Submitted:
25 February 2026
Posted:
28 February 2026
You are already at the latest version
Abstract
Keywords:
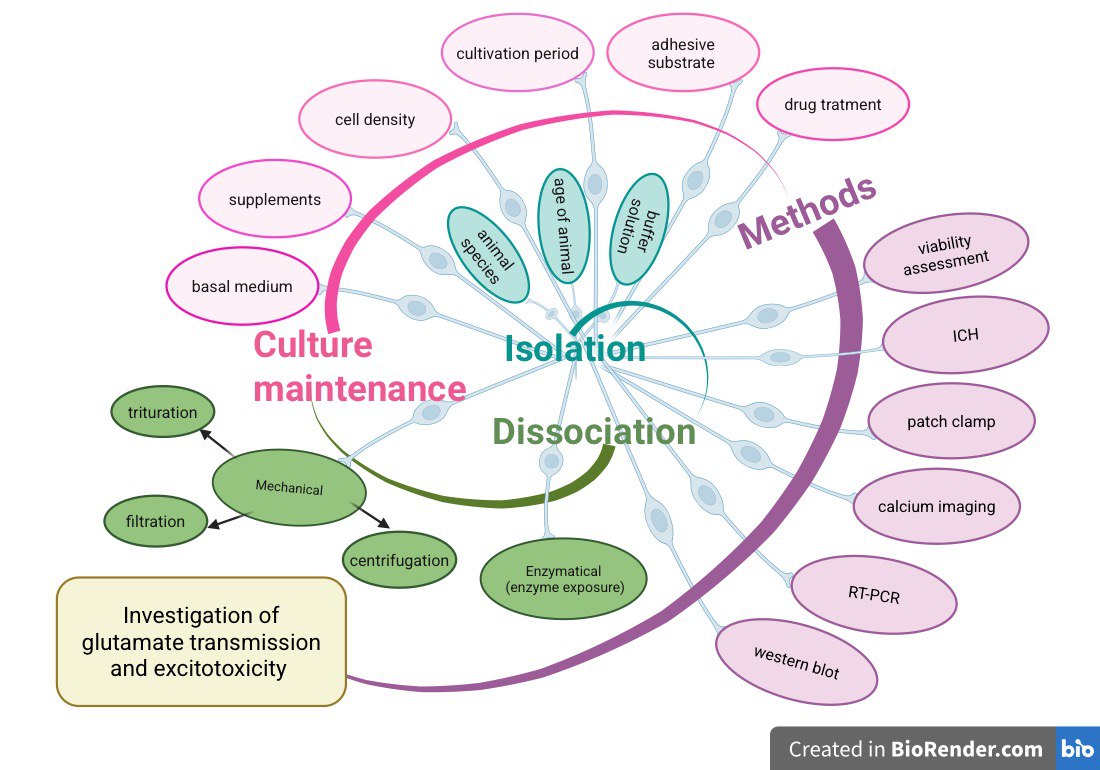
1. Introduction
2. Results
2.1. Cochlea Extraction and Modiolus Tissue Isolation
2.2. Dissociation
2.3. Culture Maintenance
2.4. Functional Properties of the Glutamatergic System in Isolated Spiral Ganglion Neurons
2.4.1. Acutely Isolated Cells
2.4.2. Primary SGCs Culture
3. Discussion
4. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 7-CKA | 7-chlorokynurenic acid |
| ACPD | (1S,3R)-aminocyclopentane-1,3-dicarboxylic acid |
| AIDA | (RS)-1-aminoindan-1,5-dicarboxylic acid |
| AIF | Apoptosis-inducing factor |
| AMPA | α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid |
| AMPARs | α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors |
| Asp | Aspartic acid |
| AraC | Cytosine β-D-arabinofuranoside |
| BDNF | Brain derived neurotrophic factor |
| CI-AMPARs | Calcium-impermeable AMPARs |
| CNQX | 6-cyano-7-nitroquinoxaline-2,3-dione |
| CP-AMPARs | Calcium-permeable AMPARs |
| DAP-5 | 2-amino-5-phosphonovalerate |
| DHPG | (S)-3,5-Dihydroxyphenylglycine |
| diCl-HQC | 6,7-dichloro-3-hydroxy-2-quinoxalinecarboxylic acid |
| DNQX | 6,7-dinitroquinoxaline-2,3-dione |
| EPSP | Excitatory postsynaptic potential |
| Glu | Glutamate |
| GRM7 | Metabotropic glutamate receptor 7 |
| GYKI 53784 | 1-(4-aminophenyl)-4-methyl-7, 8-methylenedioxy-4,5-dihydro-3-methylcarbamoyl-2,3-benzodiazepine |
| iGluRs | Ionotropic glutamate receptors |
| KA | Kainic acid |
| KARs | Kainic acid receptors |
| Kyn | Kynuranic acid |
| mGluRs | Metabotropic glutamate receptors |
| MK-801 | Dizocilpine |
| NBM | Neurobasal medium |
| NMDA | N-methyl-D-aspartate |
| NMDARs | N-methyl-D-aspartate receptors |
| NSAIDs | Nonsteroidal anti-inflammatory drug |
| NT-4/5 | Neurotrophin-4/5 |
| NT3 | Neurotrophin-3 |
| OME | Otitis media with effusion (OME) |
| QA | Quisqualic acid |
| ROS | Reactive oxygen species |
| SGCs | Spiral ganglion cells |
| SGNs | Spiral ganglion neurons |
References
- Chadha, S.; Kamenov, K.; Cieza, A. The World Report on Hearing, 2021. Bull. World Health Organ. 2021, 99, 242–242A. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, N.; Batts, S.; Stankovic, K.M. Noise-Induced Hearing Loss. J. Clin Med. 2023, 12, 2347. [Google Scholar] [CrossRef]
- Pasdelou, M.-P.; Byelyayeva, L.; Malmström, S.; Pucheu, S.; Peytavy, M.; Laullier, H.; Hodges, D.B.; Tzafriri, A.R.; Naert, G. Ototoxicity: A High Risk to Auditory Function That Needs to Be Monitored in Drug Development. Front. Mol. Neurosci. 2024, 17, 1379743. [Google Scholar] [CrossRef]
- Yang, W.; Zhao, X.; Chai, R.; Fan, J. Progress on Mechanisms of Age-Related Hearing Loss. Front. Neurosci. 2023, 17, 1253574. [Google Scholar] [CrossRef] [PubMed]
- Bing, X. Development of the Inner Ear and Regeneration of Hair Cells after Hearing Impairment. Fundam. Res. 2025, 5, 203–214. [Google Scholar] [CrossRef]
- Blankenship, C.; Zhang, F.; Keith, R. Behavioral Measures of Temporal Processing and Speech Perception in Cochlear Implant Users. J. Am. Acad. Audiol. 2016, 27, 701–713. [Google Scholar] [CrossRef] [PubMed]
- Lesica, N.A. Why Do Hearing Aids Fail to Restore Normal Auditory Perception? Trends Neurosci. 2018, 41, 174–185. [Google Scholar] [CrossRef]
- Barclay, M.; Ryan, A.F.; Housley, G.D. Type I vs Type II Spiral Ganglion Neurons Exhibit Differential Survival and Neuritogenesis during Cochlear Development. Neural. Dev. 2011, 6, 33. [Google Scholar] [CrossRef]
- Nayagam, B.A.; Muniak, M.A.; Ryugo, D.K. The spiral ganglion: connecting the peripheral and central auditory systems. Hear. Res. 2011, 278, 2–20. [Google Scholar] [CrossRef]
- Coate, T.M.; Kelley, M.W. Making Connections in the Inner Ear: Recent Insights into the Development of Spiral Ganglion Neurons and Their Connectivity with Sensory Hair Cells. Semin. Cell Dev. Biol. 2013, 24, 460–469. [Google Scholar] [CrossRef]
- Niedzielski, A.; Wenthold, R. Expression of AMPA, Kainate, and NMDA Receptor Subunits in Cochlear and Vestibular Ganglia. J. Neurosci. 1995, 15, 2338–2353. [Google Scholar] [CrossRef]
- Peng, B.G.; Li, Q.X.; Ren, T.Y.; Ahmad, S.; Chen, S.P.; Chen, P.; Lin, X. Group I Metabotropic Glutamate Receptors in Spiral Ganglion Neurons Contribute to Excitatory Neurotransmissions in the Cochlea. Neuroscience 2004, 123, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Reijntjes, D.O.J.; Pyott, S.J. The Afferent Signaling Complex: Regulation of Type I Spiral Ganglion Neuron Responses in the Auditory Periphery. Hear. Res. 2016, 336, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Ruel, J.; Chen, C.; Pujol, R.; Bobbin, R.P.; Puel, J.L. AMPA-preferring Glutamate Receptors in Cochlear Physiology of Adult Guinea-pig. J. Physiol. 1999, 518, 667–680. [Google Scholar] [CrossRef]
- Glowatzki, E.; Fuchs, P.A. Transmitter Release at the Hair Cell Ribbon Synapse. Nat. Neurosci. 2002, 5, 147–154. [Google Scholar] [CrossRef]
- Eybalin, M.; Caicedo, A.; Renard, N.; Ruel, J.; Puel, J. Transient Ca2+ -permeable AMPA Receptors in Postnatal Rat Primary Auditory Neurons. Eur. J. Neurosci. 2004, 20, 2981–2989. [Google Scholar] [CrossRef]
- Wang, J.; Serratrice, N.; Lee, C.J.; François, F.; Sweedler, J.V.; Puel, J.-L.; Mothet, J.-P.; Ruel, J. Physiopathological Relevance of D-Serine in the Mammalian Cochlea. Front. Cell. Neurosci. 2021, 15, 733004. [Google Scholar] [CrossRef]
- Paoletti, P.; Bellone, C.; Zhou, Q. NMDA Receptor Subunit Diversity: Impact on Receptor Properties, Synaptic Plasticity and Disease. Nat. Rev. Neurosci. 2013, 14, 383–400. [Google Scholar] [CrossRef] [PubMed]
- Sebe, J.Y.; Cho, S.; Sheets, L.; Rutherford, M.A.; Von Gersdorff, H.; Raible, D.W. Ca2+ -Permeable AMPARs Mediate Glutamatergic Transmission and Excitotoxic Damage at the Hair Cell Ribbon Synapse. J. Neurosci. 2017, 37, 6162–6175. [Google Scholar] [CrossRef]
- Hu, N.; Rutherford, M.A.; Green, S.H. Protection of Cochlear Synapses from Noise-Induced Excitotoxic Trauma by Blockade of Ca2+ -Permeable AMPA Receptors. Proc. Natl. Acad. Sci. U.S.A. 2020, 117, 3828–3838. [Google Scholar] [CrossRef]
- Walia, A.; Lee, C.; Hartsock, J.; Goodman, S.S.; Dolle, R.; Salt, A.N.; Lichtenhan, J.T.; Rutherford, M.A. Reducing Auditory Nerve Excitability by Acute Antagonism of Ca2+-Permeable AMPA Receptors. Front. Synaptic Neurosci. 2021, 13, 680621. [Google Scholar] [CrossRef]
- Steinbach, S.; Lutz, J. Glutamate Induces Apoptosis in Cultured Spiral Ganglion Explants. Biochem. Biophys. Res. Commun. 2007, 357, 14–19. [Google Scholar] [CrossRef]
- Lewerenz, J; Maher, P. Chronic Glutamate Toxicity in Neurodegenerative Diseases-What is the Evidence? Front Neurosci. 2015, 16, 469. [Google Scholar] [CrossRef]
- Pinelis, V.; Krasilnikova, I.; Bakaeva, Z.; Surin, A.; Boyarkin, D.; Fisenko, A.; Krasilnikova, O.; Pomytkin, I. Insulin Diminishes Superoxide Increase in Cytosol and Mitochondria of Cultured Cortical Neurons Treated with Toxic Glutamate. Int. J. Mol. Sci. 2022, 23, 12593. [Google Scholar] [CrossRef] [PubMed]
- Bakaeva, Z.; Goncharov, M.; Frolov, F.; Krasilnikova, I.; Sorokina, E.; Zgodova, A.; Smolyarchuk, E.; Zavadskiy, S.; Andreeva, L.; Myasoedov, N.; Fisenko, A.; Savostyanov, K. Regulatory Peptide Pro-Gly-Pro Accelerates Neuroregeneration of Primary Neuroglial Culture after Mechanical Injury in Scratch Test. Int. J. Mol. Sci. 2024, 25, 10886. [Google Scholar] [CrossRef] [PubMed]
- Shedenkova, M.; Gurianova, A.; Krasilnikova, I.; Sudina, A.; Karpulevich, E.; Maksimov, Y.; Samburova, M.; Guguchkin, E.; Nefedova, Z.; Babenko, V.; Frolov, D.; Savostyanov, K.; Fatkhudinov, T.; Goldshtein, D.; Bakaeva, Z.; Salikhova, D. Extracellular Vesicles from iPSC-Derived Glial Progenitor Cells Prevent Glutamate-Induced Excitotoxicity by Stabilising Calcium Oscillations and Mitochondrial Depolarisation. Cells 2025, 14, 1915. [Google Scholar] [CrossRef]
- Yamakura, T.; Shimoji, K. Subunit- and Site-Specific Pharmacology of the NMDA Receptor Channel. Prog. Neurobiol. 1999, 59, 279–298. [Google Scholar] [CrossRef]
- Nakagawa, T.; Komune, S.; Uemura, T.; Akaike, N. Excitatory Amino Acid Response in Isolated Spiral Ganglion Cells of Guinea Pig Cochlea. J. Neurophysiol. 1991, 65, 715–723. [Google Scholar] [CrossRef]
- Shimozono, M.; Tono, T.; Morimitsu, T.; Nakagawa, T.; Komune, S. Measurement of intracellular free Ca2+ concentration in guinea pig spiral ganglion cells. Neuroreport. 1995, 6, 421–424. [Google Scholar] [CrossRef]
- Hong, J.; Chen, Y.; Zhang, Y.; Li, J.; Ren, L.; Yang, L.; Shi, L.; Li, A.; Zhang, T.; Li, H.; et al. N-Methyl-D-Aspartate Receptors Involvement in the Gentamicin-Induced Hearing Loss and Pathological Changes of Ribbon Synapse in the Mouse Cochlear Inner Hair Cells. Neural. Plasticity 2018, 2018, 1–16. [Google Scholar] [CrossRef]
- Guitton, M.J.; Caston, J.; Ruel, J.; Johnson, R.M.; Pujol, R.; Puel, J.-L. Salicylate Induces Tinnitus through Activation of Cochlear NMDA Receptors. J. Neurosci. 2003, 23, 3944–3952. [Google Scholar] [CrossRef]
- Ruel, J.; Chabbert, C.; Nouvian, R.; Bendris, R.; Eybalin, M.; Leger, C. L.; Bourien, J.; Mersel, M.; Puel, J. L. Salicylate enables cochlear arachidonic-acid-sensitive NMDA receptor responses. J Neurosci. 2008, 28, 7313–7323. [Google Scholar] [CrossRef]
- Bing, D.; Lee, S.C.; Campanelli, D.; Xiong, H.; Matsumoto, M.; Panford-Walsh, R.; Wolpert, S.; Praetorius, M.; Zimmermann, U.; Chu, H.; Knipper, M.; Rüttiger, L.; Singer, W. Cochlear NMDA Receptors as a Therapeutic Target of Noise-Induced Tinnitus. Cell Physiol. Biochem. 2015, 35, 1905–1923. [Google Scholar] [CrossRef]
- Tang, X.; Zhu, X.; Ding, B.; Walton, J.P.; Frisina, R.D.; Su, J. Age-Related Hearing Loss: GABA, Nicotinic Acetylcholine and NMDA Receptor Expression Changes in Spiral Ganglion Neurons of the Mouse. Neuroscience 2014, 259, 184–193. [Google Scholar] [CrossRef]
- Chen, Z.; Kujawa, S.G.; Sewell, W.F. Auditory Sensitivity Regulation via Rapid Changes in Expression of Surface AMPA Receptors. Nat. Neurosci. 2007, 10, 1238–1240. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Peppi, M.; Kujawa, S.G.; Sewell, W.F. Regulated Expression of Surface AMPA Receptors Reduces Excitotoxicity in Auditory Neurons. J. Neurophysiol. 2009, 102, 1152–1159. [Google Scholar] [CrossRef]
- Sanchez, J.T.; Ghelani, S.; Otto-Meyer, S. From Development to Disease: Diverse Functions of NMDA-Type Glutamate Receptors in the Lower Auditory Pathway. Neuroscience 2015, 285, 248–259. [Google Scholar] [CrossRef]
- Zhang-Hooks, Y.; Agarwal, A.; Mishina, M.; Bergles, D.E. NMDA Receptors Enhance Spontaneous Activity and Promote Neuronal Survival in the Developing Cochlea. Neuron 2016, 89, 337–350. [Google Scholar] [CrossRef]
- Klotz, L.; Wendler, O.; Frischknecht, R.; Shigemoto, R.; Schulze, H.; Enz, R. Localization of Group II and III Metabotropic Glutamate Receptors at Pre- and Postsynaptic Sites of Inner Hair Cell Ribbon Synapses. FASEB J. 2019, 33, 13734–13746. [Google Scholar] [CrossRef]
- Lu, Y. Metabotropic Glutamate Receptors in Auditory Processing. Neuroscience 2014, 274, 429–445. [Google Scholar] [CrossRef]
- Kleinlogel, S.; Oestreicher, E.; Arnold, T.; Ehrenberger, K.; Felix, D. Metabotropic Glutamate Receptors Group I Are Involved in Cochlear Neurotransmission. Neuroreport. 1999, 10, 1879–1882. [Google Scholar] [CrossRef] [PubMed]
- Friedman, R.A.; Van Laer, L.; Huentelman, M.J.; Sheth, S.S.; Van Eyken, E.; Corneveaux, J.J.; Tembe, W.D.; Halperin, R.F.; Thorburn, A.Q.; Thys, S.; et al. GRM7 Variants Confer Susceptibility to Age-Related Hearing Impairment. Hum. Mol. Genet. 2009, 18, 785–796. [Google Scholar] [CrossRef]
- Klotz, L.; Enz, R. MGluR7 Is a Presynaptic Metabotropic Glutamate Receptor at Ribbon Synapses of Inner Hair Cells. FASEB J. 2021, 35. [Google Scholar] [CrossRef]
- Ma, X.; Guo, J.; Fu, Y.; Shen, C.; Jiang, P.; Zhang, Y.; Zhang, L.; Yu, Y.; Fan, J.; Chai, R. G Protein-Coupled Receptors in Cochlea: Potential Therapeutic Targets for Hearing Loss. Front. Mol. Neurosci. 2022, 15, 1028125. [Google Scholar] [CrossRef] [PubMed]
- Liberman, M.C.; Kujawa, S.G. Cochlear Synaptopathy in Acquired Sensorineural Hearing Loss: Manifestations and Mechanisms. Hear. Res. 2017, 349, 138–147. [Google Scholar] [CrossRef]
- Ma, K.; Zhang, A.; She, X.; Yang, H.; Wang, K.; Zhu, Y.; Gao, X.; Cui, B. Disruption of Glutamate Release and Uptake-Related Protein Expression After Noise-Induced Synaptopathy in the Cochlea. Front. Cell Dev. Biol. 2021, 9, 720902. [Google Scholar] [CrossRef]
- Moverman, D.J.; Liberman, L.D.; Kraemer, S.; Corfas, G.; Liberman, M.C. Ultrastructure of Noise-Induced Cochlear Synaptopathy. Sci.Rep. 2023, 13, 19456. [Google Scholar] [CrossRef]
- Wang, M.; Xu, L.; Han, Y.; Wang, X.; Chen, F.; Lu, J.; Wang, H.; Liu, W. Regulation of Spiral Ganglion Neuron Regeneration as a Therapeutic Strategy in Sensorineural Hearing Loss. Front. Mol. Neurosci. 2022, 14, 829564. [Google Scholar] [CrossRef]
- Safina, D.R.; Surin, A.M.; Pinelis, V.G.; Kostrov, S.V. Effect of Neurotrophin-3 Precursor on Glutamate-induced Calcium Homeostasis Deregulation in Rat Cerebellum Granule Cells. J Neurosci Res. 2015, 93, 1865–1873. [Google Scholar] [CrossRef]
- Schonkeren, S.L.; Küthe, T.T.; Idris, M.; Bon-Frauches, A.C.; Boesmans, W.; Melotte, V. The Gut Brain in a Dish: Murine Primary Enteric Nervous System Cell Cultures. Neurogastroenterol. Motil. 2022, 34, e14215. [Google Scholar] [CrossRef]
- Vargova, I.; Kriska, J.; Kwok, J.C.F.; Fawcett, J.W.; Jendelova, P. Long-Term Cultures of Spinal Cord Interneurons. Front. Cell. Neurosci. 2022, 16, 827628. [Google Scholar] [CrossRef] [PubMed]
- Weidinger, A.; Milivojev, N.; Hosmann, A.; Duvigneau, J.C.; Szabo, C.; Törö, G.; Rauter, L.; Vaglio-Garro, A.; Mkrtchyan, G.V.; Trofimova, L.; et al. Oxoglutarate Dehydrogenase Complex Controls Glutamate-Mediated Neuronal Death. Redox Biol. 2023, 62, 102669. [Google Scholar] [CrossRef] [PubMed]
- Cramer, T.M.L.; Tyagarajan, S.K. Protocol for the Culturing of Primary Hippocampal Mouse Neurons for Functional in Vitro Studies. STAR Protoc. 2024, 5, 102991. [Google Scholar] [CrossRef]
- Meas, S.J.; Nishimura, K.; Scheibinger, M.; Dabdoub, A. In Vitro Methods to Cultivate Spiral Ganglion Cells, and Purification of Cellular Subtypes for Induced Neuronal Reprogramming. Front. Neurosci. 2018, 12, 822. [Google Scholar] [CrossRef]
- Szabó, Z.; Harasztosi, C.; Szûcs, G.; Sziklai, I.; Rusznák, Z. A Detailed Procedure and Dissection Guide for the Isolation of Spiral Ganglion Cells of the Guinea Pig for Electrophysiological Experiments. Brain Res. Brain Res. Protoc. 2003, 10, 139–147. [Google Scholar] [CrossRef]
- Parker, M.; Brugeaud, A.; Edge, A.S.B. Primary Culture and Plasmid Electroporation of the Murine Organ of Corti. J. Vis. Exp. 2010, 1685. [Google Scholar]
- Grant, L.; Yi, E.; Goutman, J.D.; Glowatzki, E. Postsynaptic Recordings at Afferent Dendrites Contacting Cochlear Inner Hair Cells: Monitoring Multivesicular Release at a Ribbon Synapse. J. Vis. Exp. 2011, 2442. [Google Scholar]
- Montgomery, S.C.; Cox, B.C. Whole Mount Dissection and Immunofluorescence of the Adult Mouse Cochlea. J. Vis. Exp. 2016, 53561. [Google Scholar]
- May-Simera, H. Evaluation of Planar-Cell-Polarity Phenotypes in Ciliopathy Mouse Mutant Cochlea. J. Vis. Exp. 2016, 53559. [Google Scholar]
- Landegger, L.D.; Dilwali, S.; Stankovic, K.M. Neonatal Murine Cochlear Explant Technique as an In Vitro Screening Tool in Hearing Research. J. Vis. Exp. 2017, 55704. [Google Scholar]
- Sakamoto, A.; Kuroda, Y.; Kanzaki, S.; Matsuo, K. Dissection of the Auditory Bulla in Postnatal Mice: Isolation of the Middle Ear Bones and Histological Analysis. J. Vis. Exp. 2017, 55054. [Google Scholar]
- Fang, Q.-J.; Wu, F.; Chai, R.; Sha, S.-H. Cochlear Surface Preparation in the Adult Mouse. J. Vis. Exp. 2019, 10, 3791/60299. [Google Scholar]
- Ballesteros, A.; Swartz, K.J. Dextran Labeling and Uptake in Live and Functional Murine Cochlear Hair Cells. J. Vis. Exp. 2024, 10.3791/60769. [Google Scholar]
- Hahnewald, S.; Roccio, M.; Tscherter, A.; Streit, J.; Ambett, R.; Senn, P. Spiral Ganglion Neuron Explant Culture and Electrophysiology on Multi Electrode Arrays. J. Vis. Exp. 2016, 54538. [Google Scholar] [CrossRef] [PubMed]
- Schwieger, J.; Esser, K.-H.; Lenarz, T.; Scheper, V. Establishment of a Long-Term Spiral Ganglion Neuron Culture with Reduced Glial Cell Number: Effects of AraC on Cell Composition and Neurons. J. Neurosci. Methods 2016, 268, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; Zhou, X.; Shi, L.; Xia, M.; Wang, Y.; Guo, N.; Hu, H.; Zhang, P.; Yang, H.; Zhu, F.; et al. Shikonin Attenuates Cochlear Spiral Ganglion Neuron Degeneration by Activating Nrf2-ARE Signaling Pathway. Front. Mol. Neurosci. 2022, 15, 829642. [Google Scholar] [CrossRef]
- Zhai, S.-Q.; Wang, D.-J.; Wang, J.-L.; Han, D.-Y.; Yang, W.-Y. Basic Fibroblast Growth Factor Protects Auditory Neurons and Hair Cells from Glutamate Neurotoxicity and Noise Exposure. Acta Otolaryngol. 2004, 124, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Anacker, A.; Esser, K.-H.; Lenarz, T.; Paasche, G. Purification of Fibroblasts From the Spiral Ganglion. Front. Neurol. 2022, 13, 877342. [Google Scholar] [CrossRef]
- Lie, M.; Grover, M.; Whitlon, D.S. Accelerated Neurite Growth from Spiral Ganglion Neurons Exposed to the Rho Kinase Inhibitor H-1152. Neuroscience 2010, 169, 855–862. [Google Scholar] [CrossRef]
- Machado, L.; Relaix, F.; Mourikis, P. Stress Relief: Emerging Methods to Mitigate Dissociation-Induced Artefacts. Trends Cell Biol. 2021, 31, 888–897. [Google Scholar] [CrossRef]
- Uniken Venema, W.T.C.; Ramírez-Sánchez, A.D.; Bigaeva, E.; Withoff, S.; Jonkers, I.; McIntyre, R.E.; Ghouraba, M.; Raine, T.; Weersma, R.K.; Franke, L.; et al. Gut Mucosa Dissociation Protocols Influence Cell Type Proportions and Single-Cell Gene Expression Levels. Sci. Rep 2022, 12, 9897. [Google Scholar] [CrossRef] [PubMed]
- Feltz, P.; Rasminsky, M. A Model for the Mode of Action of GABA on Primary Afferent Terminals: Depolarizing Effects of GABA Applied Iontophoretically to Neurones of Mammalian Dorsal Root Ganglia. Neuropharmacology 1974, 13, 553–563. [Google Scholar] [CrossRef]
- Sun, G.; Liu, W.; Fan, Z.; Zhang, D.; Han, Y.; Xu, L.; Qi, J.; Zhang, S.; Gao, B.T.; Bai, X.; et al. The Three-Dimensional Culture System with Matrigel and Neurotrophic Factors Preserves the Structure and Function of Spiral Ganglion Neuron In Vitro. Neural. Plasticity 2016, 2016, 1–15. [Google Scholar]
- Zuo, W.-Q.; Hu, Y.-J.; Yang, Y.; Zhao, X.-Y.; Zhang, Y.-Y.; Kong, W.; Kong, W.-J. Sensitivity of Spiral Ganglion Neurons to Damage Caused by Mobile Phone Electromagnetic Radiation Will Increase in Lipopolysaccharide-Induced Inflammation in Vitro Model. J. Neuroinflammation 2015, 12, 105. [Google Scholar] [CrossRef]
- Vieira, M.; Christensen, B.L.; Wheeler, B.C.; Feng, A.S.; Kollmar, R. Survival and Stimulation of Neurite Outgrowth in a Serum-Free Culture of Spiral Ganglion Neurons from Adult Mice. Hear. Res. 2007, 230, 17–23. [Google Scholar] [CrossRef]
- Whitlon, D.S.; Ketels, K.V.; Coulson, M.T.; Williams, T.; Grover, M.; Edpao, W.; Richter, C.P. Survival and Morphology of Auditory Neurons in Dissociated Cultures of Newborn Mouse Spiral Ganglion. Neuroscience 2006, 138, 653–662. [Google Scholar] [CrossRef]
- Kaiser, O.; Paasche, G.; Stöver, T.; Ernst, S.; Lenarz, T.; Kral, A.; Warnecke, A. TGF-Beta Superfamily Member Activin A Acts with BDNF and Erythropoietin to Improve Survival of Spiral Ganglion Neurons in Vitro. Neuropharmacology 2013, 75, 416–425. [Google Scholar] [CrossRef]
- Schwieger, J.; Warnecke, A.; Lenarz, T.; Esser, K.-H.; Scheper, V. Neuronal Survival, Morphology and Outgrowth of Spiral Ganglion Neurons Using a Defined Growth Factor Combination. PLoS One 2015, 10, e0133680. [Google Scholar] [CrossRef]
- Hegarty, J.L.; Kay, A.R.; Green, S.H. Trophic Support of Cultured Spiral Ganglion Neurons by Depolarization Exceeds and Is Additive with That by Neurotrophins or cAMP and Requires Elevation of [Ca2+ ]i within a Set Range. J. Neurosci. 1997, 17, 1959–1970. [Google Scholar] [CrossRef]
- Harada, N.; Han, D.-Y.; Komeda, M.; Yamashita, T. Glutamate-Induced Intracellular Ca2+ Elevation in Isolated Spiral Ganglion Cells of the Guinea Pig Cochlea. Acta Otolaryngol. 1994, 114, 609–612. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Zhang, C.; Chen, A.; Liu, W.; Li, J.; Sun, Q.; Wang, H. Protective Effect of Edaravone on Glutamate-Induced Neurotoxicity in Spiral Ganglion Neurons. Neural. Plasticity 2016, 2016, 1–10. [Google Scholar] [CrossRef]
- Han, D.-Y.; Yamashita, T.; Harada, N.; Kumazawa, T. Calcium Mobilization in Isolated Cochlear Spiral Ganglion Cells of the Guinea Pig. Acta Otolaryngol. 1993, 113, 26–29. [Google Scholar] [CrossRef]
- Ding, Z.-J.; Chen, X.; Tang, X.-X.; Wang, X.; Song, Y.-L.; Chen, X.-D.; Wang, J.; Wang, R.-F.; Mi, W.-J.; Chen, F.-Q.; et al. Apoptosis-Inducing Factor and Calpain Upregulation in Glutamate-Induced Injury of Rat Spiral Ganglion Neurons. Mol. Med. Rep. 2015, 12, 1685–1692. [Google Scholar] [CrossRef]
- Xiao, H.; Yang, C.; He, Y.; Zheng, N. Neurotoxicity of Quinolinic Acid to Spiral Ganglion Cells in Rats. J. Huazhong Univ. Sci. Technolog. Med. Sci. 2010, 30, 397–402. [Google Scholar] [CrossRef]
- Sun, F.; Zhou, K.; Tian, K. Y.; Zhang, X. Y.; Liu, W.; Wang, J.; Zhong, C. P.; Qiu, J. H.; Zha, D. J. Atrial Natriuretic Peptide Promotes Neurite Outgrowth and Survival of Cochlear Spiral Ganglion Neurons In vitro Through NPR-A/cGMP/PKG Signaling. Front Cell Dev Biol. 2021, 9, 681421. [Google Scholar] [CrossRef]
- Wright, T.; Gillespie, L.N.; O’Leary, S.J.; Needham, K. Firing Frequency and Entrainment Maintained in Primary Auditory Neurons in the Presence of Combined BDNF and NT3. Sci. Rep. 2016, 6, 28584. [Google Scholar] [CrossRef] [PubMed]
- Brewer, G.J. Serum-free B27/Neurobasal Medium Supports Differentiated Growth of Neurons from the Striatum, Substantia Nigra, Septum, Cerebral Cortex, Cerebellum, and Dentate Gyrus. J. Neurosci. Res. 1995, 42, 674–683. [Google Scholar] [CrossRef]
- Xie, C.; Markesbery, W.R.; Lovell, M.A. Survival of Hippocampal and Cortical Neurons in a Mixture of MEM+ and B27-Supplemented Neurobasal Medium. Free Radic. Biol. Med. 2000, 28, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.H.; Ngwainmbi, J.; Grider, J.R.; Dewey, W.L.; Akbarali, H.I. An In-Vitro Preparation of Isolated Enteric Neurons and Glia from the Myenteric Plexus of the Adult Mouse. J. Vis. Exp. 2013, 50688. [Google Scholar] [PubMed]
- Vincent, P.F.Y.; Young, E.D.; Edge, A.S.B.; Glowatzki, E. Auditory Hair Cells and Spiral Ganglion Neurons Regenerate Synapses with Refined Release Properties in Vitro. Proc. Natl. Acad. Sci. U.S.A. 2024, 121, e2315599121. [Google Scholar] [CrossRef]
- Lefebvre, P.P.; Weber, T.; Leprince, P.; Rigo, J.-M.; Delre’e, P.; Rogister, B.; Moonen, G. Kainate and NMDA Toxicity for Cultured Developing and Adult Rat Spiral Ganglion Neurons: Further Evidence for a Glutamatergic Excitatory Neurotransmission at the Inner Hair Cell Synapse. Brain Res. 1991, 555, 75–83. [Google Scholar] [CrossRef]
- Zheng, J.; Stewart, R.; Gao, W. Neurotrophin-4/5 Enhances Survival of Cultured Spiral Ganglion Neurons and Protects Them from Cisplatin Neurotoxicity. J. Neurosci. 1995, 15, 5079–5087. [Google Scholar] [CrossRef]
- Bailey, E.M.; Green, S.H. Postnatal Expression of Neurotrophic Factors Accessible to Spiral Ganglion Neurons in the Auditory System of Adult Hearing and Deafened Rats. J. Neurosci. 2014, 34, 13110–13126. [Google Scholar] [CrossRef]
- Needham, K.; Nayagam, B.A.; Minter, R.L.; O’Leary, S.J. Combined Application of Brain-Derived Neurotrophic Factor and Neurotrophin-3 and Its Impact on Spiral Ganglion Neuron Firing Properties and Hyperpolarization-Activated Currents. Hear. Res. 2012, 291, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Balazs, R. Trophic Effect of Glutamate. Curr. Top. Med. Chem. 2006, 6, 961–968. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zou, Q.; Zhang, J. Vincamine exerts protective effect on spiral ganglion neurons in endolymphatic hydrops guinea pig models. Am. J. Transl. Res. 2018, 10, 3650–3663. [Google Scholar]
- McDonald, J. W.; Johnston, M. V. Physiological and pathophysiological roles of excitatory amino acids during central nervous system development. Brain Res. Brain Res. Rev. 1990, 15, 41–70. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.C.; Appler, J.M.; Houseman, E.A.; Goodrich, L.V. Developmental Profiling of Spiral Ganglion Neurons Reveals Insights into Auditory Circuit Assembly. J. Neurosci. 2011, 31, 10903–10918. [Google Scholar] [CrossRef]
- Lu, J.; Liu, H.; Lin, S.; Li, C.; Wu, H. Electrophysiological Characterization of Acutely Isolated Spiral Ganglion Neurons in Neonatal and Mature Sonic Hedgehog Knock-in Mice. Neurosci. Lett. 2020, 714, 134536. [Google Scholar] [CrossRef]
- Yamaguchi, K.; Ohmori, H. Voltage-gated and Chemically Gated Ionic Channels in the Cultured Cochlear Ganglion Neurone of the Chick. J. Physiol. 1990, 420, 185–206. [Google Scholar] [CrossRef]
- Ding, Z. J.; Chen, X.; Tang, X. X.; Wang, X.; Song, Y. L.; Chen, X. D.; Mi, W. J.; Wang, J.; Lin, Y.; Chen, F. Q.; Qiu, J. H. Calpain inhibitor PD150606 attenuates glutamate induced spiral ganglion neuron apoptosis through apoptosis inducing factor pathway in vitro. PloS One. 2015, 10, e0123130. [Google Scholar] [CrossRef] [PubMed]
- Carricondo, F.; Bartolomé, M. V.; Vicente-Torres, M. A.; Fernández-Pacheco, P.; Rodríguez, T.; Gil-Loyzaga, P. Sensitivity to glutamate neurotoxicity in different developmental periods of the rat cochlea. Adv. Otorhinolaryngol. 2002, 59, 91–95. [Google Scholar] [PubMed]
- Frisch, S.; Francis, H. Disruption of Epithelial Cell-Matrix Interactions Induces Apoptosis. J. Cell Biol. 1994, 124, 619–626. [Google Scholar] [CrossRef] [PubMed]
| Study | Animal species and age | Enzyme type | Enzyme concentration | units | Duration [min] | Temperature [˚C] |
|---|---|---|---|---|---|---|
|
Yamaguchi and Ohmori, 1990 [100] |
Chick embryo (16-19 embryonic days) |
Collagenase Papain |
0.3 10 |
units/ml units/ml |
30 25 |
37 |
|
Lefebvre et al., 1991 [91] |
Rats (P5) |
Trypsin Collagenase DNAse |
0.1 0.l 0.01 |
% % % |
25 | 37 |
| Adult rats | Collagenase Trypsin |
5 0.1 |
% % |
20 17 |
||
| Nakagawa et al., 1991 [28] |
Adult guinea pigs |
Collagenase or Dispase |
0,5 500 |
mg/ml IU/ml |
30-60 30 |
31 |
| Chickens from 2 to 5 wk post-hatch of either sex | Collagenase Trypsin type IX or Dispase |
1.0 0.5 1000 |
mg/ml mg/ml IU/ml |
30 30 60-90 |
||
| Harada et al., 1994 [80] |
Adult guinea pigs | Collagenase | 0.1 | mg/ml | 30 | NA |
| Shimozono et al., 1995 [29] |
Adult guinea pigs | Collagenase or Dispase |
0.5 500 |
mg/ml IU/ml |
30-60 30 |
31 |
| Peng et al., 2004 [12] |
Mice (P0, adult) |
Collagenase type IV Trypsin |
0.5 2.5 |
mg/ml mg/ml |
30 + 30 |
On ice 37 |
| Zhai et al., 2004 [67] |
Mice (P3) |
Collagenase Trypsin |
0.25 0.25 |
% % |
25 | 37 |
| Chen et al., 2007 [35] |
Mice (P6-8) |
Collagenase type IV Trypsin |
0.5 2.5 |
mg/ml mg/ml |
25 | 37 |
| Chen et al., 2009 [36] |
Mice (P3-5) |
Collagenase type IV Trypsin |
0.5 2.5 |
mg/ml mg/ml |
25 | 37 |
| Xiao et al., 2010 [84] |
Rats (P3-7) |
Trypsin | NA | NA | 8 + 6 |
37 low temperature |
| Ding et al., 2015 [101] |
Rats (P0-3) |
Trypsinase | 0.125 | % | 15 | 37 |
|
Bai et al., 2016 [81] |
Rats (P<5) |
Trypsinase | 0.125 | % | 15 | 37 |
|
Li et al., 2018 [96] |
Rats (embryonic day-18) |
Trypsin | 1.25 | mg/ml | 10 | 37 |
| Sun et al., 2021 [85] |
Rats (P3) |
Collagenase type IV Trypsin |
0.1 0.25 |
% % |
20 | 37 |
| Wang et al., 2021 [17] |
Rats (P1-P4) |
Collagenase type IV Trypsin |
0.5 2.5 |
mg/ml mg/ml |
60 | 37 |
| Study | Animal species and age | Long-term/short term culture | Coating substrate | Medium and supplements | Pre- experimental cultivation time/ Maximum incubation period |
|---|---|---|---|---|---|
| Yamaguchi and Ohmori, 1990 [100] |
Chick embryo (16-19 embryonic days) |
Long-term culture | Collagen Poly-D-lysine |
DMEM+F12 calf serum |
5-14 days/5-14 days |
| Short- term culture | Poly-D-lysine/ concanavalin A |
DMEM+F12 calf serum |
<1 day/ <1 day | ||
| Lefebvre et al., 1991 [91] |
Rats (P5) |
Long- term culture | Poly-ornithine; Laminin; |
DMEM+ N1 cocktail: bovine insulin, progesterone, putrescine, transferrin, selenium |
5 days/6 days |
| Adult rats | Long-term culture | Poly-ornithine; Laminin; Astrocyte-conditioned medium |
DMEM+ N1 cocktail: bovine insulin, progesterone, putrescine, transferrin, selenium |
3 days/4 days | |
| Peng et al., 2004 [12] |
Mice (P0, adult) |
Long-term culture | NA |
DMEM+ FBS penicillin streptomycin |
2 days/ 2 days and more |
| Zhai et al., 2004 [67] |
Mice (P3) |
Long-term culture | Rat-tail collagen | Not fully specified | 24 h/14 days |
| Chen et al., 2007 [35] |
Mice (P6-P8) |
Short-term culture | Gelatin |
DMEM+ F12 FBS, horse serum, NT-3, BDNF, penicillin and streptomycin |
15-18 h/ 15-18 h + ~1 h |
| Chen et al., 2009 [36] |
Mice (P3-P5) |
Short-term culture | Poly-L-ornithine |
DMEM+ F12 FBS, horse serum, NT-3, BDNF, B-27 supplement, penicillin and streptomycin |
15-18 h/15-18 h+ ~24h |
| Xiao et al., 2010 [84] |
Rats (P3-7) |
Long-term culture | NA |
NBM B27 AraC |
72 h/96 h |
| Ding et al., 2015 [101] |
Rats (P1) |
Long-term culture | NA |
DMEM B27, BDNF, penicillin |
24 h/72 h |
| Ding et al., 2015 [83] |
Rats (P0-3) |
Long-term culture | NA |
DMEM B27, BDNF, penicillin |
NA |
| Bai et al., 2016 [81] |
Rats (<P5) |
Short-term culture | Poly-L-lysine |
DMEM 10% FBS |
NA |
| Li et al., 2018 [96] |
Rats, (embryonic day-18) | Short-term culture | Poly-L-lysine |
DMEM 10% FBS |
NA |
| Sun et al., 2021 [85] |
Rats (P3) |
Long-term culture | Poly-L-lysine | NA |
4h/5 days |
| Study | Animal species and age | Methods | Applications | Glu receptors agonists (concentration) | Glu receptors antagonists (concentration) |
|
|---|---|---|---|---|---|---|
| Yamaguchi and Ohmori, 1990 [100] |
Chick embryo (16-19 embryonic days) | Patch-clamp | Measurement of ionic currents | Glu (30 µM) KA (100 µM) Asp (100 µM) NMDA (100 µM) |
APV (100 µM) | |
| Fluorescent microscopy |
Cell morphology | |||||
| Lefebvre et al., 1991 [91] |
Rats (P5); |
Immunohistochemical staining | Survival assessment; Cell morphology |
Glu 10-8 - 10-3 M NMDA 10-8 - 10-3 M Kainic acid 10-8 - 10-3 M Quisqualic acid 10-8 - 10-3 M |
DAP-5 (10-9 -10-3 M) Kyn (10-9-10-6 M) |
|
| Adult rats |
Glu (10-4 M) NMDA (10-4 M) Kainic acid (10-4 M) Quisqualic acid (10-4 M) |
DAP-5 (10-4 M) Kyn (10-4 M) |
||||
| Nakagawa et al., 1991 [28] |
Adult guinea pigs; |
Patch-clamp |
Measurement of ionic currents |
Glu (3х10-6 - 10-2 M) QA (3x10-7- 3x10-4 M) KA (3х10-6-3х10-4 M) Asp (3x10-3 M) NMDA (3x10-3 M) |
CNQX (10-8-10-5 M); DNQX (10-8-10-5 M); diCl-HQC (10-7-10-4 M); Kyn (3x10-6-3x10-3 M); APV (3х10-5 M) |
|
|
Phase-contrast microscopy |
Cell morphology | |||||
| Chickens from 2 to 5 wk post-hatch | Glu (3х10-4 M) QA (3х10-5 M) KA (10-4 M) Asp (10-3 M) NMDA (3x10-3 M) |
APV (3х10-5 M) | ||||
| Harada et al., 1994 [80] |
Adult guinea pigs | Calcium imaging | Intracellular calcium [Ca²⁺]i concentration measurement | Glu (100 µM) | NA | |
|
Phase-contrast microscopy |
Cell morphology | |||||
| Shimozono et al., 1995 [29] |
Adult guinea pigs | Patch-clamp | Measurement of ionic currents | Glu (10, 50, 100 µM) KA (100 µM); NMDA (100 µM); QA (100 µM) |
APV (NA); |
|
| Calcium imaging | Intracellular calcium [Ca²⁺]i concentration measurement | |||||
| Peng et al., 2004 [12] |
Mice (P0, adult) |
Patch-clamp | Measurement of ionic currents | KA (100 µM); Glu (300 µM) DHPG (100 µM); ACPD (100 µM) |
AIDA (300 µM) DNQX (200 µM) |
|
| Calcium imaging | Intracellular calcium [Ca²⁺]i concentration measurement | |||||
| Zhai et al., 2004 [67] |
Mice (P3) |
Histochemical staining | Survival assessment; Neurite length measurement |
Glu (20 mM) | NA | |
| Chen et al., 2007 [35] |
Mice (P6-P8) |
Immunofluorescence staining | Assessment of surface GluR2 receptors expression levels; Viability assessment |
Glu (20 µM) AMPA (20 µM) NMDA (20 µM) |
DNQX (20 µM) APV (50 µM) |
|
| Chen et al., 2009 [36] |
Mice (P3-P5) |
Immunofluorescence staining | Assessment of surface GluR2 receptors expression levels; Viability assessment |
AMPA (20 µM) NMDA (20 µM) AMPA (300 µM) NMDA (300 µM) |
NA | |
| Xiao et al., 2010 [84] |
Rats (P3-7) |
Fluorescent microscopy |
Cell morphology; Detection of apoptosis |
Quinolinic acid (100; 1000 µM/L) |
MK-801 (20 µM/L) | |
| Laser confocal microscopy | Intracellular calcium [Ca²⁺]i concentration measurement | |||||
| Ding et al., 2015 [101] |
Rats (P1) |
Stereoscopic microscopy |
Cell morphology |
Glu (20 µM) | NA | |
|
Immunofluorescence staining; |
AIF distribution |
|||||
| TUNEL assay | Apoptotic cells counting | |||||
| Western Blot | Assessment of AIF and calpain expression Levels |
|||||
| RT-PCR | Assessment of mRNA expression of AIF, calpain and caspase-3 |
|||||
| Ding et al., 2015 [83] |
Rats (P0-3) |
Immunofluorescence staining | AIF distribution |
Glu (10 mM, 20 mM, 40 mM) | NA | |
| RT-PCR | Assessment of mRNA expression of AIF, calpain and caspase-3 |
|||||
| Bai et al., 2016 [81] |
Rats (P<5) |
MTT assay | Cell viability assessment |
Glu (2 mM) | NA | |
| Trypan blue staining | Cell viability assessment |
|||||
| Ho.33342 and Propidium iodide double staining | Detection of apoptosis and necrosis | |||||
| Spectrophotometry | Detection of GSH content, SOD activity, MDA level | |||||
| Western Blot | Assessment of AKT, p-AKT, Bax, Bcl-2 genes expression levels | |||||
| Li et al., 2018 [96] |
Rats embryo (18 embryonic days) | MTT assay |
Cell viability assessment | Glu (2 mM) | NA | |
| Ho.33342 and Propidium iodide double staining | Detection of apoptosis and necrosis | |||||
| Sun et al., 2021 [85] |
Rats (P3) | Immunofluorescence staining | Neuronal number and neurite length measurement |
Glu (100 µM) | NA | |
| Wang et al., 2021 [17] |
Rats (P1-P4) |
Calcium imaging | Intracellular calcium [Ca²⁺]i concentration measurement | AMPA (100 µM) Glu (100 µM) NMDA (100 µM) Glu (100 µM) + D-serine (100 µM) |
7-CKA (50 µM) D-APV (50 µM) GYKI 53784 (40 µM) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




