4. Discussion
This study explored the association between systemic inflammatory markers and OCT-based structural severity in patients with acute NA-CRAO. To our knowledge, it is among the first studies to relate these indices to the extent of retinal tissue injury captured by OCT. We found that a higher systemic inflammatory burden, reflected by elevated NLR and SII, was associated with more extensive ischemic damage. These findings suggest that certain inflammatory markers may reflect underlying pathophysiological processes contributing to retinal tissue injury, and thus hold potential value for early risk stratification. By integrating systemic inflammation markers with structural OCT findings, our results support the concept that systemic inflammation is not merely a consequence but potentially a contributor to ischemic burden in CRAO.
Our findings are consistent with a recent meta-analysis demonstrating significantly elevated NLR and PLR levels in patients with retinal artery occlusion compared with controls. The pooled analysis of eight retrospective studies including 1,444 participants reported a moderate to large effect size for NLR and a smaller but significant effect for PLR, despite substantial between-study heterogeneity [
7].
Systemic inflammatory markers derived from the complete blood count have been widely investigated in CRAO, although their clinical utility varies substantially by marker and context. Among them, NLR has been most consistently reported to be significantly higher in CRAO patients versus controls [
17,
18,
19,
20]. As a composite marker reflecting neutrophilia and relative lymphopenia, NLR is considered a surrogate of innate immune activation and systemic stress, processes that are mechanistically relevant to acute arterial occlusion.
In our cohort, higher NLR values were observed with increasing ischemic severity, supporting a graded link between systemic inflammation and the extent of retinal damage assessed on structural imaging. This pattern suggests that NLR captures more than disease presence alone, instead reflecting an inflammatory response that scales with ischemic burden. This inflammatory milieu is thought to promote endothelial dysfunction, embolic propensity, and microvascular vulnerability, thereby predisposing to more extensive ischemic injury. Taken together, these findings point to a more prominent role of the neutrophil-related inflammatory response in the most severe CRAO presentations.
Central macular thickness represents a direct structural manifestation of retinal ischemic injury following abrupt perfusion failure [
21,
22,
23,
24]. The absence of correlation between NLR and CMT in our cohort, together with their additive effects in multivariable analysis, supports a complementary mechanistic framework in which systemic inflammatory burden and local retinal tissue response operate through distinct but convergent pathways. Notably, NLR emerged as the dominant systemic predictor, with each one-unit increase associated with an 8.398-fold higher odds of belonging to a more severe OCT-defined ischemia category, whereas CMT showed a smaller but stable independent association reflecting local tissue injury. Thus, while NLR may influence vascular vulnerability and microvascular injury, CMT provides tissue-level quantification of ischemic damage, and their combined assessment offers a more comprehensive stratification of disease severity in acute NA-CRAO.
Notably, Elbeyli et al. reported that red cell distribution width (RDW) showed greater discriminatory performance than NLR and PLR and was the only independent predictor of CRAO in multivariable analysis, underscoring that different inflammatory markers may capture distinct aspects of the systemic response to retinal ischemia [
18].
Platelet-to-lymphocyte ratio has been proposed as an index reflecting platelet activation alongside relative lymphopenia, linking inflammatory and prothrombotic pathways. However, evidence regarding PLR in RAO remains heterogeneous. While some studies reported higher PLR in affected patients than in controls, others found no meaningful differences [
17,
19]. Consistent with this variability, PLR was not associated with ischemic severity in our cohort, suggesting it may lack sensitivity in capturing the extent of retinal tissue damage.
Systemic immune-inflammation index has emerged as a versatile and accessible biomarker that reflects the interplay between systemic inflammation and immune status. Its clinical relevance has been demonstrated across multiple domains, including oncology, cardiovascular and cerebrovascular diseases, infectious conditions, and autoimmune disorders, where it correlates with disease severity, mortality risk, and prognosis [
25,
26,
27,
28]. In our cohort, higher SII values were associated with greater ischemic severity, reflecting the possible contribution of systemic inflammatory and thrombotic activity to the extent of retinal ischemic involvement. SII may thus serve as a useful tool for risk stratification and clinical monitoring in acute NA-CRAO.
In contrast, MLR and SIRI showed no significant association with ischemic severity, reinforcing the need to prioritize markers more closely linked to the underlying pathophysiology.
The present study provides novel insight into the clinical interpretation of systemic inflammatory indices in CRAO. While previous studies have primarily focused on the presence and elevation of inflammatory markers in CRAO, our study advances this perspective by demonstrating that these indices are not only elevated, but also proportionally associated with the degree of ischemic retinal injury, as assessed by OCT. This distinction is clinically meaningful, as it reframes inflammatory markers from simple indicators of disease presence to potential tools for quantifying tissue damage and stratifying acute ischemic severity.
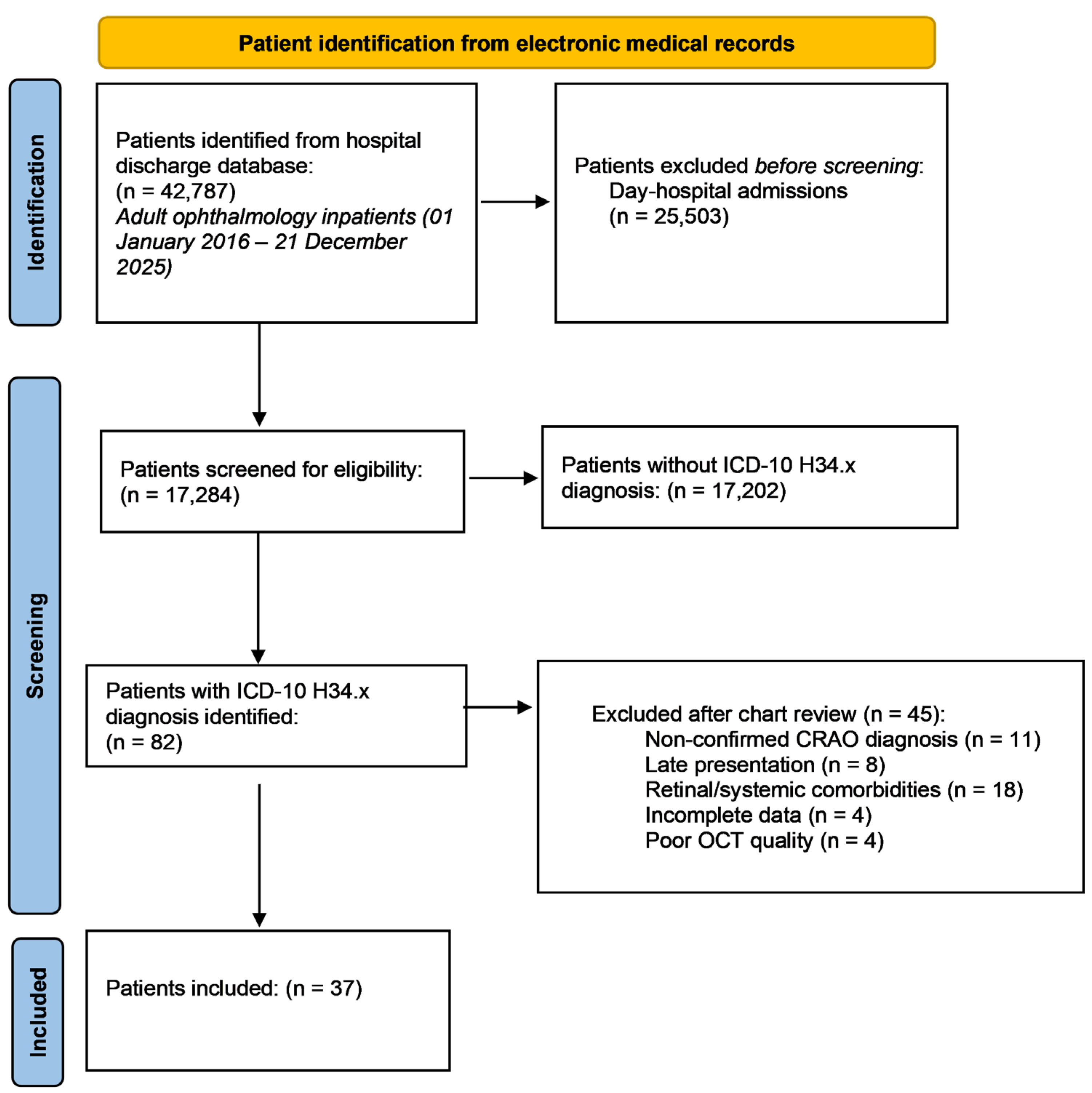
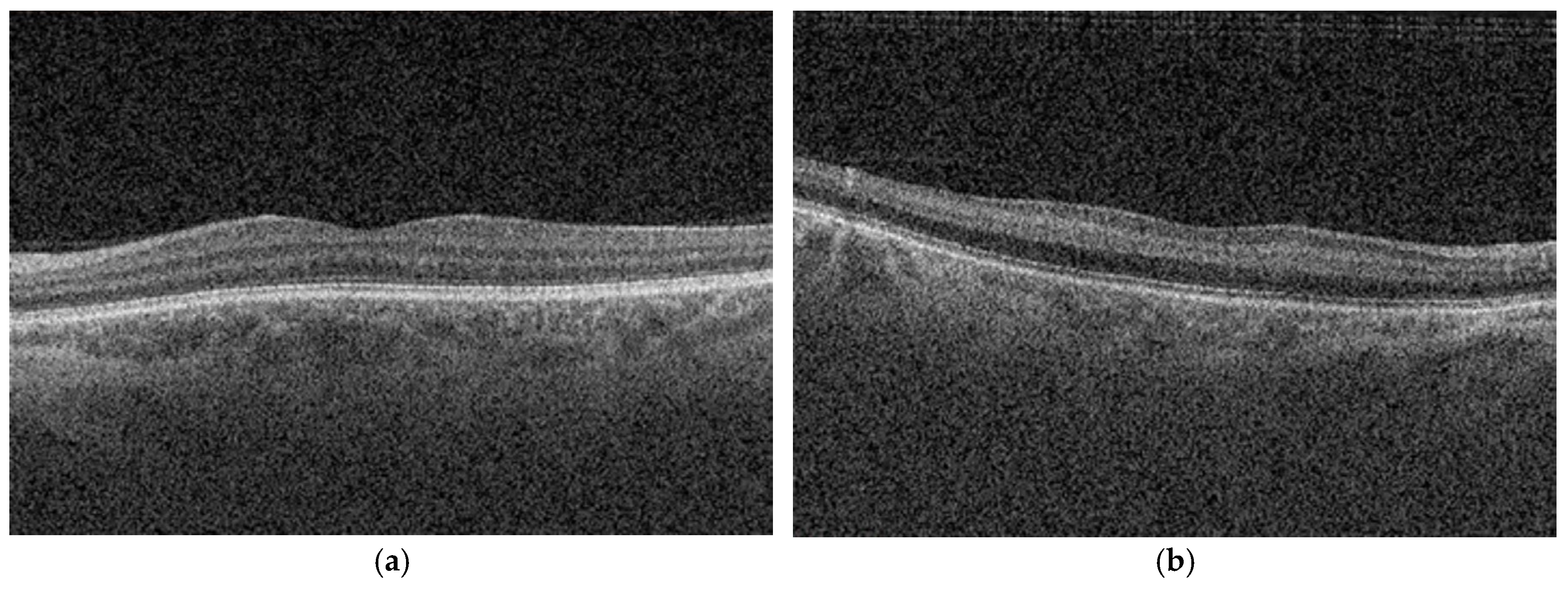
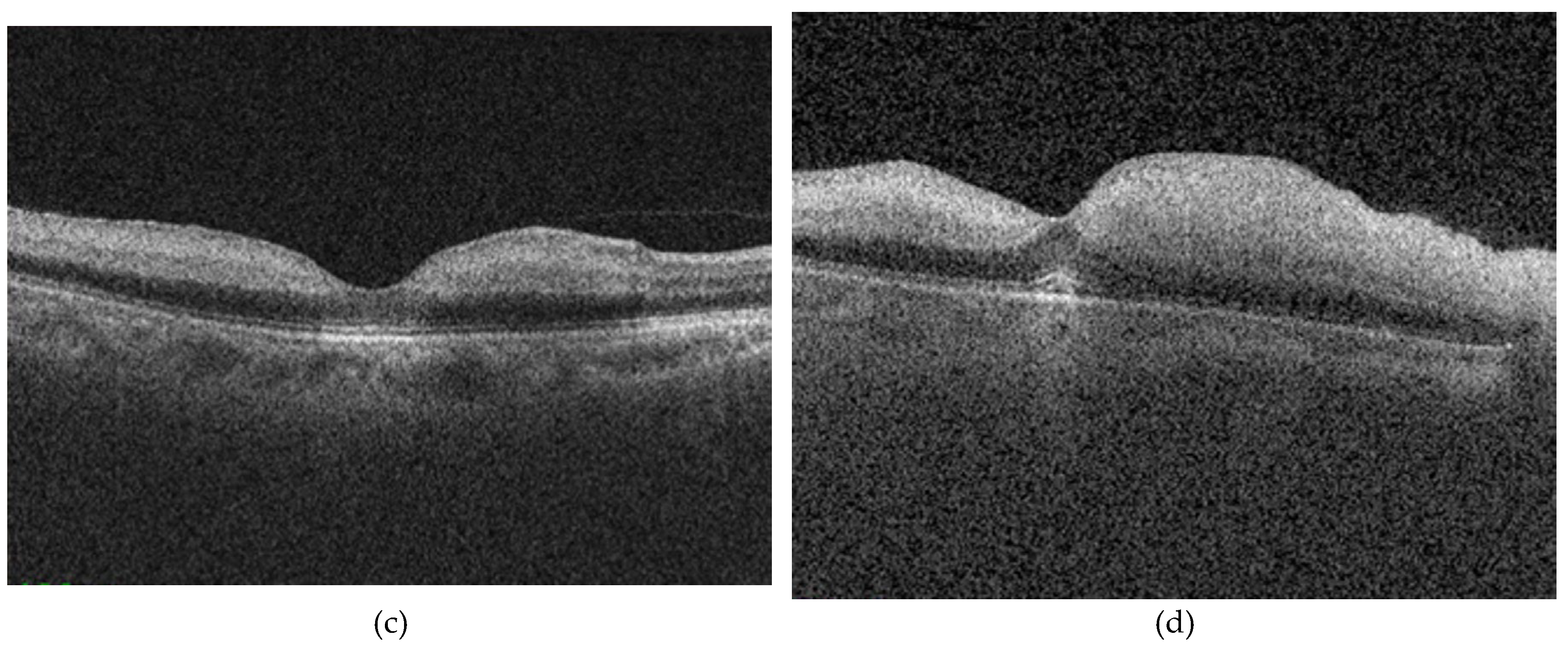
Optical coherence tomography enables high-resolution, layer-specific visualization of ischemic injury in CRAO which directly reflects the extent of retinal damage [
24,
29,
30,
31,
32]. Furthermore, recent studies recognize OCT as the primary modality for assessing ischemic injury severity, due to its capacity to detect early retinal edema, hyperreflectivity, and disorganization of inner retinal layers [
33,
34,
35].
Mangla et al. [
12] introduced a purely OCT based morphological classification system for CRAO, addressing the heterogeneity and subjectivity of previous models, which relied on mixed clinical, angiographic, and imaging criteria and commonly categorized CRAO severity as incomplete, subtotal, or total. Their grading reflects a continuous progression of structural and pathological changes, closely aligned with capillary plexus involvement and the extent of ischemic retinal injury. Furthermore, the described changes follow a consistent temporal evolution, allowing for early detection and providing a practical framework for longitudinal assessment of disease severity and potential treatment response. For our study, we adopted this grading because it uses objective, routinely available features in acute care, providing a reproducible and quantifiable measure of severity.
In a recent retrospective study, Hu et al. [
8] examined peripheral blood inflammatory indices in CRAO and related them to disease severity using a composite grading system that integrated fluorescein fundus angiography, funduscopic cherry-red spot, and OCT characteristics to classify cases as incomplete, subtotal, or total CRAO. NLR, SII, and MHR were higher in CRAO than in controls, but across severity grades only MHR (driven by lower HDL-C) was associated with total CRAO, whereas NLR and SII did not significantly differ between subtypes.
While Hu et al. primarily characterize disease severity at the vascular and clinical level our study focuses on the severity of tissue level ischemic damage, quantified on OCT. By defining severity exclusively through OCT-based morphological criteria we sought to capture the downstream retinal consequences of ischemia, rather than upstream perfusion abnormalities. In this context, systemic inflammatory indices are interpreted in relation to structural retinal damage, providing a complementary perspective that does not depend on perfusion imaging. Thus, the two frameworks capture different dimensions of CRAO severity and are best viewed as complementary.
From a clinical perspective, integrating structural retinal injury on OCT with inflammatory indices adds contextual information that supports severity assessment and may inform subsequent systemic evaluation.This is feasible in routine acute care, since both OCT and complete blood count parameters are typically available without delaying workflows.
Our findings should be interpreted in light of several limitations. The retrospective design and modest sample size may limit statistical power and generalizability. Blood samples were obtained upon presentation but not standardized by time of day or fasting state, which may influence inflammatory indices. OCT grading was focused on structural findings without correlating with long-term visual outcomes. Furthermore, strict exclusion criteria may reduce applicability to broader populations. Future prospective studies are needed to validate these associations and assess their prognostic utility.