Submitted:
24 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
Introduction
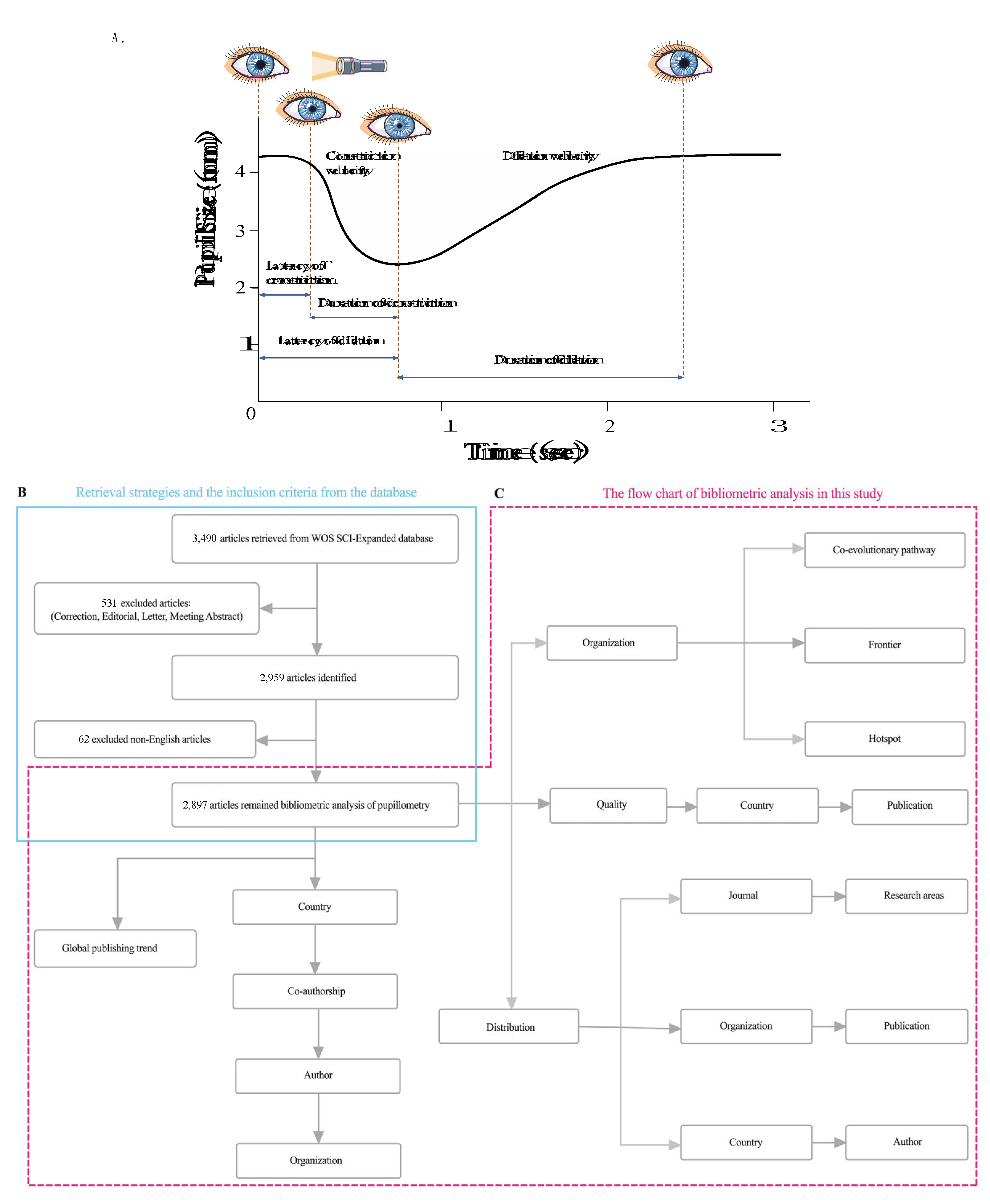
Materials and Methods
Data Source
Search Strategy
Mapping Analysis
Results
Publication Output and Development Trend
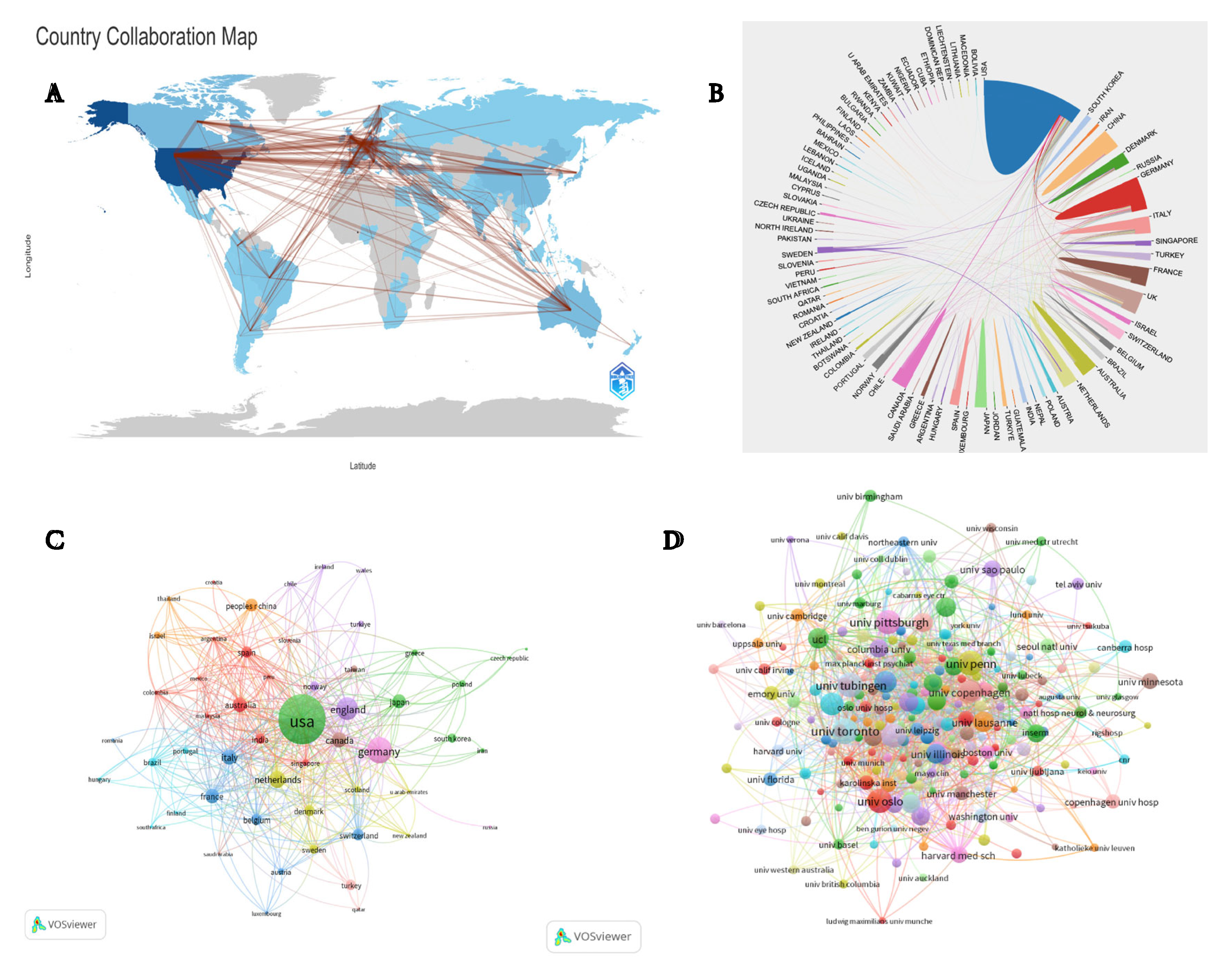
Analysis of Leading Countries/Regions
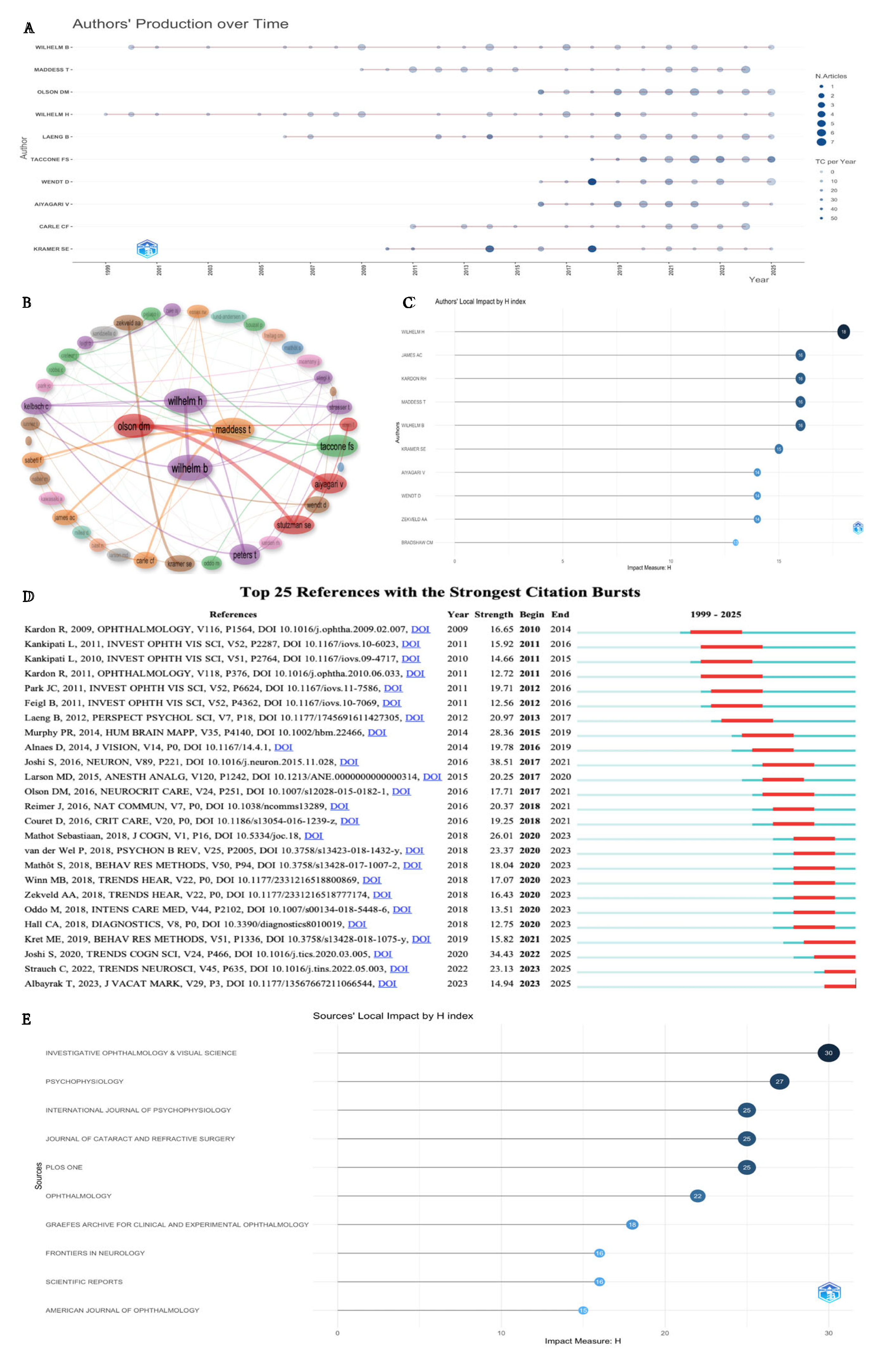
Analysis of Leading Organizations
Distribution of Journals
Distribution of Co-Cited References
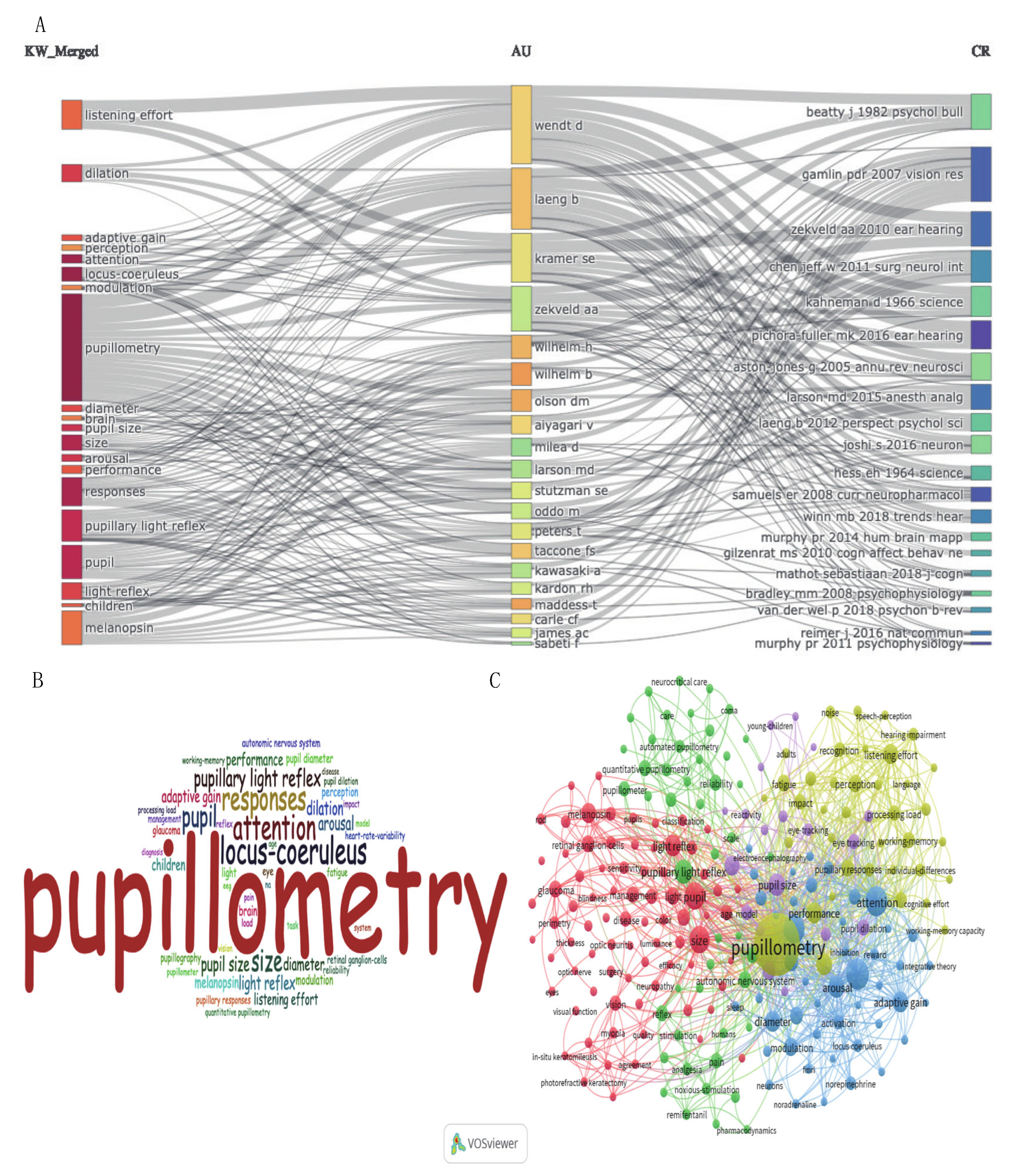
Distribution of Key Words: Hotspots of Pupillometry Study
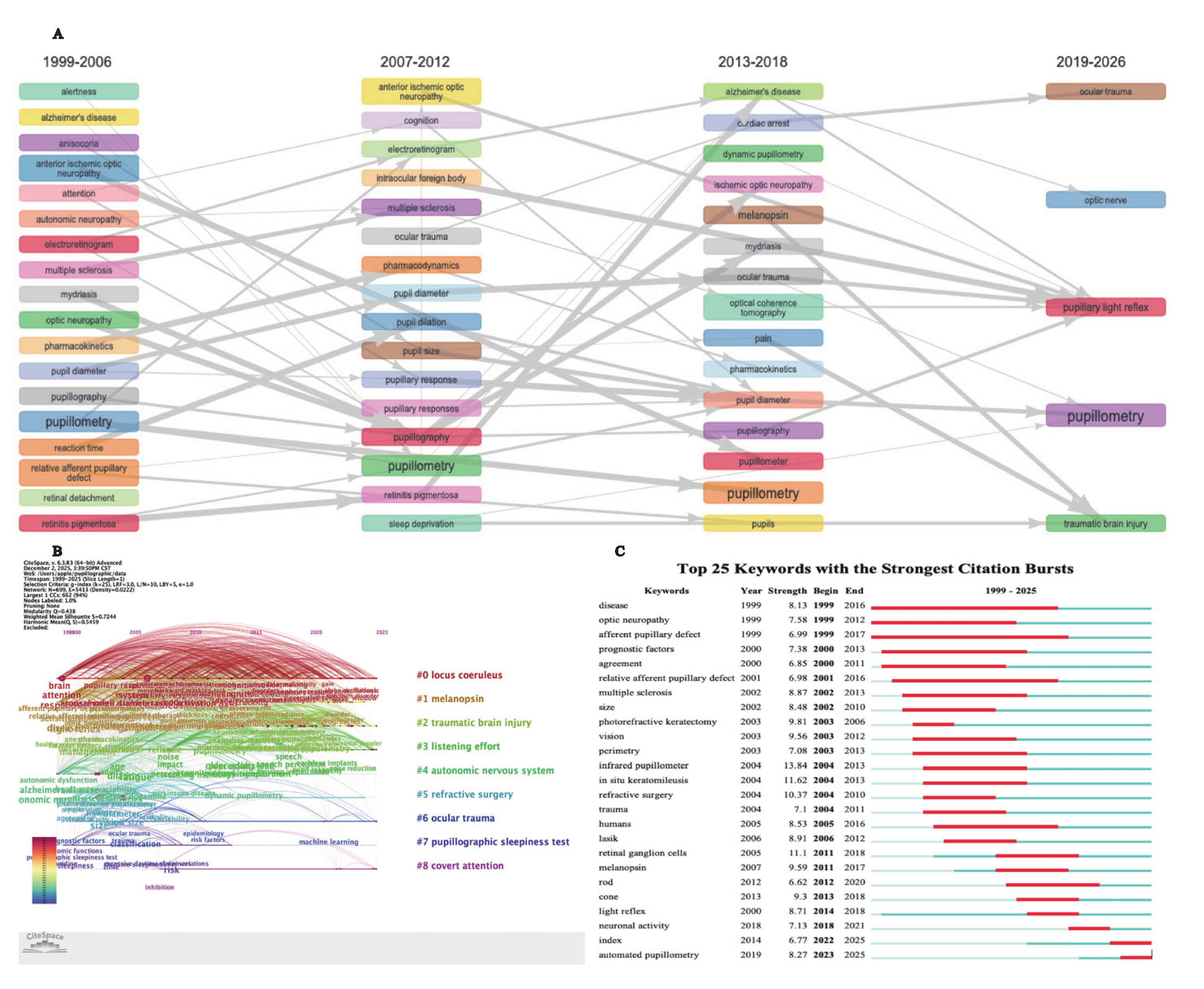
Co-Evolutionary Pathway in the Study of Pupillometry
Discussion
General Information
Clinical Applications of Pupillometry
Ocular Accommodation and Convergence Eye Movements
Relative Afferent Pupillary Defect (RAPD)
Retinal and Macular Diseases
Refractive and Cataract Surgery
Pediatric and Pharmacologic Ophthalmology
Neurological Disorder
Psychological and Psychiatric Disorders
Sleepiness
Anesthesiology
Critical Care and Emergency Medicine
Keyword Analysis of Pupillometry Study
Limitations
Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Ethical Approval
References
- Loewenfeld, IE; Lowenstein, O. The Pupil: Anatomy, Physiology, and Clinical Applications; 1999. [Google Scholar]
- Gonzalez, RC WR. Digital Image Processing, 2nd ed.; Prentice-Hall: NJ, 2002. [Google Scholar]
- DC, B. Neuroophthalmology of the pupil and accommodation. In Principles and Practice of Ophthalmology; Albert, DM, Jakobiec, FA, Eds.; W.B. Saunders: Philadelphia, 1994. [Google Scholar]
- Bremner, FD. Pupil assessment in optic nerve disorders. Eye (Lond) 2004, 18(11), 1175–81. [Google Scholar] [CrossRef]
- Sirois, S; Brisson, J. Pupillometry. Wiley Interdiscip Rev Cogn Sci. 2014, 5(6), 679–92. [Google Scholar] [CrossRef]
- Wilhelm, H; Kardon, RH. The pupillary light reflex pathway; Neuro-Ophthalmology, 1997. [Google Scholar]
- Kardon, RH. Pupil perimetry. Curr Opin Ophthalmol. 1992, 3(5), 565–70. [Google Scholar] [CrossRef]
- Yoshitomi, T; Matsui, T; Tanakadate, A; Ishikawa, S. Comparison of threshold visual perimetry and objective pupil perimetry in clinical patients. J Neuroophthalmol. 1999, 19(2), 89–99. [Google Scholar] [CrossRef] [PubMed]
- Tan, L; Kondo, M; Sato, M; Kondo, N; Miyake, Y. Multifocal pupillary light response fields in normal subjects and patients with visual field defects. Vision Res. 2001, 41(8), 1073–84. [Google Scholar] [CrossRef] [PubMed]
- Barbur, JL; Forsyth, PM. Can the pupil response be used as a measure of the visual input associated with the geniculo-striate pathway? 1986. [Google Scholar]
- Gamlin, PD. The pretectum: connections and oculomotor-related roles. Prog Brain Res. 2006, 151, 379–405. [Google Scholar]
- Tekin, K; Sekeroglu, MA; Kiziltoprak, H; Doguizi, S; Inanc, M; Yilmazbas, P. Static and dynamic pupillometry data of healthy individuals. Clin Exp Optom. 2018, 101(5), 659–65. [Google Scholar] [CrossRef]
- Lowenstein, O; Loewenfeld, IE. Electronic pupillography; a new instrument and some clinical applications. AMA Arch Ophthalmol. 1958, 59(3), 352–63. [Google Scholar] [CrossRef]
- Diamond, JP. The Pupil. Anatomy, Physiology and Clinical Applications. Br J Ophthalmol. 2001, 85(1), 121e. [Google Scholar] [CrossRef]
- Maddess, T; Carle, CF; Kolic, M; Saraç, Ö; Essex, RW; Rohan, EMF; et al. Diagnostic Power and Reproducibility of Objective Perimetry in Glaucoma. J Glaucoma 2024, 33(12), 940–50. [Google Scholar] [CrossRef] [PubMed]
- Bayraktar, S; Hondur, G; Şekeroğlu, MA; Şen, E. Evaluation of Static and Dynamic Pupillary Functions in Early-Stage Primary Open Angle Glaucoma. J Glaucoma 2023, 32(7), e90–e4. [Google Scholar] [CrossRef]
- Bhowmik, S; Arjunan, SP; Sarossy, M; Radcliffe, P; Kumar, DK. Pupillometric recordings to detect glaucoma. Physiol Meas. 2021, 42(4). [Google Scholar] [CrossRef] [PubMed]
- Weisberg, LA. Mesencephalic hemorrhages: clinical and computed tomographic correlations. Neurology 1986, 36(5), 713–6. [Google Scholar] [CrossRef]
- Du, R; Meeker, M; Bacchetti, P; Larson, MD; Holland, MC; Manley, GT. Evaluation of the portable infrared pupillometer. Neurosurgery 2005, 57(1), 198-203; discussion 198-203. [Google Scholar] [CrossRef]
- Larson, MD; Behrends, M. Portable infrared pupillometry: a review. Anesth Analg. 2015, 120(6), 1242–53. [Google Scholar] [CrossRef]
- Solyman, O; Abushanab, MMI; Carey, AR; Henderson, AD. Pilot Study of Smartphone Infrared Pupillography and Pupillometry. Clin Ophthalmol. 2022, 16, 303–10. [Google Scholar] [CrossRef]
- Halperin, A; Pajuelo, M; Tornheim, JA; Vu, N; Carnero, AM; Galdos-Cardenas, G; et al. Pupillary Light Reflexes are Associated with Autonomic Dysfunction in Bolivian Diabetics But Not Chagas Disease Patients. Am J Trop Med Hyg. 2016, 94(6), 1290–8. [Google Scholar] [CrossRef]
- Negi, R; Kalivemula, M; Bisht, K; Bhate, M; Sachdeva, V; Bharadwaj, SR. Diagnostic accuracy of a modularized, virtual-reality-based automated pupillometer for detection of relative afferent pupillary defect in unilateral optic neuropathies. Front Ophthalmol (Lausanne) 2024, 4, 1396511. [Google Scholar] [CrossRef]
- Hayes, TR; Petrov, AA. Mapping and correcting the influence of gaze position on pupil size measurements. Behav Res Methods 2016, 48(2), 510–27. [Google Scholar] [CrossRef] [PubMed]
- Holland, E; Karpecki, P; Fingeret, M; Schaeffer, J; Gupta, P; Fram, N; et al. Efficacy and Safety of CSF-1 (0.4% Pilocarpine Hydrochloride) in Presbyopia: Pooled Results of the NEAR Phase 3 Randomized, Clinical Trials. Clin Ther. 2024, 46(2), 104–13. [Google Scholar] [CrossRef]
- Brown, SM; Bradley, JC. Comparison of 2 monocular pupillometers and an autorefractor for measurement of the dark-adapted pupil diameter. J Cataract Refract Surg. 2011, 37(4), 660–4. [Google Scholar] [CrossRef]
- Patwari, PP; Stewart, TM; Rand, CM; Carroll, MS; Kuntz, NL; Kenny, AS; et al. Pupillometry in congenital central hypoventilation syndrome (CCHS): quantitative evidence of autonomic nervous system dysregulation. Pediatr Res. 2012, 71(3), 280–5. [Google Scholar] [CrossRef] [PubMed]
- Muppidi, S; Adams-Huet, B; Tajzoy, E; Scribner, M; Blazek, P; Spaeth, EB; et al. Dynamic pupillometry as an autonomic testing tool. Clin Auton Res. 2013, 23(6), 297–303. [Google Scholar] [CrossRef] [PubMed]
- McAnany, JJ; Smith, BM; Garland, A; Kagen, SL. iPhone-based Pupillometry: A Novel Approach for Assessing the Pupillary Light Reflex. Optom Vis Sci. 2018, 95(10), 953–8. [Google Scholar] [CrossRef] [PubMed]
- Yoo, YJ; Yang, HK; Hwang, JM. Efficacy of digital pupillometry for diagnosis of Horner syndrome. PLoS One 2017, 12(6), e0178361. [Google Scholar] [CrossRef]
- Chang, DS; Arora, KS; Boland, MV; Supakontanasan, W; Friedman, DS. Development and validation of an associative model for the detection of glaucoma using pupillography. Am J Ophthalmol. 2013, 156(6), 1285–96.e2. [Google Scholar] [CrossRef]
- Park, JC; Moss, HE; McAnany, JJ. The Pupillary Light Reflex in Idiopathic Intracranial Hypertension. Invest Ophthalmol Vis Sci. 2016, 57(1), 23–9. [Google Scholar]
- Marshall, LF; Barba, D; Toole, BM; Bowers, SA. The oval pupil: clinical significance and relationship to intracranial hypertension. J Neurosurg. 1983, 58(4), 566–8. [Google Scholar] [CrossRef]
- Mittal, MK; Rabinstein, AA; Wijdicks, EF. Pearls & oy-sters: oval pupil: two observations. Neurology 2013, 81(17), e124-5. [Google Scholar]
- Jefferis, JM; Littlewood, RA; Pepper, IM; Hickman, SJ; Salvi, SM. Optic nerve sheath fenestration via a supero-medial eyelid skin crease approach for the treatment of idiopathic intracranial hypertension in a UK population. Eye (Lond) 2021, 35(5), 1418–26. [Google Scholar] [CrossRef]
- Atchison, DA; Girgenti, CC; Campbell, GM; Dodds, JP; Byrnes, TM; Zele, AJ. Influence of field size on pupil diameter under photopic and mesopic light levels. Clin Exp Optom. 2011, 94(6), 545–8. [Google Scholar] [CrossRef]
- Ferrari, GL; Marques, JL; Gandhi, RA; Heller, SR; Schneider, FK; Tesfaye, S; et al. Using dynamic pupillometry as a simple screening tool to detect autonomic neuropathy in patients with diabetes: a pilot study. Biomed Eng Online 2010, 9, 26. [Google Scholar] [CrossRef]
- Prasad, S; Baccon, J; Galetta, SL. Mydriatic pupil in giant cell arteritis. J Neurol Sci. 2009, 284(1-2), 196–7. [Google Scholar] [CrossRef]
- Wang, Y; Zekveld, AA; Naylor, G; Ohlenforst, B; Jansma, EP; Lorens, A; et al. Parasympathetic Nervous System Dysfunction, as Identified by Pupil Light Reflex, and Its Possible Connection to Hearing Impairment. PLoS One 2016, 11(4), e0153566. [Google Scholar] [CrossRef]
- McCall, WV; Sareddy, S; Youssef, NA; Miller, BJ; Rosenquist, PB. The pupillary light reflex as a point-of-care test for suicide risk: Preliminary results. Psychiatry Res. 2021, 295, 113582. [Google Scholar] [CrossRef] [PubMed]
- Strauch, C; Wang, CA; Einhäuser, W; Van der Stigchel, S; Naber, M. Pupillometry as an integrated readout of distinct attentional networks. Trends Neurosci. 2022, 45(8), 635–47. [Google Scholar] [CrossRef] [PubMed]
- Oster, J; Huang, J; White, BJ; Radach, R; Itti, L; Munoz, DP; et al. Pupillary responses to differences in luminance, color and set size. Exp Brain Res. 2022, 240(6), 1873–85. [Google Scholar] [CrossRef] [PubMed]
- Larson, MD; Singh, V. Portable infrared pupillometry in critical care. Crit Care 2016, 20(1), 161. [Google Scholar] [CrossRef]
- Zhao, W; Stutzman, S; DaiWai, O; Saju, C; Wilson, M; Aiyagari, V. Inter-device reliability of the NPi-100 pupillometer. J Clin Neurosci. 2016, 33, 79–82. [Google Scholar] [CrossRef]
- Anderson, M; Elmer, J; Shutter, L; Puccio, A; Alexander, S. Integrating Quantitative Pupillometry Into Regular Care in a Neurotrauma Intensive Care Unit. J Neurosci Nurs. 2018, 50(1), 30–6. [Google Scholar] [CrossRef]
- Godau, J; Bharad, K; Rösche, J; Nagy, G; Kästner, S; Weber, K; et al. Automated Pupillometry for Assessment of Treatment Success in Nonconvulsive Status Epilepticus. Neurocrit Care 2022, 36(1), 148–56. [Google Scholar] [CrossRef]
- Zyoud, SH; Zyoud, AH. Coronavirus disease-19 in environmental fields: a bibliometric and visualization mapping analysis. Environ Dev Sustain. 2021, 23(6), 8895–923. [Google Scholar] [CrossRef]
- Li, X; Zhang, T; Zhao, L; Liu, Q; Shi, C; Zhang, R. Mapping of Global Research on Electronic Cigarettes: A Bibliometric Analysis. Front Public Health 2022, 10, 856257. [Google Scholar] [CrossRef] [PubMed]
- van Eck, NJ; Waltman, L. Software survey: VOSviewer, a computer program for bibliometric mapping. Scientometrics 2010, 84(2), 523–38. [Google Scholar] [CrossRef]
- Kulkarni, AV; Aziz, B; Shams, I; Busse, JW. Comparisons of citations in Web of Science, Scopus, and Google Scholar for articles published in general medical journals. Jama 2009, 302(10), 1092–6. [Google Scholar] [CrossRef]
- Martinez-Pulgarin, DF; Acevedo-Mendoza, WF; Cardona-Ospina, JA; Rodríguez-Morales, AJ; Paniz-Mondolfi, AE. A bibliometric analysis of global Zika research. Travel Med Infect Dis. 2016, 14(1), 55–7. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Calderón, S; Nasner-Posso, KM; Alfaro-Toloza, P; Paniz-Mondolfi, AE; Rodríguez-Morales, AJ. A bibliometric analysis of global Ebola research. Travel Med Infect Dis. 2015, 13(2), 202–4. [Google Scholar] [CrossRef] [PubMed]
- Gao, M; Gao, D; Sun, H; Cheng, X; An, L; Qiao, M. Trends in Research Related to Premenstrual Syndrome and Premenstrual Dysphoric Disorder From 1945 to 2018: A Bibliometric Analysis. Front Public Health 2021, 9, 596128. [Google Scholar] [CrossRef]
- Joshi, S; Li, Y; Kalwani, RM; Gold, JI. Relationships between Pupil Diameter and Neuronal Activity in the Locus Coeruleus, Colliculi, and Cingulate Cortex. Neuron 2016, 89(1), 221–34. [Google Scholar] [CrossRef]
- Joshi, S; Gold, JI. Pupil Size as a Window on Neural Substrates of Cognition. Trends Cogn Sci. 2020, 24(6), 466–80. [Google Scholar] [CrossRef]
- Murphy, PR; O'Connell, RG; O'Sullivan, M; Robertson, IH; Balsters, JH. Pupil diameter covaries with BOLD activity in human locus coeruleus. Hum Brain Mapp. 2014, 35(8), 4140–54. [Google Scholar] [CrossRef] [PubMed]
- Mathôt, S. Pupillometry: Psychology, Physiology, and Function. J Cogn. 2018, 1(1), 16. [Google Scholar] [CrossRef] [PubMed]
- van der Wel, P; van Steenbergen, H. Pupil dilation as an index of effort in cognitive control tasks: A review. Psychon Bull Rev. 2018, 25(6), 2005–15. [Google Scholar] [CrossRef]
- Laeng, B; Sirois, S; Gredebäck, G. Pupillometry: A Window to the Preconscious? Perspect Psychol Sci. 2012, 7(1), 18–27. [Google Scholar] [CrossRef]
- Hall, CA; Chilcott, RP. Eyeing up the Future of the Pupillary Light Reflex in Neurodiagnostics. In Diagnostics (Basel); 2018; 1, p. 8. [Google Scholar]
- Kret, ME; Sjak-Shie, EE. Preprocessing pupil size data: Guidelines and code. Behav Res Methods 2019, 51(3), 1336–42. [Google Scholar] [CrossRef]
- Aston-Jones, G; Cohen, JD. An integrative theory of locus coeruleus-norepinephrine function: adaptive gain and optimal performance. Annu Rev Neurosci. 2005, 28, 403–50. [Google Scholar] [CrossRef]
- Beatty, J. Task-evoked pupillary responses, processing load, and the structure of processing resources. Psychol Bull. 1982, 91(2), 276–92. [Google Scholar] [CrossRef] [PubMed]
- Kahneman, D; Beatty, J. Pupil diameter and load on memory. Science 1966, 154(3756), 1583–5. [Google Scholar] [CrossRef]
- Bradley, MM; Miccoli, L; Escrig, MA; Lang, PJ. The pupil as a measure of emotional arousal and autonomic activation. Psychophysiology 2008, 45(4), 602–7. [Google Scholar] [CrossRef]
- Gilzenrat, MS; Nieuwenhuis, S; Jepma, M; Cohen, JD. Pupil diameter tracks changes in control state predicted by the adaptive gain theory of locus coeruleus function. Cogn Affect Behav Neurosci. 2010, 10(2), 252–69. [Google Scholar] [CrossRef] [PubMed]
- Hess, EH; Polt, JM. Pupil Size in Relation to Mental Activity during Simple Problem-Solving. Science 1964, 143(3611), 1190–2. [Google Scholar] [CrossRef]
- Chen, JW; Gombart, ZJ; Rogers, S; Gardiner, SK; Cecil, S; Bullock, RM. Pupillary reactivity as an early indicator of increased intracranial pressure: The introduction of the Neurological Pupil index. Surg Neurol Int. 2011, 2, 82. [Google Scholar] [CrossRef] [PubMed]
- Redondo, B; Serramito, M; Vera, J; Alguacil-Espejo, M; Rubio-Martínez, M; Molina, R; et al. Diurnal Variation in Accommodation, Binocular Vergence, and Pupil Size. Optom Vis Sci. 2023, 100(12), 847–54. [Google Scholar] [CrossRef] [PubMed]
- Alpern, M; Mason, GL; Jardinico, RE. Vergence and accommodation. V. Pupil size changes associated with changes in accommodative vergence. Am J Ophthalmol. 1961, 52, 762–7. [Google Scholar] [CrossRef]
- Bharadwaj, SR; Roy, S; Satgunam, P. Spasm of Near Reflex: Objective Assessment of the Near-Triad. Invest Ophthalmol Vis Sci. 2020, 61(8), 18. [Google Scholar] [CrossRef]
- Dutta, P; Atiya, A; Vittal, S; Ambika, S; Hussaindeen, JR. Pupillary dynamics and accommodative response in mild traumatic brain injury. Taiwan J Ophthalmol. 2024, 14(2), 248–55. [Google Scholar] [CrossRef]
- Dutta, P. Characteristics of binocular vision and oculomotor function among sports-concussed athletes. Indian J Ophthalmol. 2023, 71(5), 2076–82. [Google Scholar] [CrossRef]
- Busby, A; Ciuffreda, KJ. The effect of apparent depth in pictorial images on accommodation. Ophthalmic Physiol Opt. 2005, 25(4), 320–7. [Google Scholar] [CrossRef]
- Marquart, G; Winter, JCFD. Workload assessment for mental arithmetic tasks using the task-evoked pupillary response. PeerJ 2015. [Google Scholar] [CrossRef]
- Kawata, H; Ohtsuka, K. Dynamic asymmetries in convergence eye movements under natural viewing conditions. Jpn J Ophthalmol. 2001, 45(5), 437–44. [Google Scholar] [CrossRef]
- Balaban, CD; Kiderman, A; Szczupak, M; Ashmore, RC; Hoffer, ME. Patterns of Pupillary Activity During Binocular Disparity Resolution. Front Neurol. 2018, 9, 990. [Google Scholar] [CrossRef]
- McDougal, DH; Gamlin, PD. Autonomic control of the eye. Compr Physiol. 2015, 5(1), 439–73. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, H. Disorders of the pupil. Handb Clin Neurol. 2011, 102, 427–66. [Google Scholar]
- Kawasaki, A. Disorders of the Pupil. Continuum (Minneap Minn) 2025, 31(2), 527–42. [Google Scholar] [CrossRef]
- Koshikawa, Y; Sato, A; Umeda, R; Ichibayashi, R. Diagnosis of Acute Glaucoma Attack Using a Quantitative Pupillometer: A Case Report. Cureus 2024, 16(11), e72850. [Google Scholar] [CrossRef] [PubMed]
- Herbst, K; Sander, B; Lund-Andersen, H; Wegener, M; Hannibal, J; Milea, D. Unilateral anterior ischemic optic neuropathy: chromatic pupillometry in affected, fellow non-affected and healthy control eyes. Front Neurol. 2013, 4, 52. [Google Scholar] [CrossRef] [PubMed]
- Kim, HM; Yang, HK; Hwang, JM. Quantitative analysis of pupillometry in isolated third nerve palsy. PLoS One 2018, 13(11), e0208259. [Google Scholar] [CrossRef]
- Casey, F; Van, HMT; Donovan, J; Nghia, HDT; Oanh, PKN; Thwaites, CL; et al. Automated pupillometry and optic nerve sheath diameter ultrasound to define tuberculous meningitis disease severity and prognosis. J Neurol Sci. 2023, 453, 120808. [Google Scholar] [CrossRef]
- Gil-Casas, A; Piñero, DP; Molina-Martín, A. Dynamic Pupillary Response in Multiple Sclerosis Patients with and without Optic Neuritis. Biomedicines 2023, 11(12). [Google Scholar] [CrossRef]
- Wilhelm, B; Lüdtke, H; Peters, T; Schmid, R; Wilhelm, H; Zrenner, E. Automated swinging flashlight test in patients with optic nerve diseases. Klin Monbl Augenheilkd 2001, 218(1), 21–5. [Google Scholar] [CrossRef]
- Lavertu, G; Tremblay, M; Michael, R; You, E; Chabot, G. Validation of a new cell phone application for quick screening of relative afferent pupillary defect. Can J Ophthalmol. 2025, 60(5), e680–e8. [Google Scholar] [CrossRef] [PubMed]
- Sarker, P; Zaman, N; Ong, J; Paladugu, P; Aldred, M; Waisberg, E; et al. Test-Retest Reliability of Virtual Reality Devices in Quantifying for Relative Afferent Pupillary Defect. Transl Vis Sci Technol. 2023, 12(6), 2. [Google Scholar] [CrossRef] [PubMed]
- Barbur, JL; Harlow, AJ; Sahraie, A. Pupillary responses to stimulus structure, colour and movement. Ophthalmic Physiol Opt. 1992, 12(2), 137–41. [Google Scholar] [CrossRef]
- Gamlin, PD; Zhang, H; Harlow, A; Barbur, JL. Pupil responses to stimulus color, structure and light flux increments in the rhesus monkey. Vision Res. 1998, 38(21), 3353–8. [Google Scholar] [CrossRef] [PubMed]
- Shindler, KS; Revere, K; Dutt, M; Ying, GS; Chung, DC. In vivo detection of experimental optic neuritis by pupillometry. Exp Eye Res. 2012, 100, 1–6. [Google Scholar] [CrossRef]
- Jakobsen, J. Pupillary function in multiple sclerosis. Acta Neurol Scand. 1990, 82(6), 392–5. [Google Scholar] [CrossRef]
- Bhowmik, S; Motin, MA; Sarossy, M; Radcliffe, P; Kumar, D. Sample entropy analysis of pupillary signals in glaucoma patients and control via light-induced pupillometry. Annu Int Conf IEEE Eng Med Biol Soc. 2020, 2020, 280–3. [Google Scholar]
- Volpe, NJ; Plotkin, ES; Maguire, MG; Hariprasad, R; Galetta, SL. Portable pupillography of the swinging flashlight test to detect afferent pupillary defects. Ophthalmology 2000, 107(10), 1913–21; discussion 22. [Google Scholar] [CrossRef]
- Volpe, NJ; Dadvand, L; Kim, SK; Maguire, MG; Ying, GS; Moster, ML; et al. Computerized binocular pupillography of the swinging flashlight test detects afferent pupillary defects. Curr Eye Res. 2009, 34(7), 606–13. [Google Scholar] [CrossRef]
- Bruegger, D; Grabe, HM; Vicini, R; Dysli, M; Lussi, D; Abegg, M. Detection of Relative Afferent Pupillary Defects Using Eye Tracking and a VR Headset. Transl Vis Sci Technol. 2023, 12(6), 22. [Google Scholar] [CrossRef]
- Kelbsch, C; Lange, J; Wilhelm, H; Wilhelm, B; Peters, T; Kempf, M; et al. Chromatic Pupil Campimetry Reveals Functional Defects in Exudative Age-Related Macular Degeneration with Differences Related to Disease Activity. Transl Vis Sci Technol. 2020, 9(6), 5. [Google Scholar] [CrossRef]
- Kardon, R; Anderson, SC; Damarjian, TG; Grace, EM; Stone, E; Kawasaki, A. Chromatic pupillometry in patients with retinitis pigmentosa. Ophthalmology 2011, 118(2), 376–81. [Google Scholar] [CrossRef]
- Nakayama, M; Nowak, W; Ishikawa, H; Asakawa, K; Ichibe, Y. Discovering irregular pupil light responses to chromatic stimuli using waveform shapes of pupillograms. EURASIP J Bioinform Syst Biol. 2014, 2014, 18. [Google Scholar] [CrossRef] [PubMed]
- Xu, W; Jia, F; Liu, J; Li, J; Zhao, J; Lin, S; et al. Digital Pupillometry and Centroid Shift Changes in Dominant and Nondominant Eyes. Klin Monbl Augenheilkd 2025, 242(1), 62–9. [Google Scholar] [CrossRef]
- Kiziltoprak, H; Tekin, K; Yetkin, E; Sekeroglu, MA. Static and Dynamic Pupil Characteristics in Myopic Anisometropic Amblyopia. Beyoglu Eye J. 2020, 5(2), 86–92. [Google Scholar] [CrossRef] [PubMed]
- Tekin, K; Kiziltoprak, H; Sekeroglu, MA; Yetkin, E; Bayraktar, S; Yilmazbas, P. Static and dynamic pupil characteristics in pseudoexfoliation syndrome and glaucoma. Clin Exp Optom. 2020, 103(3), 332–8. [Google Scholar] [CrossRef]
- de Vries, L; Fouquaet, I; Boets, B; Naulaers, G; Steyaert, J. Autism spectrum disorder and pupillometry: A systematic review and meta-analysis. Neurosci Biobehav Rev. 2021, 120, 479–508. [Google Scholar] [CrossRef]
- Baranowski, P; Karolewicz, B; Gajda, M; Pluta, J. Ophthalmic drug dosage forms: characterisation and research methods. ScientificWorldJournal 2014, 2014, 861904. [Google Scholar] [CrossRef]
- Farkouh, A; Frigo, P; Czejka, M. Systemic side effects of eye drops: a pharmacokinetic perspective. Clin Ophthalmol. 2016, 10, 2433–41. [Google Scholar] [CrossRef] [PubMed]
- Oliva, M. Pupil size and search performance in low and high perceptual load. Cogn Affect Behav Neurosci 2019, 19(2), 366–76. [Google Scholar] [CrossRef]
- Traylor, JI; El Ahmadieh, TY; Bedros, NM; Al Adli, N; Stutzman, SE; Venkatachalam, AM; et al. Quantitative pupillometry in patients with traumatic brain injury and loss of consciousness: A prospective pilot study. J Clin Neurosci. 2021, 91, 88–92. [Google Scholar] [CrossRef]
- Romagnoli, M; Stanzani Maserati, M; De Matteis, M; Capellari, S; Carbonelli, M; Amore, G; et al. Chromatic Pupillometry Findings in Alzheimer's Disease. Front Neurosci. 2020, 14, 780. [Google Scholar] [CrossRef]
- Giza, E; Fotiou, D; Bostantjopoulou, S; Katsarou, Z; Karlovasitou, A. Pupil light reflex in Parkinson's disease: evaluation with pupillometry. Int J Neurosci. 2011, 121(1), 37–43. [Google Scholar] [CrossRef] [PubMed]
- İşcan, D; Türkoğlu, C; Arslan, E. Evaluation of autonomic involvement in Parkinson's disease using pupillometry. Neurol Sci. 2025, 46(8), 3685–90. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, E; Muhafız, E; Erdoğan, CE; Nizamoğulları, Ş; Toprak, G; Meydan, B. Pupillographic evaluation accompanying structural and functional assessment of the optic nerve in patients with Parkinson's disease. BMC Ophthalmol. 2025, 25(1), 397. [Google Scholar] [CrossRef]
- Lin, TP; Rigby, H; Adler, JS; Hentz, JG; Balcer, LJ; Galetta, SL; et al. Abnormal visual contrast acuity in Parkinson's disease. J Parkinsons Dis. 2015, 5(1), 125–30. [Google Scholar] [CrossRef]
- Schwartz, R; Rothermich, K; Kotz, SA; Pell, MD. Unaltered emotional experience in Parkinson's disease: Pupillometry and behavioral evidence. J Clin Exp Neuropsychol. 2018, 40(3), 303–16. [Google Scholar] [CrossRef] [PubMed]
- Serbest Ceylanoglu, K; Sen, EM; Sekeroglu, MA. Static and dynamic pupillary features in graves' ophthalmopathy. Clin Exp Optom. 2023, 106(5), 551–5. [Google Scholar] [CrossRef]
- Kuskonmaz, SM; Kocer, AM; Onder, CE; Sekeroglu, MA. Static and dynamic pupillary features in graves disease without orbitopathy. Int Ophthalmol. 2024, 44(1), 428. [Google Scholar] [CrossRef]
- Lerner, AG; Bernabé-Ortiz, A; Ticse, R; Hernandez, A; Huaylinos, Y; Pinto, ME; et al. Type 2 diabetes and cardiac autonomic neuropathy screening using dynamic pupillometry. Diabet Med. 2015, 32(11), 1470–8. [Google Scholar] [CrossRef]
- Nuske, HJ; Vivanti, G; Hudry, K; Dissanayake, C. Pupillometry reveals reduced unconscious emotional reactivity in autism. Biol Psychol. 2014, 101, 24–35. [Google Scholar] [CrossRef]
- Jackson, I; Sirois, S. Infant cognition: going full factorial with pupil dilation. Dev Sci. 2009, 12(4), 670–9. [Google Scholar] [CrossRef]
- Jones, NP; Siegle, GJ; Mandell, D. Motivational and emotional influences on cognitive control in depression: A pupillometry study. Cogn Affect Behav Neurosci. 2015, 15(2), 263–75. [Google Scholar] [CrossRef]
- Yang, X; Fridman, AJ; Unsworth, N; Casement, MD. Pupillary motility responses to affectively salient stimuli in individuals with depression or elevated risk of depression: A systematic review and meta-analysis. Neurosci Biobehav Rev. 2023, 148, 105125. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, AL; Garcia, JB; Viera, EB; Santos, AM; Bertrand, RH. Pupillometry: the influence of gender and anxiety on the pain response. Pain Physician 2013, 16(3), E257-66. [Google Scholar]
- Siegle, GJ; Steinhauer, SR; Friedman, ES; Thompson, WS; Thase, ME. Remission prognosis for cognitive therapy for recurrent depression using the pupil: utility and neural correlates. Biol Psychiatry 2011, 69(8), 726–33. [Google Scholar] [CrossRef]
- Roecklein, K; Wong, P; Ernecoff, N; Miller, M; Donofry, S; Kamarck, M; et al. The post illumination pupil response is reduced in seasonal affective disorder. Psychiatry Res. 2013, 210(1), 150–8. [Google Scholar] [CrossRef]
- Laurenzo, SA; Kardon, R; Ledolter, J; Poolman, P; Schumacher, AM; Potash, JB; et al. Pupillary response abnormalities in depressive disorders. Psychiatry Res. 2016, 246, 492–9. [Google Scholar] [CrossRef] [PubMed]
- Fattal, J; Brascamp, JW; Slate, RE; Lehet, M; Achtyes, ED; Thakkar, KN. Blunted pupil light reflex is associated with negative symptoms and working memory in individuals with schizophrenia. Schizophr Res. 2022, 248, 254–62. [Google Scholar] [CrossRef] [PubMed]
- Aslan, MG; Uzun, F; Fındık, H; Kaçar, M; Okutucu, M; Hocaoğlu, Ç. Pupillometry measurement and its relationship to retinal structural changes in children with attention deficit hyperactivity disorder. Graefes Arch Clin Exp Ophthalmol. 2020, 258(6), 1309–17. [Google Scholar] [CrossRef]
- Segers, M; Bebko, JM; Zapparoli, BL; Stevenson, RA. A pupillometry study of multisensory social and linguistic processing in autism and typical development. Dev Psychol. 2020, 56(11), 2080–94. [Google Scholar] [CrossRef]
- Lowenstein, O; Feinberg, R; Loewenfeld, IE. Pupillary Movements During Acute and Chronic Fatigue A New Test for the Objective Evaluation of Tiredness. In Invest Ophthalmol; 1963. [Google Scholar]
- Regen, F; Dorn, H; Danker-Hopfe, H. Association between pupillary unrest index and waking electroencephalogram activity in sleep-deprived healthy adults. Sleep Med. 2013, 14(9), 902–12. [Google Scholar] [CrossRef]
- Urschitz, MS; Heine, K; Brockmann, PE; Peters, T; Durst, W; Poets, CF; et al. Subjective and objective daytime sleepiness in schoolchildren and adolescents: results of a community-based study. Sleep Med. 2013, 14(10), 1005–12. [Google Scholar] [CrossRef]
- Frohnhofen, H; Fulda, S; Frohnhofen, K; Popp, R. Validation of the Essener Questionnaire of Age and Sleepiness in the elderly using pupillometry. Adv Exp Med Biol. 2013, 755, 125–32. [Google Scholar] [PubMed]
- Wilhelm, B; Giedke, H; Lüdtke, H; Bittner, E; Hofmann, A; Wilhelm, H. Daytime variations in central nervous system activation measured by a pupillographic sleepiness test. J Sleep Res. 2001, 10(1), 1–7. [Google Scholar] [CrossRef] [PubMed]
- Danker-Hopfe, H; Kraemer, S; Dorn, H; Schmidt, A; Ehlert, I; Herrmann, WM. Time-of-day variations in different measures of sleepiness (MSLT, pupillography, and SSS) and their interrelations. Psychophysiology 2001, 38(5), 828–35. [Google Scholar] [CrossRef] [PubMed]
- Li, Z; Cai, J; Li, J; Xu, X; Zheng, L. Comparative evaluation of the bispectral index (BIS) and BISpro during propofol anaesthesia. J Int Med Res. 2021, 49(4), 3000605211001705. [Google Scholar] [CrossRef]
- Vide, S; Kreuzer, M; Ferreira, A; Couto, M; Agustí, M; Jaramillo, S; et al. Cortical, subcortical, brainstem and autonomic responses to nociception under total intravenous anesthesia. J Clin Anesth. 2025, 103, 111825. [Google Scholar] [CrossRef]
- Dallemagne, A; Anderloni, M; Havaux, M; Duranteau, O; Taccone, FS. Influence of Sevoflurane on the Neurological Pupil Index in Surgical and Critically Ill Patients: A Pilot Study. Brain Sci. 2024, 14(3). [Google Scholar] [CrossRef]
- Sabourdin, N; Diarra, C; Wolk, R; Piat, V; Louvet, N; Constant, I. Pupillary Pain Index Changes After a Standardized Bolus of Alfentanil Under Sevoflurane Anesthesia: First Evaluation of a New Pupillometric Index to Assess the Level of Analgesia During General Anesthesia. Anesth Analg. 2019, 128(3), 467–74. [Google Scholar] [CrossRef]
- Barvais, L; Engelman, E; Eba, JM; Coussaert, E; Cantraine, F; Kenny, GN. Effect site concentrations of remifentanil and pupil response to noxious stimulation. Br J Anaesth. 2003, 91(3), 347–52. [Google Scholar] [CrossRef]
- Larson, MD; Berry, PD; May, J; Bjorksten, A; Sessler, DI. Autonomic effects of epidural and intravenous fentanyl. Br J Anaesth. 2007, 98(2), 263–9. [Google Scholar] [CrossRef]
- Guglielminotti, J; Mentré, F; Gaillard, J; Ghalayini, M; Montravers, P; Longrois, D. Assessment of pain during labor with pupillometry: a prospective observational study. Anesth Analg. 2013, 116(5), 1057–62. [Google Scholar] [CrossRef]
- Saliman, NH; Belli, A; Blanch, RJ. Afferent Visual Manifestations of Traumatic Brain Injury. J Neurotrauma 2021, 38(20), 2778–89. [Google Scholar] [CrossRef]
- Stoll, J; Chatelle, C; Carter, O; Koch, C; Laureys, S; Einhäuser, W. Pupil responses allow communication in locked-in syndrome patients. Curr Biol. 2013, 23(15), R647–8. [Google Scholar] [CrossRef] [PubMed]
- Opic, P; Rüegg, S; Marsch, S; Gut, SS; Sutter, R. Automated Quantitative Pupillometry in the Critically Ill: A Systematic Review of the Literature. Neurology 2021, 97(6), e629–e42. [Google Scholar] [CrossRef] [PubMed]
- Jaquet, P; Couffignal, C; Tardivon, C; Godard, V; Bellot, R; Assouline, B; et al. PupillOmetry for preDIction of DeliriUM in ICU (PODIUM): protocol for a prospective multicentre cohort study. BMJ Open. 2023, 13(7), e072095. [Google Scholar] [CrossRef] [PubMed]
- Santini, T; Fuhl, W; Kasneci, E. PuReST: robust pupil tracking for real-time pervasive eye tracking; ACM, 2018. [Google Scholar]
- Villanueva, A; Cabeza, R; Porta, S. Eye tracking: Pupil orientation geometrical modeling. Image & Vision Computing 2006, 24(7), 663–79. [Google Scholar]
- Cavanagh, JF; Wiecki, TV; Kochar, A; Frank, MJ. Eye tracking and pupillometry are indicators of dissociable latent decision processes. J Exp Psychol Gen. 2014, 143(4), 1476–88. [Google Scholar] [CrossRef]
- Fountas, KN; Kapsalaki, EZ; Machinis, TG; Boev, AN; Robinson, JS; Troup, EC. Clinical implications of quantitative infrared pupillometry in neurosurgical patients. Neurocrit Care 2006, 5(1), 55–60. [Google Scholar] [CrossRef]
- Bitirgen, G; Korkmaz, C; Zamani, A; Iyisoy, MS; Kerimoglu, H; Malik, RA. Abnormal quantitative pupillary light responses following COVID-19. Int Ophthalmol. 2022, 42(9), 2847–54. [Google Scholar] [CrossRef]
- Serbest Ceylanoglu, K; Sen, EM; Sekeroglu, MA. Static and dynamic pupillary features in graves' ophthalmopathy. Clin Exp Optom. 2022, 1–5. [Google Scholar] [CrossRef]
- Crippa, SV; Pedrosa Domellöf, F; Kawasaki, A. Chromatic Pupillometry in Children. Front Neurol. 2018, 9, 669. [Google Scholar] [CrossRef] [PubMed]
- Sandroni, C; Citerio, G; Taccone, FS. Automated pupillometry in intensive care. Intensive Care Med 2022. [Google Scholar] [CrossRef] [PubMed]
- Mathôt, S; Vilotijević, A. Methods in cognitive pupillometry: Design, preprocessing, and statistical analysis. Behav Res Methods 2023, 55(6), 3055–77. [Google Scholar] [CrossRef]
- Favre, E; Rahmaty, Z; Ben-Hamouda, N; Miroz, JP; Abed-Maillard, S; Rusca, M; et al. Nociception assessment with videopupillometry in deeply sedated intensive care patients: Discriminative and criterion validations. Aust Crit Care 2024, 37(1), 84–90. [Google Scholar] [CrossRef] [PubMed]
- Fink, L; Simola, J; Tavano, A; Lange, E; Wallot, S; Laeng, B. From pre-processing to advanced dynamic modeling of pupil data. Behav Res Methods 2024, 56(3), 1376–412. [Google Scholar] [CrossRef]
- Mizera, J; Sova, M; Genzor, S; Krejci, T; Vachutka, J; Vanek, J; et al. Pupillography in contemporary sleep medicine - A narrative review. Neuro Endocrinol Lett. 2023, 44(5), 297–308. [Google Scholar]
- Nissen, C; Sander, B; Milea, D; Kolko, M; Herbst, K; Hamard, P; et al. Monochromatic Pupillometry in Unilateral Glaucoma Discloses no Adaptive Changes Subserved by the ipRGCs. Front Neurol 2014, 5, 15. [Google Scholar] [CrossRef]
- Martucci, A; Cesareo, M; Napoli, D; Sorge, RP; Ricci, F; Mancino, R; et al. Evaluation of pupillary response to light in patients with glaucoma: a study using computerized pupillometry. Int Ophthalmol. 2014, 34(6), 1241–7. [Google Scholar] [CrossRef]
- Park, HL; Jung, SH; Park, SH; Park, CK. Detectping autonomic dysfunction in patients with glaucoma using dynamic pupillometry. Medicine (Baltimore) 2019, 98(11), e14658. [Google Scholar] [CrossRef]
- Rukmini, AV; Chew, MC; Finkelstein, MT; Atalay, E; Baskaran, M; Nongpiur, ME; et al. Effects of low and moderate refractive errors on chromatic pupillometry. Sci Rep. 2019, 9(1), 4945. [Google Scholar] [CrossRef] [PubMed]
- Mabed, IS; Saad, A; Guilbert, E; Gatinel, D. Measurement of pupil center shift in refractive surgery candidates with caucasian eyes using infrared pupillometry. J Refract Surg. 2014, 30(10), 694–700. [Google Scholar] [CrossRef]
- Vilotijević, A; Mathôt, S. Functional benefits of cognitively driven pupil-size changes. Wiley Interdiscip Rev Cogn Sci. 2024, 15(3), e1672. [Google Scholar] [CrossRef]
- Murphy, PR; Robertson, IH; Balsters, JH; O'Connell, R G. Pupillometry and P3 index the locus coeruleus-noradrenergic arousal function in humans. Psychophysiology 2011, 48(11), 1532–43. [Google Scholar] [CrossRef] [PubMed]
- Lynch, G. Using Pupillometry to Assess the Atypical Pupillary Light Reflex and LC-NE System in ASD. Behav Sci (Basel) 2018, 8(11). [Google Scholar] [CrossRef] [PubMed]
- Jahns, FP; Miroz, JP; Messerer, M; Daniel, RT; Taccone, FS; Eckert, P; et al. Quantitative pupillometry for the monitoring of intracranial hypertension in patients with severe traumatic brain injury. Crit Care 2019, 23(1), 155. [Google Scholar] [CrossRef]
- El Ahmadieh, TY; Bedros, N; Stutzman, SE; Nyancho, D; Venkatachalam, AM; MacAllister, M; et al. Automated Pupillometry as a Triage and Assessment Tool in Patients with Traumatic Brain Injury. World Neurosurg. 2021, 145, e163–e9. [Google Scholar] [CrossRef]
- Fotiou, DF; Stergiou, V; Tsiptsios, D; Lithari, C; Nakou, M; Karlovasitou, A. Cholinergic deficiency in Alzheimer's and Parkinson's disease: evaluation with pupillometry. Int J Psychophysiol. 2009, 73(2), 143–9. [Google Scholar] [CrossRef]
- El Haj, M; Chapelet, G; Moustafa, AA; Boutoleau-Bretonnière, C. Pupil size as an indicator of cognitive activity in mild Alzheimer's disease. Excli j. 2022, 21, 307–16. [Google Scholar] [PubMed]
- Oddo, M; Taccone, FS; Petrosino, M; Badenes, R; Blandino-Ortiz, A; Bouzat, P; et al. The Neurological Pupil index for outcome prognostication in people with acute brain injury (ORANGE): a prospective, observational, multicentre cohort study. Lancet Neurol. 2023, 22(10), 925–33. [Google Scholar] [CrossRef]
- Choi, J; Corcoran, CM; Fiszdon, JM; Stevens, M; Javitt, DC; Deasy, M; et al. Pupillometer-based neurofeedback cognitive training to improve processing speed and social functioning in individuals at clinical high risk for psychosis. Psychiatr Rehabil J. 2017, 40(1), 33–42. [Google Scholar] [CrossRef] [PubMed]
- Mestanikova, A; Ondrejka, I; Mestanik, M; Cesnekova, D; Visnovcova, Z; Bujnakova, I; et al. Pupillary light reflex is altered in adolescent depression. Physiol Res. 2017, 66 (Suppl 2), S277–s84. [Google Scholar] [CrossRef]
- Berman, G; Muttuvelu, D; Berman, D; Larsen, JI; Licht, RW; Ledolter, J; et al. Decreased retinal sensitivity in depressive disorder: a controlled study. Acta Psychiatr Scand. 2018, 137(3), 231–40. [Google Scholar] [CrossRef]
- Barry, C; Wang, E. Racially fair pupillometry measurements for RGB smartphone cameras using the far red spectrum. Sci Rep. 2023, 13(1), 13841. [Google Scholar] [CrossRef] [PubMed]
- Pelagatti, C; Blini, E; Vannucci, M. Catching Mind Wandering With Pupillometry: Conceptual and Methodological Challenges. Wiley Interdiscip Rev Cogn Sci. 2025, 16(1), e1695. [Google Scholar] [CrossRef]
- Vinclair, M; Schilte, C; Roudaud, F; Lavolaine, J; Francony, G; Bouzat, P; et al. Using Pupillary Pain Index to Assess Nociception in Sedated Critically Ill Patients. Anesth Analg. 2019, 129(6), 1540–6. [Google Scholar] [CrossRef]
- Vide, S; Castro, A; Correia, R; Cabral, T; Lima, D; Nunes, CS; et al. Foreseeing postoperative pain in neurosurgical patients: pupillometry predicts postoperative pain ratings-an observational study. J Clin Monit Comput. 2021, 35(5), 1111–8. [Google Scholar] [CrossRef]
- Zugni, N; Guadrini, L; Rasulo, F. Noninvasive neuromonitoring in the operating room and its role in the prevention of delirium. Best Pract Res Clin Anaesthesiol. 2021, 35(2), 191–206. [Google Scholar] [CrossRef]
- Paramanathan, S; Grejs, AM; Søreide, E; Duez, CHV; Jeppesen, AN; Reinertsen Å, J; et al. Quantitative pupillometry in comatose out-of-hospital cardiac arrest patients: A post-hoc analysis of the TTH48 trial. Acta Anaesthesiol Scand. 2022, 66(7), 880–6. [Google Scholar] [CrossRef]
- Fratino, S; Garré, A; Garufi, A; Hafidi, S; Migliorino, E; Stropeni, S; et al. Evaluation of nociception in unconscious critically ill patients using a multimodal approach. Anaesth Crit Care Pain Med. 2023, 42(2), 101175. [Google Scholar] [CrossRef]
- Kornilov, E; Gehlen, L; Yacobi, D; Soehle, M; Kowark, A; Thudium, M. Pupillary Pain Index Predicts Postoperative Pain but Not the Effect of Peripheral Regional Anaesthesia in Patients Undergoing Total Hip or Total Knee Arthroplasty: An Observational Study. Medicina (Kaunas) 2023, 59(5). [Google Scholar] [CrossRef]
- Singh, A; Akhileshwar; Kumar, N; De, RR; Bahadur, R; Shekhar, S. The Role of Pupillometry in the Assessment of Pain in Children Under General Anesthesia: A Prospective Single-Blinded Observational Study. Cureus 2023, 15(8), e43894. [Google Scholar] [CrossRef]
- Nyholm, B; Grand, J; Obling, LER; Hassager, C; Møller, JE; Schmidt, H; et al. Quantitative pupillometry for neuroprognostication in comatose post-cardiac arrest patients: A protocol for a predefined sub-study of the Blood pressure and Oxygenations Targets after Out-of-Hospital Cardiac Arrest (BOX)-trial. Resusc Plus 2023, 16, 100475. [Google Scholar] [CrossRef] [PubMed]
- Ma, X; Monfared, R; Grant, R; Goh, YM. Determining Cognitive Workload Using Physiological Measurements: Pupillometry and Heart-Rate Variability. Sensors (Basel) 2024, 24(6). [Google Scholar] [CrossRef]
- Krafthöfer, J; Fabig, SC; Baron, R; Gierthmühlen, J. Pupillometry as a Potential Objective Measurement of Pain Assessment in Healthy Volunteers. J Pain Res. 2024, 17, 2037–42. [Google Scholar] [CrossRef]
- Zhang, Y; Lim, D; Yao, Y; Dong, C; Feng, Z. Global Research Trends in Radiotherapy for Gliomas: A Systematic Bibliometric Analysis. World Neurosurg. 2022, 161, e355–e62. [Google Scholar] [CrossRef] [PubMed]
- Fu, HZ; Wang, MH; Ho, YS. The most frequently cited adsorption research articles in the Science Citation Index (Expanded). J Colloid Interface Sci. 2012, 379(1), 148–56. [Google Scholar] [CrossRef] [PubMed]
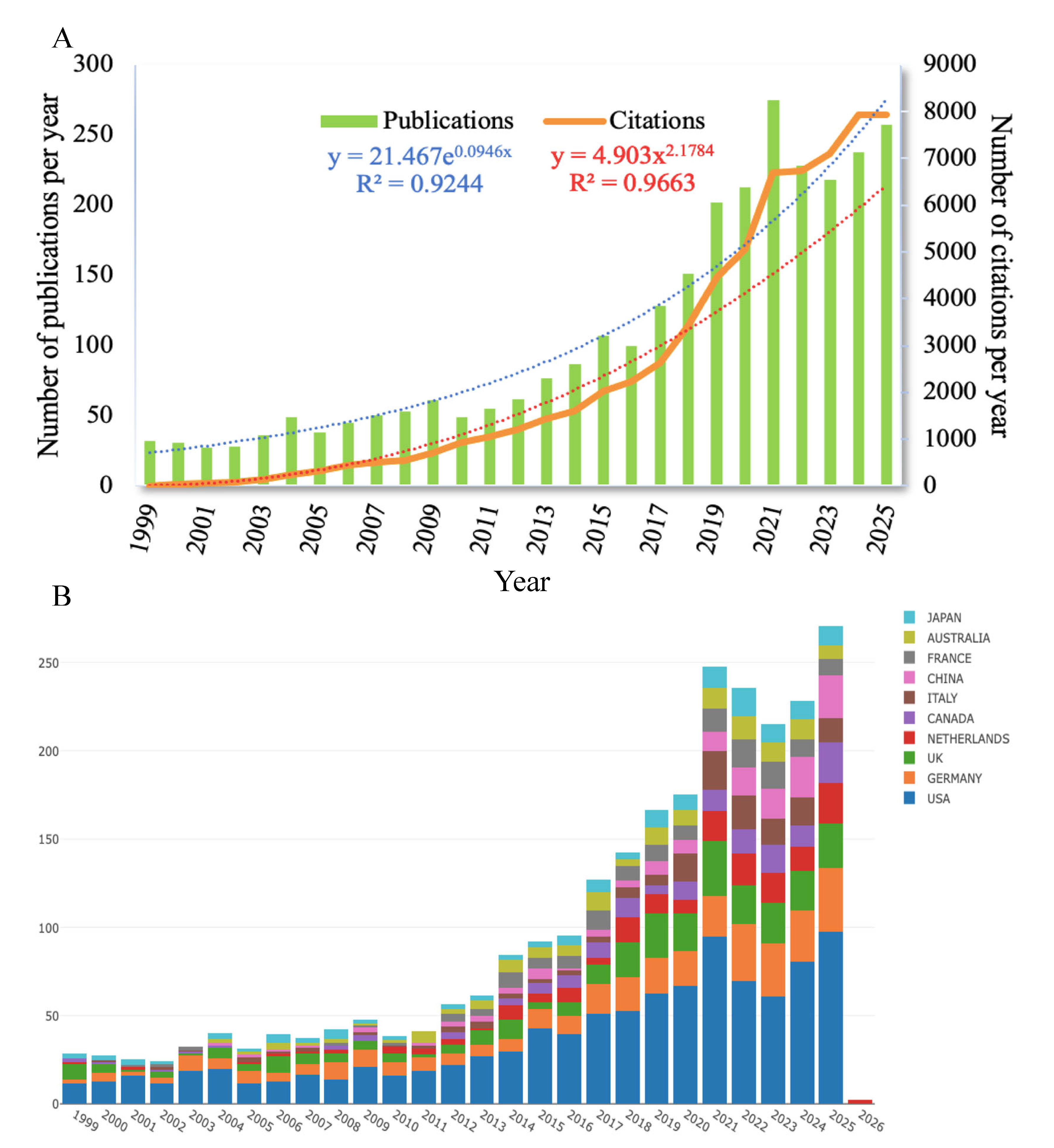
| Year | Publications | Citations |
|---|---|---|
| 1999 | 32 | 6 |
| 2000 | 31 | 32 |
| 2001 | 27 | 60 |
| 2002 | 28 | 86 |
| 2003 | 36 | 147 |
| 2004 | 49 | 249 |
| 2005 | 38 | 331 |
| 2006 | 45 | 452 |
| 2007 | 50 | 501 |
| 2008 | 53 | 548 |
| 2009 | 61 | 704 |
| 2010 | 49 | 931 |
| 2011 | 55 | 1,049 |
| 2012 | 62 | 1,217 |
| 2013 | 77 | 1,434 |
| 2014 | 87 | 1,601 |
| 2015 | 107 | 2,020 |
| 2016 | 100 | 2,230 |
| 2017 | 128 | 2,629 |
| 2018 | 151 | 3,417 |
| 2019 | 202 | 4,453 |
| 2020 | 213 | 5,050 |
| 2021 | 275 | 6,686 |
| 2022 | 228 | 6,732 |
| 2023 | 218 | 7,093 |
| 2024 | 238 | 7,941 |
| 2025 | 257 | 7,923 |
| Total | 2,897 | 65,552 |
| Rank | Country | Documents | Citations | Citation/ Document (%) | Centrality | Total link strength |
|---|---|---|---|---|---|---|
| 1 | USA | 1,086 | 29,243 | 26.92 | 0.38 | 440 |
| 2 | Germany | 351 | 8,482 | 24.16 | 0.12 | 274 |
| 3 | England | 267 | 7,380 | 27.64 | 0.30 | 254 |
| 4 | Netherlands | 168 | 6,251 | 37.20 | 0.10 | 211 |
| 5 | Canada | 146 | 3,332 | 22.82 | 0.01 | 127 |
| 6 | Italy | 145 | 3,435 | 23.68 | 0.13 | 192 |
| 7 | France | 136 | 3,483 | 25.61 | 0.07 | 132 |
| 8 | Australia | 135 | 3,784 | 28.02 | 0.23 | 124 |
| 9 | Japan | 122 | 1,703 | 13.95 | 0.04 | 56 |
| 10 | China | 106 | 1,003 | 9.46 | 0.05 | 47 |
| Rank | Organization | Country | Documents | Citations | Citation/ Document (%) (%) | Centrality |
|---|---|---|---|---|---|---|
| 1 | University of Toronto | Canada | 46 | 1,134 | 24.65 | 0.05 |
| 2 | University of Pennsylvania | USA | 43 | 2,517 | 58.53 | 0.08 |
| 3 | University of Tubingen | Germany | 43 | 980 | 22,79 | 0.03 |
| 4 | University of Oslo | Norway | 41 | 1,047 | 25.53 | 0.03 |
| 5 | Johns Hopkins University | USA | 40 | 770 | 19.25 | 0.11 |
| 6 | University of Pittsburgh | USA | 40 | 1,099 | 27.47 | 0.05 |
| 7 | Australian National University | Australia | 37 | 580 | 15.67 | 0.01 |
| 8 | University of Copenhagen | Denmark | 36 | 377 | 10.47 | 0.02 |
| 9 | University of California San Francisco | USA | 35 | 1,162 | 33.20 | 0.06 |
| 10 | Technical University of Denmark | Denmark | 33 | 938 | 28.42 | 0.01 |
| Rank | Author | Documents | Citations | H-index | Co-cited author | Citations | Total link strength |
|---|---|---|---|---|---|---|---|
| 1 | Olson Daiwai M | 34 | 703 | 13 | Beatty J | 527 | 14,690 |
| 2 | Meddess Ted | 33 | 554 | 16 | Kahneman D | 515 | 14,743 |
| 3 | Laeng Bruno | 29 | 956 | 13 | Zekveld AA | 494 | 16,363 |
| 4 | Wilhelm Barbara | 28 | 398 | 16 | Larson MD | 457 | 6,607 |
| 5 | Wihelm Helmut | 27 | 553 | 18 | Aston-jones G | 454 | 13,492 |
| 6 | Wendt Dorothea | 26 | 788 | 14 | Laeng B | 414 | 10,003 |
| 7 | Aiyagari Venkatesh | 25 | 696 | 14 | Joshi S | 412 | 11,224 |
| 8 | Peters Tobias | 22 | 442 | 13 | Murphy PR | 374 | 11,651 |
| 9 | Stutzman Sonja E | 22 | 423 | 12 | Unsworth N | 366 | 12,517 |
| 10 | Kramer Sophia E | 21 | 1,735 | 15 | Mathot S | 340 | 8,967 |
| Rank | Journal | Country | Documents (%) | Citations | Total link strength | IF (2024) | Co-cited journal | Citations | Total link strength |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Psychophysiology | USA | 79 (2.72) | 3,106 | 913 | 2.9 | IOVS | 3,008 | 150,007 |
| 2 | Scientific Reports | England | 74 (2.55) | 897 | 636 | 2.9 | Psychophysiology | 2,593 | 157,620 |
| 3 | Plos One | USA | 74 (2.55) | 1,993 | 621 | 3.8 | Ophthalmology | 2,045 | 84,424 |
| 4 | IOVS | USA | 69 (2.38) | 2,656 | 940 | 5.0 | Journal of Neuroscience | 1,987 | 135,029 |
| 5 | Journal of Cataract and Refractive Surgery | USA | 53 (1.82) | 1,473 | 358 | 2.6 | Plos One | 1,760 | 108,990 |
| 6 | Graefes Archive for Clinical and Experimental Ophthalmology | USA | 48 (1.65) | 848 | 392 | 2.3 | Science | 1,629 | 96,516 |
| 7 | International Journal of Psychophysiology | Netherlands | 46 (1.58) | 1,714 | 422 | 2.7 | Neuroimage | 1,526 | 102,829 |
| 8 | Frontiers in Neurology | Switzerland | 42 (1.44) | 986 | 602 | 2.9 | American Journal of Ophthalmology | 1,426 | 97,259 |
| 9 | Journal of Neuro-ophthalmology | USA | 37 (1.27) | 721 | 152 | 2.6 | Neuron | 1,414 | 55,775 |
| 10 | Ophthalmic Plastic and Reconstructive Surgery | USA | 35 (1.20) | 330 | 599 | 1.3 | Ear and Hearing | 1,346 | 75,559 |
| Rank | Cited reference | Title | Citations | Total link strength |
|---|---|---|---|---|
| 1 | aston-jones g, 2005, annu rev neurosci, v28, p403, doi 10.1146/annurev.neuro.28.061604.135709 | An integrative theory of locus coeruleus-norepinephrine function: adaptive gain and optimal performance | 359 | 6,684 |
| 2 | joshi s, 2016, neuron, v89, p221, doi 10.1016/j.neuron.2015.11.028 | Relationships between Pupil Diameter and Neuronal Activity in the Locus Coeruleus, Colliculi, and Cingulate Cortex | 271 | 4,667 |
| 3 | beatty j, 1982, psychol bull, v91, p276, doi 10.1037/0033-2909.91.2.276 | Task-evoked pupillary responses, processing load, and the structure of processing resources | 263 | 4,362 |
| 4 | kahneman d, 1966, science, v154, p1583, doi 10.1126/science.154.3756.1583 | Pupil diameter and load on memory | 260 | 4,642 |
| 5 | bradley mm, 2008, psychophysiology, v45, p602, doi 10.1111/j.1469-8986.2008.00654.x | The pupil as a measure of emotional arousal and autonomic activation | 220 | 3,081 |
| 6 | laeng b, 2012, perspect psychol sci, v7, p18, doi 10.1177/1745691611427305 | Pupillometry: A Window to the Preconscious? | 214 | 3,010 |
| 7 | murphy pr, 2014, hum brain mapp, v35, p4140, doi 10.1002/hbm.22466 | Pupil diameter covaries with BOLD activity in human locus coeruleus | 193 | 3,765 |
| 8 | hess eh, 1964, science, v143, p1190, doi 10.1126/science.143.3611.1190 | Pupil Size in Relation to Mental Activity during Simple Problem-Solving | 179 | 3,302 |
| 9 | gilzenrat ms, 2010, cogn affect behav ne, v10, p252, doi 10.3758/cabn.10.2.252 | Pupil diameter tracks changes in control state predicted by the adaptive gain theory of locus coeruleus function | 169 | 3,530 |
| 10 | Sebastiaan, Mathôt.(2018). Pupillometry: Psychology, Physiology, and Function. J Cogn, 1(1), 16. doi:10.5334/joc.18 | Pupillometry: Psychology, Physiology, and Function | 157 | 2,267 |
| Cluster 1 | Cluster 2 | Cluster 3 | Cluster 4 | Cluster 5 |
|---|---|---|---|---|
| Pupillometry (1069) | Pupil (239) | Locus-coeruleus (304) | Cognition (60) | Heart rate variability (99) |
| Pupillary light reflex (237) | Retinal ganglion cells (135) | Attention (240) | Speech perception (49) | Pain (66) |
| Eye tracking (112) | Eye (113) | Brain (97) | Cognitive control (48) | Analgesia (51) |
| Infrared pupillometry (87) | Melanopsin (111) | Autonomic nervous system (96) | Autism spectrum disorder (46) | Heart rate (46) |
| Pupillography (83) | Glaucoma (83) | Working-memory (96) | Anxiety (45) | Cardiac arrest (41) |
| Quantitative pupillometry (70) | Afferent pupillary defect (73) | Traumatic brain injury (85) | Depression (41) | Anesthesia (36) |
| Pupillometer (68) | Cone (51) | Intracranial pressure (57) | Stress (31) | Morphine (27) |
| Automated pupillometry (61) | Rod (42) | Alzheimer’s disease (53) | Sustained attention (28) | Pain assessment (18) |
| Dynamic pupillometry (41) | Optic neuritis (35) | Multiple sclerosis (53) | Schizophrenia (24) | Postoperative pain (15) |
| Chromatic pupillometry (22) | Myopia (35) | Parkinson's disease (36) | Dementia (18) | Diazepam (9) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




