Submitted:
17 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
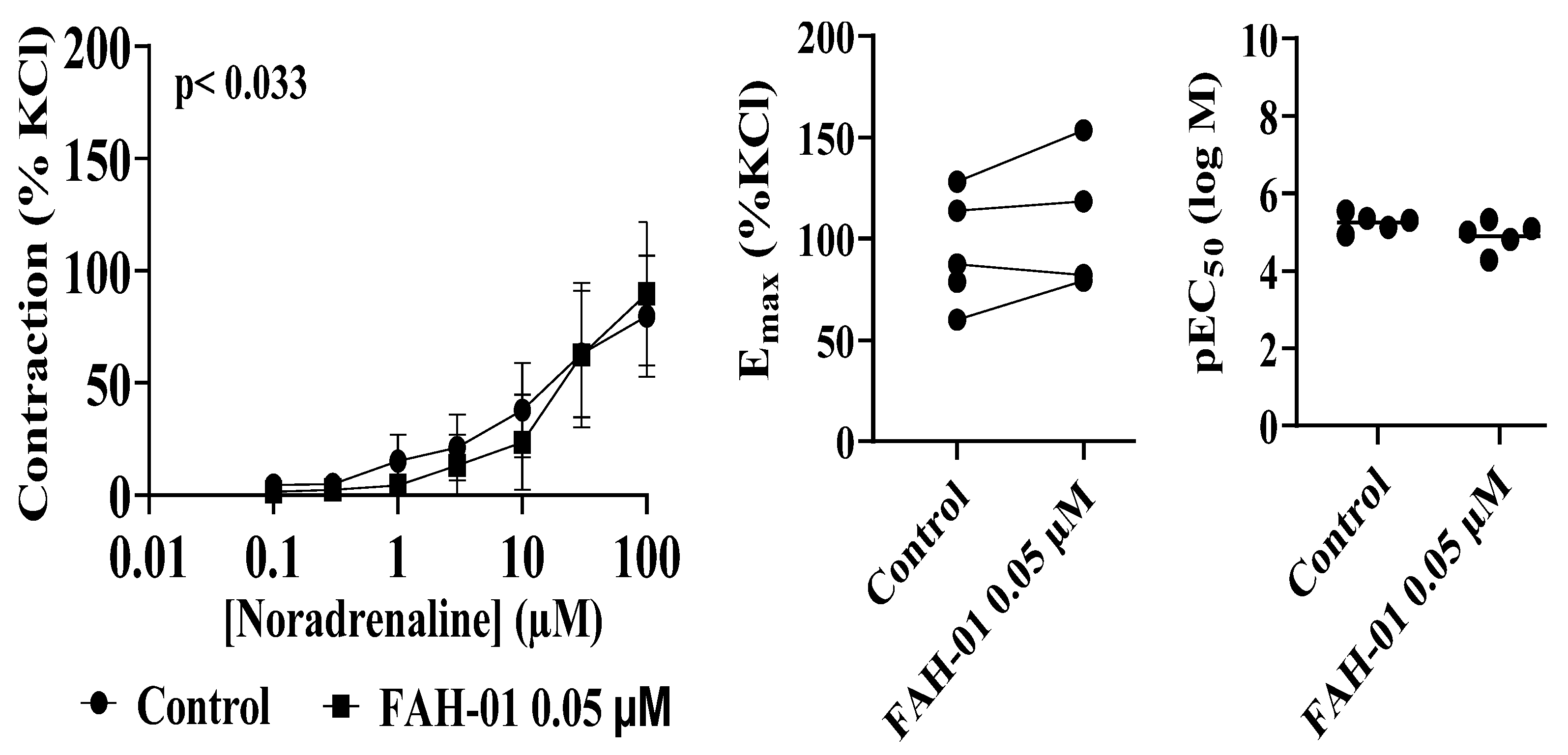
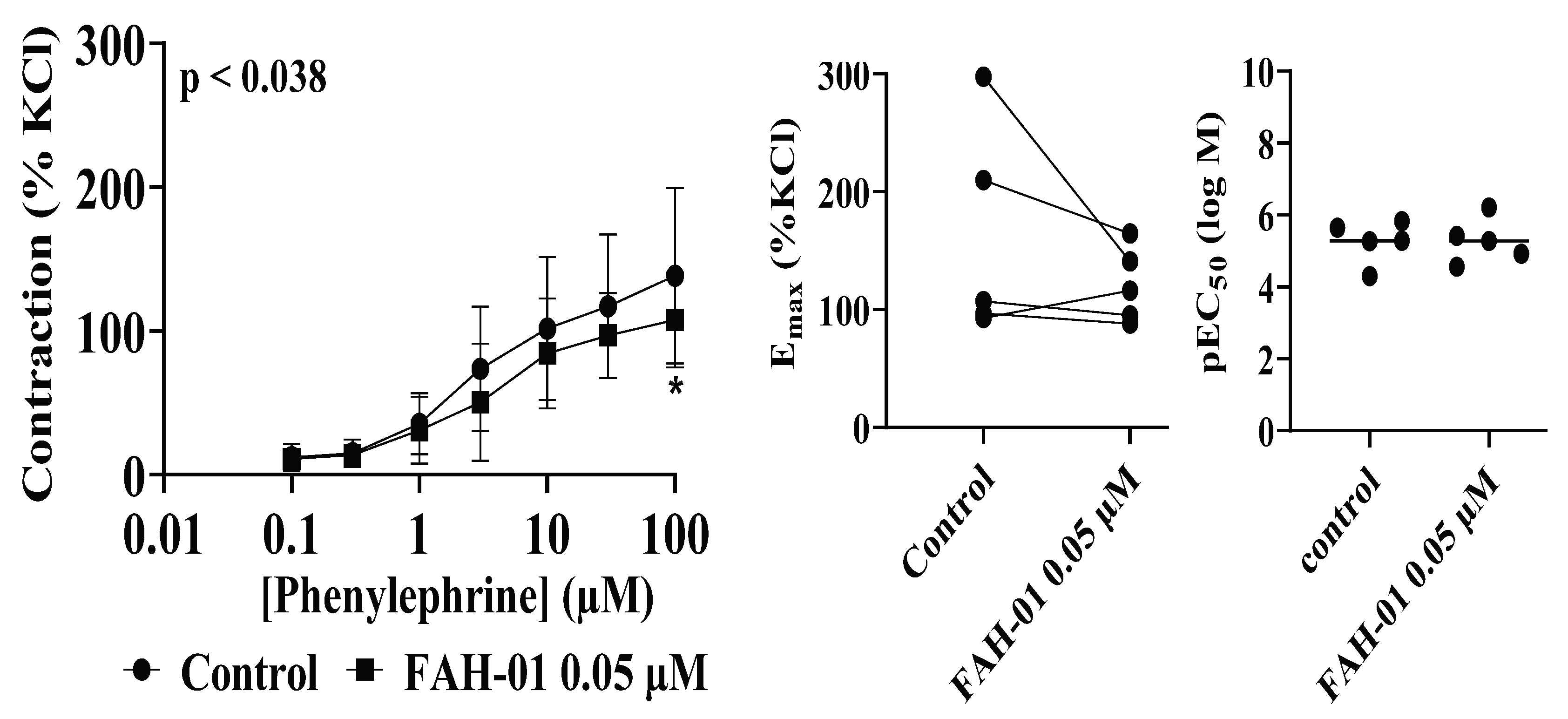
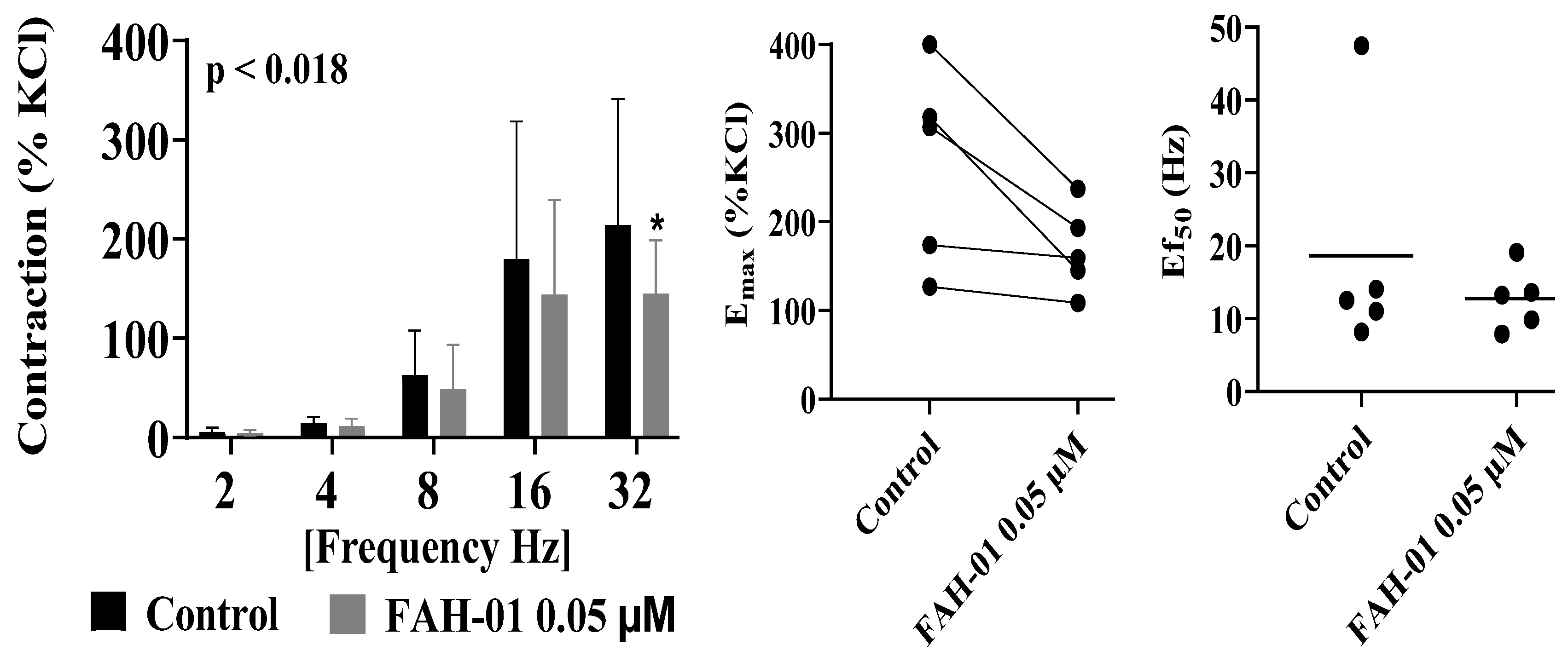
The roots of Acridocarpus smeathmannii were identified as a natural source of the benzyl-terpene 2-(5-isopropyl-4-methoxy-2-methylbenzyl)phenol (FAH-01, chamanen), which was isolated and structurally characterized by chromatographic and spectroscopic methods, including two-dimensional NMR analysis. Functionally, FAH-01 exerted pronounced inhibitory effects on human prostate smooth muscle contractility. In organ bath experiments, FAH-01 reduced noradrenaline-induced contractions by up to 72% and phenylephrine-induced contractions by up to 63%, without affecting agonist potency (pEC50). During electrical field stimulation (2 - 32 Hz), FAH-01 significantly suppressed neurogenic contractile responses, indicating interference with adrenergic and nerve-mediated signaling pathways. Beyond smooth muscle modulation, FAH-01 showed antioxidant activity in the DPPH radical-scavenging assay and produced toxicity in the Artemia salina cysts. Collectively, these findings identify FAH-01 as a bioactive natural product with potent inhibitory effects on adrenergic and neurogenic contraction in human prostate smooth muscle, supporting its therapeutic potential in conditions associated with increased smooth muscle tone. Further preclinical studies to elucidate its mechanisms of action, safety profile, and in vivo efficacy are essential.

Keywords:
1. Introduction
2. Results
2.1. Structural Analysis of 2-(5-Isopropyl-4-methoxy-2-methylbenzyl)phenol (FAH-01, chamanen)
2.2. Anti-Contractility Effects of FAH-01 on Noradrenaline- and Phenylephrine-Induced Contractions of Human Prostate Tissues
2.3. Anti-Contractility Effects of FAH-01 on Electrical Field Stimulation-Induced Contraction of Human Prostate Tissues
2.4. Acute Toxicity in Artemia salina
2.5. In Vitro Antioxidant Assay
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Plant Collection and Authentication
4.3. Extraction and Liquid-Liquid Fractionations
4.4. Chromatography Studies
4.5. Gas Chromatography-Mass Spectrometry (GC-MS) and High Resolution Mass Spectrometry Analysis (HR-MS)
4.6. Nuclear Magnetic Resonance Analysis
4.7. Reverse-Phase High-Performance Column Chromatography (RP-HPLC)
4.8. Biological Actions of Isolate on Human Prostate Tissue Contraction
4.9. Effects of Isolates on the Prostate Smooth Muscle Contractile Activity
4.10. Electrical Field Stimulation
4.11. In Vitro Antioxidant Assay
4.12. Acute Toxicity Potential in Artemia salina
4.13. Data and Statistical Analyses
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DEPT | Distortionless Enhancement by Polarization Transfer |
| GC-MS | Gas-Chromatography Mass Spectrometry |
| HMBC | Heteronuclear Multiple Bond Correlation |
| HSQC | Heteronuclear single quantum coherence |
| HRMS | High-resolution mass spectrometry |
| COSY | Homonuclear correlation spectroscopy |
| pEC50 | Negative logarithms of the molar concentration for agonists |
| NA | Noradrenaline |
| NMR | Nuclear magnetic resonance |
| NOESY | Nuclear Overhauser Effect Spectroscopy |
| PHE | Phenylephrine |
| rPx | Radical prostatectomy |
| Rf | Retention factor |
| TLC | Thin layer chromatography |
References
- Narayanankutty, A.; Famurewa, A. C.; Oprea, E. Natural bioactive compounds and human health. Molecules 2024, 29, 3372. [Google Scholar] [CrossRef]
- Singh, K.; Gupta, J. K.; Chanchal, D. K.; Shinde, M. G.; Kumar, S.; Jain, D.; Almarhoon, Z.M.; Alshahrani, A.M.; Calina, D.; Sharifi-Rad, J.; Tripathi, A. Natural products as drug leads: Exploring their potential in drug discovery and development. Naunyn-Schmiedeberg’s Archives of Pharmacology 2025, 398, 4673–4687. [Google Scholar] [CrossRef]
- Catarino, L.; Havik, P. J.; Romeiras, M. M. Medicinal plants of Guinea-Bissau: Therapeutic applications, ethnic diversity and knowledge transfer. Journal of Ethnopharmacology 2016, 183, 71–94. [Google Scholar] [CrossRef]
- Kale, O. E.; Hu, S.; Huber, C.; Schierholz, F.; Ciotkowska, A.; Tamalunas, A.; Stief, C.G.; Eisenreich, W.; Hennenberg, M. Acridocarpus smeathmannii root extracts inhibit human prostate and bladder smooth muscle contraction, porcine arterial vasoconstriction, and cytotoxicity of prostate stromal cells. Frontiers in Pharmacology 2025, 16, 1621346. [Google Scholar] [CrossRef] [PubMed]
- Kale, O. E.; Rauanov, I.; Huber, C.; Tamalunas, A.; Stief, C. G.; Eisenreich, W.; Hennenberg, M. Benzyl Benzoate Isolation from Acridocarpus smeathmannii (DC.) Guill. & Perr Roots and Its Bioactivity on Human Prostate Smooth Muscle Contractions. Pharmaceuticals 2025, 18, 687. [Google Scholar] [CrossRef]
- Lasswell, WL, Jr.; Hufford, Charles D. “Aromatic constituents from Uvaria chamae.”. Phytochemistry 1977, 16, 1439–1441. [Google Scholar] [CrossRef]
- Anam, E. M.; Ekpa, O. D.; Gariboldi, P. V. 3-Isoproyl-9α-methyl-1, 2, 4a, 9a tetrahydroxanthene, benzyl benzoates and chamanen from. Xylopia africana 1994, 179–181. [Google Scholar]
- Christianson, D. W. Structural and chemical biology of terpenoid cyclases. Chemical Reviews 2017, 117, 11570–11648. [Google Scholar] [CrossRef]
- Zhang, H.; Guo, J.; Hu, J.; Zhou, M. Terpenoid-based supramolecular materials: fabrications, performances, applications. Supramolecular Chemistry 2023, 34, 105–131. [Google Scholar] [CrossRef]
- Cardoso-Teixeira, A. C.; Ferreira-da-Silva, F. W.; Peixoto-Neves, D.; Oliveira-Abreu, K.; Pereira-Gonçalves, Á.; Coelho-de-Souza, A. N.; Leal-Cardoso, J. H. Hydroxyl group and vasorelaxant effects of perillyl alcohol, carveol, limonene on aorta smooth muscle of rats. Molecules 2018, 23, 1430. [Google Scholar] [CrossRef]
- Kanhed, A. M.; Patel, D. V.; Patel, N. R.; Sinha, A.; Thakor, P. S.; Patel, K. B.; Prajapati, N.K.; Patel, K.V.; Yadav, M. R. Indoloquinoxaline derivatives as promising multi-functional anti-Alzheimer agents. Journal of Biomolecular Structure and Dynamics 2022, 40, 2498–2515. [Google Scholar] [CrossRef]
- Bonifazi, A.; Newman, A. H.; Keck, T. M.; Gervasoni, S.; Vistoli, G.; Del Bello, F.; Giorgioni, G.; Pavletić, P.; Quaglia, W.; Piergentili, A. Scaffold hybridization strategy leads to the discovery of dopamine D3 receptor-selective or multitarget bitopic ligands potentially useful for central nervous system disorders. ACS Chemical NNeuroscience 2021, 12, 3638–3649. [Google Scholar] [CrossRef]
- Câmara, J. S.; Perestrelo, R.; Ferreira, R.; Berenguer, C. V.; Pereira, J. A.; Castilho, P. C. Plant-derived terpenoids: A plethora of bioactive compounds with several health functions and industrial applications-A comprehensive overview. Molecules 2024, 29, 3861. [Google Scholar] [CrossRef]
- Kamal El-sagheir, A. M.; Abdelmesseh Nekhala, I.; Abd El-Gaber, M. K.; Aboraia, A. S.; Persson, J.; Schäfer, A. B.; Omar, F. A. N4-substituted piperazinyl norfloxacin derivatives with broad-spectrum activity and multiple mechanisms on gyrase, topoisomerase IV, and bacterial cell wall synthesis. ACS bio & med chem Au 2023, 3, 494–506. [Google Scholar]
- Kale, O. E.; Onyeka, O. C.; Oluwatobunmi, A. A.; Victoria, F. O.; Opeyemi, A. A.; Tamalunas, A.; Hennenberg, M.; Akindele, A.J.; Awodele, O. Ameliorative potential of Acridocarpus smeathmannii extracts on testosterone propionate and sleep deprivation-induced benign prostate hyperplasia rats. Clinical Traditional Medicine and Pharmacology 2025, 200231. [CrossRef]
- Hu, S.; Xu, Y.; Brandstetter, M.; Tamalunas, A.; Kale, O. E.; Keller, P.; Stadelmeier, L.F.; Weinhold, P.; Stief, C.G.; Hennenberg, M. Target and off-target effects of vibegron on smooth muscle contraction of human detrusor and prostate tissues. Naunyn-Schmiedeberg’s Archives of Pharmacology 2025, 1–19. [Google Scholar] [CrossRef]
- Baliyan, S.; Mukherjee, R.; Priyadarshini, A.; Vibhuti, A.; Gupta, A.; Pandey, R. P.; Chang, C. M. Determination of antioxidants by DPPH radical scavenging activity and quantitative phytochemical analysis of Ficus religiosa. Molecules 2022, 27, 1326. [Google Scholar] [CrossRef]
- Okumu, M. O.; Mbaria, J. M.; Gikunju, J. K.; Mbuthia, P. G.; Madadi, V. O.; Ochola, F. O.; Jepkorir, M. S. Artemia salina as an animal model for the preliminary evaluation of snake venom-induced toxicity. Toxicon X 2021, 12, 100082. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Jia, H.; Zhu, L.; Zhang, H.; Wang, Y. Toxicity of α-Fe2O3 nanoparticles to Artemia salina cysts and three stages of larvae. Science of the Total Environment 2017, 598, 847–855. [Google Scholar] [CrossRef] [PubMed]
- Michel, M. C.; Murphy, T. J.; Motulsky, H. J. New author guidelines for displaying data and reporting data analysis and statistical methods in experimental biology. Mol. Pharmacol. 2020, 97, 49–60. [Google Scholar] [CrossRef]
- Kumar, M.; Ahmad, A.; Aguiar, A. C. C.; Maluf, S. E. C.; Shamim, A.; Ferrer, M.; Dias, L. C. Indole-2-carboxamides Optimization for Antiplasmodial Activity. ACS Bio & Med Chem Au 2025, 5, 821–839. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Liu, G.; Kale, O.; Zhu, W.; Xu, Y.; Keller, P.; Weinhold, P.; Tamalunas, A.; Stief, C.G; Hennenberg, M. Antagonism of prostate α1A-adrenoceptors by verapamil in human prostate smooth muscle contraction. The Journal of Pharmacology and Experimental Therapeutics 2025, 103603. [Google Scholar] [CrossRef] [PubMed]
- Hennenberg, M.; Michel, M. C. Adrenoceptors in the lower urinary tract. Adrenoceptors 2023, 333–367. [Google Scholar]
- Lemly, A. D. Teratogenic effects and monetary cost of selenium poisoning of fish in Lake Sutton, North Carolina. Ecotoxicology and Environmental Safety 2014, 104, 160–167. [Google Scholar] [CrossRef]
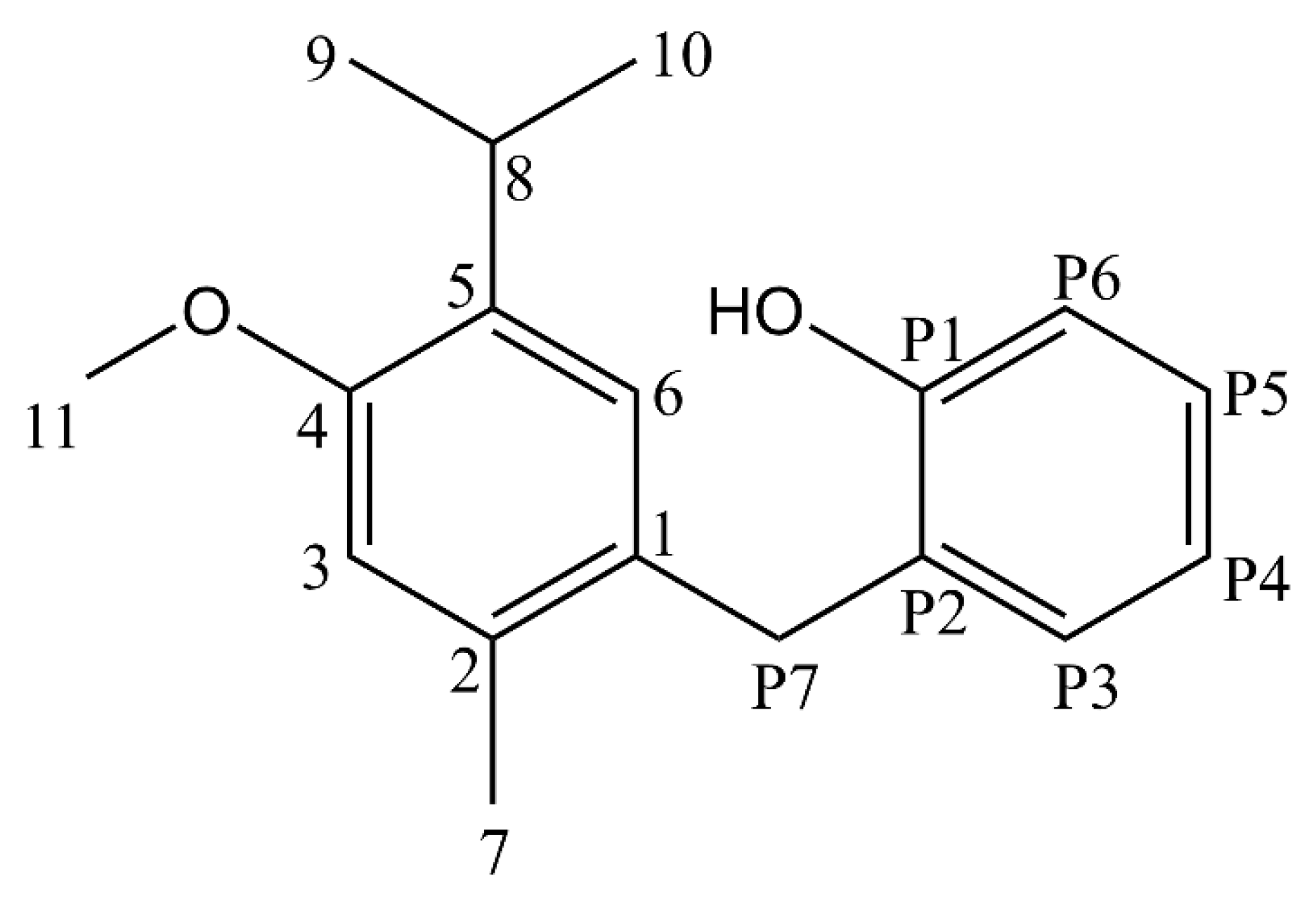
| Atom Position | 13C Chemical Shift in ppm | Predicted 13C chemical shift in ppm | CH3, CH2, CH, or C | 1H Chemical Shift in ppm | HH Coupling Constant in Hz | 1H Signal Integral | HH COSY Correlation to H# | HH NOESY Correlation to H# | HMBC Correlation from indexed C to H# |
|---|---|---|---|---|---|---|---|---|---|
| P1 | 153.85 | 155.3 | C | P5, P3, P6 | |||||
| P2 | 126.71 | 128.2 | C | P4, P6, OH, P7 | |||||
| P3 | 130.25 | 130.7 | CH | 6.93 (d) | 7.7 | 1 | P7 (w) | 6 | P5, P7 |
| P4 | 120.72 | 121.8 | CH | 6.84 (t) | 7.7 | 1 | P5 | P6 | |
| P5 | 127.44 | 127.6 | CH | 7.12 (t) | 7.7 | 1 | P4 | P3 | |
| P6 | 115.49 | 116.4 | CH | 6.79 (d) | 7.7 | 1 | P4, OH | ||
| P7 | 33.59 | 32.9 | CH2 | 3.91 (s) | 2 | 6 (w), P3 (w) | 6, OH, 7 | 6 | |
| 1 | 135.22 | 131.0 | C | 6, P7, 7 | |||||
| 2 | 128.81 | 134.2 | C | 3, P7, 7 | |||||
| 3 | 112.98 | 113.3 | CH | 6.68 (s) | 1 | 11, 7 | 7 | ||
| 4 | 155.48 | 155.4 | C | 3, 6, 11, 8 | |||||
| 5 | 134.70 | 135.2 | C | 3, 8, 9, 10 | |||||
| 6 | 127.44 | 128.1 | CH | 6.96 (s) | 1 | P7 (w), 7 (w) | P7, P3, 9, 10 | P7, 8, 7 | |
| 7 | 19.56 | 19.4 | CH3 | 2.22 (s) | 3 | 6 (w) | 3, P7 | 3, 6 | |
| 8 | 26.56 | 27.6 | CH | 3.24 (sep) | 6.9 | 1 | 9, 10 | 9, 10 | 6, 9, 10 |
| 9 | 22.70 | 23.6 | CH3 | 1.15 (d) | 6.5 | 3 | 8 | 6, 8 | 8 |
| 10 | 22.70 | 23.6 | CH3 | 1.15 (d) | 6.5 | 3 | 8 | 6, 8 | 8 |
| 11 | 55.43 | 56.1 | CH3 | 3.81 (s) | 3 | 3 | |||
| OH | 4.81 | 1 | P7 | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




