Submitted:
26 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
Introduction
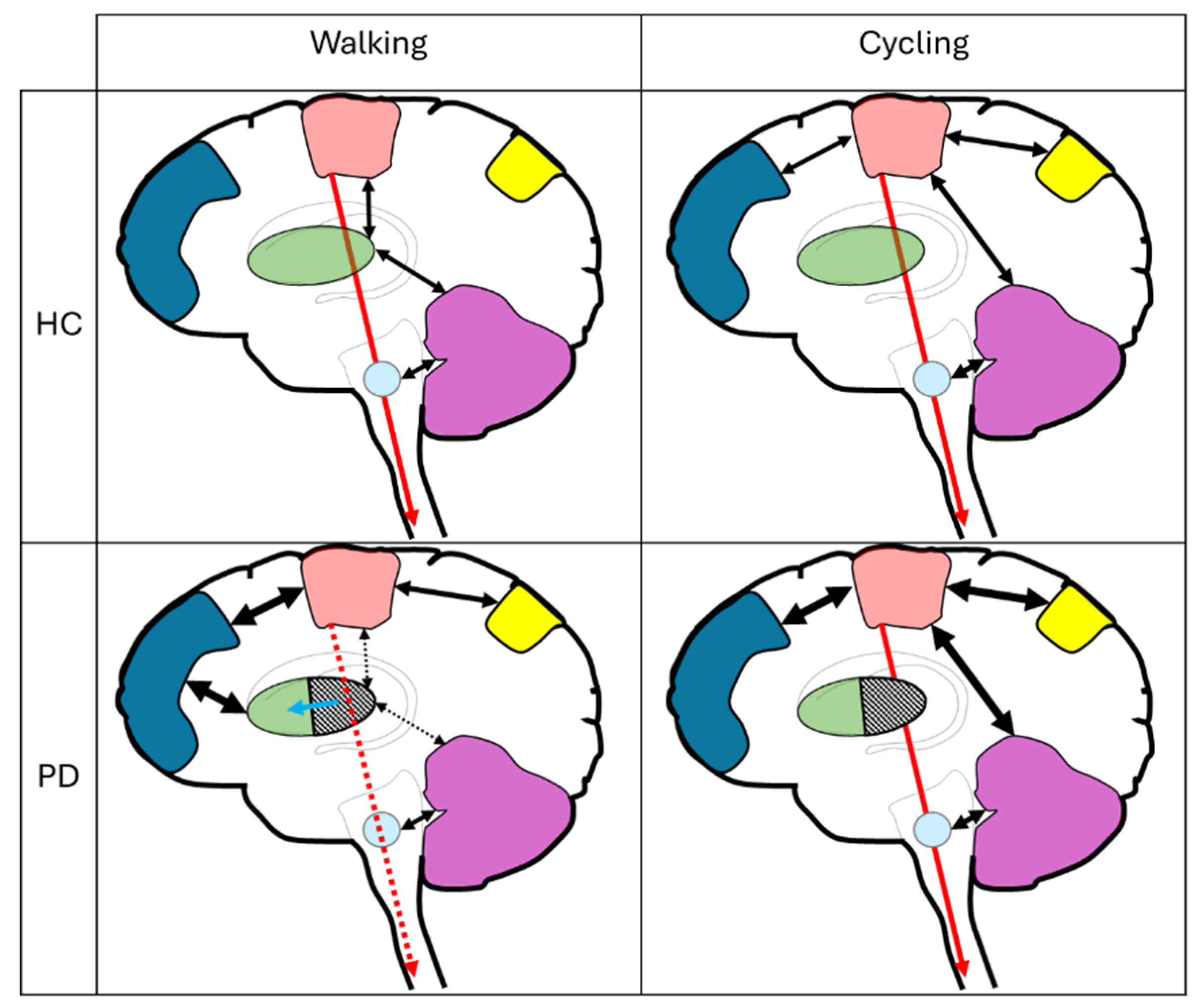
Box 1: Beta-band oscillatory activity
Box 2: Feedback versus feed-forward motor control
Symptomatic Effects of Stationary Cycling Exercise in PD
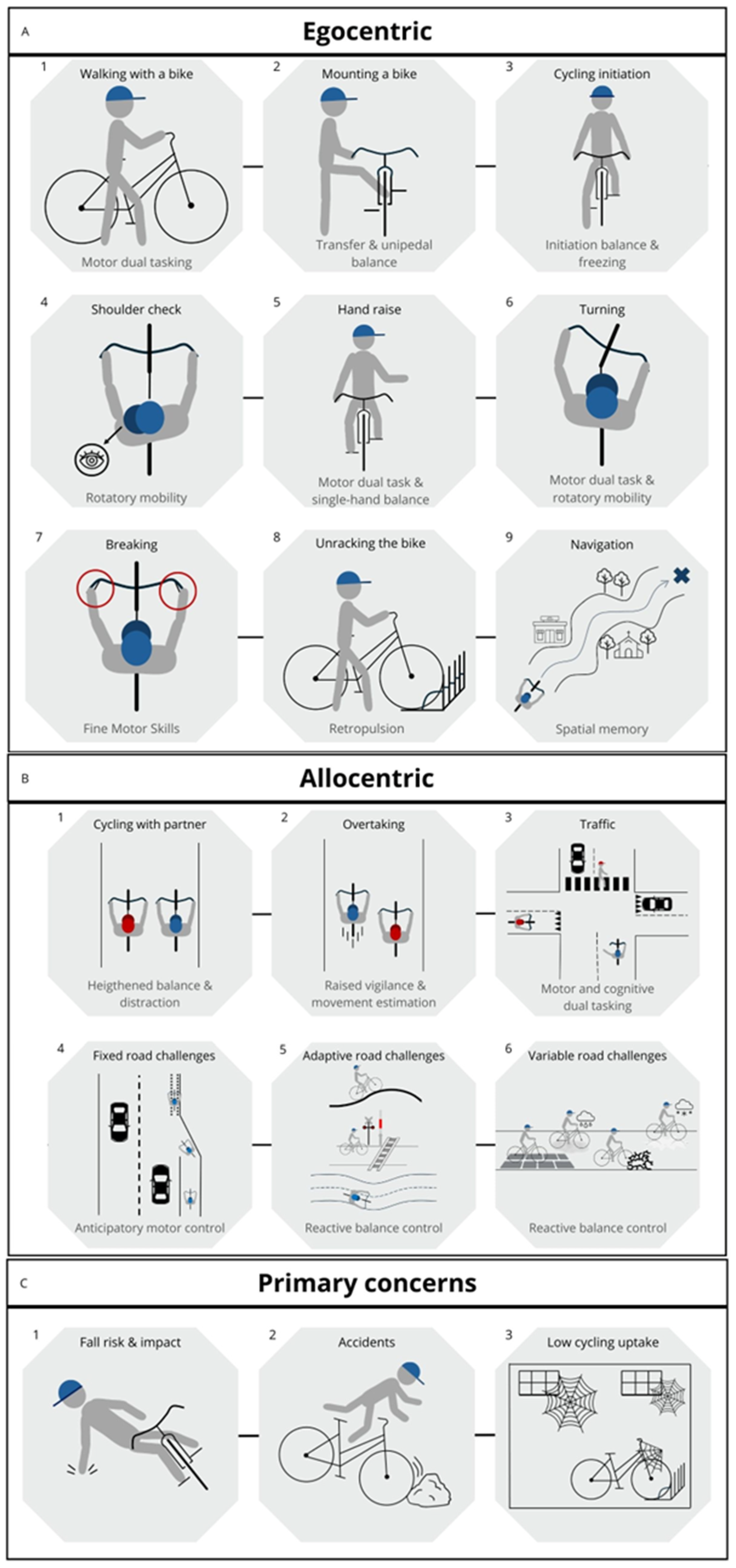
Outdoor Cycling Challenges as Alternative form of Physical Activity?
Gaps and Future Directions
Conclusion
Author contributions
Funding statements
Data availability
Acknowledgements
Statements and declarations
References
- Kalia L V., Lang AE. Parkinson’s disease. Lancet 2015; 386: 896–912.
- Dorsey ER, Sherer T, Okun MS, et al. The Emerging Evidence of the Parkinson Pandemic. J Parkinsons Dis 2018; 8: S3–S8.
- Wang S, Che Y, Lin Y, et al. Epidemiology of Parkinson’s disease – Global burden of disease research from 1990 to 2021 and future trend predictions. Clin Park Relat Disord 2026; 14: 100421.
- Wu T, Hallett M, Chan P. Motor automaticity in Parkinson’s disease. Neurobiol Dis 2015; 82: 226–234.
- Gilat M, Bell PT, Ehgoetz Martens KA, et al. Dopamine depletion impairs gait automaticity by altering cortico-striatal and cerebellar processing in Parkinson’s disease. Neuroimage 2017; 152: 207–220.
- Bloem BR, Okun MS, Klein C. Parkinson’s disease. The Lancet 2021; 397: 2284–2303.
- Redgrave P, Rodriguez M, Smith Y, et al. Goal-directed and habitual control in the basal ganglia: implications for Parkinson’s disease. Nat Rev Neurosci 2010; 11: 760–772.
- Tosserams A, Fasano A, Gilat M, et al. Management of freezing of gait — mechanism-based practical recommendations. Nature Reviews Neurology 2025 21:6 2025; 21: 327–344.
- Hassan A, Wu SS, Schmidt P, et al. The Profile of Long-term Parkinson’s Disease Survivors with 20 Years of Disease Duration and Beyond. J Parkinsons Dis 2015; 5: 313–319.
- Mirelman A, Bonato P, Camicioli R, et al. Gait impairments in Parkinson’s disease. Lancet Neurol 2019; 18: 697–708.
- Radder DLM, Lígia Silva de Lima A, Domingos J, et al. Physiotherapy in Parkinson’s Disease: A Meta-Analysis of Present Treatment Modalities. Neurorehabil Neural Repair 2020; 34: 871–880.
- Osborne JA, Botkin R, Colon-Semenza C, et al. Physical Therapist Management of Parkinson Disease: A Clinical Practice Guideline From the American Physical Therapy Association. Phys Ther; 102. Epub ahead of print 1 April 2022. [CrossRef]
- Caspersen C, Powell K, Gregory C. Physical activity, exercise, and physical fitness: definitions and distinctions for health-related research. Public Health Reports 1985; 100: 126.
- Speelman AD, Van De Warrenburg BP, Van Nimwegen M, et al. How might physical activity benefit patients with Parkinson disease? Nat Rev Neurol 2011; 7: 528–534.
- Mammen JR, Tyo M, Cadorette J, et al. Understanding what aspects of Parkinson’s disease matter most to patients and families. Scientific Reports 2024 14:1 2024; 14: 21171-.
- Hulbert S, Chivers-Seymour K, Summers R, et al. ‘PDSAFE’ - a multi-dimensional model of falls-rehabilitation for people with Parkinson’s. A mixed methods analysis of therapists’ delivery and experience. Physiotherapy 2021; 110: 77–84.
- Linder SM, Baron E, Learman K, et al. An 8-week aerobic cycling intervention elicits improved gait velocity and biomechanics in persons with Parkinson’s disease. Gait Posture 2022; 98: 313–315.
- Rosenfeldt AB, Koop MM, Penko AL, et al. Components of a successful community-based exercise program for individuals with Parkinson’s disease: Results from a participant survey. Complement Ther Med; 70. Epub ahead of print 1 November 2022. [CrossRef]
- Alberts JL, Rosenfeldt AB, Lopez-Lennon C, et al. Effectiveness of a Long-Term, Home-Based Aerobic Exercise Intervention on Slowing the Progression of Parkinson Disease: Design of the Cyclical Lower Extremity Exercise for Parkinson Disease II (CYCLE-II) Study. Phys Ther; 101. Epub ahead of print 1 November 2021. [CrossRef]
- Makahleh HY, Taamneh MM, Dissanayake D. Promoting Sustainable Transport: A Systematic Review of Walking and Cycling Adoption Using the COM-B Model. Future Transportation 2025; 5: 79.
- Foley L, O’Mahony S, Ryan-Fogarty Y, et al. Cycling among people experiencing socio-economic disadvantage: a scoping review protocol [version 1; peer review: 2 approved]. HRB Open Res; 8. Epub ahead of print 1 January 2025. [CrossRef]
- Logan G, Somers C, Baker G, et al. Benefits, risks, barriers, and facilitators to cycling: a narrative review. Front Sports Act Living 2023; 5: 1168357.
- EUR-Lex - 32024C02377 - EN - EUR-Lex, https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=OJ%3AC_202402377 (accessed 26 February 2026).
- Huang B, Wüst H, de Haas M. Assessing the E-bike trends and impact on sustainable mobility: A national-level study in the Netherlands. Journal of Cycling and Micromobility Research 2024; 2: 100027.
- Snijders AH, Toni I, Ružička E, et al. Bicycling breaks the ice for freezers of gait. Mov Disord 2011; 26: 367–371.
- Licen T, Rakusa M, Bohnen NI, et al. Brain Dynamics Underlying Preserved Cycling Ability in Patients With Parkinson’s Disease and Freezing of Gait. Front Psychol 2022; 13: 847703.
- Storzer L, Butz M, Hirschmann J, et al. Bicycling suppresses abnormal beta synchrony in the Parkinsonian basal ganglia. Ann Neurol 2017; 82: 592–601.
- Bougou V, Vanhoyland M, Decramer T, et al. Active and Passive Cycling Decrease Subthalamic β Oscillations in Parkinson’s Disease. Mov Disord 2024; 39: 85–93.
- Bohnen NI, Jahn K. Imaging: what can it tell us about parkinsonian gait? Mov Disord 2013; 28: 1492.
- Engel AK, Fries P. Beta-band oscillations — signalling the status quo? Curr Opin Neurobiol 2010; 20: 156–165.
- Nieuwboer A. Cueing for freezing of gait in patients with Parkinson’s disease: A rehabilitation perspective. Movement Disorders 2008; 23: S475–S481.
- Zoetewei D, Herman T, Ginis P, et al. On-Demand Cueing for Freezing of Gait in Parkinson’s Disease: A Randomized Controlled Trial. Movement Disorders 2024; 39: 876–886.
- Canning CG. The effect of directing attention during walking under dual-task conditions in Parkinson’s disease. Parkinsonism Relat Disord 2005; 11: 95–99.
- Rutz DG, Benninger DH. Physical Therapy for Freezing of Gait and Gait Impairments in Parkinson Disease: A Systematic Review. PM R 2020; 12: 1140–1156.
- Cosentino C, Putzolu M, Mezzarobba S, et al. One cue does not fit all: A systematic review with meta-analysis of the effectiveness of cueing on freezing of gait in Parkinson’s disease. Neurosci Biobehav Rev; 150. Epub ahead of print 1 July 2023. [CrossRef]
- Dubuc R, Cabelguen JM, Ryczko D. Locomotor pattern generation and descending control: a historical perspective. 2023; 130: 401–416. [CrossRef]
- Wolpert DM, Landy MS. Motor control is decision-making. Curr Opin Neurobiol 2012; 22: 996–1003.
- Ijspeert AJ, Daley MA. Integration of feedforward and feedback control in the neuromechanics of vertebrate locomotion: a review of experimental, simulation and robotic studies. Journal of Experimental Biology; 226. Epub ahead of print 1 August 2023. [CrossRef]
- Gilat M, Hall JM, Ehgoetz Martens KA, et al. Staircase climbing is not solely a visual compensation strategy to alleviate freezing of gait in Parkinson’s disease. J Neurol 2017; 264: 174–176.
- Ehgoetz Martens KA, Pieruccini-Faria F, Almeida QJ. Could Sensory Mechanisms Be a Core Factor That Underlies Freezing of Gait in Parkinson’s Disease? PLoS One 2013; 8: e62602.
- Keijsers NLW, Admiraal MA, Cools AR, et al. Differential progression of proprioceptive and visual information processing deficits in Parkinson’s disease. European Journal of Neuroscience 2005; 21: 239–248.
- Alshehri MA, Alzahrani H, van den Hoorn W, et al. Trunk postural control during unstable sitting among individuals with and without low back pain: A systematic review with an individual participant data meta-analysis. PLoS One 2024; 19: e0296968.
- Reeves NP, Sal y Rosas Celi VG, Ramadan A, et al. Quantifying trunk neuromuscular control using seated balancing and stability threshold. J Biomech 2020; 112: 110038.
- Nguyen KP, Person AL. Cerebellar circuit computations for predictive motor control. Nature Reviews Neuroscience 2025 26:9 2025; 26: 538–553.
- Maschke M, Gomez CM, Tuite PJ, et al. Dysfunction of the basal ganglia, but not the cerebellum, impairs kinaesthesia. Brain 2003; 126: 2312–2322.
- Arber S, Costa RM. Networking brainstem and basal ganglia circuits for movement. Nature Reviews Neuroscience 2022 23:6 2022; 23: 342–360.
- Carpenter MG, Allum JHJ, Honegger F, et al. Postural abnormalities to multidirectional stance perturbations in Parkinson’s disease. J Neurol Neurosurg Psychiatry 2004; 75: 1245–1254.
- Bekkers EMJ, Dijkstra BW, Heremans E, et al. Balancing between the two: Are freezing of gait and postural instability in Parkinson’s disease connected? Neurosci Biobehav Rev 2018; 94: 113–125.
- Jacobs J V., Nutt JG, Carlson-Kuhta P, et al. Knee trembling during freezing of gait represents multiple anticipatory postural adjustments. Exp Neurol 2009; 215: 334–341.
- Rocchi L, Chiari L, Mancini M, et al. Step initiation in Parkinson’s disease: Influence of initial stance conditions. Neurosci Lett 2006; 406: 128–132.
- Salamci M, Guclu-Gunduz A. Anticipatory postural adjustments during gait initiation in people with parkinson’s disease: a systematic review. Acta Neurol Belg. Epub ahead of print 2025. [CrossRef]
- Kang S, Park JW, Kim Y, et al. Biomechanical analysis of patients with mild Parkinson’s disease during indoor cycling training. J Neuroeng Rehabil; 22. Epub ahead of print 1 December 2025. [CrossRef]
- D’Cruz N, Seuthe J, De Somer C, et al. Dual Task Turning in Place: A Reliable, Valid, and Responsive Outcome Measure of Freezing of Gait. Mov Disord 2022; 37: 269–278.
- Goris M, Ginis P, Hansen C, et al. Is the freezing index a valid outcome to assess freezing of gait during turning in Parkinson’s disease? Front Neurol 2024; 15: 1508800.
- Zoetewei D, Ginis P, Goris M, et al. Which Gait Tasks Produce Reliable Outcome Measures of Freezing of Gait in Parkinson’s Disease? J Parkinsons Dis 2024; 14: 1163–1174.
- Cefis M, Chaney R, Wirtz J, et al. Molecular mechanisms underlying physical exercise-induced brain BDNF overproduction. Front Mol Neurosci 2023; 16: 1275924.
- Mahalakshmi B, Maurya N, Lee S Da, et al. Possible Neuroprotective Mechanisms of Physical Exercise in Neurodegeneration. Int J Mol Sci 2020; 21: 1–17.
- Mansoor M, Ibrahim A, Hamide A, et al. Exercise-Induced Neuroplasticity: Adaptive Mechanisms and Preventive Potential in Neurodegenerative Disorders. Physiologia 2025, Vol 5, Page 13 2025; 5: 13.
- Petzinger GM, Fisher BE, McEwen S, et al. Exercise-enhanced Neuroplasticity Targeting Motor and Cognitive Circuitry in Parkinson’s Disease. Lancet neurology 2013; 12: 716.
- Ellis T, Rochester L. Mobilizing Parkinson’s Disease: The Future of Exercise. J Parkinsons Dis 2018; 8: S95–S100.
- Langeskov-Christensen M, Franzén E, Hvid LG, et al. Exercise as medicine in Parkinson’s disease Movement disorders. J Neurol Neurosurg Psychiatry 2024; 95: 1077–1088.
- Kaagman DGM, van Wegen EEH, Cignetti N, et al. Effects and Mechanisms of Exercise on Brain-Derived Neurotrophic Factor (BDNF) Levels and Clinical Outcomes in People with Parkinson’s Disease: A Systematic Review and Meta-Analysis. Brain Sci 2024; 14: 194.
- Patterson CG, Joslin E, Gil AB, et al. Study in Parkinson’s disease of exercise phase 3 (SPARX3): study protocol for a randomized controlled trial. Trials 2022; 23: 855.
- van der Kolk NM, de Vries NM, Kessels RPC, et al. Effectiveness of home-based and remotely supervised aerobic exercise in Parkinson’s disease: a double-blind, randomised controlled trial. Lancet Neurol 2019; 18: 998–1008.
- Schootemeijer S, van der Kolk NM, Bloem BR, et al. Current Perspectives on Aerobic Exercise in People with Parkinson’s Disease. Neurotherapeutics 2020; 17: 1418–1433.
- Alberts JL, Rosenfeldt AB. The Universal Prescription for Parkinson’s Disease: Exercise. J Parkinsons Dis 2020; 10: S21–S27.
- Tiihonen M, Westner BU, Butz M, et al. Parkinson’s disease patients benefit from bicycling - a systematic review and meta-analysis. NPJ Parkinsons Dis; 7. Epub ahead of print 1 December 2021. [CrossRef]
- Schootemeijer S, Darweesh SKL, De Vries NM. Clinical Trial Highlights - Aerobic Exercise for Parkinson’s Disease. J Parkinsons Dis 2022; 12: 2297–2306.
- Goetz CG, Tilley BC, Shaftman SR, et al. Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord 2008; 23: 2129–2170.
- Rosenfeldt AB, Lopez-Lennon C, Suttman E, et al. Use of a Home-Based, Commercial Exercise Platform to Remotely Monitor Aerobic Exercise Adherence and Intensity in People With Parkinson Disease. Phys Ther 2024; 104: 1–11.
- Palmieri JL, Jones L, Schenkman M, et al. Bicycling for Rehabilitation of Persons With Parkinson Disease: A Scoping Review. J Neurol Phys Ther 2024; 48: 125–139.
- O’neal SK, Miller SA, Eikenberry MC, et al. A BACKWARD CYCLING PROGRAMME FOR PEOPLE WITH PARKINSON’S DISEASE: A FEASIBILITY AND PRELIMINARY RESULTS STUDY. J Rehabil Med 2024; 56: 17738.
- Warburton DER, Nicol CW, Bredin SSD. Health benefits of physical activity: the evidence. CMAJ : Canadian Medical Association Journal 2006; 174: 801.
- Tsukita K, Sakamaki-Tsukita H, Takahashi R. Long-term Effect of Regular Physical Activity and Exercise Habits in Patients With Early Parkinson Disease. Neurology 2022; 98: e859.
- Paul SS, Allen NE, Sherrington C, et al. Risk Factors for Frequent Falls in People with Parkinson’s Disease. J Parkinsons Dis 2014; 4: 699–703.
- Oguh O, Eisenstein A, Kwasny M, et al. Back to the basics: Regular exercise matters in Parkinson’s disease: Results from the National Parkinson Foundation QII Registry study. Parkinsonism Relat Disord 2014; 20: 1221–1225.
- Wallén MB, Franzén E, Nero H, et al. Levels and Patterns of Physical Activity and Sedentary Behavior in Elderly People With Mild to Moderate Parkinson Disease. Phys Ther 2015; 95: 1135–1141.
- Nimwegen M Van, Speelman AD, Hofman-Van Rossum EJM, et al. Physical inactivity in Parkinson’s disease. J Neurol 2011; 258: 2214.
- Vaugoyeau M, Viallet F, Aurenty R, et al. Axial rotation in Parkinson’s disease. J Neurol Neurosurg Psychiatry 2006; 77: 815–821.
- Korkmaz B, Yaşa ME, Sonkaya R. Upper extremity functions, spinal posture, and axial rigidity in patients with parkinson’s disease. Acta Neurol Belg. Epub ahead of print 2024. [CrossRef]
- Maarten Afschrift •, Matthijs A, De Ryck T, et al. Turning the head while biking makes older people lose cycling direction and balance. bioRxiv 2022; 2022.03.01.481993.
- Park J-H, Kang Y-J, Horak FB. What Is Wrong with Balance in Parkinson’s Disease? J Mov Disord 2015; 8: 109.
- Snijders AH, Toni I, Ružička E, et al. Bicycling breaks the ice for freezers of gait. Movement Disorders 2011; 26: 367–371.
- Gebhardt A, Vanbellingen T, Baronti F, et al. Poor dopaminergic response of impaired dexterity in Parkinson’s disease: Bradykinesia or limb kinetic apraxia? Movement Disorders 2008; 23: 1701–1706.
- Horak FB, Dimitrova D, Nutt JG. Direction-specific postural instability in subjects with Parkinson’s disease. Exp Neurol 2005; 193: 504–521.
- García-Navarra S, Llana T, Méndez M. Spatial memory deficits in Parkinson’s disease: neural mechanisms and assessment. Am J Neurodegener Dis 2025; 14: 67.
- Burgos PI, Silva-Batista C, Ragothaman A, et al. Cognition is associated with daily-life mobility in people with Parkinson’s disease. Clin Park Relat Disord 2025; 13: 100393.
- Aarsland D, Batzu L, Halliday GM, et al. Parkinson disease-associated cognitive impairment. Nature Reviews Disease Primers 2021 7:1 2021; 7: 1–21.
- Micca L, Albouy G, King BR, et al. The Effect of a Post-Learning Nap on Motor Memory Consolidation in People With Parkinson’s Disease: A Randomised Controlled Trial. J Sleep Res 2025; e70203.
- Van Biesen D, Pineda RC, Scheerder J, et al. Het Vlaamse G-sportlandschap anno 2024. 2024.
- Schootemeijer S, Van Der Kolk NM, Ellis T, et al. Barriers and Motivators to Engage in Exercise for Persons with Parkinson’s Disease. 2020; 10: 1293–1299. [CrossRef]
- Kim M, Kim E, Kim M, et al. Motivators and Barriers Affecting Exercise in Patients With Parkinson’s Disease. Journal of Clinical Neurology 2025; 21: 13–20.
- Mak MK, Wong-Yu IS, Shen X, et al. Exploring behavior, motivation, and barriers to exercise in Parkinson disease. Clin Park Relat Disord 2025; 13: 100362.
- Kovácsová N, de Winter JCF, Schwab AL, et al. Riding performance on a conventional bicycle and a pedelec in low speed exercises: Objective and subjective evaluation of middle-aged and older persons. Transp Res Part F Traffic Psychol Behav 2016; 42: 28–43.
- Maurissens T, Orban De Xivry J-J, De Groote F, et al. MaestroPD: Sensorimotor assessment of (e-)cycling in people with Parkinson’s disease. Epub ahead of print 7 August 2025. [CrossRef]

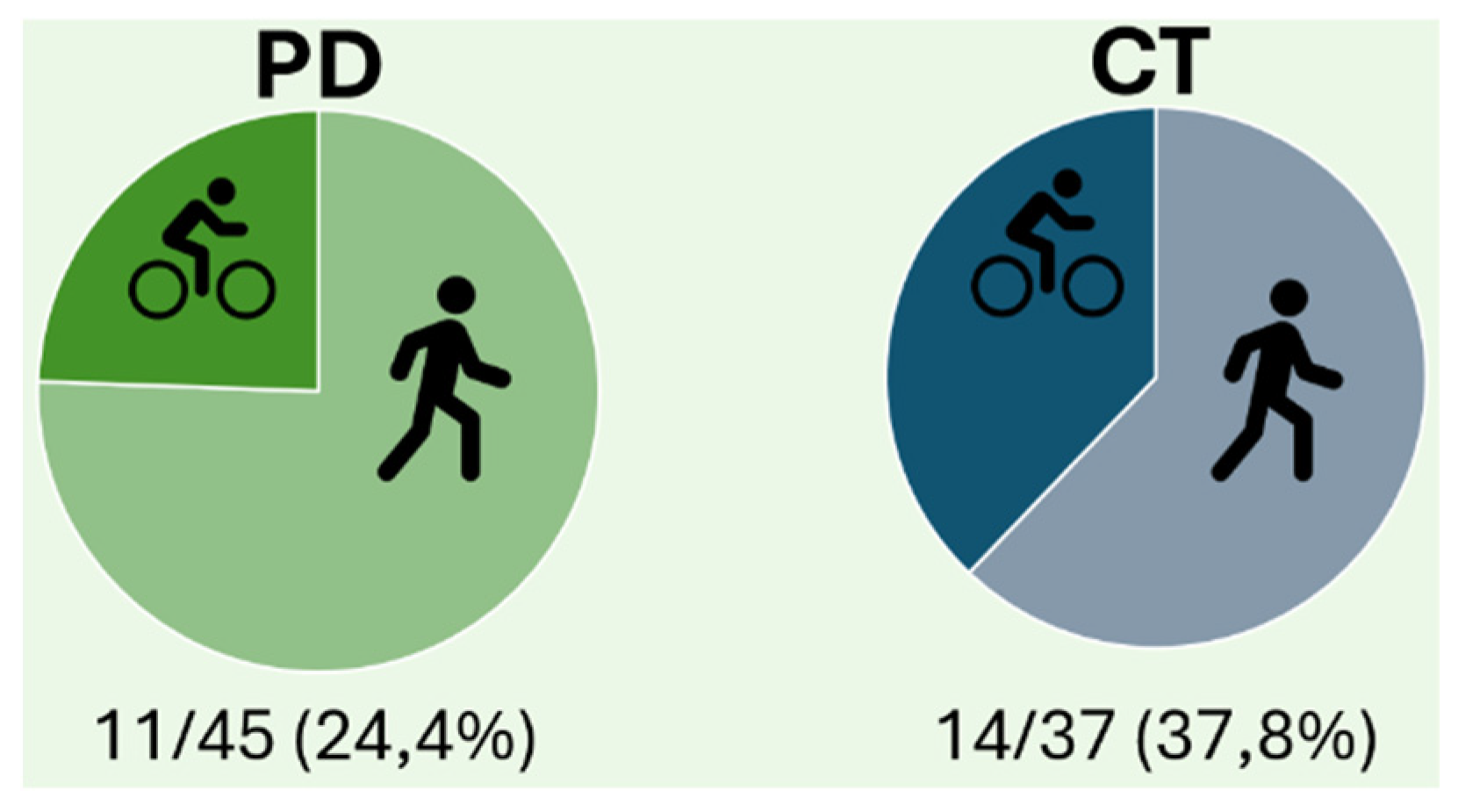
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



