Submitted:
24 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
Introduction
- Drug toxicity (calcineuron inhibitors, methotrexate, busulfan, other cytotoxic agents, radiation, azoles),
- Opportunistic infections,
- Disruption of metabolic homeostasis due to sepsis or organ failure,
- Recurrence of underlying malignancy in the central nervous system.
Methods
Eligibility Criteria
Data Collection
Transplantation Procedures
Conditioning Regimens
GVHD Prophylaxis and Therapy
Definitions
Calculation of Immune-Inflammatory Indices
Statistical Analyses
Results
Patient, Transplantation, and Graft Characteristics
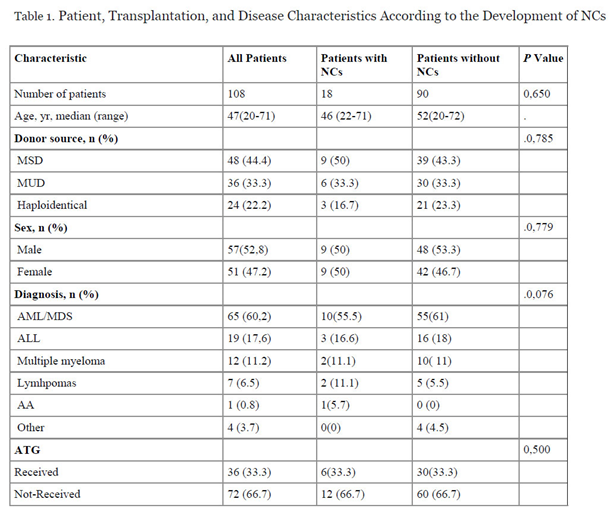
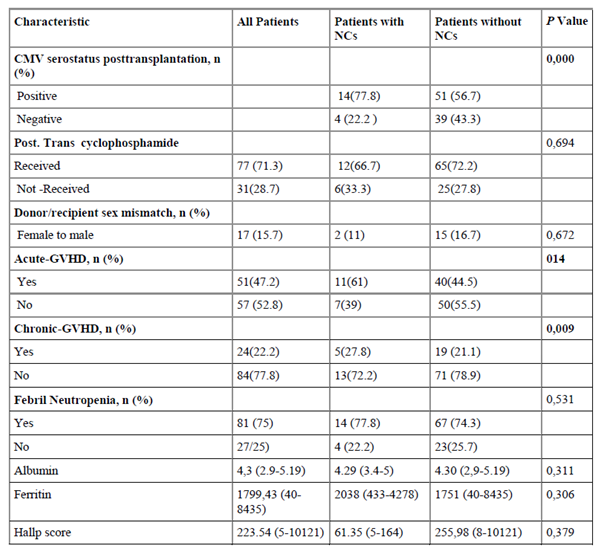
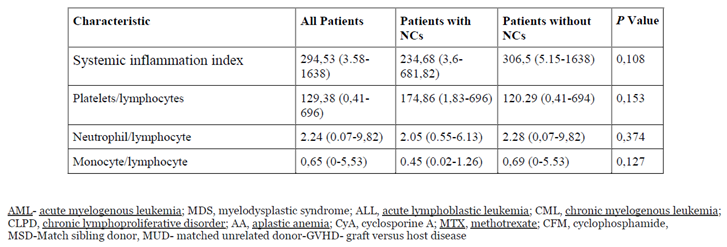
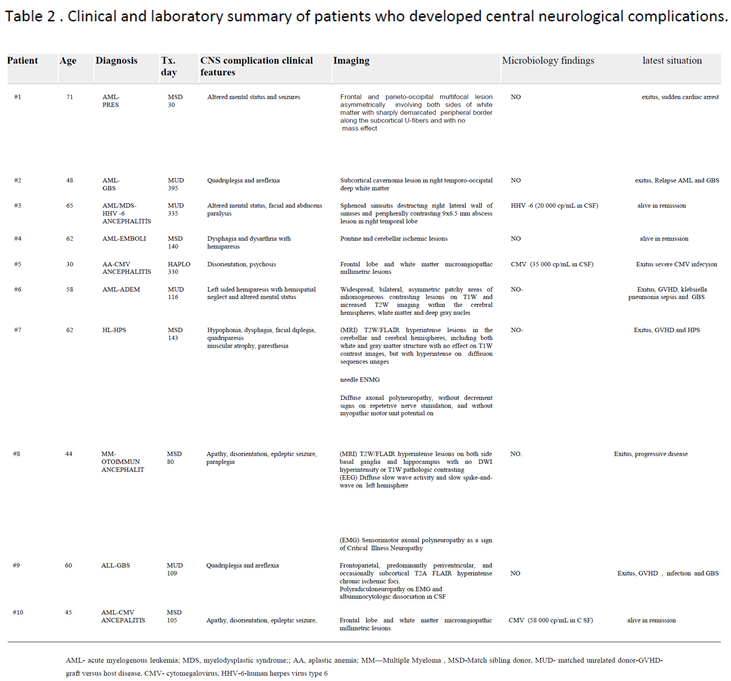
Discussion
Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pruitt, AA; Graus, F; Rosenfeld, MR. Neurological complications of transplantation: Part 1: Hematopoietic cell transplantation. Neurohospitalist 2013, 3, 24–38. [Google Scholar] [CrossRef] [PubMed]
- Maffini, E; Festuccia, M; Brunello, L; Boccadoro, M; Giaccone, L; Bruno, B. Neurologic complications after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2017, 23, 388–397. [Google Scholar] [CrossRef]
- Antonini, G; Ceschin, V; Morino, S; et al. Early neurologic complications following allogeneic bone marrow transplant for leukemia: a prospective study. Neurology 1998, 50, 1441–1445. [Google Scholar] [CrossRef] [PubMed]
- Sostak, P; Padovan, CS; Yousry, TA; Ledderose, G; Kolb, HJ; Straube, A. Prospective evaluation of neurological complications after allogeneic bone marrow transplantation. Neurology 2003, 60, 842–848. [Google Scholar] [CrossRef]
- Kang, JM; Kim, YJ; Kim, JY; et al. Neurologic complications after allogeneic hematopoietic stem cell transplantation in children: analysis of prognostic factors. Biol Blood Marrow Transplant. 2015, 21, 1091–1098. [Google Scholar] [CrossRef]
- Straathof, K; Anoop, P; Allwood, Z; et al. Long-term outcome following cyclosporine-related neurotoxicity in paediatric allogeneic haematopoietic stem cell transplantation. Bone Marrow Transplant. 2017, 52(1), 159–162. [Google Scholar] [CrossRef]
- Schmidt-Hieber, M; Silling, G; Schalk, E; et al. CNS infections in patients with hematological disorders (including allogeneic stem-cell transplantation)-Guidelines of the Infectious Diseases Working Party (AGIHO) of the German Society of Hematology and Medical Oncology (DGHO). Ann Oncol 2016, 27(7), 1207–1225. [Google Scholar] [CrossRef]
- Hanajiri, R; Kobayashi, T; Yoshioka, K; et al. Central nervous system infection following allogeneic hematopoietic stem cell transplantation. Hematol Oncol Stem Cell Ther. 2017, 10(1), 22–28. [Google Scholar] [CrossRef]
- Schmidt-Hieber, M; Engelhard, D; Ullmann, A; et al. Central nervous system disorders after hematopoietic stem cell transplantation: a prospective study of the Infectious Diseases Working Party of EBMT. J Neurol. 2020, 267(2), 430–439. [Google Scholar] [CrossRef]
- Behre, G; Becker, M; Christopeit, M. BK virus encephalitis in an allogeneic hematopoietic stem cell recipient. Bone Marrow Transplant. 2008, 42(7), 499. [Google Scholar] [CrossRef] [PubMed]
- Behre, G; Christopeit, M; Weber, T. Successful treatment of Candida spp. encephalitis occurring under Caspofungin treatment after allogeneic stem cell transplantation. Ann Hematol. 2009, 88(1), 93–94. [Google Scholar] [CrossRef]
- Christopeit, M; Grundhoff, A; Rohde, H; et al. Suspected encephalitis with Candida tropicalis and Fusarium detected by unbiased RNA sequencing. Ann Hematol. 2016, 95(11), 1919–1921. [Google Scholar] [CrossRef]
- Chu, B.; Chen, Y.; Pan, J. Prognostic significance of systemic immune inflammation index for ovarian cancer: An updated systematic review and meta-analysis. J. Ovarian Res. 2025, 18, 41. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, Y.; Guo, C.; Li, S.; Huang, C. Systemic immune-inflammation index as a predictor of survival in non-small cell lung cancer patients undergoing immune checkpoint inhibition: A systematic review and meta-analysis. Crit. Rev. Oncol. Hematol. 2025, 210, 104669. [Google Scholar] [CrossRef]
- Tan, Y.; Hu, B.; Li, Q.; Cao, W. Prognostic value and clinicopathological significance of pre-and post-treatment systemic immune-inflammation index in colorectal cancer patients: A meta-analysis. World J. Surg. Oncol. 2025, 23, 11. [Google Scholar] [CrossRef]
- Hai-Jing, Y.; Shan, R.; Jie-Qiong, X. Prognostic significance of the pretreatment pan-immune-inflammation value in cancer patients: An updated meta-analysis of 30 studies. Front. Nutr. 2023, 10, 1259929. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.C.; Liu, H.; Liu, D.C.; Tong, C.; Liang, X.W.; Chen, R.H. Prognostic value of pan-immune-inflammation value in colorectal cancer patients: A systematic review and meta-analysis. Front. Oncol. 2022, 12, 1036890. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Chen, H.; Chen, Z.; Tan, J.; Wu, F.; Li, X. Prognostic value of neutrophil-to-lymphocyte ratio in patients with hepatocellular carcinoma receiving curative therapies: A systematic review and meta-analysis. BMC Cancer 2025, 25, 571. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wang, Y.; Jiang, Y.; Yang, J.; Zhang, Y. The prognostic impact of neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio on patients with small cell lung cancer receiving first-line platinum-based chemotherapy: A systematic review and meta-analysis. BMC Pulm. Med. 2024, 24, 630. [Google Scholar] [CrossRef]
- Zhang, J.H.; Huang, D.H.; Chen, Z.Y. Prognostic role of systemic immune-inflammation index in solid tumors: A systematic review and meta-analysis. Oncotarget 2017, 8, 75381–75388. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhao, Y.; Wen, J.; Wang, Y.; Li, J. Impact of systemic immune-inflammation index and its evaluation of optimal threshold in patients with limited-stage small cell lung cancer: A retrospective study based on 572 cases. Transl. Cancer Res. 2025, 14, 371–382. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Yin, P.; Jiao, B.; Shi, Z.; Qiao, F.; Xu, J. Detecting the preoperative peripheral blood systemic immune-inflammation index (SII) as a tool for early diagnosis and prognosis of gallbladder cancer. BMC Immunol. 2025, 26, 7. [Google Scholar] [CrossRef]
- Bal, O.; Acikgoz, Y.; Yildiz, B.; Kos, F.T.; Algin, E.; Dogan, M. Simple and easily accessible prognostic markers in ewing sarcoma; neutrophil-lymphocyte ratio, neutrophil-platelet score and systemic-inflammation index. J. Cancer Res. Ther. 2023, 19, 1241–1247. [Google Scholar] [CrossRef] [PubMed]
- Yi, J.; Xue, J.; Yang, L.; Xia, L.; He, W. Predictive value of prognostic nutritional and systemic immune-inflammation indices for patients with microsatellite instability-high metastatic colorectal cancer receiving immunotherapy. Front. Nutr. 2023, 10, 1094189. [Google Scholar] [CrossRef]
- Chen, X.; Hong, X.; Chen, G.; Xue, J.; Huang, J.; Wang, F.; Ali, W.; Li, J.; Zhang, L. The Pan-Immune-Inflammation Value predicts the survival of patients with anaplastic lymphoma kinase-positive non-small cell lung cancer treated with first-line ALK inhibitor. Transl. Oncol. 2022, 17, 101338. [Google Scholar] [CrossRef]
- Fu, F.; Deng, C.; Wen, Z.; Gao, Z.; Zhao, Y.; Han, H.; Zheng, S.; Wang, S.; Li, Y.; Hu, H.; et al. Systemic immune-inflammation index is a stage-dependent prognostic factor in patients with operable non-small cell lung cancer. Transl. Lung Cancer Res. 2021, 10, 3144–3154. [Google Scholar] [CrossRef]
- Howard, R.; Kanetsky, P.A.; Egan, K.M. Exploring the prognostic value of the neutrophil-to-lymphocyte ratio in cancer. Sci. Rep. 2019, 9, 19673. [Google Scholar] [CrossRef]
- Yang, R.; Chang, Q.; Meng, X.; Gao, N.; Wang, W. Prognostic value of Systemic immune-inflammation index in cancer: A meta-analysis. J. Cancer 2018, 9, 3295–3302. [Google Scholar] [CrossRef]
- Hou, Y.; Li, X.; Yang, Y.; Shi, H.; Wang, S.; Gao, M. Serum cytokines and neutrophil-to-lymphocyte ratio as predictive biomarkers of benefit from PD-1 inhibitors in gastric cancer. Front. Immunol. 2023, 14, 1274431. [Google Scholar] [CrossRef]
- Guan, Y.; Xiong, H.; Feng, Y.; Liao, G.; Tong, T.; Pang, J. Revealing the prognostic landscape of neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio in metastatic castration-resistant prostate cancer patients treated with abiraterone or enzalutamide: A meta-analysis. Prostate Cancer Prostatic Dis. 2020, 23, 220–231. [Google Scholar] [CrossRef] [PubMed]
- Sanz, J.; Boluda, J.C.; Martín, C.; et al. Single-unit umbilical cord blood transplantation from unrelated donors in patients with hematological malignancy using busulfan, thiotepa, fludarabine and ATG as myeloablative conditioning regimen. Bone Marrow Transplant. 2012, 47, 1287–1293. [Google Scholar] [CrossRef]
- Sanz, G.F.; Saavedra, S.; Planelles, D.; et al. Standardized, unrelated donor cord blood transplantation in adults with hematologic malignancies Blood. 2001, 98, 2332–2338. [Google Scholar] [CrossRef]
- Seigers, R.; Fardell, J.E. Neurobiological basis of chemotherapy-induced cognitive impairment: A review of rodent research. Neurosci. Biobehav. Rev. 2011, 35, 729–741. [Google Scholar] [CrossRef]
- Uckan, D.; Çetinkaya, D.U.; Yigitkanli, I.; Tezcan, I.; Tuncer, M.; Karasimav, D.; Oguz, K.K.; Topcu, M. Life-threatening neurological complications after bone marrow transplantation in children. Bone Marrow Transplant. 2004, 35, 71–7. [Google Scholar] [CrossRef]
- Koh, K.N.; Park, M.; Kim, B.E.; Im, H.J.; Seo, J.J. Early central nervous system complications after allogeneic hematopoietic stem cell transplantation in children. Korean J. Hematol. 2010, 45, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Sanz, J.; Picardi, A.; Hernández Boluda, J.C.; et al. Impact of graft-versus-host disease prophylaxis on outcomes after myeloablative single-unit umbilical cord blood transplantation. Biol Blood Marrow Transplant. 2013, 19, 1387–1392. [Google Scholar] [CrossRef]
- Hinchey, J.; Chaves, C.; Appignani, B.; et al. A reversible posterior leukoencephalopathy syndrome N. Engl J Med. 1996, 334, 494–500. [Google Scholar] [CrossRef]
- Feske, S.K. Posterior reversible encephalopathy syndrome: a review. Semin Neurol 2011, 31, 202–215. [Google Scholar] [CrossRef]
- Balaguer-Rosello, A.; Bataller, L.; Lorenzo, I.; et al. Infections of the central nervous system after unrelated donor umbilical cord blood transplantation or human leukocyte antigen-matched sibling transplantation. Biol Blood Marrow Transplant. 2017, 23, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Gray, R.J. A class of K-sample tests for comparing the cumulative incidence of a competing risk. Ann Stat. 1988, 16, 1141–1154. [Google Scholar] [CrossRef]
- Evans, LE; Taylor, JL; Smith, CJ; et al. Cardiovascular comorbidities, inflammation, and cerebral small vessel disease. Cardiovasc Res. 2021, 117(13), 2575–2588. [Google Scholar] [CrossRef]
- Rawish, E; Nording, H; Münte, T; et al. Platelets as mediators of neuroinflammation and thrombosis. Front Immunol. 2020, 11, 548631. [Google Scholar] [CrossRef]
- Cox, D.R. Regression models and life-tables. J R Stat Soc Ser B Methodol. 1972, 34, 187–202. [Google Scholar] [CrossRef]
- Mantel, N. Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemother Rep. 1966, 50, 163–170. [Google Scholar]
- Siegal, D.; Keller, A.; Xu, W.; et al. Central nervous system complications after allogeneic hematopoietic stem cell transplantation: incidence, manifestations, and clinical significance. Biol Blood Marrow Transplant. 2007, 13, 1369–1379. [Google Scholar] [CrossRef] [PubMed]
- Narimatsu, H.; Miyamura, K.; Iida, H.; et al. Early central nervous complications after umbilical cord blood transplantation for adults. Biol Blood Marrow Transplant. 2009, 15, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Husain, S. Infections of the central nervous system in transplant recipients. Transpl Infect Dis. 2000, 2, 101–111. [Google Scholar] [CrossRef]
- Beitinjaneh, A.; McKinney, A.M.; Cao, Q.; et al. Toxic leukoencephalopathy following fludarabine-associated hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2011, 17, 300–308. [Google Scholar] [CrossRef]
- Rambaldi, A.; Grassi, A.; Masciulli, A. Busulfan plus cyclophosphamide versus busulfan plus fludarabine as a preparative regimen for allogeneic haemopoietic stem-cell transplantation in patients with acute myeloid leukaemia: an open-label, multicentre, randomised, phase 3 trial. Lancet Oncol. 2015, 16, 1525–1536. [Google Scholar] [CrossRef]
- Hernández-Boluda, J.C.; Lis, M.J.; Goterris, R.; et al. Guillain-Barré syndrome associated with cytomegalovirus infection after allogeneic hematopoietic stem cell transplantation. Transpl Infect Dis. 2005, 7, 93–96. [Google Scholar] [CrossRef] [PubMed]
- Antonini, G.; Ceschin, V.; Morino, S.; et al. Early neurologic complications following allogeneic bone marrow transplant for leukemia: a prospective study. Neurology 1998, 50, 1441–1445. [Google Scholar] [CrossRef] [PubMed]
- Barba, P.; Piñana, J.L.; Valcárcel, D.; et al. Early and late neurological complications after reduced-intensity conditioning allogeneic stem cell transplantation. Biol Blood Marrow Transplant. 2009, 15, 1439–1446. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



