Submitted:
22 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
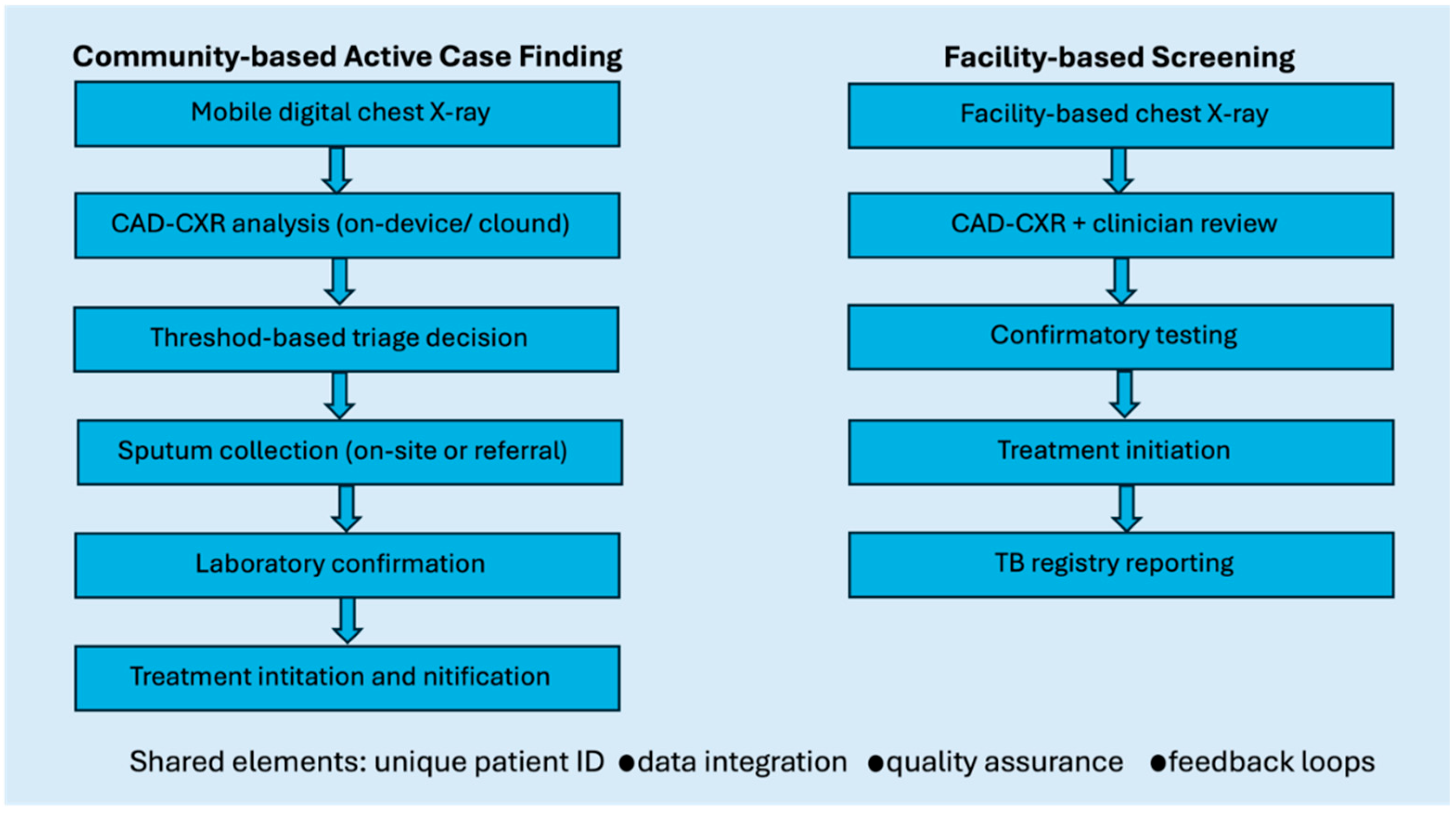
3. CAD-CXR for Tuberculosis Screening and Triage
3.1. Policy Landscape and Scope of WHO Recommendations
3.2. Evidence Underpinning Policy Recommendations
3.2.1. Diagnostic Performance in Screening Contexts
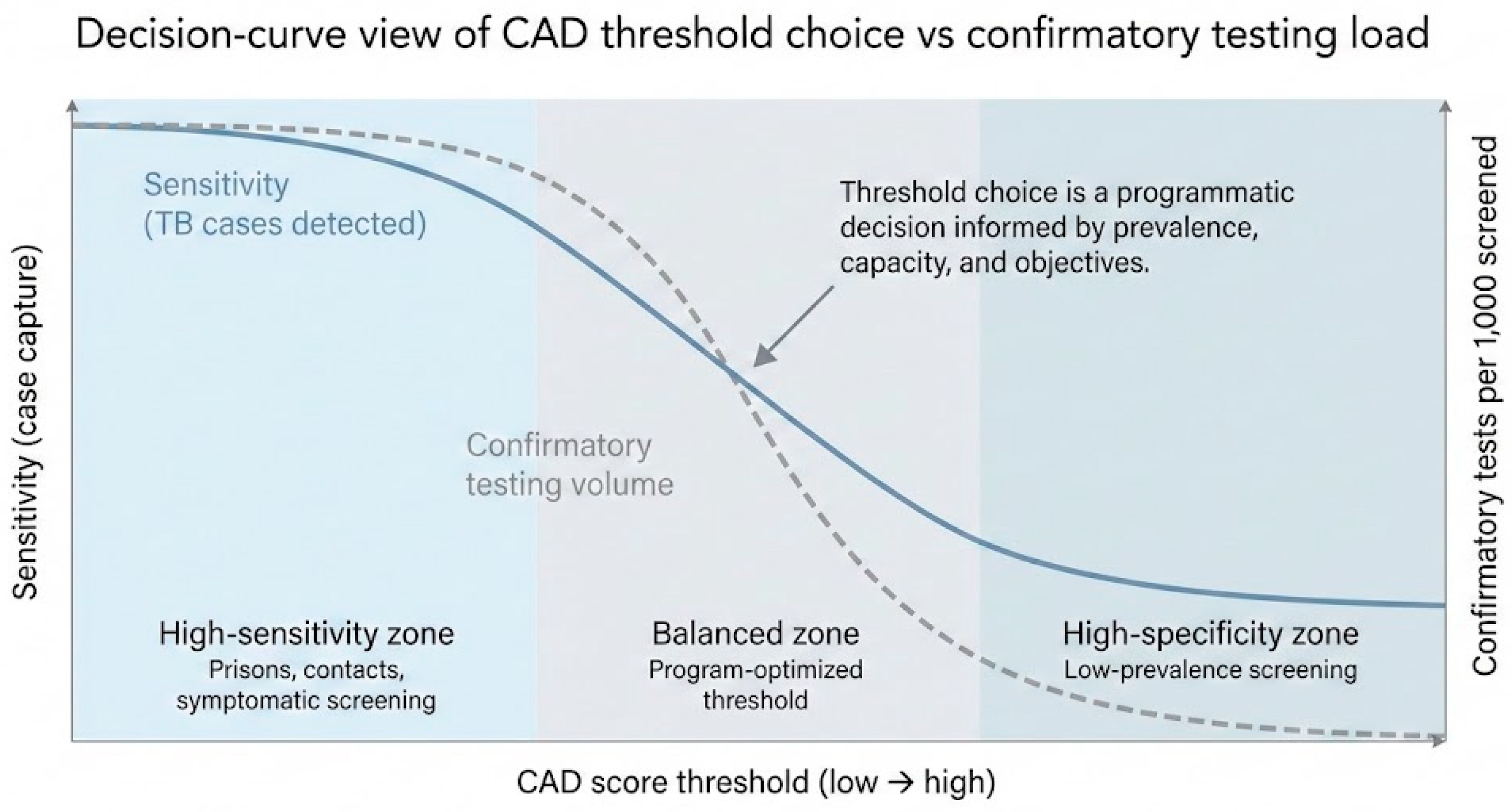
3.2.2. Threshold Calibration as a Programmatic Decision
3.2.3. Subgroup Performance and Sources of Variability
3.2.4. Linking Diagnostic Performance to Patient- and Population-Level Outcomes
3.3. Validation, Benchmarking, and Local Verification

3.4. Economics and Procurement
3.5. Implementation, Governance and Equity

4. Beyond CXR: CT, Ultrasound, Cough Sound, and Digital Stethoscopes
4.1. AI-Assisted Computed Tomography
4.2. Point-of-Care Ultrasound
4.3. Cough Sound Analysis
4.4. Digital Stethoscope and Lung Sound Analysis
4.5. AI-Enabled Data Analytic Tools for Tuberculosis Risk Stratification
4.6. AI-Assisted Interpretation of Genomic Data on TB Drug-Resistance
4.7. Summary of Readiness and Programmatic Implications Across Modalities
5. Future Directions and Research Agenda
5.1. Expanding Evidence to Priority Populations
5.2. Moving Beyond Accuracy to Patient- and Population-Level Important Outcomes
5.3. Strengthening Evidence for Emerging AI Modalities
5.4. Governance, Safety, and Lifecycle Evaluation
5.5. Integrating AI into Comprehensive TB Care
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Storla, D.G.; Yimer, S.; Bjune, G.A. A Systematic Review of Delay in the Diagnosis and Treatment of Tuberculosis. BMC Public Health 2008, 8, 1–9. [CrossRef] [PubMed]
- Team, W.; Global Programme on Tuberculosis and Lung Health (GTB) Global Tuberculosis Report 2025; 2025.
- Team, W.; Global Programme on Tuberculosis and Lung Health (GTB), G.R.C. WHO Consolidated Guidelines on Tuberculosis: Module 2: Screening: Systematic Screening for Tuberculosis Disease; 2021.
- Team, W.; (GTB), G.P. on T. and L.H. Use of Computer-Aided Detection Software for Tuberculosis Screening: WHO Policy Statement; 2025.
- Qin, Z.Z.; Sander, M.S.; Rai, B.; Titahong, C.N.; Sudrungrot, S.; Laah, S.N.; Adhikari, L.M.; Carter, E.J.; Puri, L.; Codlin, A.J.; et al. Using Artificial Intelligence to Read Chest Radiographs for Tuberculosis Detection: A Multi-Site Evaluation of the Diagnostic Accuracy of Three Deep Learning Systems. Sci. Rep. 2019, 9, 1–10. [CrossRef] [PubMed]
- Moodley, N.; Velen, K.; Saimen, A.; Zakhura, N.; Churchyard, G.; Charalambous, S. Digital Chest Radiography Enhances Screening Efficiency for Pulmonary Tuberculosis in Primary Health Clinics in South Africa. Clin. Infect. Dis. 2022, 74, 1650–1658. [CrossRef] [PubMed]
- Subbaraman, R.; Nathavitharana, R.R.; Mayer, K.H.; Satyanarayana, S.; Chadha, V.K.; Arinaminpathy, N.; Pai, M. Constructing Care Cascades for Active Tuberculosis: A Strategy for Program Monitoring and Identifying Gaps in Quality of Care. PLoS Med. 2019, 16, 1–18. [CrossRef] [PubMed]
- Bartl, L.; Zeeb, M.; Kälin, M.; Loosli, T.; Notter, J.; Furrer, H.; Hoffmann, M.; Hirsch, H.H.; Zangerle, R.; Grabmeier-Pfistershammer, K.; et al. Machine Learning-Based Prediction of Active Tuberculosis in People with HIV Using Clinical Data. Clin. Infect. Dis. 2025, 81, 521–530. [CrossRef] [PubMed]
- Bigio, J.; Kohli, M.; Klinton, J.S.; MacLean, E.; Gore, G.; Small, P.M.; Ruhwald, M.; Weber, S.F.; Jha, S.; Pai, M. Diagnostic Accuracy of Point-of-Care Ultrasound for Pulmonary Tuberculosis: A Systematic Review. PLoS One 2021, 16, 1–14. [CrossRef] [PubMed]
- Bradley, P.; Gordon, N.C.; Walker, T.M.; Dunn, L.; Heys, S.; Huang, B.; Earle, S.; Pankhurst, L.J.; Anson, L.; De Cesare, M.; et al. Rapid Antibiotic-Resistance Predictions from Genome Sequence Data for Staphylococcus Aureus and Mycobacterium Tuberculosis. Nat. Commun. 2015, 6. [CrossRef] [PubMed]
- Ma, L.; Wang, Y.; Guo, L.; Zhang, Y.; Wang, P.; Pei, X.; Qian, L.; Jaeger, S.; Ke, X.; Yin, X.; et al. Developing and Verifying Automatic Detection of Active Pulmonary Tuberculosis from Multi-Slice Spiral CT Images Based on Deep Learning. J. Xray. Sci. Technol. 2020, 28, 939–951. [CrossRef] [PubMed]
- Rajasekar, S.J.S.; Balaraman, A.R.; Balaraman, D.V.; Mohamed Ali, S.; Narasimhan, K.; Krishnasamy, N.; Perumal, V. Detection of Tuberculosis Using Cough Audio Analysis: A Deep Learning Approach with Capsule Networks. Discov. Artif. Intell. 2024, 4. [CrossRef]
- FIND Validation Platform for AI-Based Diagnostic Evaluation. 2023.
- Kagujje, M.; Kerkhoff, A.D.; Nteeni, M.; Dunn, I.; Mateyo, K.; Muyoyeta, M. The Performance of Computer-Aided Detection Digital Chest X-Ray Reading Technologies for Triage of Active Tuberculosis Among Persons With a History of Previous Tuberculosis. Clin. Infect. Dis. 2023, 76, E894–E901. [CrossRef] [PubMed]
- Garg, T.; John, S.; Abdulkarim, S.; Ahmed, A.D.; Kirubi, B.; Rahman, M.T.; Ubochioma, E.; Creswell, J. Implementation Costs and Cost-Effectiveness of Ultraportable Chest X-Ray with Artificial Intelligence in Active Case Finding for Tuberculosis in Nigeria. PLOS Digit. Heal. 2025, 4, 1–13. [CrossRef] [PubMed]
- Velen, K.; Sathar, F.; Hoffmann, C.J.; Hausler, H.; Fononda, A.; Govender, S.; Lerefolo, M.; Govender, A.; Charalambous, S. Digital Chest X-Ray with Computer-Aided Detection for Tuberculosis Screening within Correctional Facilities. Ann. Am. Thorac. Soc. 2022, 19, 1313–1319. [CrossRef] [PubMed]
- Signorell, A.; Van Heerden, A.; Ayakaka, I.; Jacobs, B.K.; Antillon, M.; Tediosi, F.; Verjans, A.; Brugger, C.; Harkare, H.V.; Labhardt, N.D.; et al. Effectiveness and Cost-Effectiveness of Community-Based TB Screening Algorithms Using Computer-Aided Detection (CAD) Technology Alone Compared with CAD Combined with Point-of-Care C Reactive Protein Testing in Lesotho and South Africa: Protocol for a Paired Screen-Positive Trial. BMJ Open 2025, 15, 1–14. [CrossRef]
- Team, W.; Chief Scientist and Science Division (SCI), H.E.& G. (HEG) Ethics and Governance of Artificial Intelligence for Health: Guidance on Large Multi-Modal Models; 2025.
- Yan, C.; Wang, L.; Lin, J.; Xu, J.; Zhang, T.; Qi, J.; Li, X.; Ni, W.; Wu, G.; Huang, J.; et al. A Fully Automatic Artificial Intelligence–Based CT Image Analysis System for Accurate Detection, Diagnosis, and Quantitative Severity Evaluation of Pulmonary Tuberculosis. Eur. Radiol. 2022, 32, 2188–2199. [CrossRef] [PubMed]
- Mangione, S.; Nieman, L.Z. Pulmonary Auscultatory Skills during Training in Internal Medicine and Family Practice. Am. J. Respir. Crit. Care Med. 1999, 159, 1119–1124. [CrossRef] [PubMed]
- Lawn, D.S.D.; Wood, R.; Cock, K.M. De; Kranzer, K.; Lewis, J.J.; Gavin J Churchyard Antiretrovirals and Isoniazid Preventive Therapy in the Prevention of HIV-Associated Tuberculosis in Settings with Limited Health-Care Resources. Lancet Infect Dis 2010, 10, 489–498. [CrossRef] [PubMed]
- Jamal, S.; Khubaib, M.; Gangwar, R.; Grover, S.; Grover, A.; Hasnain, S.E. Artificial Intelligence and Machine Learning Based Prediction of Resistant and Susceptible Mutations in Mycobacterium Tuberculosis. Sci. Rep. 2020, 10, 1–16. [CrossRef] [PubMed]
- Pruthi, S.S.; Billows, N.; Thorpe, J.; Campino, S.; Phelan, J.E.; Mohareb, F.; Clark, T.G. Leveraging Large-Scale Mycobacterium Tuberculosis Whole Genome Sequence Data to Characterise Drug-Resistant Mutations Using Machine Learning and Statistical Approaches. Sci. Rep. 2024, 14, 1–10. [CrossRef] [PubMed]
| Study | Setting and population | AI application | Study design | Key patient / program-level outcomes | Main findings |
| Moodley et al., 2022 | Primary health clinics, South Africa | CAD-enabled digital CXR for TB screening | Prospective implementation study | Screening throughput; confirmatory testing yield | CAD-supported CXR improved screening efficiency and throughput in routine clinic settings, with acceptable referral volumes for confirmatory testing. |
| Velen et al., 2022 | Correctional facilities, South Africa | CAD-enabled digital CXR | Prospective screening evaluation | TB yield; referral volume; operational feasibility | Use of CAD in prisons identified additional TB cases compared with symptom-based screening and supported high-volume screening in a congregate setting. |
| Garg et al., 2025 | Community-based active case finding, Nigeria | Ultraportable CXR with CAD | Economic evaluation alongside implementation | Cost per TB case detected; program costs | CAD-enabled screening was associated with lower cost per TB case detected than symptom-based screening in settings with substantial asymptomatic TB. |
| Qin et al., 2019 | Facility- and community-based screening, multiple countries | CAD-CXR | Comparative diagnostic study with operational implications | Inter-reader variability; workflow implications | CAD reduced inter-reader variability and achieved diagnostic performance comparable to human readers, supporting its potential use as a standardized triage aid in screening workflows. |
| Signorell et al., 2025 (protocol) | Community screening, Lesotho and South Africa | CAD alone vs. CAD + point-of-care CRP | Paired screen-positive pragmatic trial (protocol) | Time to treatment initiation; cost-effectiveness | Designed to evaluate downstream patient- and program-level outcomes beyond diagnostic accuracy; results pending. |
| Bartl et al., 2025 | HIV care cohorts, sub-Saharan Africa | ML-based clinical TB risk model | Retrospective cohort analysis | Incident TB risk stratification | Risk model identified PLHIV at higher risk of developing TB than conventional screening approaches, suggesting potential to improve prioritization for testing or preventive therapy. |
| Kagujje et al., 2023 | Adults with prior TB, Zambia | CAD-CXR for triage | Diagnostic accuracy study with subgroup analysis | False-positive referrals; subgroup performance | CAD performance differed in individuals with prior TB due to residual lung changes, highlighting implications for referral volume and threshold calibration. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




