy) TRIALS. J Am Coll Cardiol. 2014, 63(18), 1845–54. [Google Scholar]
- Dangas, G; Iakovou, I; Nikolsky, E; Aymong, ED; Mintz, GS; Kipshidze, NN; et al. Contrast-induced nephropathy after percutaneous coronary interventions in relation to chronic kidney disease and hemodynamic variables. Am J Cardiol. 2005, 95(1), 13–9. [Google Scholar] [CrossRef]
- Saltzman, AJ; Stone, GW; Claessen, BE; Narula, A; Leon-Reyes, S; Weisz, G; et al. Long-term impact of chronic kidney disease in patients with ST-segment elevation myocardial infarction treated with primary percutaneous coronary intervention: the HORIZONS-AMI (Harmonizing Outcomes With Revascularization and Stents in Acute Myocardial Infarction) trial. JACC Cardiovasc Interv. 2011, 4(9), 1011–9. [Google Scholar]
- Appleby, CE; Ivanov, J; Lavi, S; Mackie, K; Horlick, EM; Ing, D; et al. The adverse long-term impact of renal impairment in patients undergoing percutaneous coronary intervention in the drug-eluting stent era. Circ Cardiovasc Interv. 2009, 2(4), 309–16. [Google Scholar] [CrossRef]
- Barbato, E; Carrié, D; Dardas, P; Fajadet, J; Gaul, G; Haude, M; et al. European expert consensus on rotational atherectomy. EuroIntervention 2015, 11(1), 30–6. [Google Scholar] [CrossRef]
- Jung, J; Her, S-H. Rotational Atherectomy: A to Z. J Cardiovasc Interv. 2025, 4(1), 29–39. [Google Scholar] [CrossRef]
- Matsuo, S; Imai, E; Horio, M; Yasuda, Y; Tomita, K; Nitta, K; et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009, 53(6), 982–92. [Google Scholar] [CrossRef] [PubMed]
- Levine, GN; Bates, ER; Blankenship, JC; Bailey, SR; Bittl, JA; Cercek, B; et al. 2011 ACCF/AHA/SCAI guideline for percutaneous coronary intervention: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Society for Cardiovascular Angiography and Interventions. Catheter Cardiovasc Interv. 2013, 82(4), E266–355. [Google Scholar] [CrossRef] [PubMed]
- Levine, GN; Bates, ER; Bittl, JA; Brindis, RG; Fihn, SD; Fleisher, LA; et al. 2016 ACC/AHA Guideline Focused Update on Duration of Dual Antiplatelet Therapy in Patients With Coronary Artery Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines: An Update of the 2011 ACCF/AHA/SCAI Guideline for Percutaneous Coronary Intervention, 2011 ACCF/AHA Guideline for Coronary Artery Bypass Graft Surgery, 2012 ACC/AHA/ACP/AATS/PCNA/SCAI/STS Guideline for the Diagnosis and Management of Patients With Stable Ischemic Heart Disease, 2013 ACCF/AHA Guideline for the Management of ST-Elevation Myocardial Infarction, 2014 AHA/ACC Guideline for the Management of Patients With Non-ST-Elevation Acute Coronary Syndromes, and 2014 ACC/AHA Guideline on Perioperative Cardiovascular Evaluation and Management of Patients Undergoing Noncardiac Surgery. Circulation 2016, 134(10), e123–55. [Google Scholar] [PubMed]
- Moussa, ID; Klein, LW; Shah, B; Mehran, R; Mack, MJ; Brilakis, ES; et al. Consideration of a new definition of clinically relevant myocardial infarction after coronary revascularization: an expert consensus document from the Society for Cardiovascular Angiography and Interventions (SCAI). Catheter Cardiovasc Interv. 2014, 83(1), 27–36. [Google Scholar] [CrossRef]
- Liu, X; Wan, L; Wu, X; Gu, Y; Hu, L. Low Preoperative Mean Arterial Pressure as a Risk Factor for Contrast-Induced Nephropathy After Rotational Atherectomy. Rev Cardiovasc Med. 2025, 26(11), 43418. [Google Scholar] [CrossRef]
- Kumar, S; Kearney, KE; Chung, CJ; Elison, D; Steinberg, ZL; Lombardi, WL; et al. Risk of acute kidney injury after percutaneous coronary intervention with plaque modification. Cardiovasc Revasc Med. 2025, 77, 12–9. [Google Scholar] [CrossRef]
- Kawamoto, H; Latib, A; Ruparelia, N; Ielasi, A; D’Ascenzo, F; Pennacchi, M; et al. In-hospital and midterm clinical outcomes of rotational atherectomy followed by stent implantation: the ROTATE multicentre registry. EuroIntervention 2016, 12(12), 1448–56. [Google Scholar] [CrossRef]
- Crimi, G; De Marzo, V; De Marco, F; Conrotto, F; Oreglia, J; D’Ascenzo, F; et al. Acute Kidney Injury After Transcatheter Aortic Valve Replacement Mediates the Effect of Chronic Kidney Disease. J Am Heart Assoc. 2022, 11(19), e024589. [Google Scholar] [CrossRef]
- Li, SX; Patel, NK; Flannery, LD; Cigarroa, RJ; Shaqdan, AW; Erickson, P; et al. Impact of bleeding after transcatheter aortic valve replacement in patients with chronic kidney disease. Catheter Cardiovasc Interv. 2021, 97(1), E172–e8. [Google Scholar] [CrossRef]
- Demir, OM; Poletti, E; Lombardo, F; Laricchia, A; Beneduce, A; Maccagni, D; et al. Rotational atherectomy-based percutaneous coronary intervention and the risk of contrast-induced nephropathy. Minerva Cardioangiol. 2020, 68(2), 137–45. [Google Scholar] [CrossRef]
- Jubelirer, SJ. Hemostatic abnormalities in renal disease. Am J Kidney Dis. 1985, 5(5), 219–25. [Google Scholar] [CrossRef]
- Yeh, RW; Secemsky, EA; Kereiakes, DJ; Normand, SL; Gershlick, AH; Cohen, DJ; et al. Development and Validation of a Prediction Rule for Benefit and Harm of Dual Antiplatelet Therapy Beyond 1 Year After Percutaneous Coronary Intervention. Jama 2016, 315(16), 1735–49. [Google Scholar] [CrossRef]
- Costa, F; van Klaveren, D; James, S; Heg, D; Räber, L; Feres, F; et al. Derivation and validation of the predicting bleeding complications in patients undergoing stent implantation and subsequent dual antiplatelet therapy (PRECISE-DAPT) score: a pooled analysis of individual-patient datasets from clinical trials. Lancet 2017, 389(10073), 1025–34. [Google Scholar] [CrossRef]
- Urban, P; Mehran, R; Colleran, R; Angiolillo, DJ; Byrne, RA; Capodanno, D; et al. Defining High Bleeding Risk in Patients Undergoing Percutaneous Coronary Intervention. Circulation 2019, 140(3), 240–61. [Google Scholar] [CrossRef]
- van Geuns, RJ; Chun-Chin, C; McEntegart, MB; Merkulov, E; Kretov, E; Lesiak, M; et al. Bioabsorbable polymer drug-eluting stents with 4-month dual antiplatelet therapy versus durable polymer drug-eluting stents with 12-month dual antiplatelet therapy in patients with left main coronary artery disease: the IDEAL-LM randomised trial. EuroIntervention 2022, 17(18), 1467–76. [Google Scholar] [CrossRef]
- Chiu, JH; Bhatt, DL; Ziada, KM; Chew, DP; Whitlow, PL; Lincoff, AM; et al. Impact of female sex on outcome after percutaneous coronary intervention. Am Heart J. 2004, 148(6), 998–1002. [Google Scholar] [CrossRef]
- Lo, WJ; Chen, WJ; Lai, CH; Chen, YW; Su, CS; Chang, WC; et al. Rotablation in Patients with Advanced Renal Insufficiency through End-Stage Renal Disease: Short- and Intermediate-Term Results. J Interv Cardiol. 2022, 2022, 7884401. [Google Scholar] [CrossRef]
| non CKD | CKD | ||
| n = 586 | n = 66 | p-value | |
| Age | 72.0±9.6 | 74.3±8.3 | 0.069 |
| Sex | 351(60.0) | 38(57.6) | 0.703 |
| BMI | 24.2±3.9 | 24.1±3.6 | 0.874 |
| Systolic BP | 131.9±24.1 | 133.5±18.2 | 0.581 |
| Diastolic BP | 74.8±12.5 | 74.3±10.9 | 0.737 |
| Smoking | 111(19.0) | 7(10.6) | 0.094 |
| HTN | 446(76.2) | 57(86.4) | 0.063 |
| DM | 317(54.2) | 52(78.8) | <0.001 |
| Dyslipidemia | 301(51.5) | 33(50.0) | 0.823 |
| CKD (no dialysis) | 0(0.0) | 66(100.0) | <0.001 |
| LV Ejection Fraction | 54.0±12.9 | 48.4±14.0 | 0.001 |
| Medical History | |||
| PCI | 136(23.3) | 19(28.8) | 0.317 |
| CABG | 24(4.1) | 4(6.1) | 0.515 |
| MI | 64(10.9) | 9(13.6) | 0.511 |
| CVA | 93(15.9) | 10(15.2) | 0.875 |
| PVD | 38(6.5) | 14(21.2) | <0.001 |
| Chronic lung disease | 34(5.8) | 8(12.1) | 0.061 |
| Heart failure | 73(12.5) | 12(18.2) | 0.192 |
| Atrial fibrillation | 42(7.2) | 10(15.2) | 0.024 |
| Clinical_diagnosis (STEMI/NSTEMI) |
152(26.0) | 26(39.4) | 0.021 |
| non CKD | CKD | ||
| n = 586 | n = 66 | p-value | |
| Labaratory analyses | |||
| Hb | 12.6±2.3 | 11.1±1.8 | <0.001 |
| Platelet | 220.8±65.5 | 219.8±93.0 | 0.910 |
| Triglyceride | 119.0±74.2 | 123.1±66.0 | 0.686 |
| Total cholesterol | 146.5±40.1 | 134.6±35.3 | 0.030 |
| LDL cholesterol | 85.2±39.3 | 75.6±31.9 | 0.079 |
| HDL cholesterol | 47.1±14.7 | 40.2±14.3 | <0.001 |
| Hs-CRP | 2.6±10.6 | 2.5±6.8 | 0.971 |
| HbA1c | 6.7±1.6 | 7.4±1.5 | 0.002 |
| Medication | |||
| NOAC | 21(3.6) | 5(7.6) | 0.171 |
| DAPT | 559(95.6) | 61(92.4) | 0.230 |
| Aspirin | 571(97.6) | 62(93.9) | 0.099 |
| P2Y12_inhibitor | 574(98.1) | 66(100.0) | 0.614 |
| Cilostazol | 73(12.5) | 6(9.1) | 0.424 |
| Beta-blocker | 392(67.0) | 45(68.2) | 0.848 |
| ACEi or ARB | 377(64.4) | 39(59.1) | 0.391 |
| Statin | 552(94.4) | 62(93.9) | 0.782 |
| non CKD | CKD | ||
| n = 586 | n = 66 | p-value | |
| Lesion Classification(B2/C) | 549(93.9) | 62(93.9) | >0.999 |
| Vessel disease | |||
| 1VD | 124(21.2) | 9(13.6) | 0.312 |
| 2VD | 187(32.0) | 25(37.9) | |
| 3VD | 274(46.8) | 32(48.5) | |
| Multivessel disease (MVD) | 461(78.8) | 57(86.4) | 0.149 |
| LM disease | 86(14.7) | 11(16.7) | 0.671 |
| IVUS | 303(51.8) | 37(56.1) | 0.511 |
| Direct | 211(36.1) | 24(36.4) | 0.962 |
| Other vessel revascularization | 245(41.9) | 36(54.6) | 0.049 |
| Procedural approach | |||
| Radial | 291(49.7) | 31(47.0) | 0.669 |
| Femoral | 294(50.3) | 35(53.0) | |
| Procedure success | 555(94.9) | 64(97.0) | 0.762 |
| Technical success | 562(96.1) | 66(100.0) | 0.155 |
| Contrast (mL) | 214.1±91.9 | 189.4±95.6 | 0.044 |
| Procedural time | 78.0±51.4 | 86.5±56.4 | 0.211 |
| Radiation dose | 3,811.6±3,643.3 | 4,354.1±2,684.3 | 0.574 |
| Rotational atherectomy | |||
| Size of burr (start) | 1.5±0.2 | 1.5±0.2 | 0.951 |
| Size of burr (max) | 1.5±0.6 | 1.5±0.2 | 0.795 |
| Number of burr | 1.2±0.4 | 1.2±0.5 | 0.991 |
| Stent | |||
| Stent diameter | 3.0±0.4 | 3.0±0.3 | 0.384 |
| Total number of stent | 2.5±1.3 | 2.6±1.3 | 0.377 |
| Total length of stent | 69.6±37.7 | 74.3±38.4 | 0.343 |
| Peri-procedural complication | |||
| Coronary dissection | 192(32.8) | 19(28.8) | 0.507 |
| Coronary perforation | 21(3.6) | 2(3.0) | >0.999 |
| Urgent intervention for tamponade | 3(0.5) | 0(0.0) | >0.999 |
| Temporary pacemaker | 39(6.7) | 6(9.1) | 0.442 |
| Periprocedural MI | 54(9.2) | 10(15.2) | 0.126 |
| Inhospital outcome | |||
| Inhospital death | 18(3.1) | 2(3.0) | >0.999 |
| Inhospital CVA | 2(0.3) | 0(0.0) | >0.999 |
| Urgent CABG | 2(0.3) | 0(0.0) | >0.999 |
| Urgent PCI | 5(0.9) | 1(1.5) | 0.475 |
| non CKD | CKD | univariate | multivariate | |||||||||
| Primary outcome | n = 586 | n = 66 | p-value | OR | 95%CI (lower – upper) |
p-value | OR | 95%CI (lower – upper) |
p-value | |||
| Composite | 30(5.1) | 11(16.7) | 0.001 | 3.700 | 1.758 | 7.789 | <0.001 | 3.019 | 1.363 | 6.685 | 0.006 | |
| CIN | 10(1.7) | 10(15.2) | <0.001 | 10.269 | 4.099 | 25.727 | <0.001 | 7.184 | 2.324 | 22.210 | <0.001 | |
| In-hospital bleeding | 21(3.6) | 2(3.0) | >0.999 | 0.839 | 0.192 | 3.662 | 0.816 | 0.779 | 0.170 | 3.565 | 0.747 | |
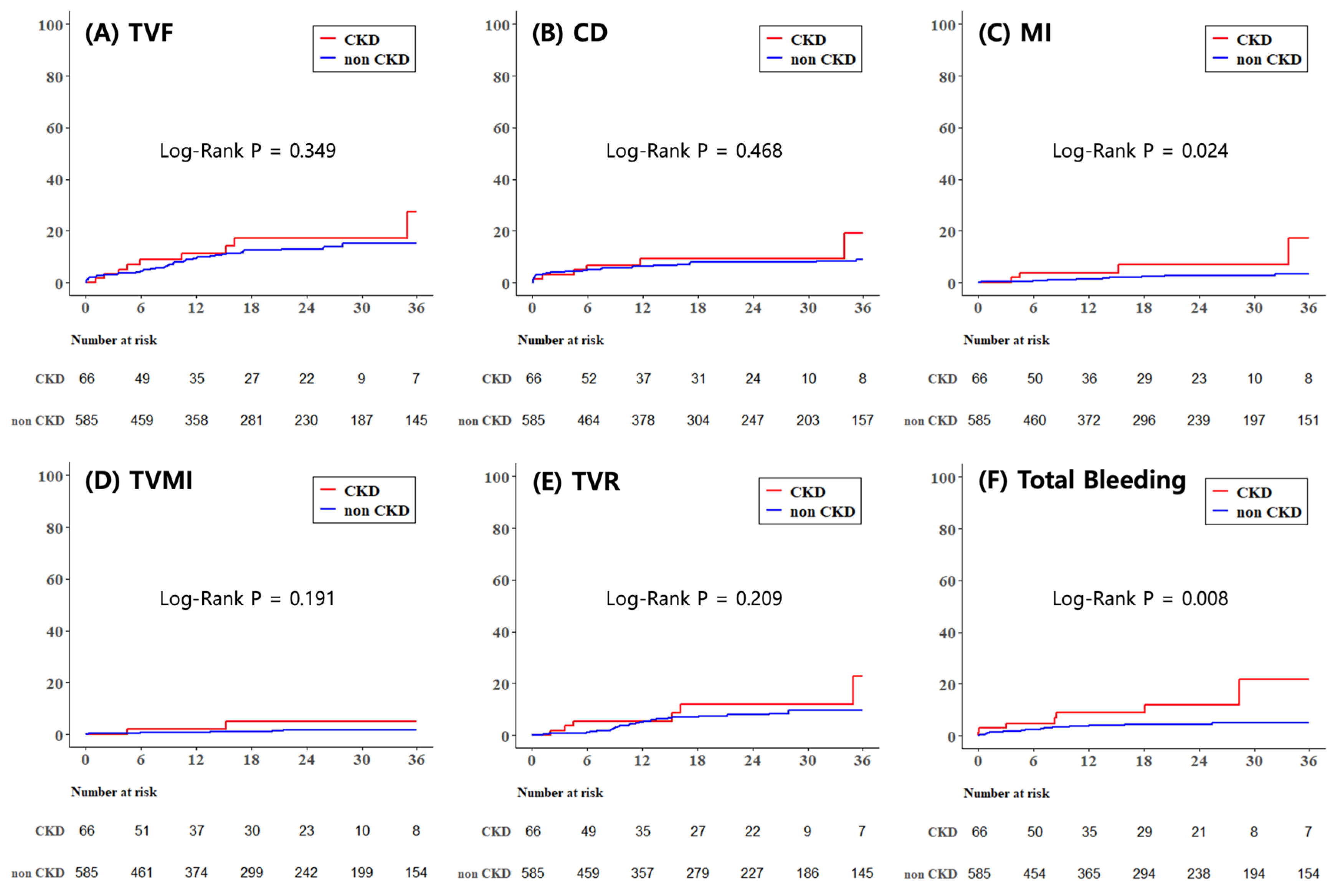
| Secondary outcome (3-year outcome) |
n = 586 | n = 66 | p-value | Log-rank p-value |
HR | 95%CI (lower – upper) |
p-value | HR | 95%CI (lower – upper) |
p-value | ||
| TVF | 63(10.8) | 9(13.6) | 0.481 | 0.349 | 1.395 | 0.693 | 2.807 | 0.351 | 1.317 | 0.639 | 2.715 | 0.456 |
| AD | 55(9.4) | 9(13.6) | 0.273 | 0.210 | 1.565 | 0.772 | 3.171 | 0.214 | 1.326 | 0.641 | 2.743 | 0.447 |
| CD | 41(7.0) | 6(9.1) | 0.461 | 0.468 | 1.373 | 0.582 | 3.236 | 0.469 | 1.260 | 0.522 | 3.043 | 0.607 |
| MI | 12(2.1) | 4(6.1) | 0.069 | 0.024 | 3.417 | 1.097 | 10.645 | 0.034 | 3.975 | 1.155 | 13.683 | 0.029 |
| TVMI | 7(1.2) | 2(3.0) | 0.229 | 0.191 | 2.735 | 0.568 | 13.175 | 0.210 | 3.904 | 0.616 | 24.735 | 0.148 |
| RR | 50(8.6) | 6(9.1) | 0.881 | 0.723 | 1.165 | 0.499 | 2.719 | 0.724 | 0.933 | 0.390 | 2.233 | 0.876 |
| TVR | 35(6.0) | 6(9.1) | 0.291 | 0.209 | 1.733 | 0.727 | 4.128 | 0.215 | 1.459 | 0.590 | 3.611 | 0.414 |
| TLR | 31(5.3) | 4(6.1) | 0.772 | 0.632 | 1.289 | 0.454 | 3.660 | 0.633 | 1.091 | 0.370 | 3.216 | 0.875 |
| NLR | 25(4.3) | 3(4.6) | 0.756 | 0.756 | 1.209 | 0.365 | 4.010 | 0.756 | 1.149 | 0.333 | 3.971 | 0.826 |
| CVA | 8(1.4) | 3(4.6) | 0.091 | 0.054 | 3.419 | 0.906 | 12.913 | 0.070 | 3.296 | 0.820 | 13.250 | 0.093 |
| ST | 5(0.9) | 1(1.5) | 0.475 | 0.603 | 1.756 | 0.205 | 15.028 | 0.607 | 1.731 | 0.179 | 16.745 | 0.635 |
| Total bleeding | 23(3.9) | 7(10.6) | 0.025 | 0.008 | 2.983 | 1.275 | 6.978 | 0.012 | 3.392 | 1.374 | 8.370 | 0.008 |
| Minor bleeding | 9(1.5) | 5(7.6) | 0.009 | <0.001 | 5.776 | 1.918 | 17.396 | 0.002 | 7.798 | 2.282 | 26.646 | 0.001 |
| Major bleeding | 9(1.5) | 1(1.5) | >0.999 | 0.940 | 1.082 | 0.137 | 8.577 | 0.940 | 1.336 | 0.151 | 11.854 | 0.795 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




