Submitted:
25 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Study Design
2.2. Search Strategy
- “Constipation” [Major Topic]
- “Aged” [MeSH Terms]
2.3. Eligibility Criteria
- Studies involving older adults (generally defined as ≥65 years of age)
- Constipation addressed as a primary research topic and designated as a MeSH Major Topic
- Original research articles, systematic reviews, epidemiological studies, or clinical studies
- Availability of an abstract
- Articles published from 2023 onward
- Studies limited to pediatric or young adult populations
- Studies in which constipation was evaluated only as a secondary outcome
- Case reports, editorials, or studies available only as conference abstracts
- Animal studies or basic experimental research without clinical data
2.4. Data Extraction and Synthesis
- Author names, year of publication, and study design
- Study population (e.g., community-dwelling older adults, residents of long-term care facilities, home care patients)
- Definition and diagnostic criteria of constipation (e.g., Rome III, Rome IV, Rome IV-TR)
- Prevalence, associated factors, and clinical outcomes
- Diagnostic and management challenges specific to older adults
2.5. Figure Design and Source Attribution
3. Definition of Constipation and Diagnostic Challenges in Older Adults
4. Epidemiology of Constipation in Older Adults
4.1. Prevalence and Age-Specific Patterns
4.2. Differences Between Community-Dwelling and Institutionalized Older Adults
4.3. Female Predominance and Age-Related Changes
4.4. Geographic and Socioeconomic Factors
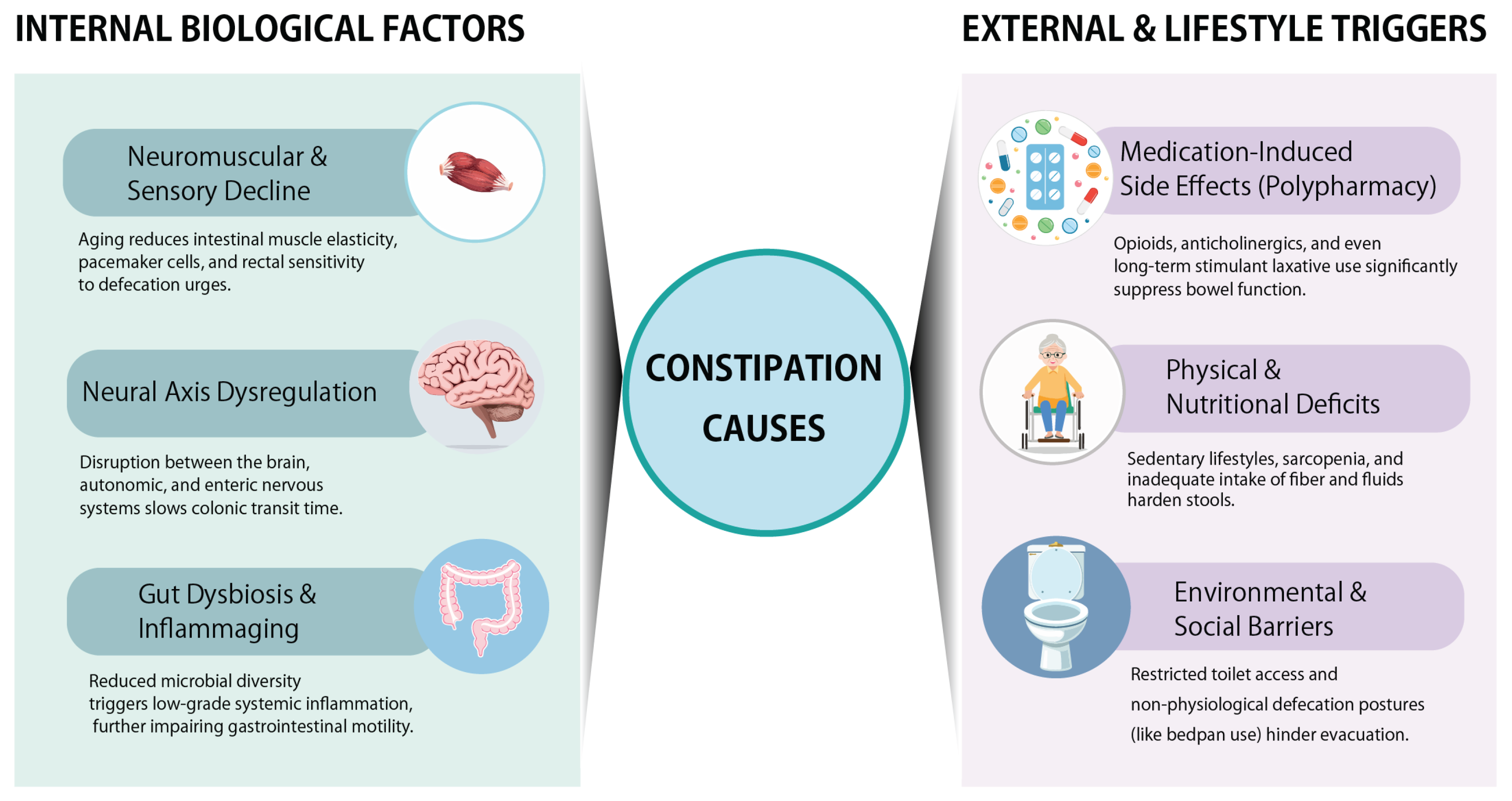
5. Pathophysiology
5.1. Peripheral Mechanisms (Smooth Muscle, ICC, Anorectal Function)
5.2. Central and Autonomic Dysregulation (CNS–ENS–ANS)
5.3. Gut Microbiota and Inflammaging
6. Risk Factors
6.1. Physical and Nutritional Factors
6.2. Medication-Related Factors
6.2.1. Opioid Analgesics
6.2.2. Anticholinergic Medications
6.2.3. Other Medications and Effects on the Gastrointestinal Environment
6.3. Environmental and Social Factors
7. Clinical Impact
7.1. Cognitive, Psychological, and Systemic Outcomes
7.2. Prognosis and Healthcare Burden
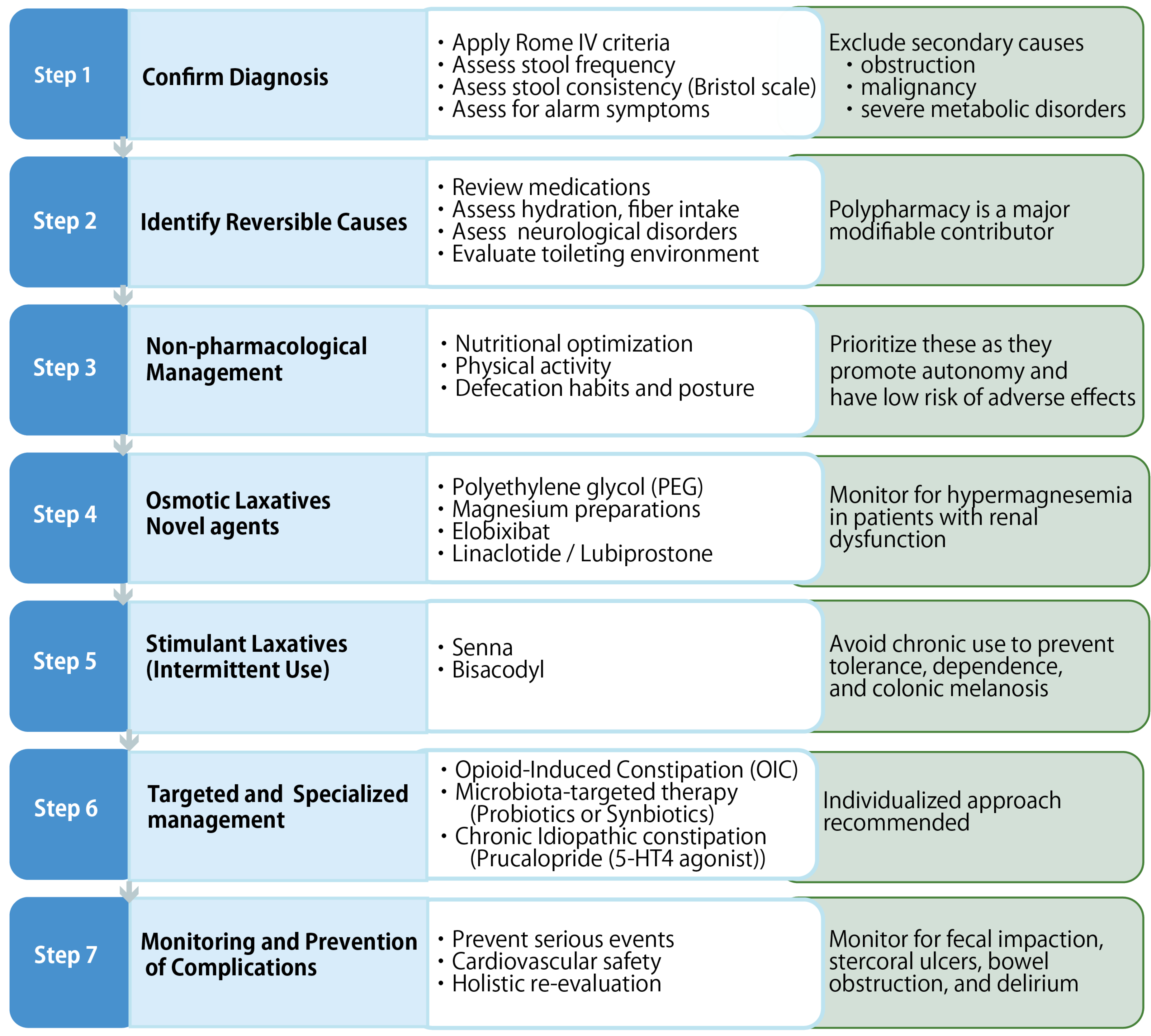
8. Management Strategies
8.1. Non-Pharmacological Interventions
8.1.1. Lifestyle and Dietary Modification
8.1.2. Physical Activity and Mechanical Stimulation
8.1.3. Toileting Environment, Posture, and Daily Rhythm
8.2. Pharmacological Therapy
8.2.1. Conventional Laxatives: Benefits and Limitations
8.2.2. Novel Secretagogues and Prokinetic Agents
8.2.3. Management of Secondary Constipation in Older Adults
8.3. Microbiota-targeted therapy
8.3.1. Mechanistic Rationale for Microbiota-Based Therapy
8.3.2. Clinical Evidence in Older Adults
8.3.3. Toward Personalized Microbiota Modulation
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, F.; Fei, M.; Hu, W.Z.; Wang, X.D.; Liu, S.; Zeng, Y.; Zhang, J.H.; Lv, Y.; Niu, J.P.; Meng, X.L.; et al. Prevalence of Constipation in Elderly and Its Association With Dementia and Mild Cognitive Impairment: A Cross-Sectional Study. Front. Neurosci. 2022, 15, 821654. [Google Scholar] [CrossRef] [PubMed]
- Schnitker, L.M.; Alrababah, S.; Young, A.; Selby, Z.; Meyer, C.; Lowthian, J.A. Best Practice to Prevent Constipation in People Living With Dementia: A Scoping Review. Int. J. Older People Nurs. 2025, 20. [Google Scholar] [CrossRef]
- Yun, Q.; Wang, S.; Li, B.; Yang, Z.; Wei, C.; Yuan, J.; Hu, J.; Li, Y.; Yang, Z.; Sha, F.; et al. Constipation Preceding Stroke, Dementia and Parkinson’s Disease in Middle-Aged and Older Adults: A Population-Based Cohort Study. Age Ageing 2025, 54. [Google Scholar] [CrossRef]
- Sommers, T.; Corban, C.; Sengupta, N.; Jones, M.; Cheng, V.; Bollom, A.; Nurko, S.; Kelley, J.; Lembo, A. Emergency Department Burden of Constipation in the United States from 2006 to 2011. American Journal of Gastroenterology 2015, 110, 572–579. [Google Scholar] [CrossRef]
- An, J.; Jiao, C.; Guo, J.; Zhang, L.; Li, Z.; Liu, G.; Zhang, Y.; Liu, W. The Association of ZJU Index with Bowel Health in US Participants: A Cross-Sectional Study. Eur. J. Med. Res. 2025, 30, 1165. [Google Scholar] [CrossRef]
- Purayil, S.; Yi, J.; Fletcher, D.; Denke, L.; Atem, F.D.; Zhang, Y.; Wingfield, S. Exploring the Correlation between Constipation and Delirium among Hospitalized Patients Aged 65 Years and Older. Geriatr. Nurs. (Minneap) 2025, 67, 103724. [Google Scholar] [CrossRef]
- Aziz, I.; Whitehead, W.E.; Palsson, O.S.; Törnblom, H.; Simrén, M. An Approach to the Diagnosis and Management of Rome IV Functional Disorders of Chronic Constipation. Expert Rev. Gastroenterol. Hepatol. 2020, 14, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Assadollahzadeh, A.; Ravari, A.; Assadollahi, Z.; Mirzaei, T. Comparative Effects of Traditional Thai Abdominal Massage versus Prune Consumption on Constipation and Quality of Life in Elderly Women: A Randomized Controlled Trial. Aging Clin. Exp. Res. 2025, 37. [Google Scholar] [CrossRef] [PubMed]
- Suares, N.C.; Ford, A.C. Prevalence of, and Risk Factors for, Chronic Idiopathic Constipation in the Community: Systematic Review and Meta-Analysis. Am. J. Gastroenterol. 2011, 106, 1582–1591. [Google Scholar] [CrossRef] [PubMed]
- Bytzer, P.; Howell, S.; Leemon, M.; Young, L.J.; Jones, M.P.; Talley, N.J. Low Socioeconomic Class Is a Risk Factor for Upper and Lower Gastrointestinal Symptoms: A Population Based Study in 15 000 Australian Adults. Gut 2001, 49, 66. [Google Scholar] [CrossRef]
- Bharucha, A.E.; Pemberton, J.H.; Locke, G.R. American Gastroenterological Association Technical Review on Constipation. Gastroenterology 2013, 144, 218–238. [Google Scholar] [CrossRef]
- Liu, R.; Chao, A.; Wang, K.; Wu, J. Incidence and Risk Factors of Medical Complications and Direct Medical Costs after Osteoporotic Fracture among Patients in China. Arch. Osteoporos. 2018, 13. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, H.; Guo, Z.; Li, S.; Fu, Q. Inverse Association of Alcohol Consumption Patterns with Constipation and Diarrhea: A Population-Based Cross-Sectional Study. BMC Gastroenterol. 2025, 25, 865. [Google Scholar] [CrossRef]
- Zhu, H.T.; Wang, H.F.; Zhang, Q.; Bai, J.F.; Wang, M.; Du, S.Y.; Zhang, Y.L. Comprehensive Microbial and Clinical Profiling of Functional Constipation: A Stratified Comparative Study of Age and Constipation Subtype. World J. Gastroenterol. 2025, 31. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Yang, X.J.; Zhu, H.M.; Tang, Z.; Li, B.Y.; Zhao, D.D. Epidemiological Study of Elderly Constipation in Beijing. World J. Gastroenterol. 2015, 21, 13368. [Google Scholar] [CrossRef] [PubMed]
- Segura-Bayona, C.; Vidal-Alaball, J.; Ramírez-Morros, A.; Miró-Catalina, Q.; Ruiz-Comellas, A. A Primary Care Program Based on Behavioral Reeducation and Abdominal Massage for Improving the Symptoms of Chronic Constipation: Protocol for a Randomized Controlled Trial. JMIR Res. Protoc. 2025, 14. [Google Scholar] [CrossRef]
- Kilgore, A.; Khlevner, J. Functional Constipation: Pathophysiology, Evaluation, and Management. Aliment. Pharmacol. Ther. 2024, 60 Suppl 1, S20–S29. [Google Scholar] [CrossRef]
- Tian, Y.; Li, Y.; Yi, T.; Wang, Q.; Jiang, X.; He, Z.X. Comparison of Tc-99 m Scintigraphic Method and Radiological Method for Colon Transmit Assessment in Patients with Functional Constipation. BMC Gastroenterol 2025, 25. [Google Scholar] [CrossRef]
- Rajasekaran, M.R.; Kanoo, S.; Fu, J.; Nguyen, M.U.L.; Bhargava, V.; Mittal, R.K. Age-Related External Anal Sphincter Muscle Dysfunction and Fibrosis: Possible Role of Wnt/β-Catenin Signaling Pathways. Am. J. Physiol. Gastrointest. Liver Physiol. 2017, 313, G581–G588. [Google Scholar] [CrossRef]
- Yang, X.; Li, W.; Xiao, Q.; Du, S.; Wang, X.; Zhang, Y.; Li, S. Predicting the Risk of Postoperative Constipation in Middle-Aged and Elderly Patients with Lower Limb Fractures Using Machine Learning Algorithms. PLoS One 2025, 20, e0336466. [Google Scholar] [CrossRef]
- Sadeghi, A.; Akbarpour, E.; Majidirad, F.; Bor, S.; Forootan, M.; Hadian, M.R.; Adibi, P. Dyssynergic Defecation: A Comprehensive Review on Diagnosis and Management. The Turkish Journal of Gastroenterology 2023, 34, 182. [Google Scholar] [CrossRef]
- Zhao, X.; Wang, X.; Quan, L. Association between Dietary Inflammatory Index and Energy-Adjusted Dietary Inflammatory Index and Constipation in US Adults. BMC Gastroenterol. 2024, 24. [Google Scholar] [CrossRef] [PubMed]
- Tazaki, T.; Yamada, H.; Sato, R.; Ishii, H.; Sugita, S.; Yanagihara, H.; Nakamura, D.; Takashio, O.; Inamoto, A.; Iwanami, A. Constipation-Associated Factors in Outpatients with Schizophrenia: A Multicenter Questionnaire Survey. Neuropsychopharmacol. Rep. 2024, 44, 604–613. [Google Scholar] [CrossRef]
- Nakagawa, H.; Takeshima, T.; Ozaka, A.; Sasaki, S.; Hamaguchi, S.; Fukuhara, S. The Association between Sarcopenia and Constipation Onset in Community-Dwelling Older Adults in Japan. Intern. Med. 2025, 64, 1965–1970. [Google Scholar] [CrossRef] [PubMed]
- Chiang, C.Y.; Lo, S.C.; Beckstead, J.W.; Yang, C.Y. Associations between Constipation Risk and Lifestyle, Medication Use, and Affective Symptoms in Patients with Schizophrenia: A Multicenter Cross-Sectional Study. Soc. Psychiatry Psychiatr. Epidemiol. 2025, 60, 427–440. [Google Scholar] [CrossRef]
- Zheng, T.; Tavares, L.C.; D’Amato, M.; Marques, F.Z. Constipation Is Associated with an Increased Risk of Major Adverse Cardiac Events in a UK Population. Am. J. Physiol. Heart Circ. Physiol. 2024, 327, H956–H964. [Google Scholar] [CrossRef]
- Xiao, C.F.; Li, Y.F.; Sun, Y.Y.; Meng, L.Y.; Wu, J.W.; Wang, M.; Cao, Y.Q.; Wang, C.; Yao, Y.B. Influence of the Examination Position and Distension Medium on the Rectal Sensory Test in Patients with Functional Constipation. BMC Gastroenterol. 2024, 24. [Google Scholar] [CrossRef]
- Guo, M.; Yao, J.; Yang, F.; Liu, W.; Bai, H.; Ma, J.; Ma, X.; Zhang, J.; Fang, Y.; Miao, Y.; et al. The Composition of Intestinal Microbiota and Its Association with Functional Constipation of the Elderly Patients. In Future Microbiol.; REQUESTEDJOURNAL:JOURNAL:IFMB20; WGROUP:STRING:PUBLICATION, 2020; Volume 15, pp. 163–175. [Google Scholar] [CrossRef]
- Strasser, B.; Ticinesi, A. Intestinal Microbiome in Normal Ageing, Frailty and Cognition Decline. Curr. Opin. Clin. Nutr. Metab. Care 2023, 26, 8–16. [Google Scholar] [CrossRef]
- Zhang, Z.J.; Gao, R.; Lu, Y.T.; Zuo, Z.L.; Li, Y.H.; Liu, S.; Song, S.Y.; Wang, Y.; Lai, H. Factors Affecting Dysbiosis of the Gut Microbiota in the Elderly and the Progress of Interventions in Traditional Chinese and Western Medicine. Front. Cell. Infect. Microbiol. 2025, 15, 1529347. [Google Scholar] [CrossRef]
- Zou, L.; da Zheng, X.; Wang, C.G.; Ma, T. Factors Associated with Constipation after Total Hip Arthroplasty and Nursing Countermeasures. Medicine 2025, 104, e45226. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Lim, J.; Baek, J.Y.; Lee, E.; Jung, H.W.; Jang, I.Y. Status of Constipation and Its Association with Sarcopenia in Older Adults: A Population-Based Cohort Study. Int. J. Environ. Res. Public Health 2021, 18. [Google Scholar] [CrossRef]
- Ishida, T.; Kawada, K.; Jobu, K.; Hamada, T.; Kubo, T.; Fukuda, H.; Hyohdoh, Y.; Kawai, K.; Nakaoka, Y.; Yabe, T.; et al. Impact of Non-cardiovascular Factors on Cardiovascular Death in Acute Decompensated Heart Failure: Enhancing Predictive Models with Frailty and Comorbid Constipation—the Constipation and Frailty Augmented AHEAD Score. Geriatr. Gerontol. Int. 2025, 25, 1341. [Google Scholar] [CrossRef]
- Qi, R.; Zhai, H.; Xu, D.; Wen, Y.; Xu, S. Association between the Planetary Health Diet Index and Chronic Constipation and Diarrhea Risk in General Population: A Cross-Sectional Analysis of NHANES. Medicine 2025, 104, e45468. [Google Scholar] [CrossRef] [PubMed]
- Hamano, J.; Higashibata, T.; Kessoku, T.; Kajiura, S.; Hirakawa, M.; Oyamada, S.; Ariyoshi, K.; Yamada, T.; Yamamoto, Y.; Takashima, Y.; et al. Naldemedine for Opioid-Induced Constipation in Patients With Cancer: A Multicenter, Double-Blind, Randomized, Placebo-Controlled Trial. J. Clin. Oncol. 2024, 42. [Google Scholar] [CrossRef]
- Liu, K.; Chen, A.; Rajaram, P.; Buning, G.; Lee, A.A.; Singh, P. PAMORAs in Opioid-Induced Constipation: Are We Following the Guidelines? Clin. Transl. Gastroenterol. 2025, 16, e00876. [Google Scholar] [CrossRef]
- Yimer, B.B.; Soomro, M.; McBeth, J.; Medina, C.R.R.; Lunt, M.; Dixon, W.G.; Jani, M. Comparative Risk of Severe Constipation in Patients Treated with Opioids for Non-Cancer Pain: A Retrospective Cohort Study in Northwest England. BMC Med. 2025, 23. [Google Scholar] [CrossRef]
- Thomé, B.I.; Scharan, K.O.; Assis, G.M.; Moser, A.D. de L. Unraveling the Frequency of Intestinal Constipation in Woman with Urinary Incontinence: A Descriptive Observational Study. Rev. Bras. Ginecol. Obstet. 2025, 47. [Google Scholar] [CrossRef] [PubMed]
- Leta, V.; Zinzalias, P.; Batzu, L.; Mandal, G.; Staunton, J.; Jernstedt, F.; Rosqvist, K.; Timpka, J.; van Vliet, T.; Trivedi, D.; et al. Effects of a Four-Strain Probiotic on Gut Microbiota, Inflammation, and Symptoms in Parkinson’s Disease: A Randomized Clinical Trial. Mov. Disord. 2025, 40, 2710–2721. [Google Scholar] [CrossRef]
- Umamahesan, C.; Pilcicka, A.; Yick, J.; Baker, K.; Smith, M.; Taylor, D.; Ma, Y.; Mullish, B.H.; Marchesi, J.R.; Gilbert, S.; et al. Interplay of Constipation, Intestinal Barrier Dysfunction and Fungal Exposome in Aetiopathogenesis of Parkinson’s Disease: Hypothesis with Supportive Data. Biochem. J. 2025, 482, 807–821. [Google Scholar] [CrossRef] [PubMed]
- Kutsuna, A.; Nishiyama, Y.; Sakamoto, Y.; Suzuki, F.; Hayashi, T.; Fujisawa, Y.; Suzuki, K.; Aoki, J.; Kimura, K. Constipation in Patients with Acute Ischemic Stroke: A Single-Center Retrospective Analysis. J. Nippon Med. Sch. 2025, 92, 154–162. [Google Scholar] [CrossRef]
- Li, M. qi; Li, Y.; Lam, W.; Yeung, W.F.; Ho, Y.S.; Li, J. ying; Sun, T.C.; Yuen, S.; Hu, Y. le; Yorke, J. Home-Based Acupressure for Managing Constipation and Subjective Well-Being in Spinal Cord Injury Survivors: A Randomized Controlled Trial. J. Integr. Med. 2025, 23, 660–669. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; Lu, L.; Liu, Y.; Yan, Y.; La, R.; Wu, Q.; Xu, J.; Zhou, X. The Association between Dietary Vitamin B1 Intake and Constipation: A Population-Based Study. BMC Gastroenterol. 2024, 24. [Google Scholar] [CrossRef]
- Komori, S.; Akiyama, J.; Tatsuno, N.; Yamada, E.; Izumi, A.; Hamada, M.; Seto, K.; Nishiie, Y.; Suzuki, K.; Hisada, Y.; et al. Prevalence and Risk Factors of Constipation Symptoms among Patients Undergoing Colonoscopy: A Single-Center Cross-Sectional Study. Digestion 2024, 105, 299–309. [Google Scholar] [CrossRef]
- Xian, X.; Wang, X.; Liu, J.; Yang, H. Investigation of Functional Constipation in Elderly Inpatients and Analysis of Its Influencing Factors: A Cross-Sectional Study. Medicine 2024, 103, e39624. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Chen, S.; Zhu, Y.; Wu, M.; Lu, B.; Zhou, X.; Zhu, Y.; Xu, X.; Liu, H.; Zhu, F.; et al. Intestinal Microbiome Changes and Mechanisms of Maintenance Hemodialysis Patients with Constipation. Front. Cell. Infect. Microbiol. 2024, 14. [Google Scholar] [CrossRef]
- Huang, K.Y.; Yu, Z.Z.; Tu, J.J.; Tang, X.Y.; Huang, J.M.; Lu, T.M.; Lu, Y.Q.; Huang, M.C.; Zhou, J.; Maier, A.B.; et al. Positive Association between Constipation and Mild Cognitive Impairment in Elders: A Cross-Sectional Study. Medicine 2024, 103, e39943. [Google Scholar] [CrossRef]
- Martin, C.R.; Osadchiy, V.; Kalani, A.; Mayer, E.A. The Brain-Gut-Microbiome Axis. Cell. Mol. Gastroenterol. Hepatol. 2018, 6, 133. [Google Scholar] [CrossRef]
- Yuan, C.; He, Y.; Xie, K.; Feng, L.; Gao, S.; Cai, L. Review of Microbiota Gut Brain Axis and Innate Immunity in Inflammatory and Infective Diseases. Front. Cell. Infect. Microbiol. 2023, 13. [Google Scholar] [CrossRef]
- Xie, J.; Bruggeman, A.; De Nolf, C.; Vandendriessche, C.; Van Imschoot, G.; Van Wonterghem, E.; Vereecke, L.; Vandenbroucke, R.E. Gut Microbiota Regulates Blood-Cerebrospinal Fluid Barrier Function and Aβ Pathology. EMBO J 2023, 42. [Google Scholar] [CrossRef] [PubMed]
- Esra Bozkurt, M.; Erdogan, T.; Fetullahoglu, Z.; Ozkok, S.; Kilic, C.; Bahat, G.; Akif Karan, M. Investigation of the Prevalence of Functional Constipation and Its Related Factors for in Older Outpatients. Acta Gastroenterol. Belg 2024, 87, 361–365. [Google Scholar] [CrossRef]
- Rudroff, C.; Madukkakuzhy, J.; Otten, J.; Schroer, J.; Richards, D.; Ludwig, S.; Henning, M. Improvement in Functional and Mental Outcomes after Resection Rectopexy for Obstructive Defecation Syndrome ODS. Sci. Rep. 2025, 15. [Google Scholar] [CrossRef]
- Namiuchi, S.; Sunamura, S.; Tanita, A.; Ogata, T.; Noda, K.; Takii, T.; Nitta, Y.; Yoshida, S. Effect of Constipation on Hospitalization Due to Heart Failure in Patients after Myocardial Infarction: A Retrospective Cohort Study. BMC Cardiovasc. Disord. 2025, 25. [Google Scholar] [CrossRef] [PubMed]
- Al Alawi, A.M.; Al Nou’mani, J.; Al Abri, N.; Al Sabbri, M.; Al-Maqbali, J.S. Impact of Constipation on Health Outcomes in Medically Hospitalized Patients: A Prospective Study on Laxative Use and Health Care Outcomes. Medicina (Kaunas) 2024, 60. [Google Scholar] [CrossRef]
- Xu, Y.; Xue, Z. The Relationship between Food Components and Constipation: A Cross-Sectional Study. BMC Gastroenterol 2024, 24. [Google Scholar] [CrossRef]
- Yu, Z.; Guo, M.; Bai, X.; Ruan, G.; Sun, Y.; Han, W.; Yang, H. Exploring the Association between Cardiovascular Health and Bowel Health. Sci. Rep. 2024, 14. [Google Scholar] [CrossRef] [PubMed]
- Kido, M.; Inoue, K.; Kobayashi, R.; Takashima, N.; Shoda, K.; Sugino, S.; Kato, R.; Itoh, Y. Seasonal Variations and a Demographic Analysis of Digital Evacuation Incidence for Constipation Management: A Japanese Population-Based Cohort Study. Intern. Med. 2025, 64, 1623–1632. [Google Scholar] [CrossRef]
- Li, S.; Wang, S.; Li, S.; Liu, C.; Sun, J.; Cao, J.; Cui, X.; Han, Q.; Li, Z.; Wu, F. Brain–Gut–Microbiota Axis: An Effective Target of Abdominal Acupuncture Therapy for Post-Stroke Functional Constipation. Complement. Ther. Med. 2025, 95, 103286. [Google Scholar] [CrossRef]
- Shen, S.; Feng, H.; Wang, C.; Yang, Y.; Chen, Z.; Wang, X.; Li, J. Association between the Dietary Index of Gut Microbiota and Abnormal Bowel Symptoms in U.S. Adults: A Cross-Sectional Study Based on NHANES 2007-2010. BMC Gastroenterol 2025, 25. [Google Scholar] [CrossRef]
- Huang, J.F.; Xiong, Y.J.; Da Meng, X.; Lv, T. Combined Association of Chewing Capacity and Depression with Constipation: A Cross-Sectional Study. BMC Gastroenterol 2025, 25. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Vimala, R.; Chui, P.L.; Hilmi, I.N. An Abdominal Vibration Combined with Walking Exercise (AVCWE) Program for Older Patients with Constipation: Development and Feasibility Study. Saudi J. Gastroenterol. 2024, 30, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.; Qin, W.; Liao, J.; Liu, Y.; Yan, L. Application of Traditional Chinese Medicine Nursing Technology: Traditional Chinese Medicine Ironing Combined with Traditional Chinese Medicine Massage in Improving Functional Constipation in Elderly Patients. Medicine 2024, 103, e39863. [Google Scholar] [CrossRef]
- Boangmanalu, E.S.; Masfuri, M.; Adam, M.; Nining, S.; Banna, T.; Pulungan, I.M. Swedish Abdominal Massage against Warm Water Therapy on Postoperative Orthopaedic Surgery Constipation: A Comparison Quasi-Experimental Study. F1000Res 2025, 13. [Google Scholar] [CrossRef] [PubMed]
- Brenner, D.M.; Rao, S.S.C.; Curtin, B.; Quigley, E. Real-World Evidence of the Long-Term Clinical Utility of a Vibrating Capsule in the Management of Chronic Idiopathic Constipation. Clin. Transl. Gastroenterol. 2025, 16. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xue, Y.; Chen, Q.; Ling, Y.; Zhou, H.; Li, M.; Xu, X.; Zeng, Y. Clinical Relevance of Transperineal Ultrasound Compared with Anorectal Manometry for the Evaluation of Female Patients with Obstructive Defecation Syndrome. BMC Gastroenterol. 2025, 25. [Google Scholar] [CrossRef]
- Zhang, G.; Wang, S.; Ma, P.; Wang, T.; Sun, X.; Zhang, X.; Li, H.; Pan, J. Association of Habitual Sleep Duration with Abnormal Bowel Symptoms: A Cross-Sectional Study of the 2005-2010 National Health and Nutrition Examination Survey. J. Health Popul. Nutr. 2024, 43. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, G.B.; Han, K.; Jung, E.J.; Suh, H.J.; Jo, K. Efficacy and Safety of Galacto-Oligosaccharide in the Treatment of Functional Constipation: Randomized Clinical Trial. Food Funct. 2024, 15, 6374–6382. [Google Scholar] [CrossRef]
- Suzuki, N.; Takeda, Y.; Kabuto, A.; Konishi, T.; Konishi, T.; Sumino, F.; Iwata, H.; Iwagami, M.; Kouchi, Y.; Hitomi, Y.; et al. Efficacy of Tenapanor in Managing Hyperphosphatemia and Constipation in Hemodialysis Patients: A Randomized Controlled Trial. PLoS One 2025, 20. [Google Scholar] [CrossRef]
- Nakajima, A.; Unno, A.; Ota, T.; Shoji, A.; Uenishi, T.; Igarashi, A. Socioeconomic Value of Treatments for Chronic Idiopathic Constipation in Japan. BMC Gastroenterol 2025, 25. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Zhang, J.; Yang, C.; Dong, X.; Wang, Y. Evaluation of Post-Market Adverse Events of Lubiprostone: A Real-World Adverse Event Analysis from the FAERS Database. BMC Gastroenterol 2025, 25. [Google Scholar] [CrossRef]
- Cash, B.D.; Lu, M.; Lembo, A.; Feuerstadt, P.; Nguyen, L.; Terasawa, E.; Ayyagari, R.; Du, S.; Pi, S.; Westermeyer, B.; et al. A Real-World Study of Persistence and Adherence to Prescription Medications in Patients with Chronic Idiopathic Constipation in the United States. J. Manag. Care Spec. Pharm. 2024, 30, 1136–1148. [Google Scholar] [CrossRef]
- Singh, R.G.; Aoki, F.; Rodriguez-Palmero Seuma, M.; Aguilo, M.; Washida, M.; Espadaler-Mazo, J.; Al-Wahsh, H.; Crowley, D.C.; Guthrie, N.; Evans, M.; et al. Efficacy of Probiotic Supplementation with Lactiplantibacillus Plantarum Strains on Gastrointestinal Tract Function - A Randomized Controlled Trial. J. Diet. Suppl. 2025, 22, 549–570. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Zhou, Y.; Xu, X.; Chen, S.; Zhang, S.; Jiang, N.; Liu, Z.; Zhang, J.; Luo, Z.; Zhang, X.; et al. Improvement of Post-Surgery Constipation in Patients with Fractures by Lactobacillus Rhamnosus JYLR-127: A Single-Blind Randomized Controlled Trial. Nutrients 2024, 16. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zhang, Y.; Yue, F.; Yu, X.; Chen, Y.; Ma, X. Weizmannia Coagulans BC01 Supplementation Improved Functional Constipation by Regulating Gut Microbiota: A Randomized, Placebo-Controlled, Clinical Study. Medicine 2025, 104, e46861. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhang, Q.; Zhao, W.; Chen, J.; Liu, Y.; Zhou, L.; Liu, Y.; Wang, L.; Guan, Y.; He, J.; et al. Effects and Persistence of Bifidobacterium Animalis Subsp. Lactis BL-99 and Fructooligosaccharides on Older Adults with Functional Constipation: A Randomized, Double-Blind, Placebo-Controlled Trial. J. Nutr. Health Aging 2025, 29. [Google Scholar] [CrossRef]
- Luk-In, S.; Leepiyasakulchai, C.; Saelee, C.; Keeratichamroen, A.; Srisangwan, N.; Ponprachanuvut, P.; Chammari, K.; Chatsuwan, T.; Wannigama, D.L.; Shein, A.M.S.; et al. Impact of Resistant Starch Type 3 on Fecal Microbiota and Stool Frequency in Thai Adults with Chronic Constipation Randomized Clinical Trial. Sci. Rep. 2024, 14. [Google Scholar] [CrossRef]
| Aspect | Non-Pharmacological Interventions | Pharmacological Therapy | Microbiota-Targeted Therapy |
| Primary Methods | Dietary fiber and fluid intake, abdominal massage (Thai, Swedish), postural adjustment, and vibrating capsules [8]. | Osmotic laxatives (Mg Oxide, PEG), stimulants (Senna), prosecretory agents (Elobixibat, Lubiprostone, Linaclotide), and PAMORAs (Naldemedine) [70]. | Probiotics (L. plantarum, W. coagulans) and synbiotics (Bifidobacteria + FOS) [72]. |
| Mechanism of action | Mechanical stimulation of colonic motility, induction of the gastrocolic reflex, and optimization of the anorectal angle [63]. | Increases intestinal fluid secretion via ion channels or triggers direct peristaltic stimulation via enteric nerves [70]. | Reshaping gut microbiota, producing Short-Chain Fatty Acids (SCFAs), and strengthening the intestinal barrier [72]. |
| Elderly-Specific Benefits | Enhances patient autonomy, reduces psychological distress (anxiety/depression), and provides sensory stimulation [8]. | Provides rapid symptom relief; specific agents like PAMORAs are highly effective for opioid-induced constipation (OIC) [36]. | Improves stool consistency and frequency sustainably while positively influencing the brain-gut-microbiota axis to reduce stress [72]. |
| Risks & Side Effects | Requires caregiver training for massage; effectiveness of exercise/diet alone can be limited by immobility [2]. | Risk of dependence, tolerance, electrolyte imbalance, and hypermagnesemia in patients with renal impairment [8]. | Generally safe and well-tolerated; however, some patients may experience transient diarrhea during the early evacuation of accumulated stool [68]. |
| Economic & Clinical Utility | Offers low-cost interventions that can be integrated into primary care to reduce pharmacological dependence [8]. | Elobixibat is associated with lower total costs and better QoL in Japan; productivity loss is a major indirect cost [69]. | Requires additional supplement costs but can contribute to long-term health maintenance and reduced medication burden [72]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




