Submitted:
27 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Controlled Ovarian Stimulation Protocol
2.3. Fertilization
2.4. Embryo Culture and Assessment
2.5. Embryo Transfer
2.6. Outcome Measurements
2.7. Ethical Consideration
2.8. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AFC | Antral Follicle Count |
| AMH | Anti-Mullerian Hormone |
| BMI | Body mass index |
| CI | Conventional Incubator |
| FSH | Follicle Stimulating Hormone |
| IVF | In Vitro Fertilization |
| LH | Luteinizing Hormone |
| MII | Metaphase II |
| TL | Time Lapse |
| TLT | Time Lapse Technology |
References
- Organization WH. Infertility. 2023.
- Organization WH. Infertility prevalence estimates, 1990–2021. 2023.
- Carson, S.A.; Kallen, A.N. Diagnosis and Management of Infertility: A Review. JAMA 2021, 326, 65-76.
- Ferraretti, A.P.; Nygren, K.; Andersen, A.N.; de Mouzon, J.; Kupka, M.; Calhaz-Jorge, C.; Wyns, C.; Gianaroli, L.; Goossens, V. Trends over 15 years in ART in Europe: an analysis of 6 million cycles. Hum Reprod Open 2017, hox012. [CrossRef]
- Dyer, S.; Chambers, G.M.; Jwa, S.C.; Baker, V.L.; Banker, M.; de Mouzon, J.; Elgindy, E.; Fu, B.; Ishihara, O.; Kupka, M.S.; Zegers-Hochschild, F.; Adamson, G.D. International Committee for Monitoring Assisted Reproductive Technologies world report: assisted reproductive technology. Hum Reprod 2021, 36, 2921-2934.
- Lacconi, V.; Massimiani, M.; Carriero, I.; Bianco, C.; Ticconi, C.; Pavone, V.; Alteri, A.; Muzii, L.; Rago, R.; Pisaturo, V.; Campagnolo, L. When the Embryo Meets the Endometrium: Identifying the Features Required for Successful Embryo Implantation. Int J Mol Sci 2024, 25. [CrossRef]
- Gimenez, C.; Conversa, L.; Murria, L.; Meseguer, M. Time-lapse imaging: Morphokinetic analysis of in vitro fertilization outcomes. Fertil Steril 2023, 120, 218-227. [CrossRef]
- Apter, S.; Ebner, T.; Freour, T.; Guns, Y.; Kovacic, B. Good practice recommendations for the use of time-lapse technology. Hum Reprod Open 2020, 2020, hoaa008. [CrossRef]
- Reignier, A.; Lefebvre, T.; Loubersac, S.; Lammers, J.; Barriere, P.; Freour, T. Time-lapse technology improves total cumulative live birth rate and shortens time to live birth as compared to conventional incubation system in couples undergoing ICSI. J Assist Reprod Genet 2021, 38, 917-923. [CrossRef]
- Kieslinger, D.C.V.C.; Ramos, L.; Arends, B.; Curfs, M.H.J.M.; Slappendel, E. Clinical outcomes of uninterrupted embryo culture with or without time-lapse-based embryo selection versus interrupted standard culture (SelecTIMO): a three-armed, multicentre, double-blind, randomised controlled trial. Lancet 2023, 401, 1438-1446. [CrossRef]
- Zhang, X.D.; Zhang, Q.; Han, W.; Liu, W.W.; Shen, X.L.; Yao, G.D.; Shi, S.L.; Hu, L.L.; Wang, S.S.; Wang, J.X.; Zhou, J.J.; Kang, W.W.; Zhang, H.D.; Luo, C.; Yu, Q.; Liu, R.Z.; Sun, Y.P.; Sun, H.X.; Wang, X.H.; Quan, S.; Huang, G.N. Comparison of embryo implantation potential between time-lapse incubators and standard incubators: A randomized controlled study. Reprod Biomed Online 2022, 45, 858-866. [CrossRef]
- Jiang, Y.W.L.; Wang, S.; Shen, H.; Wang, B.; Zheng, J. The effect of embryo selection using time-lapse monitoring on IVF/ICSI outcomes: A systematic review and meta-analysis. J Obstet Gynaecol Res 2023, 49. [CrossRef]
- Bergh, C.; Lundin, K. No benefit to pregnancy or livebirth by time-lapse-based embryo selection in IVF. Lancet 2023, 401, 1401-1403. [CrossRef]
- Cheon, K.W.; Song, S.J.; Choi, B.C.; Lee, S.C.; Lee, H.B.; Yu, S.Y.; Yoo, K.J. Comparison of clinical efficacy between a single administration of long-acting gonadotrophin-releasing hormone agonist (GnRHa) and daily administrations of short-acting GnRHa in in vitro fertilization-embryo transfer cycles. J Korean Med Sci 2008, 23, 662-666. [CrossRef]
- Sahin, G.N.; Yildirim, R.M.; Seli, E. Embryonic arrest: causes and implications. Current Opinion in Obstetrics and Gynecology 2023, 35, 184-192. [CrossRef]
- Elmerdahl Frederiksen, L.; Olgaard, S.M.; Roos, L.; Petersen, O.B.; Rode, L.; Hartwig, T. Maternal age and the risk of fetal aneuploidy: A nationwide cohort study of more than 500 000 singleton pregnancies in Denmark from 2008 to 2017. Acta Obstet Gynecol Scand 2024, 103, 351-359. [CrossRef]
- Chera-Aree, P.; Thanaboonyawat, I.; Thokha, B.; Laokirkkiat, P. Comparison of pregnancy outcomes using a time-lapse monitoring system for embryo incubation versus a conventional incubator in in vitro fertilization: An age-stratification analysis. Clin Exp Reprod Med 2021, 48, 174-183. [CrossRef]
- Franasiak, J.M.; Forman, E.J.; Hong, K.H.; Werner, M.D.; Upham, K.M.; Treff, N.R.; Scott, R.T. Jr. The nature of aneuploidy with increasing age of the female partner: a review of 15,169 consecutive trophectoderm biopsies evaluated with comprehensive chromosomal screening. Fertil Steril 2014, 101, 656-663. [CrossRef]
- Siristatidis, C.; Komitopoulou, M.A.; Makris, A.; Sialakouma, A.; Botzaki, M.; Mastorakos, G.; Salamalekis, G.; Bettocchi, S.; Palmer, G.A. Morphokinetic parameters of early embryo development via time lapse monitoring and their effect on embryo selection and ICSI outcomes: a prospective cohort study. J Assist Reprod Genet 2015, 32, 563-570. [CrossRef]
- McQueen, D.B.; Mazur, J.; Kimelman, D.; Confino, R.; Robins, J.C.; Bernardi, L.A.; Yeh, C.; Zhang, J.; Pavone, M.E. Can embryo morphokinetic parameters predict euploid pregnancy loss? Fertil Steril 2021, 115, 382-388. [CrossRef]
- Coticchio, G.; Pennetta, F.; Rizzo, R.; Tarozzi, N.; Nadalini, M.; Orlando, G.; Centonze, C.; Gioacchini, G.; Borini, A. Embryo morphokinetic score is associated with biomarkers of developmental competence and implantation. J Assist Reprod Genet 2021, 38, 1737-1743. [CrossRef]
- Armstrong, S.; Bhide, P.; Jordan, V.; Pacey, A.; Farquhar, C. Time-lapse systems for embryo incubation and assessment in assisted reproduction. Cochrane Database Syst Rev 2018, 5, CD011320.
- Wu, Y.G.; Lazzaroni-Tealdi, E.; Wang, Q.; Zhang, L.; Barad, D.H.; Kushnir, V.A.; Darmon, S.K.; Albertini, D.F.; Gleicher, N. Different effectiveness of closed embryo culture system with time-lapse imaging (EmbryoScope(TM)) in comparison to standard manual embryology in good and poor prognosis patients: a prospectively randomized pilot study. Reprod Biol Endocrinol 2016, 14, 49-54. [CrossRef]
- Nakahara, T.; Iwase, A.; Goto, M.; Harata, T.; Suzuki, M.; Ienaga, M.; Kobayashi, H.; Takikawa, S.; Manabe, S.; Kikkawa, F.; Ando, H. Evaluation of the safety of time-lapse observations for human embryos. J Assist Reprod Genet 2020, 27, 93-96. [CrossRef]
- Glujovsky, D.; Quinteiro Retamar, A.M.; Alvarez Sedo, C.R.; Ciapponi, A.; Cornelisse, S.; Blake, D. Cleavage-stage versus blastocyst-stage embryo transfer in assisted reproductive technology. Cochrane Database Syst Rev 2022, 5, CD002118. [CrossRef]
- Fernandez-Shaw, S.; Cercas, R.; Brana, C.; Villas, C.; Pons, I.O. Ongoing and cumulative pregnancy rate after cleavage-stage versus blastocyst-stage embryo transfer using vitrification for cryopreservation: Impact of age on the results. J Assist Reprod Genet 2015, 32, 177-184. [CrossRef]
- Giladi Yacobi, E.; Miller, N.; Goren Gepstein, N.; Mashiach, J.; Haikin Herzberger, E.; Levi, M.; Ghetler, Y.; Wiser, A. Does cleavage- versus blastocyst-stage embryo transfer improve fertility rates in women over 38 years of age undergoing assisted reproductive technology? Int J Gynaecol Obstet 2024, 167, 1017-1022. [CrossRef]
- Guo, Y.H.; Liu, Y.; Qi, L.; Song, W.Y.; Jin, H.X. can time-lapse incubation and monitoring be beneficial to assisted reproduction technology outcomes? A randomized controlled trial using day 3 double embryo transfer. Front Physiol 2021, 12, 794601. [CrossRef]
- Bhide, P.; Chan, D.Y.L.; Lanz, D.; Alqawasmeh, O.; Barry, E.; Baxter, D.; Gonzalez Carreras, F.; Choudhury, Y.; Cheong, Y.; Chung, J.P.W.; Collins, B.; Cong, L.; Doidge, S.; Heighway, J.; Patel, D.; Pardo, M.C.; Rattos, A.; Wright, A.; Dodds, J.; Perez, T.; Khan, K.S.; Thangaratinam, S. Clinical effectiveness and safety of time-lapse imaging systems for embryo incubation and selection in in-vitro fertilisation treatment (TILT): A multicentre, three-parallel-group, double-blind, randomised controlled trial. Lancet 2024, 404, 256-265. [CrossRef]
- Adamson, G.D.; Abusief, M.E.; Palao, L.; Witmer, J.; Palao, L.M.; Gvakharia, M. Improved implantation rates of day 3 embryo transfers with the use of an automated time-lapse-enabled test to aid in embryo selection. Fertil Steril 2016, 105, 369-375. [CrossRef]
- Goodman, L.R.; Goldberg, J.; Falcone, T.; Austin, C.; Desai, N. Does the addition of time-lapse morphokinetics in the selection of embryos for transfer improve pregnancy rates? A randomized controlled trial. Fertil Steril 2016, 105, 275-285. [CrossRef]
- Wang, X.; Wei, Q.; Huang, W.; Yin, L.; Ma, T. Can time-lapse culture combined with artificial intelligence improve ongoing pregnancy rates in fresh transfer cycles of single cleavage stage embryos? Front Endocrinol (Lausanne) 2024, 15, 1449035. [CrossRef]
| Variables | Total (n=400) | CI group (n=300) | TL group (n=100) | p-Value | |||
|---|---|---|---|---|---|---|---|
| Age (yr)* | 34.62±5.39 | 34.82±5.53 | 34.02±4.92 | 0.17 | |||
| Infertility duration (months)* | 65.12±43.36 | 63.85±43.25 | 69.00±43.71 | 0.34 | |||
| BMI (kg/m2)* | 24.34±3.83 | 24.42±3.94 | 24.13±3.53 | 0.50 | |||
| Causes of infertility** | |||||||
| Female | 135 (33.8) | 106 (35.3) | 29 (29.0) | 0.10 | |||
| Male | 176 (44.0) | 133 (44.4) | 43 (43.0) | ||||
| Combined | 66 (16.5) | 42 (14.0) | 24 (24.0) | ||||
| Idiopathic | 23 (5.7) | 19 (6.3) | 4 (4.0) | ||||
| Ovarian reserve markers | |||||||
| AFC* | 7.66±4.39 | 7.39±4.46 | 8.38±3.85 | 0.06 | |||
| AMH (ng/ml)* | 1.90±1.49 | 1.92±1.49 | 2.12±1.39 | 0.11 | |||
| FSH (mIU/ml)* | 7.88±3.30 | 8.22±3.28 | 7.74±3.30 | 0.22 | |||
| Sperm preparation** | |||||||
| Ejaculated | 355 (88.7) | 272 (90.7) | 83 (83.0) | 0.06 | |||
| Testicular | 45 (11.3) | 28 (9.3) | 17 (17.0) | ||||
| Hormone’s level on trigger day | |||||||
| Estrogen (pg/ml)* | 2042.84±1098.81 | 2049.94±1114.65 | 2008.69±1005.47 | 0.56 | |||
| LH (mIU/ml)* | 4.64±4.17 | 4.51±4.25 | 5.04±3.91 | 0.26 | |||
| Progesterone (ng/ml)* | 1.10±0.86 | 1.13±1.03 | 0.98±0.41 | 0.05 | |||
| Variables | Total (n=400) | CI groups (n=300) | TL groups (n=100) | p-Value |
|---|---|---|---|---|
| Duration of stimulation (days) | 14.29±1.91 | 14.33±1.98 | 14.18±1.65 | 0.45 |
| Total dose of gonadotropin (IU) | 2030.30±792.10 | 2040.83±780.14 | 2288.87±773.01 | 0.08 |
| Number of retrieved oocytes | 8.92±6.37 | 8.62±6.72 | 9.82±5.09 | 0.06 |
| Number of MII oocytes | 7.30±5.38 | 7.09±5.68 | 7.90±4.34 | 0.35 |
| Maturation rate (%) | 81.13±20.45 | 81.73±21.06 | 79.32±18.48 | 0.23 |
| Fertilization rate (%) | 91.99±18.68 | 91.52±15.90 | 85.52±23.71 | 0.12 |
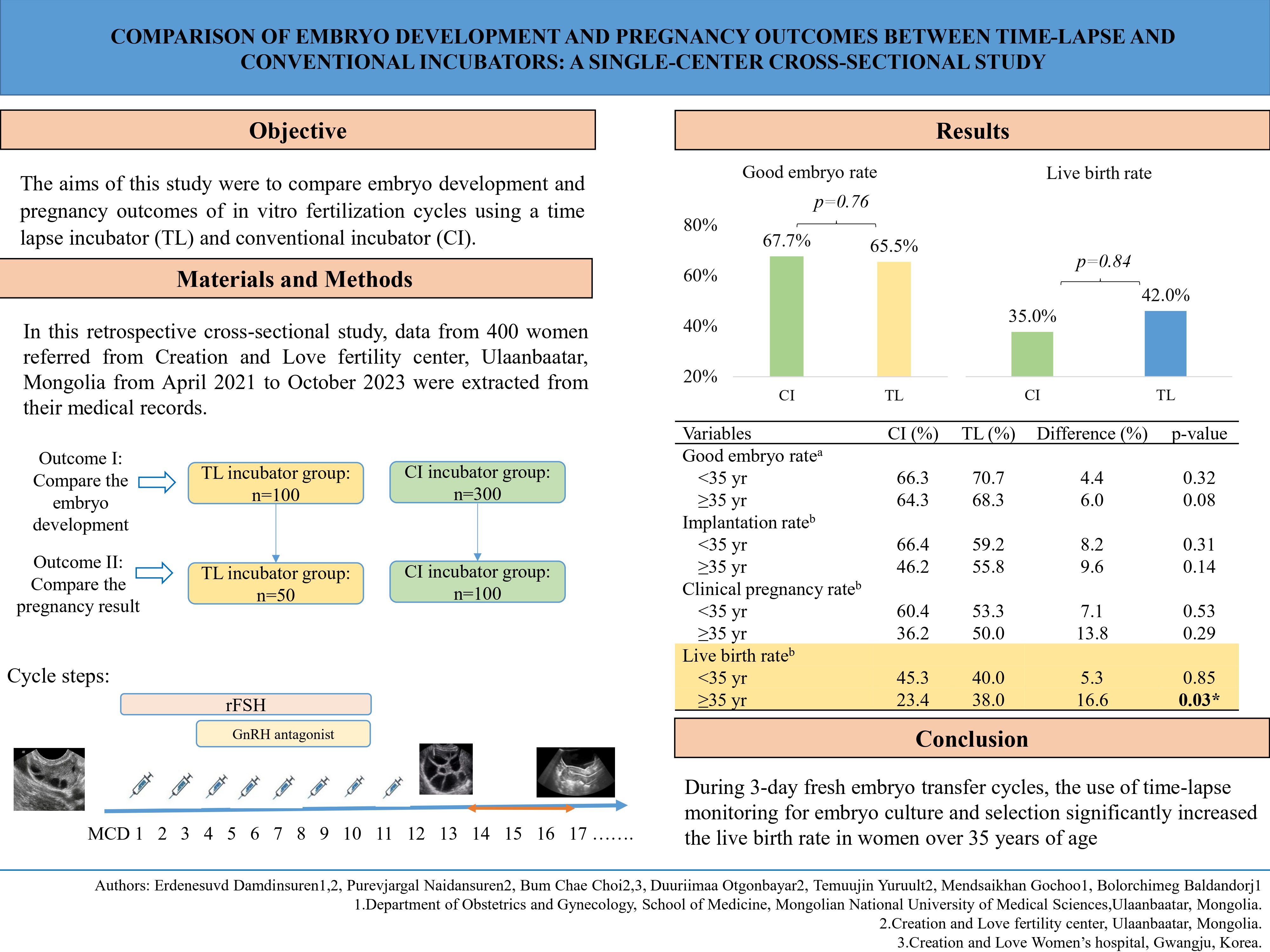
| Good embryo rate (%) | 65.85±28.69 | 67.65±27.84 | 65.47±30.59 | 0.84 |
| Variables | Total (n=150) |
CI group (n=100) | TL group (n=50) | p-Value | |
|---|---|---|---|---|---|
| Age (yr)* | 34.11±4.60 | 34.06±4.57 | 33.62±4.67 | 0.36 | |
| Age of husband (yr)* | 35.02±5.78 | 35.81±5.18 | 34.58±6.17 | 0.45 | |
| BMI (kg/m2)* | 24.72±3.81 | 24.69±3.98 | 24.78±3.52 | 0.90 | |
| AMH (ng/ml)* | 2.13±1.42 | 1.97±1.38 | 2.41±1.46 | 0.09 | |
| Causes of infertility** | |||||
| Female | 48 (32.0) | 30 (30.0) | 18 (36.0) | 0.46 | |
| Male | 67 (44.7) | 44 (44.0) | 23 (46.0) | ||
| Combined | 20 (13.3) | 14 (14.0) | 6 (12.0) | ||
| Idiopathic | 15 (10.0) | 12 (12.0) | 3 (6.0) | ||
| Sperm preparation** | |||||
| Ejaculated | 119 (79.3) | 78 (78.0) | 41 (82.0) | 0.32 | |
| Testicular | 31 (20.7) | 22 (22.0) | 9 (18.0) | ||
| Progesterone level on trigger day (ng/ml)* | 0.87±0.50 | 0.91±0.57 | 0.79±0.34 | 0.11 | |
| Endometrial thickness (mm) on trigger day* | 9.93±2.20 | 10.11±2.60 | 9.65±1.37 | 0.20 | |
| Number of transferred embryos*** | |||||
| Single embryo transfer | 12 (8.0) | 9 (9.0) | 3 (6.0) | 0.82 | |
| Double embryo transfer | 71 (47.3) | 48 (48.0) | 23 (46.0) | ||
| Triple embryo transfer | 67 (44.7) | 43 (43.0) | 24 (48.0) | ||
| Grade of transferred embryo** | |||||
| All embryos were grade I | 103 (68.7) | 68 (68.0) | 35 (70.0) | 0.73 | |
| Grade I+one or two grade II embryo/s | 26 (17.3) | 18 (18.0) | 8 (16.0) | ||
| All embryos were grade II | 21 (14.0) | 14 (14.0) | 7 (14.0) | ||
| Implantation rate (%)** | 196/337 (58.2) | 128/219 (58.4) | 68/118 (57.1) | 0.79 | |
| Clinical pregnancy rate (%)** | 75/150 (50.0) | 49/100 (49.0) | 26/50 (52.0) | 0.86 | |
| Live birth rate (%)** | 56/150 (37.3) | 35/100 (35.0) | 21/50 (42.0) | 0.76 | |
| Variables | CI (%) | TL (%) | Difference (%) | p-value |
|---|---|---|---|---|
| Maturation ratea | ||||
| <35 yr | 81.4 | 80.5 | 0.9 | 0.76 |
| ≥35 yr | 81.3 | 80.4 | 0.9 | 0.79 |
| Fertilization ratea | ||||
| <35 yr | 88.6 | 92.4 | 3.8 | 0.15 |
| ≥35 yr | 89.0 | 91.0 | 2.0 | 0.28 |
| Good embryo ratea | ||||
| <35 yr | 66.3 | 70.7 | 4.4 | 0.32 |
| ≥35 yr | 64.3 | 68.3 | 6.0 | 0.08 |
| Implantation rateb | ||||
| <35 yr | 66.4 | 59.2 | 8.2 | 0.31 |
| ≥35 yr | 46.2 | 55.8 | 9.6 | 0.14 |
| Clinical pregnancy rateb | ||||
| <35 yr | 60.4 | 53.3 | 7.1 | 0.53 |
| ≥35 yr | 36.2 | 50.0 | 13.8 | 0.29 |
| Live birth rateb | ||||
| <35 yr | 45.3 | 40.0 | 5.3 | 0.85 |
| ≥35 yr | 23.4 | 38.0 | 16.6 | 0.03 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




