Submitted:
24 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Subjects
2.2. Sample Size Calculation
2.3. Data Collection
2.4. Outcome Measures
2.5. Statistical Methods
3. Results
3.1. Baseline Characteristics of Patients
3.2. Univariate Analysis of Postoperative Complications
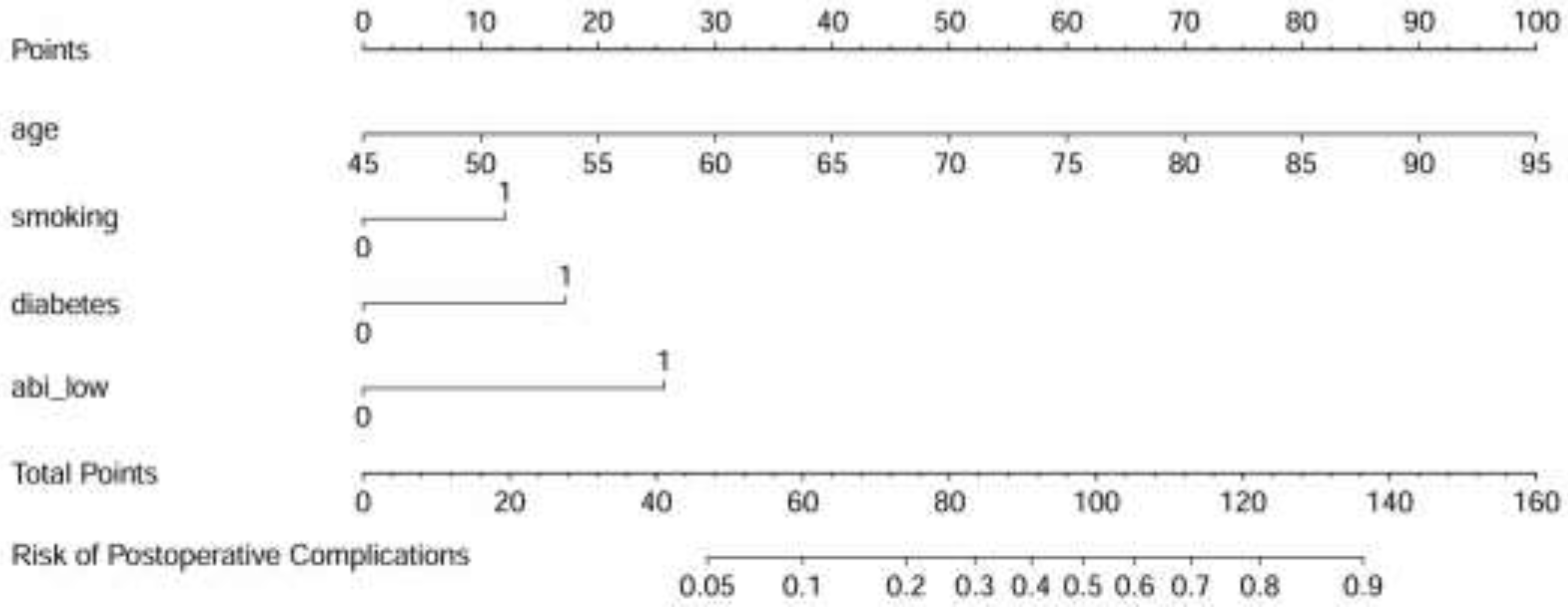
3.3. Multivariate Logistic Regression Analysis and Nomogram Model Construction
3.4. Model Performance Validation
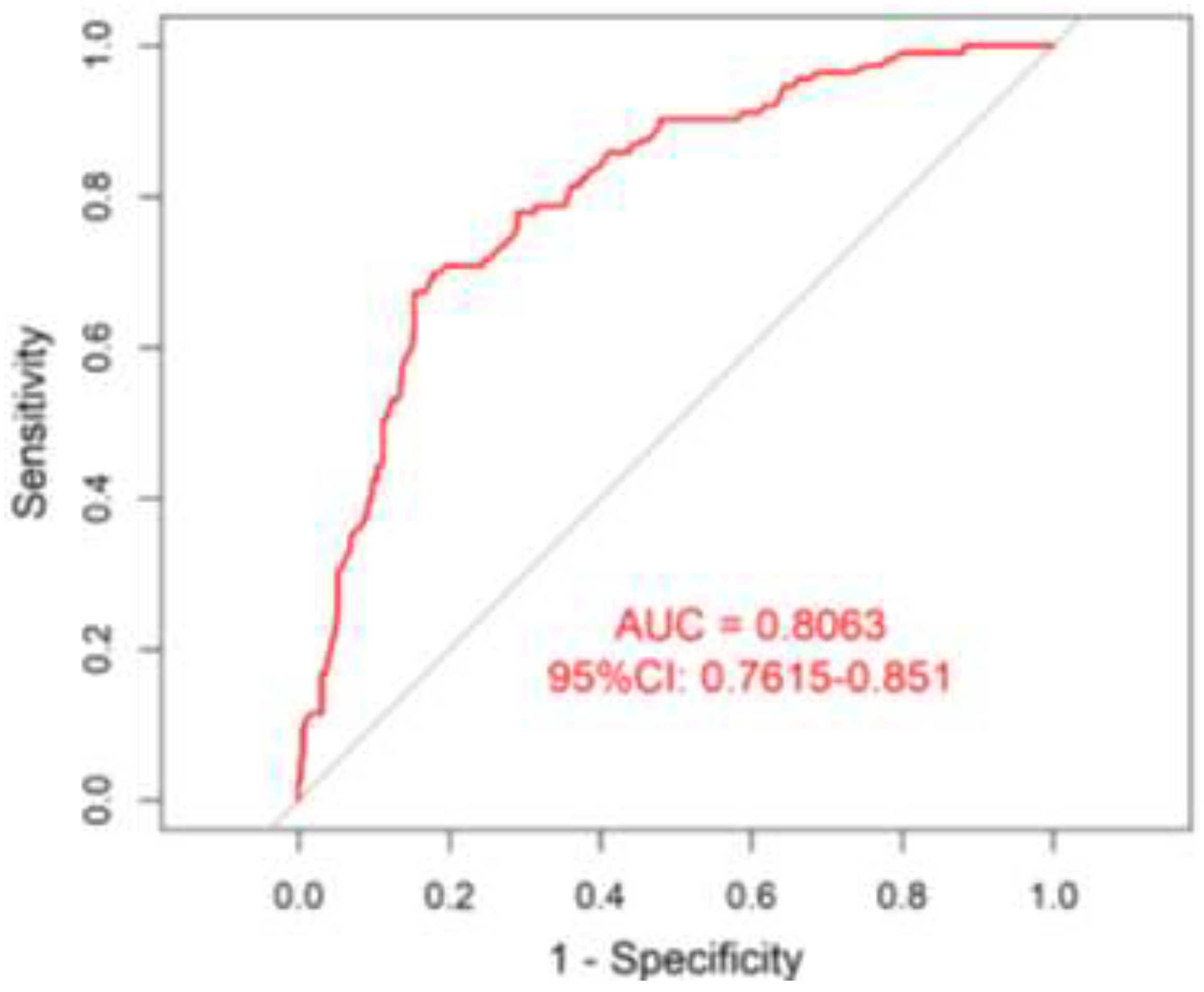
3.4.1. Discrimination Evaluation
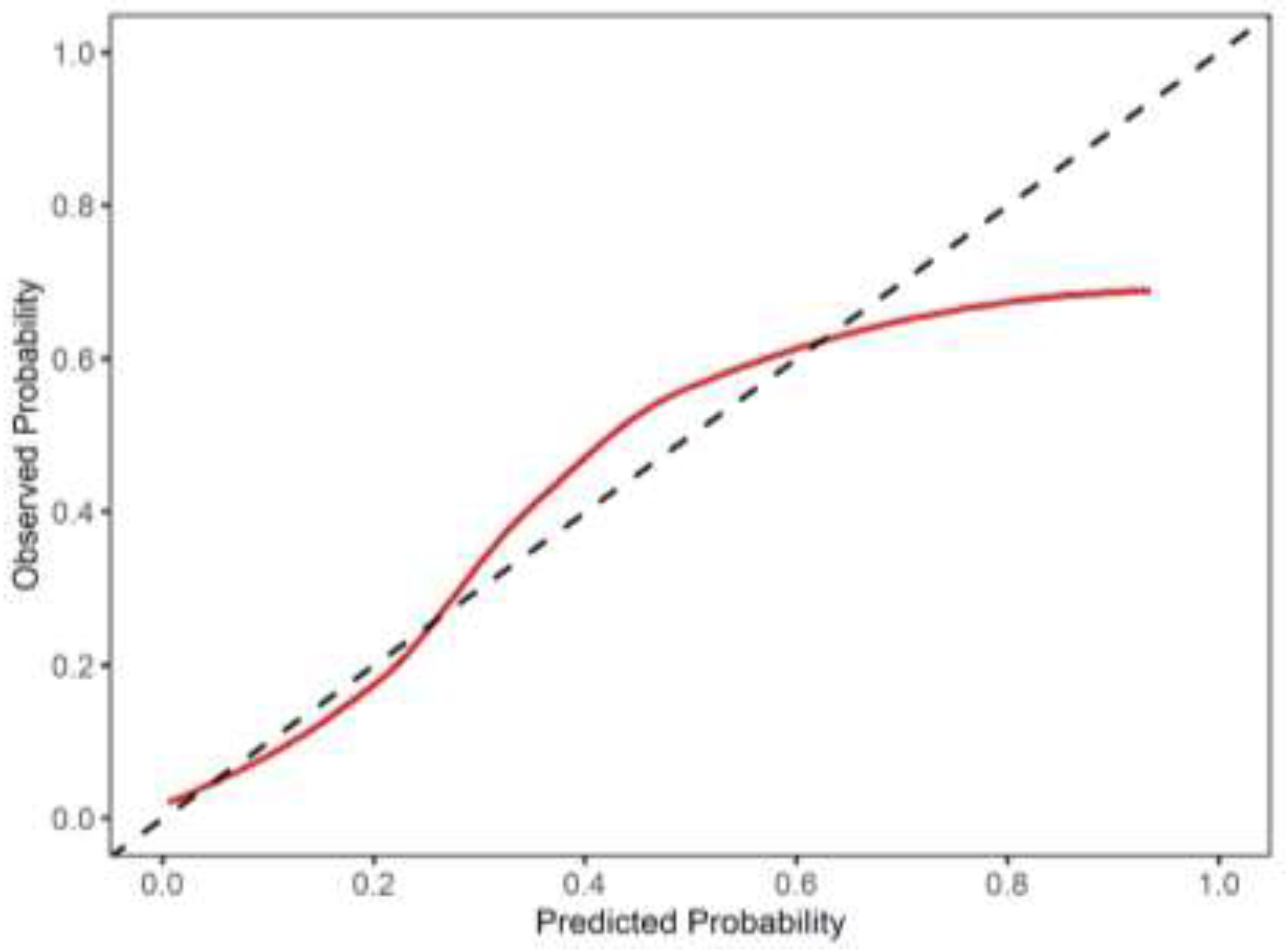
3.4.2. Calibration Evaluation
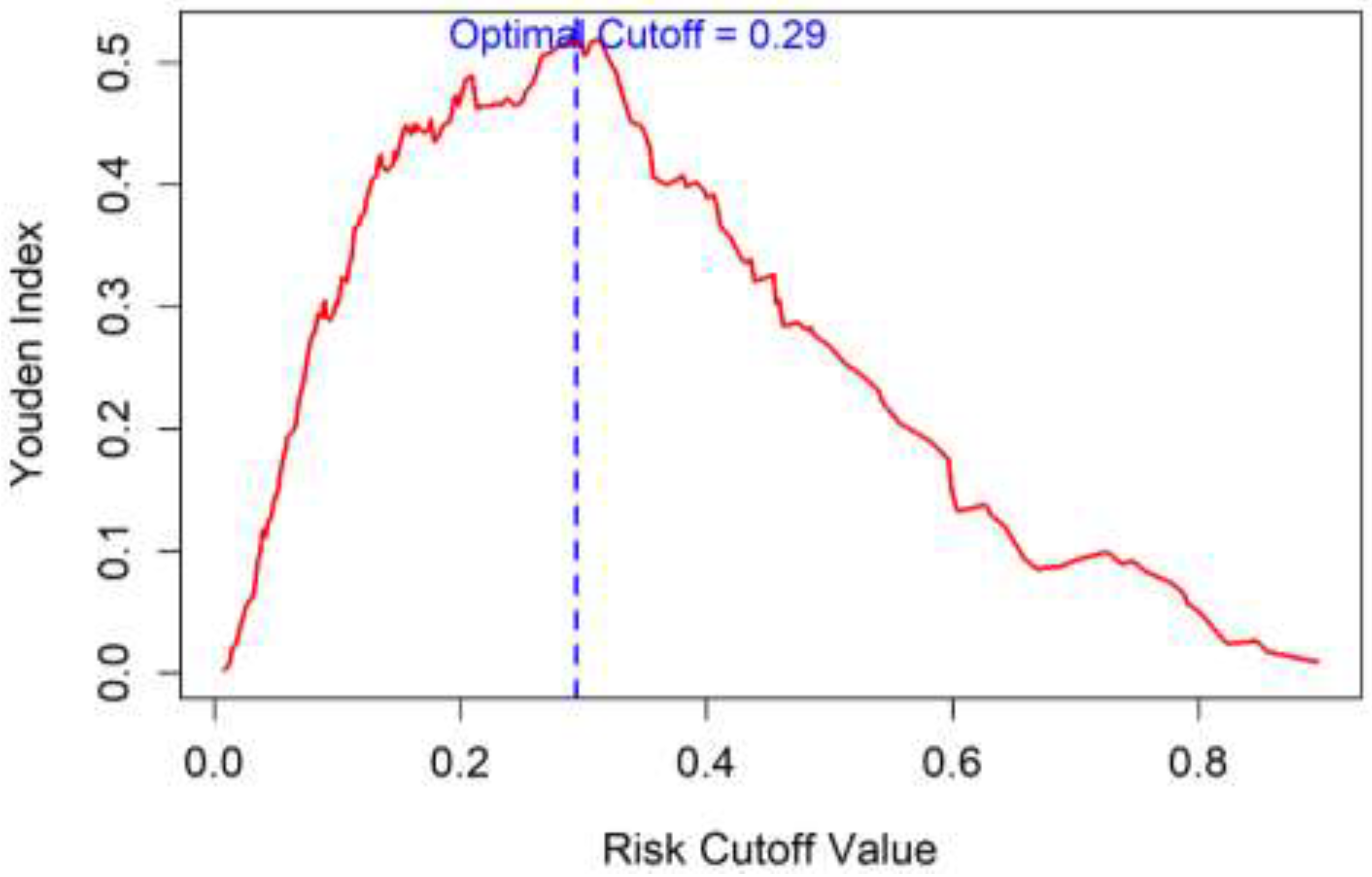
3.4.3. Optimal Cutoff Value Analysis
4. Discussion
4.1. Correlation Mechanism Between Preoperative ABI and Postoperative Complications
4.2. Clinical Significance of Other Independent Risk Factors
4.3. Clinical Application Value of the Nomogram Model
4.4. Clinical Explanation for Higher ABI in the Conservative Treatment Group
4.5. Limitations of the Study
5. Conclusion
6. Declarations
References
- Victor, A; Jean-Baptiste, R. The ‘Ten Commandments’ of 2017 ESC Guidelines on the Diagnosis and Treatment of Peripheral Arterial Diseases. European heart journal 2018, 39(9), 722. [Google Scholar]
- Jie, Z; Guojun, Z; Lin, Z. Implementation of holistic nursing interventions based on fast track surgery concept in patients with lower extremity arterial occlusive disease[J]. Medicine 2023, 102(49), e36485. [Google Scholar]
- R G F F ,D G M ,I B , et al. Ankle Brachial Index Combined With Framingham Risk Score to Predict Cardiovascular Events and Mortality: A Meta-analysis[J]. JAMA: The Journal of the American Medical Association 2008, 300(2), 197–208.
- L N ,R W H ,A J D , et al. Inter-Society Consensus for the Management of Peripheral Arterial Disease (TASC II).[J]. Journal of vascular surgery 2007, 45, Suppl SS5-67.
- Conte, S.M.; Bradbury, W.A.; Kolh, P. Global vascular guidelines on the management of chronic limb-threatening ischemia[J]. Journal of Vascular Surgery 2019, 69(6), 3S-125S.e40. [Google Scholar] [CrossRef] [PubMed]
- Moshkele, H.M.; Mirmoosavi, S.; Najafi, T.M. Low Ankle-Brachial Index Is Associated With Albuminuria and Diabetic Kidney Disease in Type 2 Diabetes; A Cross Sectional Study.[J]. Endocrinology, diabetes & metabolism 2025, 8(6), e70115. [Google Scholar]
- Bhasin, N; Scott, A J D. Ankle Brachial Pressure Index: identifying cardiovascular risk and improving diagnostic accuracy[J]. Journal of the Royal Society of Medicine 2007, 100(1), 4–5. [Google Scholar] [CrossRef] [PubMed]
- Che, D; Jiang, Z; Xiang, X. Predictors of amputation in patients with diabetic foot ulcers: a multi-centre retrospective cohort study. Endocrine 2024, 85(1), 181–189. [Google Scholar] [CrossRef] [PubMed]
- Tang, T; Shen, Y; Bai, X. Predicting surgical site infections using a novel nomogram in patients undergoing hepatectomy[J]. HPB 2019, 21(S2), S361–S361. [Google Scholar] [CrossRef]
- Lai, J; Lin, P; Zhuang, J. Development and internal validation of a nomogram based on peripheral blood inflammatory markers for predicting prognosis in nasopharyngeal carcinoma.[J]. Cancer medicine 2024, 13(7), e7135–e7135. [Google Scholar] [CrossRef] [PubMed]
- Zheng, G; Yi, Q; Tong, Y. Effect of Different Antithrombotic Regimens to Prevent Postoperative Restenosis in Patients With Lower Extremity Arteriosclerosis[J]. American Journal of Therapeutics 2021, 28(4), e517–e520. [Google Scholar]
- ZN, C; K, W; GX, Z. A nomogram to predict major adverse cardiovascular events of patients with acute chest pain, Non-ST-segment deviation, and normal troponin concentrations.[J]. European review for medical and pharmacological sciences 2020, 24(19), 10096–10106. [Google Scholar]
- Sorensen, M C; Abramowitz, D S; Malik, K R. Inter-Society Consensus for the Management of Peripheral Arterial Disease Correlates Better Than the Society for Vascular Surgery Lower Extremity Threatened Limb Classification Based on Wound, Ischemia, and foot Infection (WIfI) in Predicting Major Amputation[J]. Journal of Vascular Surgery 2016, 64(3), 837–838. [Google Scholar]
- Manabe, D; Arizumi, T; Aoyagi, H. Risk factors for post-endoscopic retrograde cholangiopancreatography complications in very elderly patients aged 90 years or older-No additional risk.[J]. Geriatrics & gerontology international 2024, 25(1), 54–60. [Google Scholar]
- G, S H; B, D S; MuhammadSaad, H. Trends In Mortality And Post-Operative Complications Among Octogenarian Patients Undergoing Carotid Endarterectomy.[J]. Journal of vascular surgery 2023, 78(1), 132–140.e2. [Google Scholar] [CrossRef] [PubMed]
| Variables | Total population(n=512) | Surgical treatment group(n=479) | Conservative treatment group(n=33) | t/χ² value | P value |
|---|---|---|---|---|---|
| Age (years, x̄±s) | 69.4±8.2 | 69.1±8.1 | 73.5±7.9 | 3.254 | 0.001 |
| Male [n (%)] | 474(92.6) | 443(92.5) | 31(93.9) | 0.102 | 0.749 |
| BMI(kg/m²,x̄±s) | 24.1±3.2 | 24.2±3.1 | 23.8±3.5 | 0.587 | 0.557 |
| Smoking history [n (%)] | 292(57.0) | 275(57.4) | 17(51.5) | 0.481 | 0.488 |
| Comorbidities [n (%)] |
— | — | — | — | — |
| Hypertension | 293(57.2) | 272(56.8) | 21(63.6) | 0.526 | 0.468 |
| Diabetes mellitus | 238(46.5) | 221(46.1) | 17(51.5) | 0.351 | 0.553 |
| Coronary heart disease | 148(28.9) | 135(28.2) | 13(39.4) | 1.762 | 0.185 |
| Preoperative ABI (x̄±s) | 0.38±0.12 | 0.38±0.12 | 0.45±0.10 | 2.987 | 0.003 |
| Variables | Non-complication group (n=366) | Complication group (n=113) | t/χ² value | P value |
|---|---|---|---|---|
| Age (years, x̄±s) | 67.3±8.2 | 70.6±7.9 | 3.450 | 0.001 |
| Male [n (%)] | 338(92.3) | 105(92.9) | 0.040 | 0.840 |
| BMI (kg/m², x̄±s) | 24.2±3.3 | 24.5±3.5 | 0.720 | 0.471 |
| Smoking history [n (%)] | 220(60.1) | 82(72.6) | 6.180 | 0.013 |
| Hypertension history [n (%)] |
203(55.5) | 62(54.9) | 0.010 | 0.920 |
| Diabetes mellitus history [n (%)] | 147(40.2) | 58(51.3) | 4.820 | 0.028 |
| Coronary heart disease history [n (%)] | 128(35.0) | 32(28.3) | 1.880 | 0.170 |
| Preoperative ABI (x̄±s) | 0.43±0.10 | 0.31±0.12 | 9.910 | <0.001 |
| Surgical method [n (%)] | — | — | 9.530 | 0.002 |
| Endovascular intervention | 338(92.4) | 92(81.4) | — | — |
| Open/hybrid surgery | 28(7.7) | 21(18.6) | — | — |
| Variables | β Value |
Standard Error | Wald χ² Value | P Value |
OR Value | 95% Confidence Interval |
|---|---|---|---|---|---|---|
| Age | 0.080 | 0.027 | 8.73 | 0.003 | 1.08 | 1.03-1.14 |
| Smoking history (Yes) | 0.632 | 0.312 | 4.10 | 0.043 | 1.88 | 1.02-3.36 |
| Diabetes mellitus history (Yes) | 0.875 | 0.315 | 7.74 | 0.005 | 2.40 | 1.36-3.47 |
| Preoperative ABI < 0.4 (Yes) | 1.112 | 0.399 | 7.78 | 0.005 | 3.04 | 1.39-6.64 |
| Surgical method (open/hybrid) | 0.049 | 0.310 | 0.02 | 0.886 | 1.05 | 0.55-2.00 |
| Risk cutoff value | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Accuracy (%) | Youden index |
|---|---|---|---|---|---|---|
| >0.10 | 91.15 | 39.07 | 31.60 | 93.46 | 51.36 | 0.302 |
| >0.20 | 77.88 | 69.13 | 43.78 | 91.01 | 71.19 | 0.470 |
| >0.29 | 69.91 | 82.24 | 54.86 | 89.85 | 79.33 | 0.522 |
| >0.40 | 50.44 | 88.52 | 57.58 | 85.26 | 79.54 | 0.390 |
| >0.50 | 33.63 | 93.17 | 60.32 | 81.97 | 79.12 | 0.268 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




