Submitted:
25 February 2026
Posted:
28 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
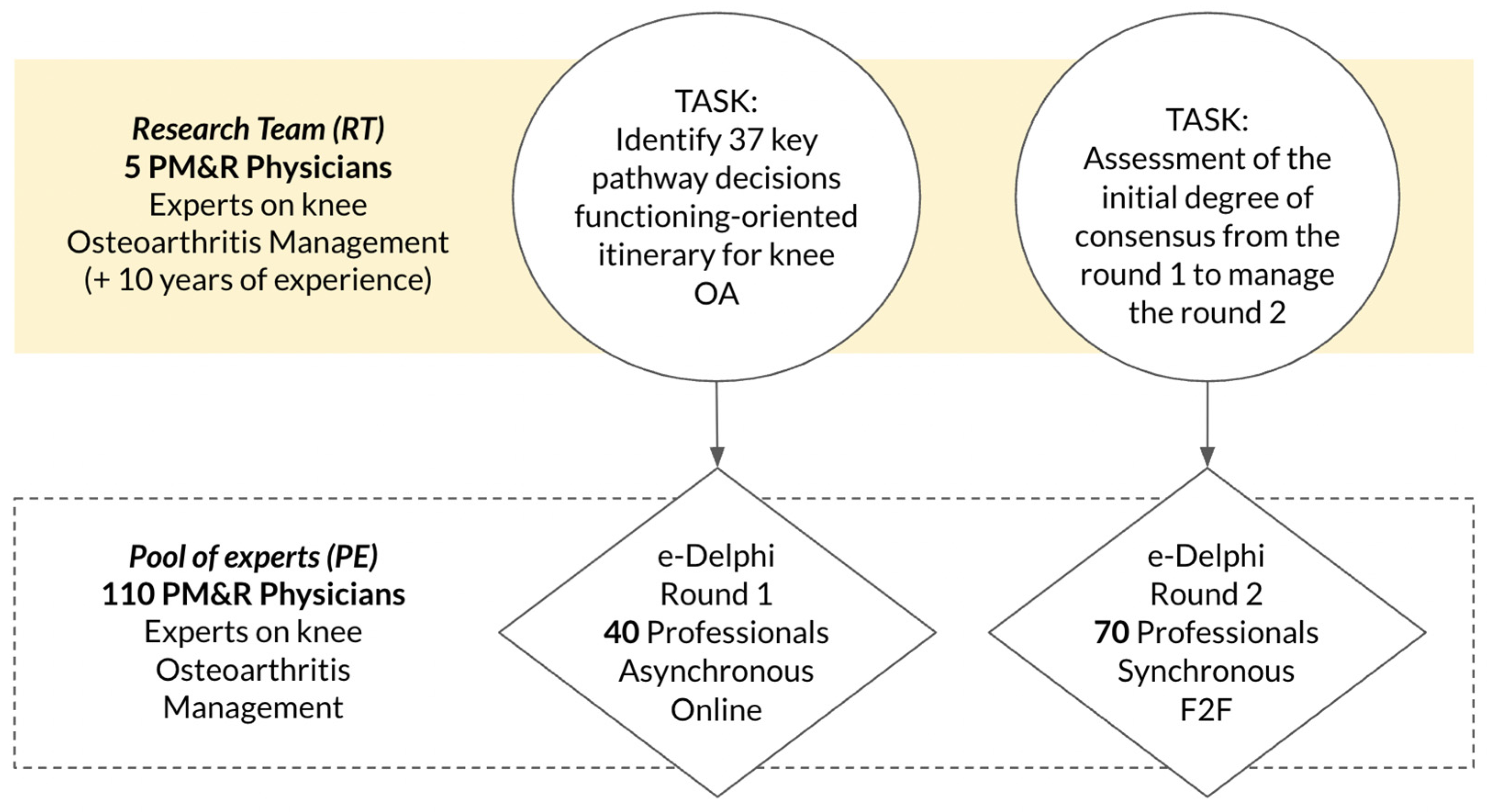
2.1. Study Design and Digital Platform
2.2. Questionnaire Development
2.3. Participants, Eligibility, and Recruitment
2.4. Delphi Procedure and Rating Scale
2.5. Data Collection
2.6. Consensus Criteria
2.7. Statistical Analysis
2.8. Ethical and Data Protection Considerations
3. Results
3.1. Delphi participants Rounds 1 and 2
3.2. Construction of the Function-Oriented KOA Rehabilitation Pathway
| # | Item (full text) | Mean | Δ | Median | SD | IQR | ||||
| R1 | R2 | Mean | R1 | R2 | R1 | R2 | R1 | R2 | ||
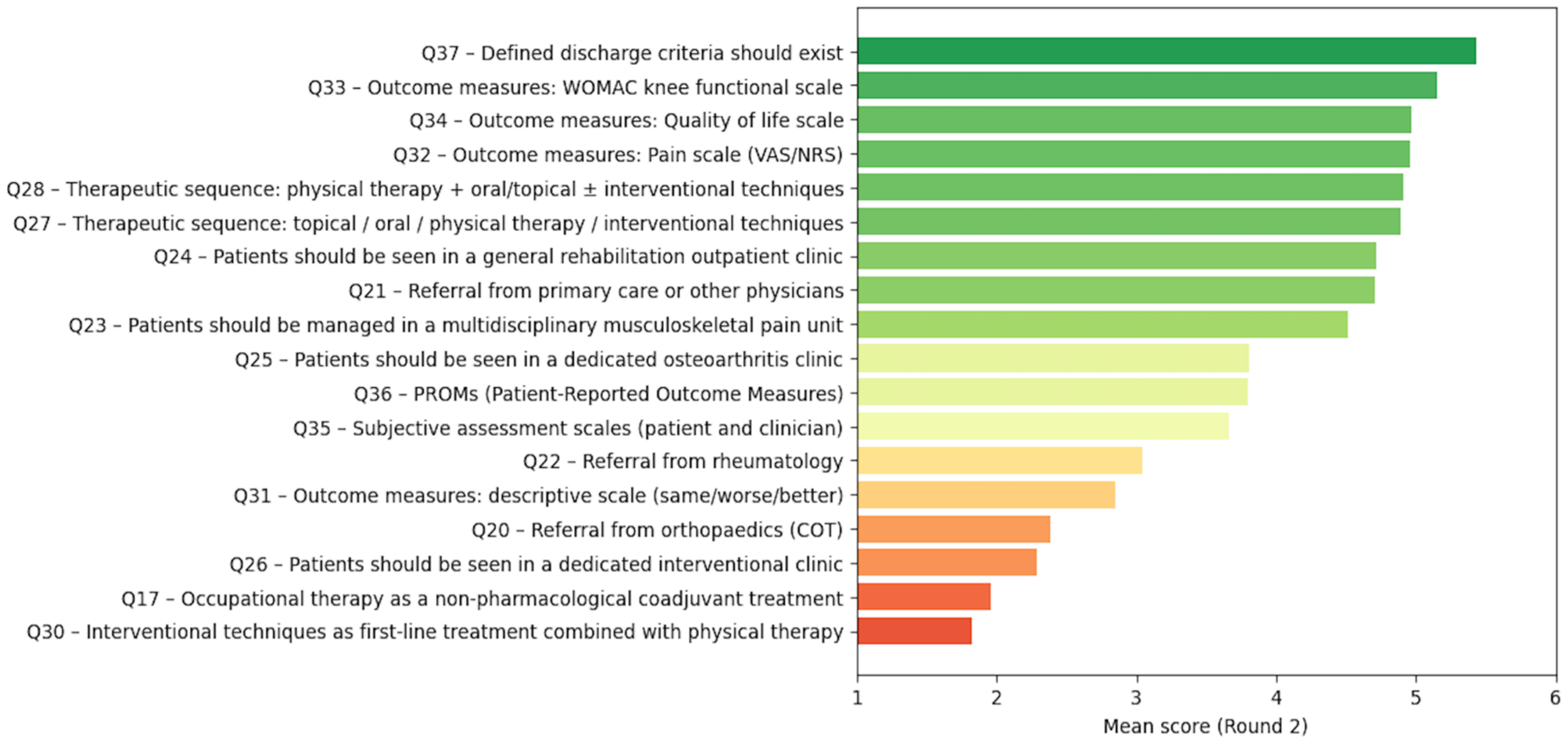
| Q37 | Ideally, defined discharge criteria should exist. | 5.20 | 5.43 | 0.23 | 5 | 5 | 0.72 | 0.63 | 1 | 1 |
| Q33 | Ideally, outcome measures should include a WOMAC knee functional scale. | 4.88 | 5.15 | 0.27 | 5 | 5 | 1.02 | 0.63 | 2 | 1 |
| Q34 | Ideally, outcome measures should include a quality of life scale. | 4.53 | 4.97 | 0.44 | 5 | 5 | 0.85 | 0.74 | 1 | 0 |
| Q32 | Ideally, outcome measures should include a pain scale (VAS/NRS). | 4.75 | 4.96 | 0.21 | 5 | 5 | 1.06 | 0.83 | 2 | 1 |
| Q28 | In knee osteoarthritis, the therapeutic sequence should include physical therapy in combination with oral and/or topical therapy and/or interventional techniques. | 4.53 | 4.91 | 0.38 | 5 | 5 | 0.93 | 0.79 | 1 | 0 |
| Q27 | In knee osteoarthritis, the therapeutic sequence should be topical therapy, oral therapy, physical therapy, and interventional techniques. | 4.63 | 4.89 | 0.26 | 5 | 5 | 1.08 | 0.99 | 1 | 2 |
| Q24 | Ideally, patients with knee osteoarthritis should be seen in a general rehabilitation outpatient clinic. | 4.35 | 4.72 | 0.37 | 5 | 5 | 1.19 | 1.04 | 1 | 1 |
| Q21 | Ideally, patients with knee osteoarthritis should be referred from primary care or other physicians. | 4.61 | 4.71 | 0.10 | 5 | 5 | 1.14 | 1.00 | 1 | 1 |
| Q23 | Ideally, patients with knee osteoarthritis should be managed in a multidisciplinary musculoskeletal pain unit. | 4.43 | 4.51 | 0.08 | 5 | 5 | 1.47 | 1.17 | 3 | 1 |
| Q25 | Ideally, patients with knee osteoarthritis should be seen in a dedicated osteoarthritis clinic. | 3.95 | 3.81 | 0.14 | 4 | 4 | 1.18 | 1.21 | 2 | 2 |
| Q36 | Ideally, outcome measures should include PROMs (Patient-Reported Outcome Measures). | 4.05 | 3.80 | 0.25 | 4 | 4 | 1.11 | 1.24 | 2 | 2 |
| Q35 | Ideally, outcome measures should include subjective assessment scales (patient and clinician). | 3.85 | 3.66 | 0.19 | 4 | 4 | 1.12 | 1.08 | 2 | 2 |
| Q22 | Ideally, patients with knee osteoarthritis should be referred from rheumatology. | 2.83 | 3.04 | 0.21 | 3 | 3 | 0.83 | 1.12 | 1 | 2 |
| Q31 | Ideally, outcome measures should include a descriptive scale (same/worse/better). | 2.93 | 2.85 | 0.08 | 3 | 3 | 0.92 | 0.97 | 1 | 1 |
| Q20 | Ideally, patients with knee osteoarthritis should be referred from orthopaedics (COT). | 2.15 | 2.38 | 0.23 | 2 | 2 | 0.82 | 0.89 | 1 | 1 |
| Q26 | Ideally, patients with knee osteoarthritis should be seen in a dedicated interventional clinic. | 2.83 | 2.29 | 0.54 | 3 | 2 | 0.98 | 0.89 | 2 | 1 |
| Q30 | Interventional techniques should be used as first-line treatment combined with physical therapy. | 2.08 | 1.82 | 0.26 | 2 | 2 | 0.73 | 0.83 | 0 | 1 |
| Mean | Δ | Median | SD | IQR | ||||||
| # | Item (full text) | R1 | R2 | Mean | R1 | R2 | R1 | R2 | R1 | R2 |
| Q15 | Ideally, treatment should include therapeutic exercise | 5.52 | 5.71 | 0.19 | 6 | 6 | 0.71 | 0.52 | 0 | 0 |
| Q14 | Ideally, treatment should include weight control | 5.41 | 5.65 | 0.24 | 6 | 6 | 0.76 | 0.60 | 0 | 0 |
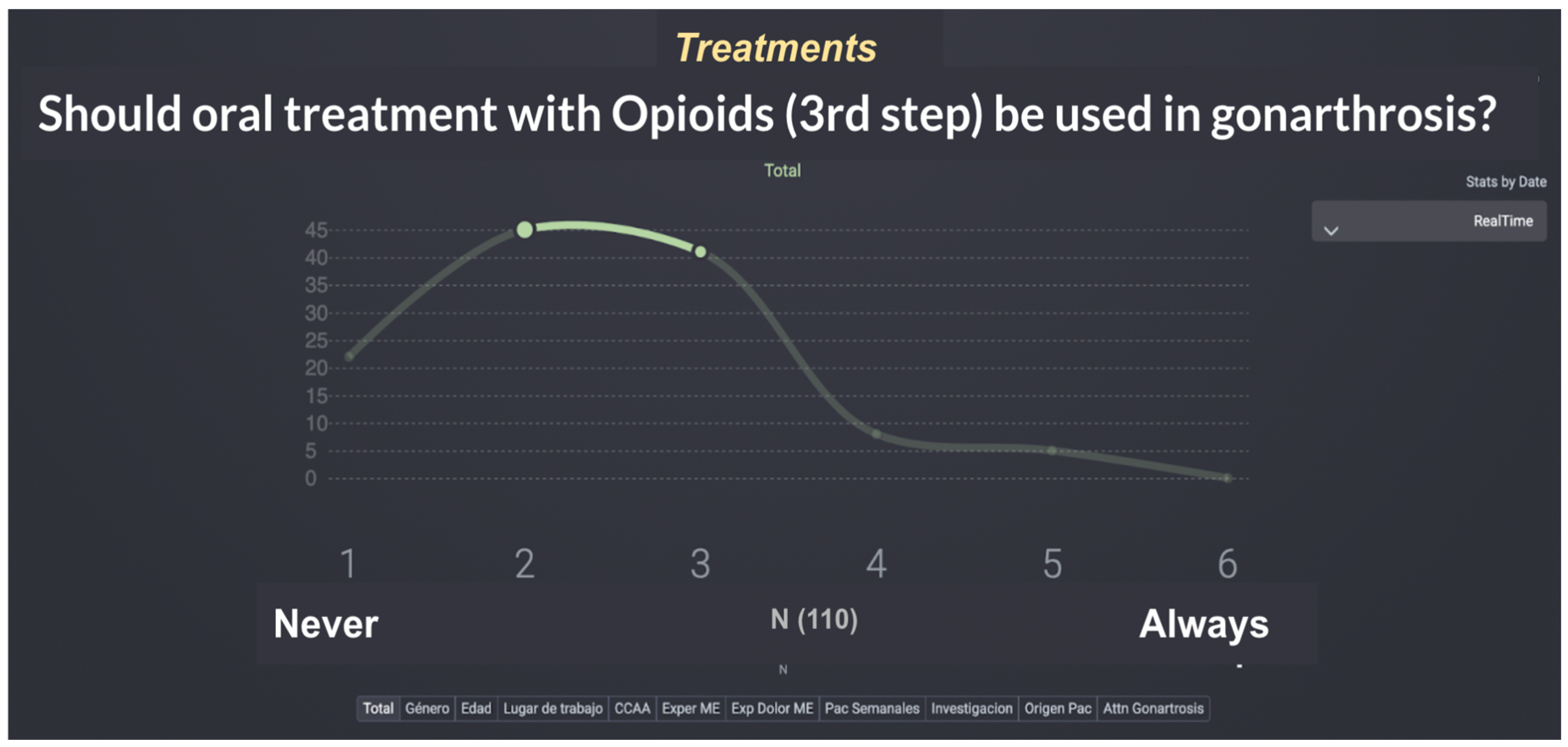
| Q9 | Ideally, treatment should include oral strong opioids | 2.32 | 2.05 | 0.27 | 2 | 2 | 0.94 | 0.81 | 1 | 1 |
| Q18 | Ideally, treatment should include orthoses | 2.56 | 2.40 | 0.16 | 2 | 2 | 1.01 | 0.95 | 1 | 1 |
| Q17 | Ideally, treatment should include occupational therapy | 2.48 | 2.36 | 0.12 | 2 | 2 | 0.98 | 0.88 | 1 | 1 |
| Q8 | Ideally, treatment should include oral tramadol | 3.21 | 3.10 | 0.11 | 3 | 3 | 1.02 | 0.93 | 1 | 1 |
| Q19 | Ideally, treatment should include heat or cold therapy | 3.98 | 4.07 | 0.09 | 4 | 4 | 1.07 | 1.00 | 2 | 2 |
| Q7 | Ideally, treatment should include oral NSAIDs | 4.02 | 4.18 | 0.16 | 4 | 4 | 1.12 | 1.05 | 2 | 2 |
| Q13 | Ideally, treatment should include topical capsaicin | 3.22 | 3.15 | 0.07 | 3 | 3 | 1.03 | 1.02 | 2 | 2 |
| Q12 | Ideally, treatment should include topical NSAIDs | 4.01 | 3.86 | 0.15 | 4 | 4 | 1.08 | 1.16 | 2 | 2 |
| Q11 | Ideally, treatment should include chondroitin or glucosamine | 3.25 | 3.12 | 0.13 | 3 | 3 | 1.15 | 1.18 | 2 | 2 |
| Q10 | Ideally, treatment should include duloxetine | 3.08 | 3.01 | 0.07 | 3 | 3 | 1.06 | 1.12 | 2 | 2 |
| Q6 | Ideally, treatment should include classical analgesic electrotherapy | 3.11 | 3.02 | 0.09 | 3 | 3 | 1.09 | 1.15 | 2 | 2 |
| Q16 | Ideally, treatment should include physiotherapy | 3.04 | 2.92 | 0.12 | 3 | 3 | 1.14 | 1.19 | 2 | 2 |
| # | Mean | Δ | Median | SD | IQR | |||||
| # | Item (full text) | R1 | R2 | Mean | R1 | R2 | R1 | R2 | R1 | R2 |
| Q1 | The ideal management of KOA patients should include early evaluation by PM&R specialists | 5.34 | 5.58 | 0.24 | 6 | 6 | 0.81 | 0.63 | 1 | 0 |
| Q2 | KOA management should prioritise functional recovery as a primary clinical objective | 5.46 | 5.69 | 0.23 | 6 | 6 | 0.77 | 0.58 | 1 | 0 |
| Q3 | KOA patient care should be coordinated through multidisciplinary management | 5.12 | 5.41 | 0.29 | 5 | 6 | 0.96 | 0.76 | 1 | 1 |
| Q4 | PM&R services should play a central coordinating role in KOA care pathways | 4.98 | 5.21 | 0.23 | 5 | 5 | 1.01 | 0.88 | 2 | 1 |
| Q5 | KOA clinical management should include structured clinical pathway protocols | 4.86 | 5.03 | 0.17 | 5 | 5 | 1.08 | 0.94 | 2 | 2 |
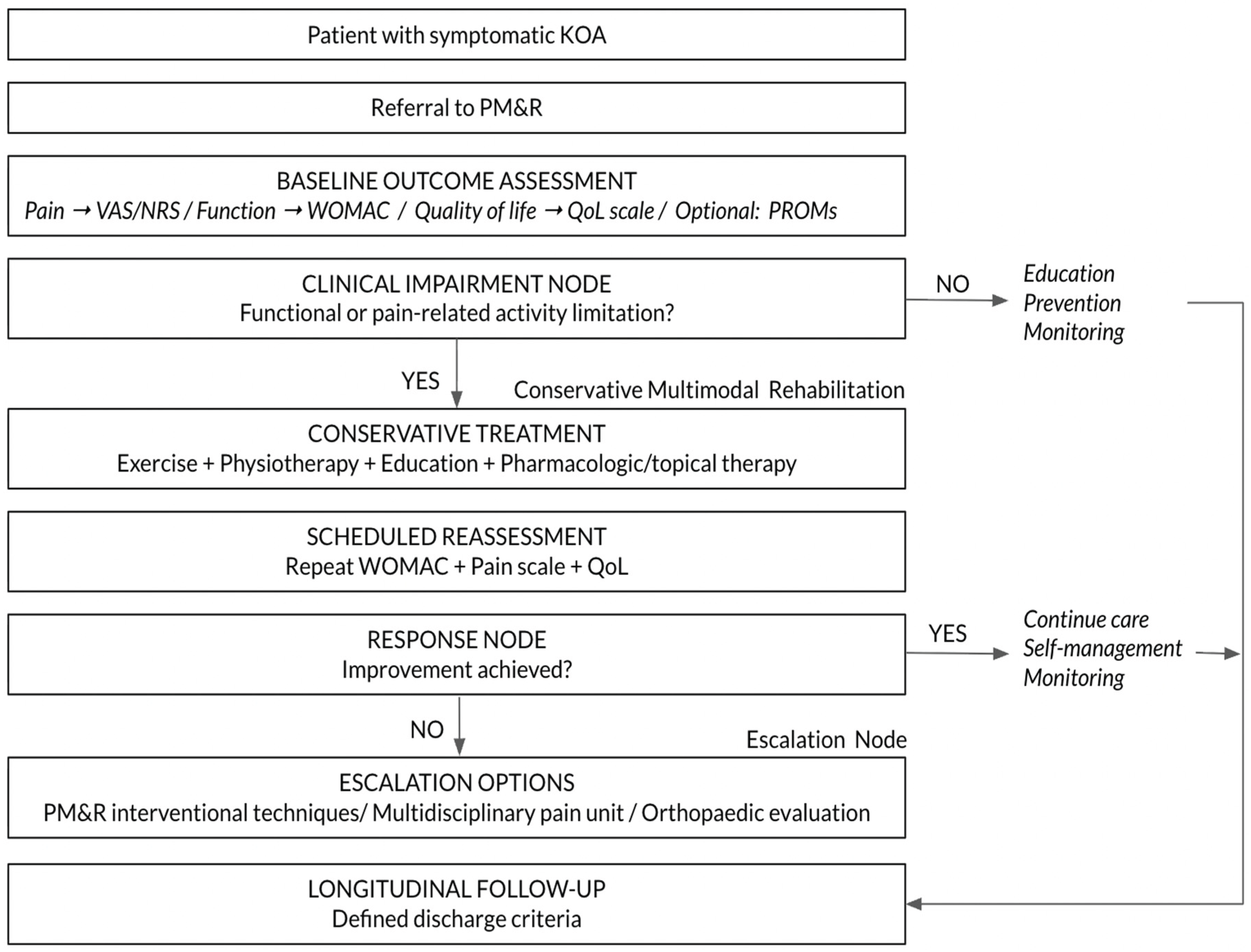
3.2.1. Entry and Referral Phase.
3.2.2. Comprehensive Functional Assessment Phase.
3.2.3. Structured Conservative Intervention Phase.
3.2.4. Escalation and Interventional Decision-Making Phase.
3.2.5. Longitudinal Monitoring and Discharge Planning Phase.
3.3. Mapping of Delphi Consensus Statements to the Rehabilitation Pathway Model
4. Discussion
4.1. Function-First Framing and Measurement Strategy
4.2. Conservative Multimodal Care as the Backbone
4.3. Escalation and Interventional Positioning
4.4. Service Configuration, Referral Routes, and Multidisciplinary Models
4.5. Discharge Criteria and Longitudinal Follow-Up as Quality Levers
4.6. Implications for Implementation
4.7. Strengths and Limitations
4.8. Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| KOA | Knee Osteoarthritis |
| PM&R | Physical Medicine and Rehabilitation |
| MSK | Musculoskeletal |
References
- Steinmetz, J.D.; et al. GBD 2021 Osteoarthritis Collaborators. Global, Regional, and National Burden of Osteoarthritis, 1990–2020 and Projections to 2050: A Systematic Analysis for the Global Burden of Disease Study 2021. Lancet Rheumatol. 2023, 5, e508–e522. [Google Scholar] [CrossRef] [PubMed]
- Moseng, T.; Vliet Vlieland, T.P.M.; Battista, S.; et al. EULAR Recommendations for the Non-Pharmacological Core Management of Hip and Knee Osteoarthritis: 2023 Update. Ann. Rheum. Dis. 2024, 83, 730–740. [Google Scholar] [CrossRef] [PubMed]
- Bannuru, R.R.; Osani, M.C.; Vaysbrot, E.E.; et al. OARSI Guidelines for the Non-Surgical Management of Knee, Hip, and Polyarticular Osteoarthritis. Osteoarthritis Cartilage 2019, 27, 1578–1589. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Care Excellence (NICE). Osteoarthritis in Over 16s: Diagnosis and Management (NG226); NICE: London, UK, 2022. [Google Scholar]
- Ackerman, I.N.; Doukas, F.; Buchbinder, R.; et al. Ensuring a Fit-for-Purpose Resource for Consumers, Clinicians and Health Services: The Updated Osteoarthritis of the Knee Clinical Care Standard. Med. J. Aust. 2024, 221, 197–200. [Google Scholar] [CrossRef] [PubMed]
- Australian Commission on Safety and Quality in Health Care (ACSQHC). Osteoarthritis of the Knee Clinical Care Standard; ACSQHC: Sydney, Australia, 2024. [Google Scholar]
- Varndell, W.; Fry, M.; Elliott, D. Applying Real-Time Delphi Methods: Development of a Pain Management Survey in Emergency Nursing. BMC Nurs. 2021, 20, 149. [Google Scholar] [CrossRef] [PubMed]
- Monguet, J.M.; Trejo, A.; Martí, T.; Escarrabill, J. Health Consensus: A Digital Adapted Delphi for Healthcare. Int. J. User-Driven Healthc. 2017, 7, 27–43. [Google Scholar] [CrossRef]
- Brophy, R.H.; Fillingham, Y.A. AAOS Clinical Practice Guideline Summary: Management of Osteoarthritis of the Knee (Nonarthroplasty), Third Edition. J. Am. Acad. Orthop. Surg. 2022, 30, e721–e729. [Google Scholar] [CrossRef] [PubMed]
- Conley, B.; Bunzli, S.; Bullen, J.; O’Brien, P.; Persaud, J.; Gunatillake, T.; Dowsey, M.M.; Choong, P.F.M.; Lin, I. Core Recommendations for Osteoarthritis Care: A Systematic Review of Clinical Practice Guidelines. Arthritis Care Res. (Hoboken) 2023, 75, 1897–1907. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, A.J.; Gray, B.; Wallis, J.A.; Taylor, N.F.; Kemp, J.L.; Hunter, D.J.; Barton, C.J. Recommendations for the Management of Hip and Knee Osteoarthritis: A Systematic Review of Clinical Practice Guidelines. Osteoarthritis Cartilage 2023, 31, 1280–1292. [Google Scholar] [CrossRef] [PubMed]
- Bonsel, J.M.; Itiola, A.J.; Huberts, A.S.; Bonsel, G.J.; Penton, H. The Use of Patient-Reported Outcome Measures to Improve Patient-Related Outcomes—A Systematic Review. Health Qual. Life Outcomes 2024, 22, 101. [Google Scholar] [CrossRef] [PubMed]
- Amat-Fernandez, C.; Pardo, Y.; Bosch, G.; Lizano-Barrantes, C.; Briseño Diaz, R.; Vernet-Tomas, M.; Fumadó, L.; Beisani, M.; Redondo Pachón, D.; Bach Pascual, A.; et al. Evaluating the Implementation of PROMs and PREMs in Routine Clinical Care: Co-Design of Tools from the Perspective of Patients and Healthcare Professionals. Health Qual. Life Outcomes 2025, 23, 15. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.D.C.; Perriman, D.M.; Fearon, A.M.; Couldrick, J.M.; Scarvell, J.M. Minimal Important Change and Difference for Knee Osteoarthritis Outcome Measurement Tools after Non-Surgical Interventions: A Systematic Review. BMJ Open 2023, 13, e063026. [Google Scholar] [CrossRef] [PubMed]
- Young, J.J.; Pedersen, J.R.; Bricca, A. Exercise Therapy for Knee and Hip Osteoarthritis: Is There an Ideal Prescription? Curr. Treatm. Opt. Rheumatol. 2023, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Lawford, B.J.; Hall, M.; Hinman, R.S.; van der Esch, M.; Harmer, A.R.; Spiers, L.; Kimp, A.; Dell’Isola, A.; Bennell, K.L. Exercise for Osteoarthritis of the Knee. Cochrane Database Syst. Rev. 2024, 12, CD004376. [Google Scholar] [CrossRef] [PubMed]
- Uson, J.; Rodriguez-García, S.C.; Castellanos-Moreira, R.; O’Neill, T.W.; Doherty, M.; Boesen, M.; Pandit, H.; Möller Parera, I.; Vardanyan, V.; Terslev, L.; et al. EULAR Recommendations for Intra-Articular Therapies. Ann. Rheum. Dis. 2021, 80, 1299–1305. [Google Scholar] [CrossRef] [PubMed]
- Pereira, T.V.; Saadat, P.; Bobos, P.; Iskander, S.M.; Bodmer, N.S.; Rudnicki, M.; Kiyomoto, H.D.; Montezuma, T.; Almeida, M.O.; Bansal, R.; et al. Effectiveness and Safety of Intra-Articular Interventions for Knee and Hip Osteoarthritis Based on Large Randomized Trials: A Systematic Review and Network Meta-Analysis. Osteoarthritis Cartilage 2025, 33, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Voorn, M.J.J.; Boymans, T.A.E.J.; Köke, A.J.A.; de Mooij, M.A.C.; Rommers, C.G.M.; de Nooijer, J.; Goossens, M.J.E.B.; Huijnen, I.P.J.; Verbunt, J.A.M.C.F. Enhancing Collaboration and Integrated Vision on Health: Key Strategies for Addressing Knee Osteoarthritis. Int. J. Integr. Care 2025, 25, 28. [Google Scholar] [CrossRef] [PubMed]
| Variable | Category | n | % |
| Sample | Complete respondents | 110 | 100 |
| Gender | |||
| Female | 71 | 64.0 | |
| Male | 39 | 35.1 | |
| Age group | |||
| 20–29 | 13 | 12.6 | |
| 30–39 | 24 | 21.6 | |
| 40–49 | 26 | 23.4 | |
| 50–59 | 31 | 27.9 | |
| 60–69 | 15 | 13.5 | |
| ≥70 | 1 | 0.9 | |
| Work setting | |||
| Tertiary / referral hospital | 57 | 51.4 | |
| Secondary hospital (~400 beds) | 30 | 27.0 | |
| General hospital (~200 beds) | 9 | 8.1 | |
| District hospital (~100 beds) | 9 | 8.1 | |
| Primary care | 2 | 2.7 | |
| Other | 3 | 2.7 | |
| Experience in musculoskeletal Rehabilitation. Medicine | |||
| <5 years | 7 | 7.2 | |
| 5–10 years | 19 | 17.1 | |
| 10–20 years | 38 | 34.2 | |
| >20 years | 46 | 41.4 | |
| Experience with interventional techniques for musculoskeletal pain management | |||
| <5 years | 13 | 12.6 | |
| 5–10 years | 27 | 24.3 | |
| 10–20 years | 43 | 38.7 | |
| >20 years | 27 | 24.3 | |
| Weekly number of patients with knee OA evaluated per physician | |||
| <20 | 15 | 14.4 | |
| 20–50 | 46 | 41.4 | |
| >50 | 49 | 44.1 | |
| Research involvement measured in publications during last 5 years | |||
| None | 35 | 32.4 | |
| <5 studies | 53 | 47.7 | |
| 5–30 studies | 15 | 13.5 | |
| >30 studies | 7 | 6.3 | |
| Where are your patients with knee osteoarthritis generally managed? | |||
| General rehabilitation clinic | 88 | 80.2 | |
| Multidisciplinary MSK pain unit | 16 | 14.4 | |
| Dedicated OA clinic | 1 | 0.9 | |
| Interventional clinic | 1 | 0.9 | |
| Other | 4 | 3.6 | |
| Pathway Phase | Clinical Purpose | Delphi Items Supporting the Phase |
| Phase 1. Entry and Referral to PM&R | Defines how patients access rehabilitation care and where initial evaluation occurs | Q21 Referral from primary care or other physicians Q24 Evaluation in general rehabilitation outpatient clinic Q20 Possible referral from orthopaedics Q22 Possible referral from rheumatology |
| Phase 2. Functional and Outcome Assessment | Establishes baseline clinical status and enables follow-up comparison using validated outcome measures | Q32 Pain scale (VAS/NRS) Q33 WOMAC functional scale Q34 Quality of life scale Q31 Descriptive global status scale Q35 Subjective clinical and patient assessment Q36 PROMs |
| Phase 3. Conservative Multimodal Treatment | Defines first-line therapeutic management combining rehabilitation and supportive therapies | Q28 Multimodal treatment combining physical therapy and pharmacological/topical therapy Q27 Sequential organisation of treatment components |
| Phase 4. Escalation and Advanced Therapeutic Options | Defines when and how advanced treatments or specialist referrals are considered | Q30 Interventional techniques should not be first-line treatment Q26 Use of interventional techniques when escalation is required Q23 Possible management in multidisciplinary MSK pain unit Q25 Dedicated OA clinics as specialised care setting |
| Phase 5. Longitudinal Monitoring and Discharge | Defines follow-up strategy and criteria for completing rehabilitation care | Q37 Defined discharge criteria |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




