Submitted:
24 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussions
2.1. Ethnobotanical Survey Outcomes
2.1.1. Characteristics of Respondents
2.1.2. Plants Species Cited by Respondents and Way of Their Use
2.1.2. Frequency of Citation of Additives
2.2. Laboratory Validation
2.2.1. Plant Selection for Laboratory Assessment
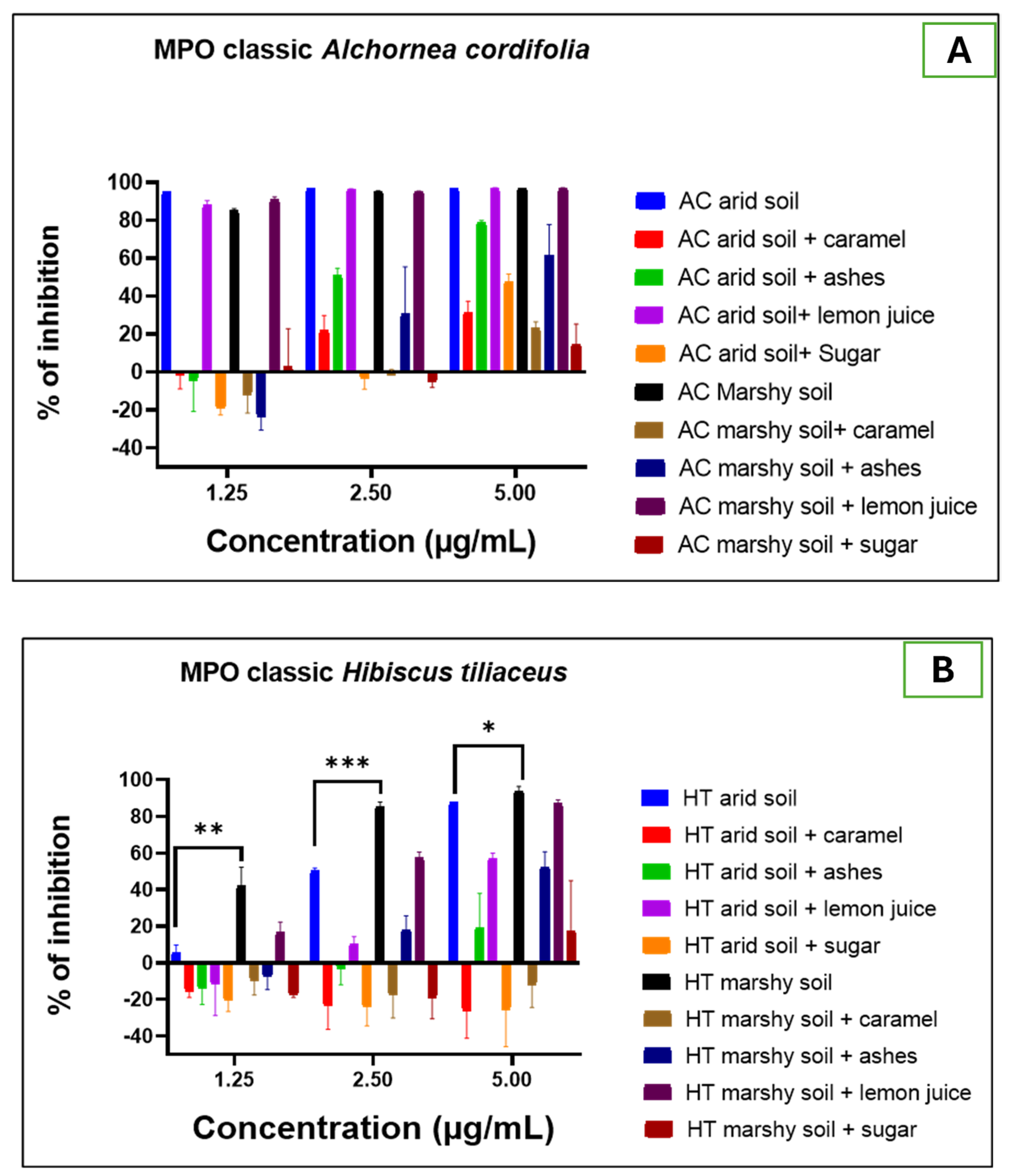
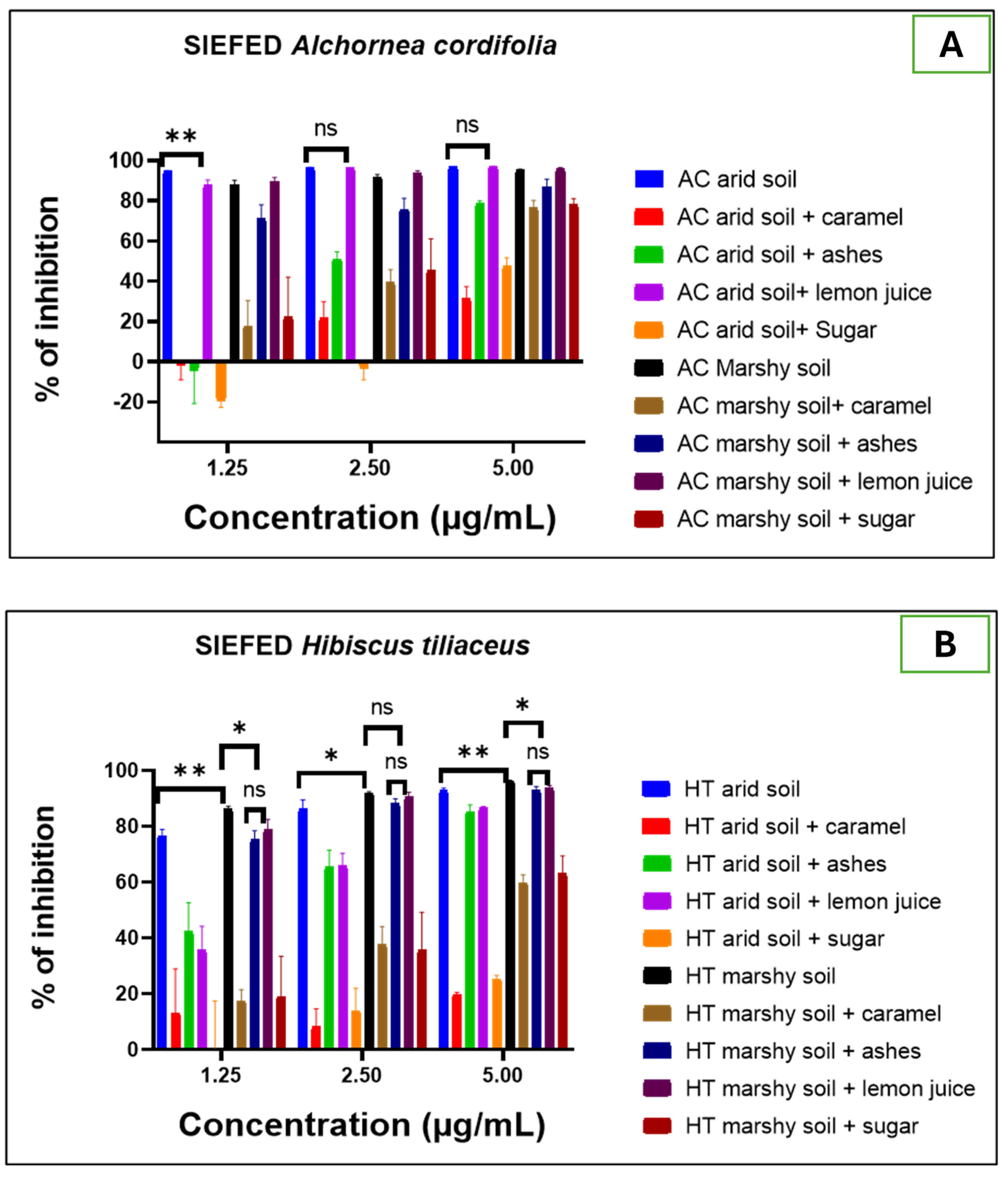
2.2.2. Biological Activities Evaluation
- Anti-inflammatory activity
3. Materials and Methods
3.1. Materials
3.1.1. Survey Area and Period
3.1.2. Vegetable Materials and Reagents for Bioassays
3.1.2.1. Vegetable Materials
3.1.2.2. Reagents
3.2. Methods
3.2.1. Ethnobotanical Survey
3.2.2. Laboratory Assessment
3.2.2.1. Plant Selection and Collection
3.2.2.2. Preparation of Plants Extracts
3.2.2.3. Biological Activities Assays
- Anti-inflammatory activities
- Classical assay of MPO activity
- SIEFED assay of MPO activity
3.2.2.4. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Ethical aspects
References
- Onimoe, G.; Rotz, S. Sickle cell disease: A primary care update. Cleve. Clin. J. Med. 2020, 87, 19–27. [Google Scholar] [CrossRef]
- Ramos-González, E.J.; Bitzer-Quintero, O.K.; Ortiz, G.; Hernández-Cruz, J.J.; Ramírez-Jirano, L.J. Relationship between inflammation and oxidative stress and its effect on multiple sclerosis. Neurología 2024, 39, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Piel, F.B.; Steinberg, M.H.; Rees, D.C. Sickle Cell Disease. N. Engl. J. Med. 2017, 376, 1561–1573. [Google Scholar] [CrossRef]
- El Hoss, S.; Brousse, V. Considering the spleen in sickle cell disease. Expert Rev. Hematol. 2019, 12, 563–573. [Google Scholar] [CrossRef] [PubMed]
- Rees, D.C.; Brousse, V.A.M.; Brewin, J.N. Determinants of severity in sickle cell disease. Blood Rev. 2022, 56, 100983. [Google Scholar] [CrossRef] [PubMed]
- Ranque, B.; Kitenge, R.; Ndiaye, D.D.; Ba, M.D.; Adjoumani, L.; Traore, H.; Coulibaly, C.; Guindo, A.; Boidy, K.; Mbuyi, D.; Ly, I.D.; Offredo, L.; Diallo, D.A.; Tolo, A.; Kafando, E.; Tshilolo, L.; Diagne, I. Estimating the risk of child mortality attributable to sickle cell anemia in sub-Saharan Africa: a retrospective, multicenter, case-control study. Lancet Haematol. 2022, 9, e208–e216. [Google Scholar] [CrossRef]
- WHO Africa region. WHO Africa releases groundbreaking guidance to boost fight against sickle cell disease. 2024. [Google Scholar]
- Tshilolo, L.; Aissi, L.M.; Lukusa, D.; Kinsiama, C.; Wembonyama, S.; Gulbis, B.; Vertongen, F. Neonatal screening for sickle cell anaemia in the Democratic Republic of the Congo: experience from a pioneer project on 31 204 newborns. J. Clin. Pathol. 2009, 62, 35–38. [Google Scholar] [CrossRef]
- Foundation Pierre Fabre The DRC introduces a national plan to combat sickle cell disease. 2020.
- Agasa, B.; Bosunga, K.; Opara, A.; Tshilumba, K.; Dupont, E.; Vertongen, F.; Cotton, F.; Gulbis, B. Prevalence of SCD in a northeastern region of the Democratic Republic of Congo: what impact on transfusion policy? Transfus. Med. 2010, 20, 62–65. [Google Scholar] [CrossRef]
- Kasai, E.T.; Gulbis, B.; Ntokamunda, J.K.; Bours, V.; Batina Agasa, S.; Marini Djang’eing’a, R.; Boemer, F.; Katenga Bosunga, G.; Ngbonda Dauly, N.; Sokoni Vutseme, L.J.; Boso Mokili, B.; Alworong’a Opara, J.P. Newborn screening for SCD in Kisangani, Democratic Republic of the Congo: an update. Hematology 2023, 28. [Google Scholar] [CrossRef]
- Cordeiro, N.J.V., 2003. Phytomedicines (medicines derived from plants) for sickle cell disease, Cochrane Database of Systematic Review. ed.
- Ofakunrin, A.O.D.; Oguche, S.; Adekola, K.; Okpe, E.S.; Afolaranmi, T.O.; Diaku-Akinwumi, I.N.; Zoakah, A.I.; Sagay, A.S. Effectiveness and Safety of Hydroxyurea in the Treatment of Sickle Cell Anaemia Children in Jos, North Central Nigeria. J. Trop. Pediatr. 2020, 66, 290–298. [Google Scholar] [CrossRef]
- Wambebe, C.O.; Bamgboye, E.A.; Badru, B.O.; Khamofu, H.; Momoh, J.A.; Ekpeyong, M.; Audu, B.S.; Njoku, S.O.; Nasipuri, N.R.; Kunle, O.O.; Okogun, J.I.; Enwerem, N.M.; Gamaniel, S.K.; Obodozie, O.O.; Samuel, B.; Fojule, G.; Ogunyale, P.O. Efficacy of Niprisan in the prophylactic management of patients with sickle cell disease. Curr. Ther. Res. 2001, 62, 26–34. [Google Scholar] [CrossRef]
- Bernaudin, F. Why, Who, When, and How? Rationale for Considering Allogeneic Stem Cell Transplantation in Children with Sickle Cell Disease. J. Clin. Med. 2019, 8, 1523. [Google Scholar] [CrossRef] [PubMed]
- Tshilolo, L.; Gonzalez, J.-P. Stigmatization of SCD across the Democratic Republic of Congo: A presentation of two cases. Int. Health Trends Perspect. 2024, 4, 181–186. [Google Scholar] [CrossRef]
- Ware, R.E. Is Sickle Cell Anemia a Neglected Tropical Disease? PLoS Negl. Trop. Dis. 2013, 7, e2120. [Google Scholar] [CrossRef] [PubMed]
- Kambale-Kombi, P.; Marini Djang’eing’a, R.; Alworong’a Opara, J.-P.; Minon, J.-M.; Boemer, F.; Bours, V.; Tonen-Wolyec, S.; Kayembe Tshilumba, C.; Batina-Agasa, S. Management of sickle cell disease: current practices and challenges in a northeastern region of the Democratic Republic of the Congo. Hematology 2021, 26, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Mukinayi, B.M.; Cibeyibeyi, G.K.; Disashi, G.T.; Gulbis, B. Sickle cell disease in the Democratic Republic of Congo: What are the obstacles to treatment with hydroxyurea? Pan. Afr. Med. J. 2021, 38, 41. [Google Scholar] [CrossRef] [PubMed]
- Ngunde-te-Ngunde, Samy. Ethno-botanical Survey on Medicinal Plants Traditionally Used to Treat Sickle Cell Anemia in Yakoma Territory (Nord-Ubangi, D. R. Congo). International Journal of Plant Science and Ecology 2020, 6, 7–13. [Google Scholar]
- Nurain, I.O.; Bewaji, C.O.; Johnson, J.S.; Davenport, R.D.; Zhang, Y. Potential of Three Ethnomedicinal Plants as Antisickling Agents. Mol. Pharm. 2017, 14, 172–182. [Google Scholar] [CrossRef]
- Sani, I.; Ukwuani-Kwaja, A.N.; Haruna, M. Ethnobotanical Survey and In vitro Antisickling Effect of Some Selected Medicinal Plants. Asian J. Res. Biochem. 2021, 1–14. [Google Scholar] [CrossRef]
- Yembeau, N.L.; Biapa Nya, P.C.; Pieme, C.A.; Tchouane, K.D.; Kengne Fotsing, C.B.; Nya Nkwikeu, P.J.; Feudjio, A.F.; Telefo, P.B. Ethnopharmacological Study of the Medicinal Plants Used in the Treatment of Sickle Cell Anemia in the West Region of Cameroon. Evid. Based Complement. Alternat. Med. 2022, 1–10. [Google Scholar] [CrossRef]
- Crocq, L.; Dalligand, L.; Villerbu, L.; Tarquinio, C.; Duchet, C.; Coq, J.M.; Chidiac, N.; Vitry, M. Traumatismes psychiques. Prise en charge psychologique des victimes; Elsevier-Masson: Issy-les-Moulineaux, 2007; p. p. 308. [Google Scholar]
- Kgatla, S.T.; Park, J. Healing in Herero culture and Namibian African independent churches. HTS Teol. Stud. Theol. Stud. 2015, 71, 9 pages. [Google Scholar] [CrossRef]
- Mutombo, P.N.; Kasilo, O.M.J.; James, P.B.; Wardle, J.; Kunle, O.; Katerere, D.; Wambebe, C.; Matsabisa, M.G.; Rahmatullah, M.; Nikiema, J.-B.; Mukankubito, I.; Sheridan, R.; Sanogo, R.; Nissapatorn, V.; Sivakorn, C.; Tripathy, S.; Goyal, R.; Dhobi, M. Experiences and challenges of African traditional medicine: lessons from COVID-19 pandemic. BMJ Glob. Health 2023, 8, e010813. [Google Scholar] [CrossRef] [PubMed]
- Shoko, T. Karanga Traditional Medicine And Healing. Afr. J. Tradit. Complement. Altern. Med. 2008, 4, 501. [Google Scholar] [CrossRef]
- Cook, J.D.; Reddy, M.B. Effect of ascorbic acid intake on nonheme-iron absorption from a complete diet. Am. J. Clin. Nutr. 2001, 73, 93–98. [Google Scholar] [CrossRef]
- Hebbel, R.P. Reconstructing sickle cell disease: A data-based analysis of the “hyperhaemolysis paradigm” for pulmonary hypertension from the perspective of evidence-based medicine. Am. J. Hematol. 2011, 86, 123–154. [Google Scholar] [CrossRef]
- Nur, E.; Biemond, B.J.; Otten, H.; Brandjes, D.P.; Schnog, J.B. the CURAMA Study Group, 2011. Oxidative stress in sickle cell disease; pathophysiology and potential implications for disease management. Am. J. Hematol. 86, 484–489. [CrossRef]
- Zhang, H.; Xu, H.; Weihrauch, D.; Jones, D.W.; Jing, X.; Shi, Y.; Gourlay, D.; Oldham, K.T.; Hillery, C.A.; Pritchard, K.A. Inhibition of myeloperoxidase decreases vascular oxidative stress and increases vasodilatation in SCD mice. J. Lipid Res. 2013, 54, 3009–3015. [Google Scholar] [CrossRef] [PubMed]
- Degotte, G.; Frederich, M.; Francotte, P.; Franck, T.; Colson, T.; Serteyn, D.; Mouithys-Mickalad, A. Targeting Myeloperoxidase Activity and Neutrophil ROS Production to Modulate Redox Process: Effect of Ellagic Acid and Analogues. Molecules 2023, 28, 4516. [Google Scholar] [CrossRef] [PubMed]
- Franck, T.; Grulke, S.; Deby-Dupont, G.; Deby, C.; Duvivier, H.; Peters, F.; Serteyn, D. Development of an Enzyme-Linked Immunosorbent Assay for Specific Equine Neutrophil Myeloperoxidase Measurement in Blood. J. Vet. Diagn. Invest. 2005, 17, 412–419. [Google Scholar] [CrossRef]
- Borive Amani, M.; Frederich, M.; Jansen, O.; Bonnet, O.; Ledoux, A.; Memvanga, P.B.; Batina Agasa, S.; Mouithys-Mickalad, A.; Djang’eing’a, R.M. Phytochemical Characterization of Hibiscus tiliaceus L. Leaves and Evaluation of Their Antisickling, Antioxidant, and Anti-Inflammatory Activities. Molecules 2025, 30, 1765. [Google Scholar] [CrossRef]
- Oruka, O.; Achuba, F.I. In vitro Antioxidant and Anti-Inflammatory Activities of Aqueous Leaf Extract of Alchornea cordifolia. J. Appl. Sci. Environ. Manag. 2023, 27, 299–304. [Google Scholar] [CrossRef]
- Poret, M.; Tran, T.; Villotte, M.; Nüsse, O. Myeloperoxidase: a clever strategist in the fight against pathogen infection. medicine/science 2017, 33, 741–743. [Google Scholar] [CrossRef]
- Manga, H.M.; Brkic, D.; Marie, D.E.P.; Quetin-Leclercq, J. In vivo anti-inflammatory activity of Alchornea cordifolia (Schumach. & Thonn.) Müll. Arg. (Euphorbiaceae). J. Ethnopharmacol. 2004, 92, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Vinh, L.B.; Nguyet, N.T.M.; Thanh, C.D.; Huong, T.T.; Tram, L.H.; Van Thong, N.; Minh, N.H.; Thao, N.P.; Hwang, I.; Yang, S.Y.; Kim, Y.H. Chemical constituents of Vietnamese mangrove Hibiscus tiliaceus with antioxidant and alpha-glucosidase inhibitory activity. Nat. Prod. Res. 2021, 35, 2899–2904. [Google Scholar] [CrossRef] [PubMed]
- Rajeswari, G.; Priyanka, B.; Amrutha, R.E.; Rajaram, C.; Kanhere, R.S.; Nelson Kumar, S. Hibiscus tiliaceus: A possible immunomodulatory agent. J. Pharm. Res. 2013, 6, 742–747. [Google Scholar] [CrossRef]
- Surana, A.R.; Kumbhare, M.R.; Gunjal, A.R.; Goswami, S.S.; Ghuge, D.M. Chemical characterization, thrombolytic and antioxidant activity of Hibiscus tiliaceus L. leaves. Nat. Prod. Res. 2022, 36, 6106–6110. [Google Scholar] [CrossRef] [PubMed]
- Djimeli, M.N.; Fodouop, S.P.C.; Njateng, G.S.S.; Fokunang, C.; Tala, D.S.; Kengni, F.; Gatsing, D. Antibacterial activities and toxicological study of the aqueous extract from leaves of Alchornea cordifolia (Euphorbiaceae). BMC Complement. Altern. Med. 2017, 17, 349. [Google Scholar] [CrossRef]
- Adounkpe, F.; Ayena, A.C.; Aholoukpe, V.; Dougnon, V.; Klotoe, J.-R.; Medehouenou, M.; Baba-Moussa, L. Use of the leaves of Alchornea cordifolia (Schumach. & Thonn.) Müll (Euphorbiaceae) and prospects for treatment of infections due to multidrug-resistant bacteria. Bull. Natl. Res. Cent. 2022, 46, 132. [Google Scholar] [CrossRef]
- Charan, J.; Biswas, T. How to Calculate Sample Size for Different Study Designs in Medical Research? Indian J. Psychol. Med. 2013, 35, 121–126. [Google Scholar] [CrossRef]
- Cochran, W.G. Sampling Techniques, 3rd Edition; John Wiley & Sons, Ltd: New York, 1977. [Google Scholar]
- Thaipong, K.; Boonprakob, U.; Crosby, K.; Cisneros-Zevallos, L.; Hawkins Byrne, D. Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J. Food Compos. Anal. 2006, 19, 669–675. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef] [PubMed]
- Widowati, W.; Rani, A.P.; Hamzah, R.A.; Arumwardana, S.; Afifah, E.; Kusuma, H.S.W.; Rihibiha, D.D.; Nufus, H.; Amalia, A. Antioxidant and Antiaging Assays of Hibiscus sabdariffa Extract and Its Compounds. Nat. Prod. Sci. 2017, 23, 192. [Google Scholar] [CrossRef]
- Nyssen, P.; Mouithys-Mickalad, A.; Minguet, G.; Sauvage, E.; Wouters, J.; Franck, T.; Hoebeke, M. Morphine, a potential inhibitor of myeloperoxidase activity. Biochim. Biophys. Acta BBA - Gen. Subj. 2018, 1862, 2236–2244. [Google Scholar] [CrossRef] [PubMed]
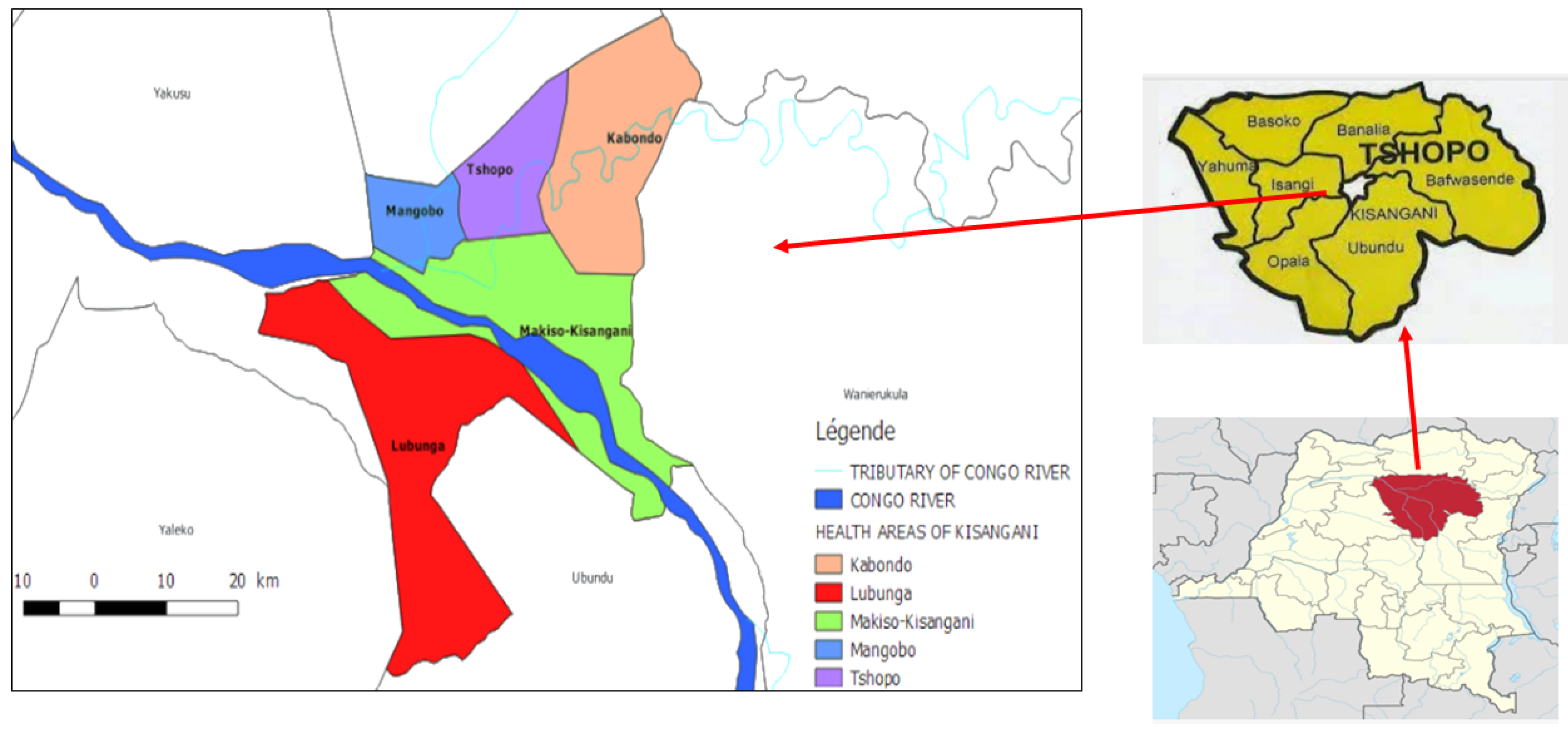
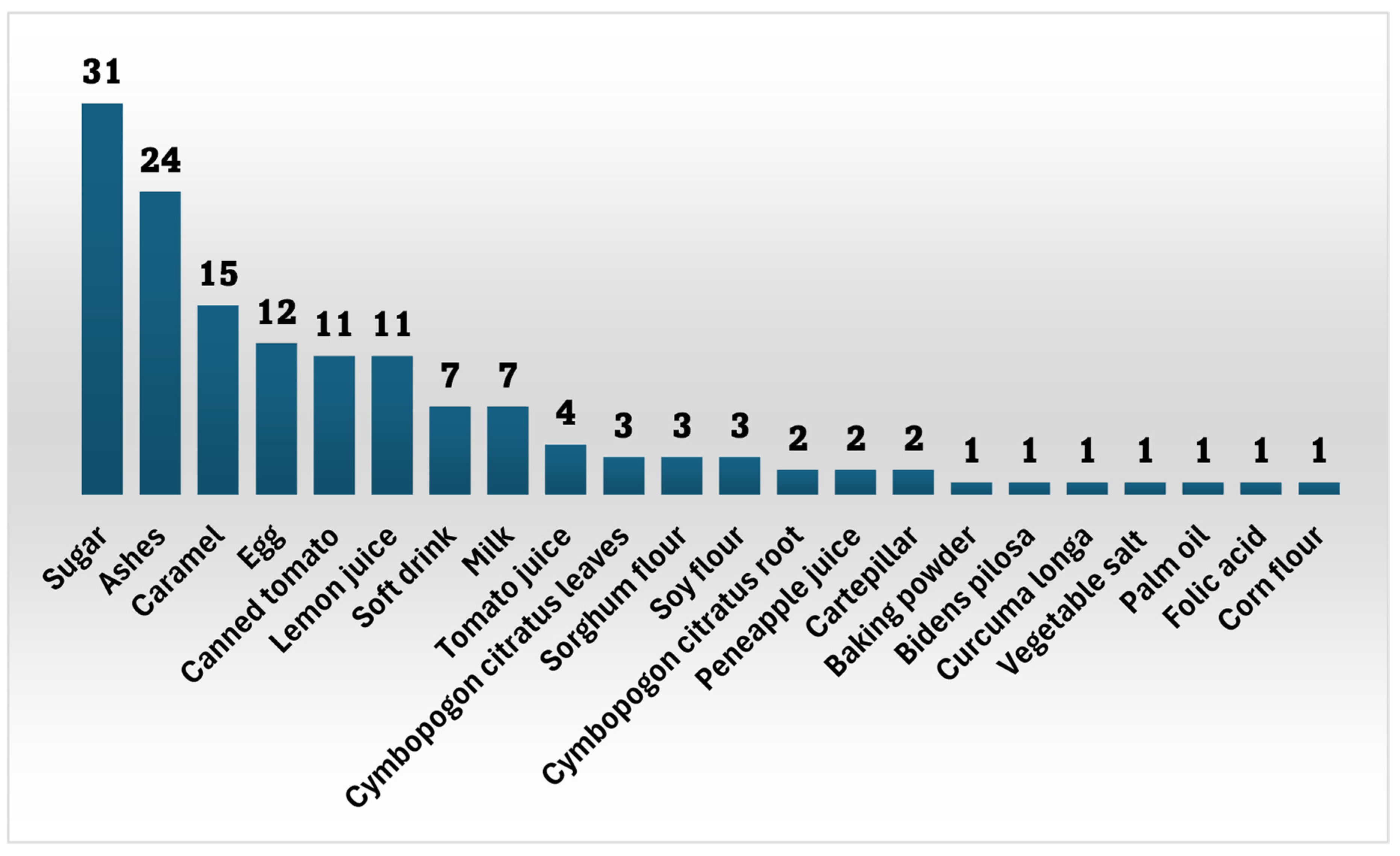
| Deposit number | Plant names | VN cited by participants | Family | Used parts | Preparation method | Administration route | Combination | FC | UR |
|---|---|---|---|---|---|---|---|---|---|
| MBA/01 | Acmella paniculata (Wall. ex DC) R.K.Jansen | Kekemu | Asteraceae | WP, FL | Dec | Rectal, Oral | nd | 2 | 2 |
| MBA/05 | Alchornea cordifolia (Schumach.) Müll.Arg. | Mabanzi | Euphorbiaceae | FL, DLT | Inf, Dec, Mac | Oral | Ash, sugar, Caramel, canned tomato, Lemon juice | 17 | 3 |
| nd | Amaranthus cruentus L. | Muchicha | Amaranthaceae | FL | Cooking | Oral | Sugar, Limon Juice | 3 | 3 |
| nd | Ananas comosus (L.) Merr. | Anana | Bromeliaceae | Fruit | Mac, EJ, Dec | Oral | Canned tomato and Soft drink, Sugar, Eggs, Caramel | 7 | 6 |
| MBA/25 | Andasonia digitata L. | Liguma | Malvaceae | FL, DF | Dec, Mac | Oral | Sugar, Sugar milk, Eggs and folic acid, Caramel, Milk and eggs, Eggs and sugar | 8 | 6 |
| MBA/35 | Anisopappus chinensis Hook.& Arn. | Nzete ya makila | Asteraceae | FL, Flower | Mac, Dec | Oral, Cutaneous | Ash | 5 | 4 |
| MBA/28 | Annona reticulata L. | Bizabibu, Libombi | Annonaceae | FL | Dec | Oral | Ash | 1 | 1 |
| nd | Annonidium manii (Oliv.) | Nzete ya bombi | Annonaceae | FL, Bark | Dec | Oral, Cutaneous | nd | 4 | 2 |
| MBA/6 | Bidens pilosa L. | Police | Asteraceae | WP | Dec | Oral | nd | 1 | 1 |
| MBA/20 | Bridelia atroviridis Müll.Arg. | Mgiangange | Phyllanthaceae | FL | Dec | Oral | nd | 1 | 1 |
| MBA/07 | Carica papaya L. | Payipayi | Caricaceae | FL, DLT |
Dec, Mac | Oral, Cutaneous | C.citratus root, Ash, Caramel, Peneapple juice, Sugar, Canned tomato, Soy flour, Caterpillar | 14 | 13 |
| MBA/23 | Catharanthus roseus (L.) G. Don | Apocynaceae | Flower | Dec | Oral | Sugar | 1 | 1 | |
| MBA/11 | Cocos nucifera L. | Cocoti | Arecaceae | Juice, FL | Mixt, Dec | Oral | Soft drink, Sugar | 3 | 3 |
| MBA/29 | Coffea robusta L. Linden | Kafé | Rubiaceae | FL, Seed | Dec, Mac, Grinding | Oral | C. citratus leaves, Sugar milk and Eggs, Caramel, Ash | 4 | 5 |
| MBA/22 | Cosmos sulphureus Cav. | Maloti | Asteraceae | Flower, leaves, Root | Mac, EJ, Dec Grinding | Oral | Ash, Sugar | 10 | 9 |
| nd | Cucurbita pepo L. | Kasa ya maboke, djurubi | Cucurbitaceae | FL | EJ, Dec, Mac | Oral | Sugar, Ash, Caramel | 6 | 6 |
| MBA/21 | Cymbopogon citratus (DC.)Stapf | Nyasi | Poaceae | FL, Root | Inf, Dec | Oral | Bidens pilosa and sugar, Curcuma longa | 3 | 3 |
| MBA/16 | Elaeis guineensis Jacq. | Ngasi, Nzete ya lito | Arecaceae | Fruit | Mac | Rectal | nd | 1 | 1 |
| nd | Fagara zanthoxyloides (Lam.) B. Zepernick & Timler | - | Rutaceae | FL | Mac, Dec | Oral | Sorghum powder | 3 | 2 |
| nd | Ficus mucuso Welw. Ex Ficalho | Apendanyoka | Moraceae | FL | Dec | Oral | nd | 1 | 1 |
| MBA/13 | Harungana madagascariensis Lam.ex Poir. | Botondolondo | Hypericaceae | FL | Dec | Oral | Sugar | 2 | 2 |
| MBA/33 | Hibiscus sabdariffa L. | Ngai-ngai | Malvaceae | FL | Dec, Cooking | Oral | C. citratus leaves, Sugar | 7 | 5 |
| MBA/30 | Hibiscus tiliaceus L. | Kasa ya makila | Malvaceae | FL | Dec, EJ, Mac, Inf | Oral, Cutaneous | Ash, Sugar; Soy flour, Corn flour , milk; Tomato juice, soft drink, lemon juice, Caterpillar, C. citratus root and leaves, Caramel, | 42 | 22 |
| MBA/26 | Ipomoea batata (L.) Lam. | Matembela | Convolvulaceae | FL | Mac | Oral | Tomato juice, Eggs and Sugar milk | 2 | 2 |
| MBA/04 | Laportea canadensis Wedd. | Ibenja ou Katolia | Urticaceae | WP | Dec | Rectal, Oral | nd | 3 | 2 |
| MBA/19 | Macaranga spinosa Müll.Arg. | - | Euphorbiaceae | FL | Dec | Oral | nd | 1 | 1 |
| MBA/17 | Macaranga stipulosa Müll. Arg. | - | Euphorbiaceae | FL | Dec | Oral | nd | 1 | 1 |
| MBA/34 | Mangifera indica L. | Manga | Anacardiaceae | FL | Dec | Oral | Eggs | 1 | 1 |
| nd | Manihot esculenta Crantz | Sombe, Mwinja | Euphorbiaceae | FL | Dec, Inf, Mac, Cooking | Oral | Lemon Juice, Milk | 8 | 7 |
| MBA/10 | Morinda morindoides (Baker) Milne-Redh. | Kongo bololo | Rubiaceae | FL | Inf | Oral | nd | 1 | 2 |
| MBA/08 | Moringa oleifera Lam. | Moringa | Moringaceae | FL | Mac, Dec, Inf | Oral | Sugar | 8 | 4 |
| MBA/24 | Musa paradisiaca L. | Makemba | Musaceae | DLT | Dec | Cutaneous, Oral | Ash, Sugar | 4 | 3 |
| MBA/09 | Myrianthus arboreus P. Beauv. | Bokomu | Moraceae | FL | Dec, Mixt | Cutaneous, Oral | Ash | 4 | 3 |
| nd | Oryza sativa L. | Loso | Poaceae | Seed | Grinding and calcined | Oral | Caramel, Sugar | 4 | 4 |
| MBA/12 | Passiflora edulis Sims | Marakuja | Passifloraceae | Fruit, FL | EJ, Dec | Oral | Soft drink, Sugar | 2 | 4 |
| MBA/18 | Persea americana Mill. | Avocati, Isandu igeboka | Lauraceae | FL, Fruit,Pit, Bark | Dec, Mac | Oral, Cutaneous | C,citratus leaves, Sugar milk; Tomato juice; Caramel; Ash; Soft drink; Eggs; Sugar; Baking powder; | 21 | 17 |
| MBA/14 | Ricinus communis L. | Mbalika | Euphorbiaceae | FL | Heating | Cutaneous | nd | 1 | 1 |
| MBA/03 | Senna alata (L.) Roxb. | Folele | Fabaceae | FL | Dec | Oral | Sugar | 1 | 2 |
| MBA/02 | Sida acuta Burm.f. | Omongo, Uende ukamuita mama | Malvaceae | Stem, Root | Grinding and calcined, Dec | Oral | Vegetable salt and palm oil; Ash | 2 | 2 |
| nd | Solanum betaceum Cav. | Damudamu | Solanaceae | FL, Fruit | Dec, EJ | Oral | Sugar , Eggs | 2 | 2 |
| nd | Solanum lycopersicum L. | Tomate | Solanaceae | Fruit | Mixt | Oral | Soft drink | 2 | 3 |
| MBA/32 | Tectona grandis L.f. | Tec | Verbenaceae | FL | Mac, Dec | Cutaneous | Tomato juice, Eggs, Soft drink, Sorghum flour, Caramel, Sugar | 20 | 12 |
| MBA/15 | Terminalia catapa L. | Madamé | Combretaceae | FL | Dec | Cutaneous | Caramel | 4 | 3 |
| MBA/27 | Theobroma cacao L. | Cacao | Malvaceae | FL, DLT | Dec | Oral | Ash, Sugar | 4 | 3 |
| MBA/31 | Zingiber officinal Roscoe | Tangawisi | Zingiberaceae | Root | Inf | Oral | nd | 1 | 3 |
| Sample | Antioxidant expressed as IC50 (in µg/mL) Mean ± Standard deviation (n=3) |
|---|---|
| Alchornea cordifolia (Schumach.) Müll.Arg. | |
| AC arid soil aqueous extract (AC AAe) | 1.76 ± 0.17 |
| AC AAe + caramel | nd |
| AC AAe+ Ash | 12.46 ± 4.51 |
| AC AAe + lemon juice | 2.89 ± 0.26 |
| AC AAe + sugar | nd |
| AC marsh soil aqueous extract (AC Mae) | 3.11 ± 0.10 |
| AC MAe + caramel | nd |
| AC MAe + Ash | 36.04 ± 25.15 |
| AC MAe + lemon juice | 4.99 ± 3.59 |
| AC MAe + sugar | nd |
| Hibiscus tiliaceus L. | |
| HT arid soil aqueous extract (HT AAe) | 18.71 ± 11.78 |
| HT AAe + caramel | nd |
| HT AAe + Ash | nd |
| HT AAe + lemon juice | nd |
| HT AAe + sugar | nd |
| HT marsh soil aqueous extract (HT Mae) | 4.85 ± 0. 86 |
| HT MAe + caramel | nd |
| HT MAe + Ash | 26.22 ± 10.02 |
| HT MAe+ lemon juice | 12.24 ± 3.51 |
| HT MAe + sugar | nd |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




