Submitted:
23 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
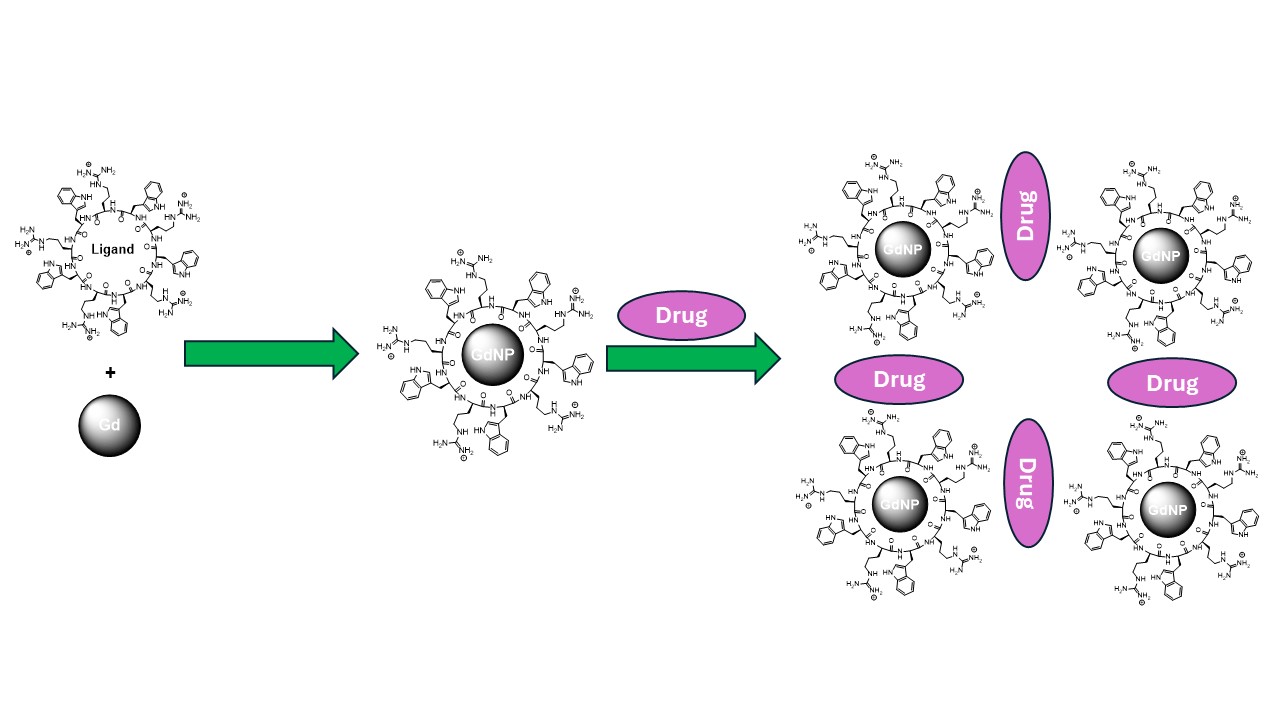
Keywords:
1. Introducion
1.1. Inorganic Nanoparticles in Drug Delivery
1.2. Metal Nanoparticles in Drug Delivery
1.3. Gadolinium (Gd)
1.4. Gadolinium Nanoparticles (GdNPs)
2. Chemistry of GdNPs
3. Absorption, Distribution, and Elimination of GdNPs
4. GdNPs in Drug Delivery
4.1. Used as Photo-Thermal Nanoparticles and Work Through Increasing Energy
4.2. Surface, Size, and Charge Modification of GdNPs for Drug Delivery
5. GdNPs in Cancer Therapy
6. Future Perspectives
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| GdNPS | Gadolinium Nanoparticles |
| REEs | Rare Earth Elements |
| ADME | Absorption, Distribution, Metabolism, and Excretion |
References
- Shirazi, A.N.; Vadlapatla, R.; Koomer, A.; Nguyen, A.; Khoury, V.; Parang, K. Peptide-Based Inorganic Nanoparticles as Efficient Intracellular Delivery Systems. Pharmaceutics 2025, 17, 1123. [Google Scholar] [CrossRef]
- Salavati-Niasari, M.; Davar, F.; Mir, N. Synthesis and Characterization of Metallic Copper Nanoparticles via Thermal Decomposition. Polyhedron 2008, 27, 3514–3518. [Google Scholar] [CrossRef]
- Ealia, S.A.M.; Saravanakumar, M.P. A Review on the Classification, Characterisation, Synthesis of Nanoparticles and Their Application. IOP Conf. Ser. Mater. Sci. Eng. 2017, 263, 032019. [Google Scholar] [CrossRef]
- Kim, H.; Mondal, S.; Jang, B.; Manivasagan, P.; Moorthy, M.S.; Oh, J. Biomimetic Synthesis of Metal–Hydroxyapatite (Au-HAp, Ag-HAp, Au-Ag-HAp): Structural Analysis, Spectroscopic Characterization and Biomedical Application. Ceram. Int. 2018, 44, 20490–20500. [Google Scholar] [CrossRef]
- Iravani, S. Green Synthesis of Metal Nanoparticles Using Plants. Green Chem. 2011, 13, 2638–2650. [Google Scholar] [CrossRef]
- Khan, M.U.; Ullah, H.; Honey, S.; Talib, Z.; Abbas, M.; Umar, A.; Ahmad, T.; Sohail, J.; Sohail, A.; Makgopa, K.; et al. Metal Nanoparticles: Synthesis Approach, Types and Applications—A Mini Review. Nano-Horizons 2023, 2, 1–21. [Google Scholar]
- Chakraborty, I.; Pradeep, T. Atomically Precise Clusters of Noble Metals: Emerging Link between Atoms and Nanoparticles. Chem. Rev. 2017, 117, 8208–8271. [Google Scholar] [CrossRef]
- Zaleska-Medynska, A.; Marchelek, M.; Diak, M.; Grabowska, E. Noble Metal-Based Bimetallic Nanoparticles: The Effect of the Structure on the Optical, Catalytic and Photocatalytic Properties. Adv. Colloid Interface Sci. 2016, 229, 80–107. [Google Scholar] [CrossRef]
- Vasić, K.; Knez, Ž.; Leitgeb, M. Multifunctional Iron Oxide Nanoparticles as Promising Magnetic Biomaterials in Drug Delivery: A Review. J. Funct. Biomater. 2024, 15, 227. [Google Scholar] [CrossRef]
- Karim, W.; Kleibert, A.; Hartfelder, U.; Bansmann, J.; Bäumer, M. Size-Dependent Redox Behavior of Iron Observed by In-Situ Single Nanoparticle Spectro-Microscopy on Well-Defined Model Systems. Sci. Rep. 2016, 6, 18818. [Google Scholar] [CrossRef]
- Tai, C.Y.; Tai, C.-T.; Chang, M.-H.; Liu, H.-S. Synthesis of Magnesium Hydroxide and Oxide Nanoparticles Using a Spinning Disk Reactor. Ind. Eng. Chem. Res. 2007, 46, 5536–5541. [Google Scholar] [CrossRef]
- Vargas-Ortiz, J.R.; Gonzalez, C.; Esquivel, K. Magnetic Iron Nanoparticles: Synthesis, Surface Enhancements, and Biological Challenges. Processes 2022, 10, 2282. [Google Scholar] [CrossRef]
- Le, N.; Zhang, M.; Kim, K. Quantum Dots and Their Interaction with Biological Systems. Int. J. Mol. Sci. 2022, 23, 10763. [Google Scholar] [CrossRef] [PubMed]
- Iravani, S.; Korbekandi, H.; Mirmohammadi, S.V.; Zolfaghari, B. Synthesis of Silver Nanoparticles: Chemical, Physical and Biological Methods. Res. Pharm. Sci. 2014, 9, 385–406. [Google Scholar]
- Gupta, A.K.; Gupta, M. Synthesis and Surface Engineering of Iron Oxide Nanoparticles for Biomedical Applications. Biomaterials 2005, 26, 3995–4021. [Google Scholar] [CrossRef]
- Genc, S.; Taghizadehghalehjoughi, A.; Yeni, Y.; Jafarizad, A.; Hacimuftuoglu, A.; Nikitovic, D.; Docea, A.O.; Mezhuev, Y.; Tsatsakis, A. Fe3O4 Nanoparticles in Combination with 5-FU Exert Antitumor Effects Superior to Those of the Active Drug in a Colon Cancer Cell Model. Pharmaceutics 2023, 15, 245. [Google Scholar] [CrossRef]
- Premanathan, M.; Karthikeyan, K.; Jeyasubramanian, K.; Manivannan, G. Selective Toxicity of ZnO Nanoparticles toward Gram-Positive Bacteria and Cancer Cells by Apoptosis through Lipid Peroxidation. Nanomedicine 2011, 7, 184–192. [Google Scholar] [CrossRef]
- Chen, G.; Yang, F.; Fan, S.; Jin, H.; Liao, K.; Li, X.; Liu, G.B.; Liang, J.; Zhang, J.; Xu, J.F.; et al. Immunomodulatory Roles of Selenium Nanoparticles: Novel Arts for Potential Immunotherapy Strategy Development. Front. Immunol. 2022, 13, 956181. [Google Scholar] [CrossRef]
- Huang, Y.; He, L.; Liu, W.; Fan, C.; Zheng, W.; Wong, Y.S.; Chen, T. Selective Cellular Uptake and Induction of Apoptosis of Cancer-Targeted Selenium Nanoparticles. Biomaterials 2013, 34, 7106–7116. [Google Scholar] [CrossRef]
- Gong, G.; Fu, B.; Ying, C.; Zhu, Z.; He, X.; Li, Y.; Shen, Z.; Xuan, Q.; Huang, Y.; Lin, Y.; et al. Targeted Delivery of Paclitaxel by Functionalized Selenium Nanoparticles for Anticancer Therapy through ROS-Mediated Signaling Pathways. RSC Adv. 2018, 8, 39957–39966. [Google Scholar] [CrossRef]
- Caravan, P.; Ellison, J.J.; McMurry, T.J.; Lauffer, R.B. Gadolinium(III) Chelates as MRI Contrast Agents: Structure, Dynamics, and Applications. Chem. Rev. 1999, 99, 2293–2352. [Google Scholar] [CrossRef] [PubMed]
- Lauffer, R.B. Paramagnetic Metal Complexes as Water Proton Relaxation Agents for NMR Imaging. Chem. Rev. 1987, 87, 901–927. [Google Scholar] [CrossRef]
- Wang, H.; Liu, Y.; Zhang, M.; Wang, J.; Wu, Z.; Li, L. Gadolinium-Based Nanoparticles for Theranostic Applications: A Review. ACS Appl. Nano Mater. 2021, 4, 4673–4691. [Google Scholar]
- Shirazi, A.N.; Sajid, M.I.; Mandal, D.; Stickley, D.; Nagasawa, S.; Long, J.; Lohan, S.; Parang, K.; Tiwari, R.K. Cyclic Peptide-Gadolinium Nanocomplexes as siRNA Delivery Tools. Pharmaceuticals 2021, 14, 1064. [Google Scholar]
- Shirazi, A.N.; Park, S.E.; Rad, S.; Baloyan, L.; Mandal, D.; Sajid, M.I.; Hall, R.; Lohan, S.; Zoghebi, K.; Parang, K.; et al. Cyclic Peptide–Gadolinium Nanoparticles for Enhanced Intracellular Delivery. Pharmaceutics 2020, 12, 792. [Google Scholar] [CrossRef]
- Shirazi, A.N.; Paquin, K.L.; Howlett, N.G.; Mandal, D.; Parang, K. Cyclic Peptide-Capped Gold Nanoparticles for Enhanced siRNA Delivery. Molecules 2014, 19, 13319–13331. [Google Scholar] [CrossRef]
- Nasrolahi Shirazi, A.; Tiwari, R.K.; Oh, D.; Sullivan, B.; McCaffrey, K.; Mandal, D.; Parang, K. Surface Decorated Gold Nanoparticles by Linear and Cyclic Peptides as Molecular Transporters. Mol. Pharm. 2013, 10, 3137–3146. [Google Scholar] [CrossRef]
- Nasrolahi Shirazi, A.; Mandal, D.; Tiwari, R.K.; Guo, L.; Lu, W.; Parang, K. Cyclic Peptide-Capped Gold Nanoparticles as Drug Delivery Systems. Mol. Pharm. 2013, 10, 500–508. [Google Scholar] [CrossRef]
- Shirazi, A.N.; Vadlapatla, R.; Koomer, A.; Yep, K.; Parang, K. Selenium Nanoparticles as Versatile Delivery Tools. Pharmaceutics 2025, 17, 1556. [Google Scholar] [CrossRef]
- Greenwood, N.N.; Earnshaw, A. Chemistry of the Elements, 2nd ed.; Butterworth-Heinemann: Oxford, UK, 1997; pp. 1227–1249. [Google Scholar]
- Pyykkö, P. Magically Magnetic Gadolinium. Nat. Chem. 2015, 7, 680–681. [Google Scholar] [CrossRef]
- Abragam, A.; Bleaney, B. Electron Paramagnetic Resonance of Transition Ions; Oxford University Press: Oxford, UK, 2012. [Google Scholar]
- Laing, M. Gadolinium: Central Metal of the Lanthanoids. J. Chem. Educ. 2009, 86, 188–189. [Google Scholar] [CrossRef]
- Arajs, S.; Colvin, R.V. Magnetic Susceptibility of Gadolinium, Terbium, and Dysprosium Metals. J. Appl. Phys. 1961, 32(3), S336–S337. [Google Scholar] [CrossRef]
- Robertson, A.G.; Rendina, L.M. Gadolinium Theranostics for the Diagnosis and Treatment of Cancer. Chem. Soc. Rev. 2021, 50, 4231–4244. [Google Scholar] [CrossRef] [PubMed]
- Ramalho, J.; Semelka, R.C.; Ramalho, M.; Nunes, R.H.; AlObaidy, M.; Castillo, M. Gadolinium-Based Contrast Agent Accumulation and Toxicity: An Update. AJNR Am. J. Neuroradiol. 2016, 37, 1192–1198. [Google Scholar] [CrossRef]
- Liu, Y.; Tegafaw, T.; Baek, A.; Ho, S.L.; Zhao, D.; Chen, X.; Mulugeta, E.; Lee, H.; Nam, H.; Kim, J.; Chang, Y.; Lee, G.H. Tumor-Targeting T₁ MRI Contrast Agents with Antioxidant Effects: Gadolinium Oxide Nanoparticles Grafted with Polydopamine- and Polyserotonin-Conjugated Polyacrylic Acids. ACS Omega 2025, 10, 58885–58899. [Google Scholar] [CrossRef]
- Bashir, A.; Gray, M.L.; Burstein, D. Gd-DTPA2− as a measure of cartilage degradation. Magn. Reson. Med. 1996, 36(5), 665–673. [Google Scholar] [CrossRef]
- Nieminen, M.T.; Rieppo, J.; Silvennoinen, J.; Töyräs, J.; Hakumäki, J.M.; Hyttinen, M.M.; Helminen, H.J.; Jurvelin, J.S. Spatial assessment of articular cartilage proteoglycans with Gd-DTPA-enhanced T1 imaging. Magn. Reson. Med. 2002, 48(4), 640–648. [Google Scholar] [CrossRef]
- Miyata, S.; Homma, K.; Numano, T.; Furukawa, K.; Tateishi, T.; Ushida, T. Assessment of fixed charge density in regenerated cartilage by Gd-DTPA-enhanced MRI. Magn. Reson. Med. Sci. 2006, 5, 73–78. [Google Scholar] [CrossRef]
- Chen, C.T.; Fishbein, K.W.; Torzilli, P.A.; Hilger, A.; Spencer, R.G.; Horton, W.E., Jr. Matrix fixed-charge density as determined by magnetic resonance microscopy of bioreactor-derived hyaline cartilage correlates with biochemical and biomechanical properties. Arthritis Rheum. 2003, 48(4), 1047–1056. [Google Scholar] [CrossRef]
- Ogan, M.D.; Schmiedl, U.; Moseley, M.E.; Grodd, W.; Paajanen, H.; Brasch, R.C. Albumin labeled with Gd-DTPA. An intravascular contrast-enhancing agent for magnetic resonance blood pool imaging: Preparation and characterization. Invest. Radiol. 1987, 22(8), 665–671. [Google Scholar] [CrossRef]
- Li, L.; Jiang, W.; Luo, K.; Song, H.; Lan, F.; Wu, Y.; Gu, Z. Superparamagnetic Iron Oxide Nanoparticles as MRI Contrast Agents for Non-Invasive Stem Cell Labeling and Tracking. Theranostics 2013, 3(8), 595–615. [Google Scholar] [CrossRef] [PubMed]
- Parfenov, V.A.; Koudan, E.V.; Bulanova, E.A.; Karalkin, P.A.; DAS Pereira, F.; Norkin, N.E.; Knyazeva, A.D.; Gryadunova, A.A.; Petrov, O.F.; Vasiliev, M.M.; Myasnikov, M.I.; Chernikov, V.P.; Kasyanov, V.A.; Marchenkov, A.Y.; Brakke, K.; Khesuani, Y.D.; Demirci, U.; Mironov, V.A. Scaffold-Free, Label-Free and Nozzle-Free Biofabrication Technology Using Magnetic Levitational Assembly. Biofabrication 2018, 10, 034104. [Google Scholar] [CrossRef] [PubMed]
- Parfenov, V.A.; Mironov, V.A.; Koudan, E.V.; Nezhurina, E.K.; Karalkin, P.A.; Pereira, F.D.A.S.; Petrov, S.V.; Krokhmal, A.A.; Aydemir, T.; Vakhrushev, I.V.; Zobkov, Y.V.; Smirnov, I.V.; Fedotov, A.Y.; Demirci, U.; Khesuani, Y.D.; Komlev, V.S. Fabrication of Calcium Phosphate 3D Scaffolds for Bone Repair Using Magnetic Levitational Assembly. Sci. Rep. 2020, 10, 4013. [Google Scholar] [CrossRef] [PubMed]
- Türker, E.; Demirçak, N.; Arslan-Yildiz, A. Scaffold-free three-dimensional cell culturing using magnetic levitation. Biomater. Sci. 2018, 6, 1745–1753. [Google Scholar] [CrossRef]
- Pan, X.; Hu, M.; Wu, L.; Wei, E.; Zhu, Q.; Lv, L.; Xv, X.; Dong, X.; Liu, H.; Liu, Y. Biomedical Applications of Gadolinium-Containing Biomaterials: Not Only MRI Contrast Agent. Adv. Sci. 2025, 12, e2501722. [Google Scholar] [CrossRef]
- Krokhicheva, P.A.; Goldberg, M.A.; Fomin, A.S.; Khayrutdinova, D.R.; Antonova, O.S.; Sadovnikova, M.A.; Mikheev, I.V.; Leonov, A.V.; Merzlyak, E.M.; Kovalishina, D.A.; Akhmedova, S.A.; Sergeeva, N.S.; Gafurov, M.R.; Barinov, S.M.; Komlev, V.S. Gadolinium-Doped Injectable Magnesium-Calcium Phosphate Bone Cements for Noninvasive Visualization. J. Magnes. Alloys 2024, 12, 3698–3716. [Google Scholar] [CrossRef]
- Chaturvedi, K.; Singhwane, A.; Jaiswal, A.; Mili, M.; Tilwari, A.; Mohapatra, R.K.; Srivastava, A.K.; Verma, S. Robust Synthesis and Characteristics of Novel One-Dimensional Gadolinium Oxide Nanorods Decorated Multiwalled Carbon Nanotubes Based Antibacterial Nanocomposites for Health Care Applications. Inorg. Chem. Commun. 2023, 157, 111324. [Google Scholar] [CrossRef]
- Zhang, Z.; Wells, C.J.R.; Liang, R.; et al. Gadolinium Doped Layered Double Hydroxides for Simultaneous Drug Delivery and Magnetic Resonance Imaging. J. Clust. Sci. 2023, 34, 385–394. [Google Scholar] [CrossRef]
- Zhang, J.; Lu, L.; Zhang, W.; Miao, Y.; Du, H.; Xia, H.; Tao, Z.; Du, Z.; Tang, Y.; Fang, Q. Gadolinium Ion-Loaded Mesoporous Organosilica Nanoplatform for Enhanced Radiotherapy in Breast Tumor Treatment. Colloids Surf. B Biointerfaces 2025, 246, 114374. [Google Scholar] [CrossRef]
- Sun, Z.; Zhou, Y.; Li, L.; Zhou, C.; Jia, W.; Liu, Y.; Cao, X.; Su, S.; Zhao, Z.; Zhen, M.; Wang, C. Gadolinium-Based Nanomaterials for Advanced Biomedical Applications. Sci. Bull. 2023, 68, 1651–1665. [Google Scholar] [CrossRef]
- Mulugeta, E.; Tegafaw, T.; Liu, Y.; Zhao, D.; Chen, X.; Baek, A.; Kim, J.; Chang, Y.; Lee, G.H. Current Status and Future Aspects of Gadolinium Oxide Nanoparticles as Positive Magnetic Resonance Imaging Contrast Agents. Nanomaterials 2025, 15, 1340. [Google Scholar] [CrossRef] [PubMed]
- Pecharsky, V.K.; Gschneidner, K.A., Jr. Magnetocaloric Effect and Magnetic Refrigeration. J. Magn. Magn. Mater. 1999, 200, 44–56. [Google Scholar] [CrossRef]
- Blomqvist, L.; Nordberg, G.F.; Nurchi, V.M.; Aaseth, J.O. Gadolinium in Medical Imaging—Usefulness, Toxic Reactions and Possible Countermeasures—A Review. Biomolecules 2022, 12, 742. [Google Scholar] [CrossRef] [PubMed]
- Henderson, I.M.; Benevidez, A.D.; Mowry, C.D.; Watt, J.; Bachand, G.D.; Kirk, M.L.; Dokładny, K.; DeAguero, J.; Escobar, G.P.; Wagner, B. Precipitation of gadolinium from magnetic resonance imaging contrast agents may be the brass tacks of toxicity. Magn. Reson. Imaging 2025, 119, 110383. [Google Scholar] [CrossRef]
- McDonald, R.J.; McDonald, J.S.; Kallmes, D.F.; Jentoft, M.E.; Murray, D.L.; Thielen, K.R.; Williamson, E.E.; Eckel, L.J. Intracranial Gadolinium Deposition after Contrast-Enhanced MR Imaging. Radiology 2015, 275(3), 772–782. [Google Scholar] [CrossRef]
- Kanda, T.; Ishii, K.; Kawaguchi, H.; Kitajima, K.; Takenaka, D. High Signal Intensity in the Dentate Nucleus and Globus Pallidus on Unenhanced T1-Weighted MR Images: Relationship with Increasing Cumulative Dose of a Gadolinium-Based Contrast Material. Radiology 2014, 270(3), 834–841. [Google Scholar] [CrossRef]
- Diagaradjane, P.; Deorukhkar, A.; Gelovani, J.G.; Maru, D.M.; Krishnan, S. Gadolinium Chloride Augments Tumor-Specific Imaging of Targeted Quantum Dots In Vivo. ACS Nano 2010, 4(7), 4131–4141. [Google Scholar] [CrossRef]
- Faucher, L.; Guay-Bégin, A.-A.; Lagueux, J.; Côté, M.-F.; Petitclerc, É.; Fortin, M.-A. Ultra-Small Gadolinium Oxide Nanoparticles to Image Brain Cancer Cells In Vivo with MRI. Contrast Media Mol. Imaging 2011, 6, 209–218. [Google Scholar] [CrossRef]
- Jiang, X.; Mietner, J.B.; Raveendran, D.; Ovchinnikov, K.V.; Sochor, B.; Mueller, S.; Boehm-Sturm, P.; Lerouge, F.; Chaput, F.; Gurikov, P.; Roth, S.V.; Navarro, J.R.G. Multifunctional Cellulose Nanofibrils–GdF3 Nanoparticles Hybrid Gel and Its Potential Uses for Drug Delivery and Magnetic Resonance Imaging. ACS Appl. Nano Mater. 2023, 6, 21182–21193. [Google Scholar] [CrossRef]
- Vecchione, D.; Aiello, M.; Cavaliere, C.; Nicolai, E.; Netti, P.A.; Torino, E. Hybrid Core–Shell Nanoparticles Entrapping Gd-DTPA and 18F-FDG for Simultaneous PET/MRI Acquisitions. Nanomedicine 2017, 12, 2223–2231. [Google Scholar]
- Fauzia, R.P.; Afriani, Z.; Haryuni, R.D.; Juliyanto, S.; Wyantuti, S.; Bahti, H.H. Effect of Size, Charge, and Surface Functionalization of Gadolinium Nanoparticles on Biocompatibility and Cellular Uptake as Magnetic Resonance Imaging Contrast Agents. Trends Sci. 2025, 22, 9330. [Google Scholar] [CrossRef]
- Zhu, D.; Liu, F.; Ma, L.; Liu, D.; Wang, Z. Nanoparticle-Based Systems for T1-Weighted Magnetic Resonance Imaging Contrast Agents. Int. J. Mol. Sci. 2013, 14, 10591–10607. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, N. Gadolinium-Loaded Nanoparticles in Theranostic Magnetic Resonance Imaging. Biomaterials 2012, 33(21), 5363–5375. [Google Scholar] [CrossRef] [PubMed]
- Šimečková, P.; Hubatka, F.; Kotouček, J.; et al. Gadolinium Labelled Nanoliposomes as the Platform for MRI Theranostics: In Vitro Safety Study in Liver Cells and Macrophages. Sci. Rep. 2020, 10, 4780. [Google Scholar] [CrossRef]
- Tran, V.-L.; Thakare, V.; Natuzzi, M.; Moreau, M.; Oudot, A.; Vrigneaud, J.M.; Courteau, A.; Louis, C.; Roux, S.; Boschetti, F.; Denat, F.; Tillement, O.; Lux, F. Functionalization of Gadolinium Chelates Silica Nanoparticle through Silane Chemistry for Simultaneous MRI/64Cu PET Imaging. Contrast Media Mol. Imaging 2018, 2018, 7938267. [Google Scholar] [CrossRef]
- Karati, D.; Meur, S.; Roy, S.; Mukherjee, S. Gadolinium Based Nanoplatform as Drug Delivery Approach for Targeted Therapy. J. Drug Deliv. Sci. Technol. 2026, 115, 107807. [Google Scholar] [CrossRef]
- Yong, Y.; Zhou, L.; Zhang, S.; et al. Gadolinium Polytungstate Nanoclusters: A New Theranostic with Ultrasmall Size and Versatile Properties for Dual-Modal MR/CT Imaging and Photothermal Therapy/Radiotherapy of Cancer. NPG Asia Mater. 2016, 8, e273. [Google Scholar] [CrossRef]
- Dutta Gupta, Y.; Mackeyev, Y.; Krishnan, S.; et al. Mesoporous Silica Nanotechnology: Promising Advances in Augmenting Cancer Theranostics. Cancer Nanotechnol. 2024, 15, 9. [Google Scholar] [CrossRef]
- Domingo, J.L.; Semelka, R.C. Gadolinium Toxicity: Mechanisms, Clinical Manifestations, and Nanoparticle Role. Arch. Toxicol. 2025, 99, 3897–3916. [Google Scholar] [CrossRef]
- Sancey, L.; Kotb, S.; Truillet, C.; Appaix, F.; Marais, A.; Thomas, E.; van der Sanden, B.; Klein, J.-P.; Laurent, B.; Cottier, M.; Antoine, R.; Dugourd, P.; Panczer, G.; Lux, F.; Perriat, P.; Motto-Ros, V.; Tillement, O. Long-Term In Vivo Clearance of Gadolinium-Based AGuIX Nanoparticles and Their Biocompatibility after Systemic Injection. ACS Nano 2015, 9, 2477–2488. [Google Scholar] [CrossRef]
- Fatima, A.; Ahmad, M.W.; Al Saidi, A.K.A.; Choudhury, A.; Chang, Y.; Lee, G.H. Recent Advances in Gadolinium Based Contrast Agents for Bioimaging Applications. Nanomaterials 2021, 11, 2449. [Google Scholar] [CrossRef]
- Lux, F.; Sancey, L.; Bianchi, A.; Crémillieux, Y.; Roux, S.; Tillement, O. Gadolinium-Based Nanoparticles for Theranostic MRI-Radiosensitization. Nanomedicine 2015, 10, 1801–1815. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ai, K.; Lu, L. Nanoparticulate Gadolinium Complexes as MRI Contrast Agents. Chem. Rev. 2014, 114, 5057–5115. [Google Scholar] [CrossRef] [PubMed]
- Rowe, M.D.; Thamm, D.H.; Kraft, S.L.; Boyes, S.G. Polymer-Modified Gadolinium Metal-Organic Framework Nanoparticles Used as Multifunctional Nanomedicines for the Targeted Imaging and Treatment of Cancer. Biomacromolecules 2009, 10, 983–993. [Google Scholar] [CrossRef] [PubMed]
- Khajuria, H.; Ladol, J.; Singh, R.; Khajuria, S.; Khajuria, H.N. Surfactant Assisted Sonochemical Synthesis and Characterization of Gadolinium Doped Zinc Oxide Nanoparticles. Acta Chim. Slov. 2015, 62, 849–858. [Google Scholar] [CrossRef]
- Li, Z.; Guo, J.; Zhang, M.; Li, G.; Hao, L. Gadolinium-Coated Mesoporous Silica Nanoparticle for Magnetic Resonance Imaging. Front. Chem. 2022, 10, 837032. [Google Scholar] [CrossRef]
- Unruh, C.; Van Bavel, N.; Anikovskiy, M.; Prenner, E.J. Benefits and Detriments of Gadolinium: From Medical Advances to Health and Ecological Risks. Molecules 2020, 25, 5762. [Google Scholar] [CrossRef]
- Kotb, S.; Detappe, A.; Lux, F.; Appaix, F.; Barbier, E.L.; Tran, V.-L.; Plissonneau, M.; Gehan, H.; Lefranc, F.; Rodriguez-Lafrasse, C.; Verry, C.; Berbeco, R.; Tillement, O.; Sancey, L. Gadolinium-Based Nanoparticles and Radiation Therapy for Multiple Brain Melanoma Metastases: Proof of Concept before Phase I Trial. Theranostics 2016, 6, 418–427. [Google Scholar] [CrossRef]
- Chen, J.S.; Chen, J.; Bhattacharjee, S.; Cao, Z.; Wang, H.; Swanson, S.D.; et al. Functionalized Nanoparticles with Targeted Antibody to Enhance Imaging of Breast Cancer In Vivo. J. Nanobiotechnol. 2020, 18, 135–139. [Google Scholar] [CrossRef]
- Usman, M.S.; Hussein, M.Z.; Fakurazi, S.; et al. Gadolinium-Based Layered Double Hydroxide and Graphene Oxide Nano-Carriers for Magnetic Resonance Imaging and Drug Delivery. Chem. Cent. J. 2017, 11, 47. [Google Scholar] [CrossRef]
- Truillet, C.; Bouziotis, P.; Tsoukalas, C.; Brugière, J.; Martini, M.; Sancey, L.; Brichart, T.; Denat, F.; Boschetti, F.; Darbost, U.; Bonnamour, I.; Stellas, D.; Anagnostopoulos, C.D.; Koutoulidis, V.; Moulopoulos, L.A.; Perriat, P.; Lux, F.; Tillement, O. Ultrasmall Particles for Gd-MRI and 68Ga-PET Dual Imaging. Contrast Media Mol. Imaging 2015, 10(4), 309–319. [Google Scholar] [CrossRef] [PubMed]
- Tillement, O.; Lux, F.; Luciani, N.; et al. Gadolinium-Based Nanoparticles for Theranostic MRI. Adv. Healthc. Mater. 2014, 3, 1769–1782. [Google Scholar]
- Lux, F.; Tran, V.L.; Thomas, E.; Dufort, S.; Rossetti, F.; Martini, M.; Truillet, C.; Doussineau, T.; Bort, G.; Denat, F.; Boschetti, F.; Angelovski, G.; Detappe, A.; Crémillieux, Y.; Mignet, N.; Doan, B.-T.; Larrat, B.; Meriaux, S.; Barbier, E.; Roux, S.; Fries, P.; Müller, A.; Abadjian, M.-C.; Anderson, C.; Canet-Soulas, E.; Bouziotis, P.; Barberi-Heyob, M.; Frochot, C.; Verry, C.; Balosso, J.; Evans, M.; Sidi-Boumedine, J.; Janier, M.; Butterworth, K.; McMahon, S.; Prise, K.; Aloy, M.-T.; Ardail, D.; Rodriguez-Lafrasse, C.; Porcel, E.; Lacombe, S.; Berbeco, R.; Allouch, A.; Perfettini, J.-L.; Chargari, C.; Deutsch, E.; Le Duc, G.; Tillement, O. AGuIX® from Bench to Bedside—Transfer of an Ultrasmall Theranostic Gadolinium-Based Nanoparticle to Clinical Medicine. Br. J. Radiol. 2019, 92, 20180365. [Google Scholar] [CrossRef] [PubMed]
- Thomas, E.; Colombeau, L.; Gries, M.; Peterlini, T.; Mathieu, C.; Thomas, N.; Boura, C.; Frochot, C.; Vanderesse, R.; Lux, F.; Barberi-Heyob, M.; Tillement, O. Ultrasmall AGuIX theranostic nanoparticles for vascular-targeted interstitial photodynamic therapy of glioblastoma. Int. J. Nanomed. 2017, 12, 7075–7088. [Google Scholar] [CrossRef]
- Bridot, J.-L.; Faure, A.-C.; Laurent, S.; Riviere, C.; Billotey, C.; Hiba, B.; Janier, M.; Josserand, V.; Coll, J.-L.; Elst, L.V.; Muller, R.; Roux, S.; Perriat, P.; Tillement, O. Hybrid Gadolinium Oxide Nanoparticles: Multimodal Contrast Agents for In Vivo Imaging. J. Am. Chem. Soc. 2007, 129, 5076–5084. [Google Scholar] [CrossRef]
- Major, J.L.; Meade, T.J. Bioresponsive, Cell-Penetrating, and Multimeric MR Contrast Agents. Acc. Chem. Res. 2009, 42, 893–903. [Google Scholar] [CrossRef]
- Longmire, M.; Choyke, P.L.; Kobayashi, H. Clearance Properties of Nano-Sized Particles and Molecules as Imaging Agents. Nanomedicine 2008, 3, 703–717. [Google Scholar] [CrossRef]
- Alexis, F.; Pridgen, E.; Molnar, L.K.; Farokhzad, O.C. Factors Affecting the Clearance and Biodistribution of Polymeric Nanoparticles. Mol. Pharm. 2008, 5, 505–515. [Google Scholar] [CrossRef]
- Owens, D.E.; Peppas, N.A. Opsonization, Biodistribution, and Pharmacokinetics of Polymeric Nanoparticles. Int. J. Pharm. 2006, 307, 93–102. [Google Scholar] [CrossRef]
- Albanese, A.; Tang, P.S.; Chan, W.C.W. The Effect of Nanoparticle Size, Shape, and Surface Chemistry on Biological Systems. Annu. Rev. Biomed. Eng. 2012, 14, 1–16. [Google Scholar] [CrossRef]
- Blanco, E.; Shen, H.; Ferrari, M. Principles of Nanoparticle Design for Overcoming Biological Barriers to Drug Delivery. Nat. Biotechnol. 2015, 33, 941–951. [Google Scholar] [CrossRef]
- Bertrand, N.; Leroux, J.-C. The Journey of a Drug-Carrier in the Body: An Anatomo-Physiological Perspective. J. Control. Release 2012, 161, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Peer, D.; Karp, J.M.; Hong, S.; Farokhzad, O.C.; Margalit, R.; Langer, R. Nanocarriers as an Emerging Platform for Cancer Therapy. Nat. Nanotechnol. 2007, 2, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Parang, K.; Nasrolahi Shirazi, A. Peptide-Mediated Cellular Delivery Systems. Chem. Soc. Rev. 2013, 42, 4120–4133. [Google Scholar]
- Roberts, D.R.; Lindhorst, S.M.; Welsh, C.T.; et al. High Levels of Gadolinium Deposition in the Skin of a Patient with Normal Renal Function. Invest. Radiol. 2016, 51, 280–289. [Google Scholar] [CrossRef]
- Roberts, D.R.; Holden, K.R. Progressive Increase of T1 Signal Intensity in the Dentate Nucleus and Globus Pallidus on Unenhanced T1-Weighted MR Images in the Pediatric Brain Exposed to Multiple Doses of Gadolinium Contrast. Brain Dev. 2016, 38(3), 331–336. [Google Scholar] [CrossRef]
- Choi, H.S.; Liu, W.; Misra, P.; et al. Renal Clearance of Quantum Dots. Nat. Biotechnol. 2007, 25, 1165–1170. [Google Scholar] [CrossRef]
- Frenzel, T.; Lengsfeld, P.; Schirmer, H.; Hütter, J.; Weinmann, H.-J. Stability of Gadolinium-Based Magnetic Resonance Imaging Contrast Agents in Human Serum at 37 °C. Invest. Radiol. 2008, 43, 817–828. [Google Scholar] [CrossRef]
- Idée, J.-M.; Port, M.; Robic, C.; Medina, C.; Sabatou, M.; Corot, C. Role of Thermodynamic and Kinetic Stability in Gadolinium Chelate Safety. Invest. Radiol. 2009, 44, 123–133. 94. [Google Scholar]
- Rogosnitzky, M.; Branch, S. Gadolinium-Based Contrast Agent Toxicity: A Review of Known and Proposed Mechanisms. Biometals 2016, 29, 365–376. [Google Scholar] [CrossRef]
- Neburkova, J.; Rulseh, A.M.; Chang, S.L.Y.; Raabova, H.; Vejpravova, J.; Dracinsky, M.; Tarabek, J.; Kotek, J.; Pingle, M.; Majer, P.; Vymazal, J.; Cigler, P. Formation of Gadolinium–Ferritin from Clinical Magnetic Resonance Contrast Agents. Nanoscale Adv. 2020, 2, 5567–5571. [Google Scholar] [CrossRef]
- McDonald, R.J.; McDonald, J.S.; Kallmes, D.F.; et al. Gadolinium Deposition in Human Brain Tissues after Contrast-Enhanced MR Imaging in Adult Patients without Intracranial Abnormalities. Radiology 2017, 285(2), 546–554. [Google Scholar] [CrossRef]
- Ayers-Ringler, J.; McDonald, J.S.; Connors, M.A.; et al. Neurologic Effects of Gadolinium Retention in the Brain after Gadolinium-Based Contrast Agent Administration. Radiology 2022, 302(3), 676–683. [Google Scholar] [CrossRef]
- Tibussek, D.; Rademacher, C.; Caspers, J.; et al. Gadolinium Brain Deposition after Macrocyclic Gadolinium Administration: A Pediatric Case-Control Study. Radiology 2017, 285(1), 223–230. [Google Scholar] [CrossRef] [PubMed]
- Stojanov, D.A.; Aracki-Trenkic, A.; Vojinovic, S.; Benedeto-Stojanov, D.; Ljubisavljevic, S. Increasing Signal Intensity within the Dentate Nucleus and Globus Pallidus on Unenhanced T1W Magnetic Resonance Images in Patients with Relapsing-Remitting Multiple Sclerosis: Correlation with Cumulative Dose of Gadobutrol. Eur. Radiol. 2016, 26(3), 807–815. [Google Scholar] [CrossRef] [PubMed]
- Bjørnerud, A.; Vatnehol, S.A.S.; Larsson, C.; Due-Tønnessen, P.; Hol, P.K.; Groote, I.R. Signal Enhancement of the Dentate Nucleus at Unenhanced MR Imaging after Very High Cumulative Doses of Gadobutrol: An Observational Study. Radiology 2017, 285(2), 434–444. [Google Scholar] [CrossRef] [PubMed]
- Radbruch, A.; Weberling, L.D.; Kieslich, P.J.; et al. High-Signal Intensity in the Dentate Nucleus and Globus Pallidus on Unenhanced T1-Weighted Images: Evaluation of Gadobutrol. Invest. Radiol. 2015, 50(12), 805–810. [Google Scholar] [CrossRef]
- Murata, N.; Gonzalez-Cuyar, L.F.; Murata, K.; et al. Macrocyclic and Other Non-Group 1 Gadolinium Contrast Agents Deposit Low Levels of Gadolinium in Brain and Bone Tissue: Preliminary Results. Invest. Radiol. 2016, 51(7), 447–453. [Google Scholar] [CrossRef]
- Kobayashi, M.; Levendovszky, S.R.; Hippe, D.S.; et al. Comparison of Human Tissue Gadolinium Retention and Elimination between Gadoteridol and Gadobenate. Radiology 2021, 300(3), 559–569. [Google Scholar] [CrossRef]
- Le Fur, M.; McDonald, R.J.; Murray, D.L.; Thielen, K.R.; Eckel, L.J.; Jentoft, M.E.; Kallmes, D.F.; Widrick, J.J.; Toland, J.A.; Wagner, B.; et al. Gadolinium-based Contrast Agent Biodistribution and Speciation in Rats and Humans: Comparison of Gadoterate, Gadoteridol, Gadobutrol, and Gadobenate Using MRI, Mass Spectrometry, Elemental Imaging, and Electron Paramagnetic Resonance Spectroscopy. Radiology 2023, 309, e230984. [Google Scholar] [CrossRef]
- Guenther, C.; Frenzel, T.; Rottmann, A.; Lohrke, J.; Janssen, W. Pharmacokinetics, Excretion, and Organ Distribution of the Novel Gadolinium-Based MRI Contrast Agent Gadoquatrane in Female Cynomolgus Monkeys. Invest. Radiol. 2025, 60, 831–839. [Google Scholar] [CrossRef]
- Kubik-Huch, R.A.; Gottstein-Aalame, N.M.; Frenzel, T.; Seifert, B.; Puchert, E.; Wittek, S.; Debatin, J.F. Gadopentetate Dimeglumine Excretion into Human Breast Milk during Lactation. Radiology 2000, 216(2), 555–558. [Google Scholar] [CrossRef] [PubMed]
- Hahn, G.; Sorge, I.; Gruhn, B.; et al. Pharmacokinetics and Safety of Gadobutrol-Enhanced Magnetic Resonance Imaging in Pediatric Patients. Invest. Radiol. 2009, 44(12), 776–783. [Google Scholar] [CrossRef]
- Lux, F.; Roux, S.; Perriat, P.; Tillement, O. Gadolinium-Based Nanoparticles for Theranostic MRI-Radiosensitization. Nanomedicine 2019, 14, 2479–2493. [Google Scholar] [CrossRef] [PubMed]
- McMahon, S.J.; Hyland, W.B.; Muir, M.F.; Coulter, J.A.; Jain, S.; Butterworth, K.T.; Schettino, G.; Dickson, G.R.; Hounsell, A.R.; O’Sullivan, J.M.; Prise, K.M.; Hirst, D.G.; Currell, F.J. Biological Consequences of Nanoscale Energy Deposition near Irradiated Heavy Atom Nanoparticles. Sci. Rep. 2016, 6, 33290. [Google Scholar] [CrossRef] [PubMed]
- Lux, F.; Tran, V.-L.; Thomas, E.; Dufort, S.; Rossetti, F.; Martini, M.; Truillet, C.; Chouikrat, R.; Boschetti, F.; Denat, F.; Roux, S.; Perriat, P.; Tillement, O. AGuIX® Gadolinium-Based Nanoprobes for Theranostic Applications. Theranostics 2014, 4, 625–635. [Google Scholar]
- Le Duc, G.; Miladi, I.; Alric, C.; Mowat, P.; Bräuer-Krisch, E.; Bouchet, A.; Khalil, E.; Billotey, C.; Janier, M.; Lux, F.; Epicier, T.; Perriat, P.; Roux, S.; Tillement, O. Toward an image-guided microbeam radiation therapy using gadolinium-based nanoparticles. ACS Nano 2011, 5, 9566–9574. [Google Scholar] [CrossRef]
- Aubrun, C.; Doussineau, T.; Carmès, L.; Meyzaud, A.; Boux, F.; Dufort, S.; Delfour, A.; De Beaumont, O.; Mirjolet, C.; Le Duc, G. Mechanisms of Action of AGuIX as a Pan-Cancer Nano-Radiosensitizer: A Comprehensive Review. Pharmaceuticals 2025, 18, 519. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, L.; Li, Y.; Wang, J. Nanomaterials for photothermal therapy: An update. Nanoscale 2016, 8, 5567–5583. [Google Scholar]
- Jaque, D.; Martínez Maestro, L.; del Rosal, B.; Haro-González, P.; Benayas, A.; Plaza, J. L.; Rodríguez, E. M.; García Solé, J. Nanoparticles for photothermal therapies. Nanoscale 2014, 6, 9494–9530. [Google Scholar] [CrossRef]
- Lv, X.; Wang, X.; Li, T.; Wei, C.; Tang, Y.; Yang, T.; Wang, Q.; Yang, X.; Chen, H.; Shen, J.; Yang, H.; Ke, H. Rationally designed monodisperse Gd₂O₃/Bi₂S₃ hybrid nanodots for efficient cancer theranostics. Small 2018, 14, 1802904. [Google Scholar] [CrossRef]
- Jain, A.; Fournier, P. G. J.; Mendoza-Lavaniegos, P.; Sengar, P.; Guerra-Olvera, F. M.; Iñiguez, E.; Kretzschmar, T. G.; Hirata, G. A.; Juárez, P. Functionalized Gd₂O₃ nanoparticles for magnetic resonance imaging and targeted photothermal therapy of cancer. J. Nanobiotechnol. 2018, 16, 26. [Google Scholar] [CrossRef]
- Hildebrandt, B.; Wust, P.; Ahlers, O.; Dieing, A.; Sreenivasa, G.; Kerner, T.; Felix, R.; Riess, H. The cellular and molecular basis of hyperthermia. Crit. Rev. Oncol. Hematol. 2002, 43, 33–56. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Jain, P. K.; El-Sayed, I. H.; El-Sayed, M. A. Plasmonic photothermal therapy (PPTT) using gold nanoparticles. Lasers Med. Sci. 2008, 23, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Yung, B.; Huang, P.; Chen, X. Nanotechnology for multimodal synergistic cancer therapy. Chem. Rev. 2017, 117, 13566–13638. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Bhattarai, P.; Dai, Z.; Chen, X. Photothermal therapy and immunotherapy: A synergistic combination. ACS Nano 2018, 12, 2493–2505. [Google Scholar]
- Zhao, P.-P.; Ge, Y.-W.; Liu, X.-L.; Ke, Q.-F.; Zhang, J.-W.; Zhu, Z.-A.; Guo, Y.-P. Ordered Arrangement of Hydrated GdPO₄ Nanorods in Magnetic Chitosan Matrix Promotes Tumor Photothermal Therapy and Bone Regeneration against Breast Cancer Bone Metastases. Chem. Eng. J. 2020, 381, 122694. [Google Scholar] [CrossRef]
- Cui, H.; Zhao, Y.-Y.; Wu, Q.; You, Y.; Lan, Z.; Zou, K.-L.; Cheng, G.-W.; Chen, H.; Han, Y.-H.; Chen, Y.; Qi, X.-D.; Meng, X.-W.; Ma, L.-M.; Yu, G.-T. Microwave-Responsive Gadolinium Metal–Organic Framework Nanosystem for MRI-Guided Cancer Thermotherapy and Synergistic Immunotherapy. Bioact. Mater. 2024, 33, 532–544. [Google Scholar]
- Jiang, Q.; Liu, L.; Li, Q.; Cao, Y.; Chen, D.; Du, Q.; Yang, X.; Huang, D.; Pei, R.; Chen, X.; Huang, G. NIR-Laser-Triggered Gadolinium-Doped Carbon Dots for Magnetic Resonance Imaging, Drug Delivery and Combined Photothermal Chemotherapy for Triple-Negative Breast Cancer. J. Nanobiotechnol. 2021, 19, 64. [Google Scholar] [CrossRef]
- Bertrand, N.; Wu, J.; Xu, X.; Kamaly, N.; Farokhzad, O.C. Cancer Nanotechnology: The Impact of Passive and Active Targeting in the Era of Modern Cancer Biology. Adv. Drug Deliv. Rev. 2014, 66, 2–25. [Google Scholar] [CrossRef]
- Mowat, P.; Mignot, A.; Rima, W.; Lux, F.; Tillement, O.; Roulin, C.; Dutreix, M.; Bechet, D.; Huger, S.; Humbert, L.; Barberi-Heyob, M.; Aloy, M.-T.; Armandy, E.; Rodriguez-Lafrasse, C.; Le Duc, G.; Roux, S.; Perriat, P. In Vitro Radiosensitizing Effects of Ultrasmall Gadolinium-Based Particles on Tumour Cells. J. Nanosci. Nanotechnol. 2011, 11, 7833–7839. [Google Scholar] [CrossRef] [PubMed]
- Detappe, A.; Thomas, E.; Tibbitt, M.W.; Kunjachan, S.; Zavidij, O.; Parnandi, N.; Reznichenko, E.; Lux, F.; Tillement, O.; Berbeco, R. Ultrasmall Silica-Based Bismuth Gadolinium Nanoparticles for Dual Magnetic Resonance–Computed Tomography Image-Guided Radiation Therapy. Nano Lett. 2017, 17, 1733–1740. [Google Scholar] [CrossRef] [PubMed]
- Siribbal, S.M.; Jurewicz, A.; Hassan, M.; Iqbal, S.; Hu, Z.; Uvdal, K.; Hussain, M.S.; Mathur, S.; Ilyas, S. Biocompatible Hollow Gadolinium Oxide Nanocarriers for the Transport of Bioactive Molecules to Cells. ACS Appl. Nano Mater. 2024, 7, 12408–12419. [Google Scholar] [CrossRef]
- Kumar, A.; Sarkar, T.; Solanki, P.R. Amine Functionalized Gadolinium Oxide Nanoparticles-Based Electrochemical Immunosensor for Cholera. Biosensors 2023, 13, 177. [Google Scholar] [CrossRef]
- Siribbal, S.M.; Ilyas, S.; Renner, A.M.; Iqbal, S.; Muñoz Vázquez, S.; Moawia, A.; Valldor, M.; Hussain, M.S.; Schomäcker, K.; Mathur, S. Click Functionalized Biocompatible Gadolinium Oxide Core–Shell Nanocarriers for Imaging of Breast Cancer Cells. RSC Adv. 2022, 12, 31830–31845. [Google Scholar] [CrossRef]
- Mi, P. Stimuli-Responsive Nanocarriers for Drug Delivery, Tumor Imaging, Therapy and Theranostics. Theranostics 2020, 10, 4557–4588. [Google Scholar] [CrossRef]
- Ganesh, S.K.; Chakravorty, A.; Raghavan, V.; Subathra Devi, C. Chitosan-Coated ZnO/TiO2/Gd2O3 Nanocomposites for Improved Rapamycin Delivery in Non-Small Cell Lung Cancer. Int. J. Biol. Macromol. 2025, 319, 145439. [Google Scholar] [CrossRef]
- Babayevska, N.; Kustrzyńska, K.; Przysiecka, Ł.; Jarek, M.; Jancelewicz, M.; Iatsunskyi, I.; Dydak, K.; Skupin-Mrugalska, P.; Janiszewska, E. Doxorubicin and ZnO-Loaded Gadolinium Oxide Hollow Spheres for Targeted Cancer Therapy and Bioimaging. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2025, 333, 125903. [Google Scholar] [CrossRef]
- Wei, Z.; Wu, M.; Li, Z.; Lin, Z.; Zeng, J.; Sun, H.; Liu, X.; Liu, J.; Li, B.; Zeng, Y. Gadolinium-Doped Hollow CeO₂–ZrO₂ Nanoplatform as Multifunctional MRI/CT Dual-Modal Imaging Agent and Drug Delivery Vehicle. Drug Deliv. 2018, 25, 353–363. [Google Scholar] [CrossRef]
- Sun, X.; Kou, B. Biocompatibility and Potential Anticancer Activity of Gadolinium Oxide (Gd2O3) Nanoparticles against Nasal Squamous Cell Carcinoma. BMC Biotechnol. 2024, 24, 53. [Google Scholar] [CrossRef]
- Butterworth, K.T.; McMahon, S.J.; Currell, F.J.; Prise, K.M. Physical Basis and Biological Mechanisms of Gold Nanoparticle Radiosensitization. Nanotechnology 2012, 23, 295101. [Google Scholar] [CrossRef]
- Rétif, P.; Pinel, S.; Toussaint, M.; Frochot, C.; Chouikrat, R.; Bastogne, T.; Barberi-Heyob, M. Nanoparticles for Radiation Therapy Enhancement: The Key Parameters. Cancer Nanotechnol. 2015, 6, 1–16. [Google Scholar] [CrossRef]
- Her, S.; Jaffray, D.A.; Allen, C. Gold Nanoparticles for Applications in Cancer Radiotherapy: Mechanisms and Recent Advancements. Theranostics 2017, 7, 225–239. [Google Scholar] [CrossRef]
- Jackson, S.P.; Bartek, J. The DNA-Damage Response in Human Biology and Disease. Nature 2009, 461, 1071–1078. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, T.; Ke, H.; Zhu, A.; Wang, Y.; Wang, J.; Shen, J.; Liu, G.; Chen, C.; Zhao, Y. Smart Albumin–Biomineralized Nanocomposites for Multimodal Imaging and Photothermal Tumor Ablation. Biomaterials 2018, 160, 57–68. [Google Scholar] [CrossRef]
- Pawlik, T.M.; Keyomarsi, K. Role of Cell Cycle in Chemotherapy Sensitivity. J. Clin. Oncol. 2004, 22, 4436–4445. [Google Scholar]
- American College of Radiology. ACR Manual on Contrast Media; Version 2024; American College of Radiology: Reston, VA, USA, 2024. [Google Scholar]
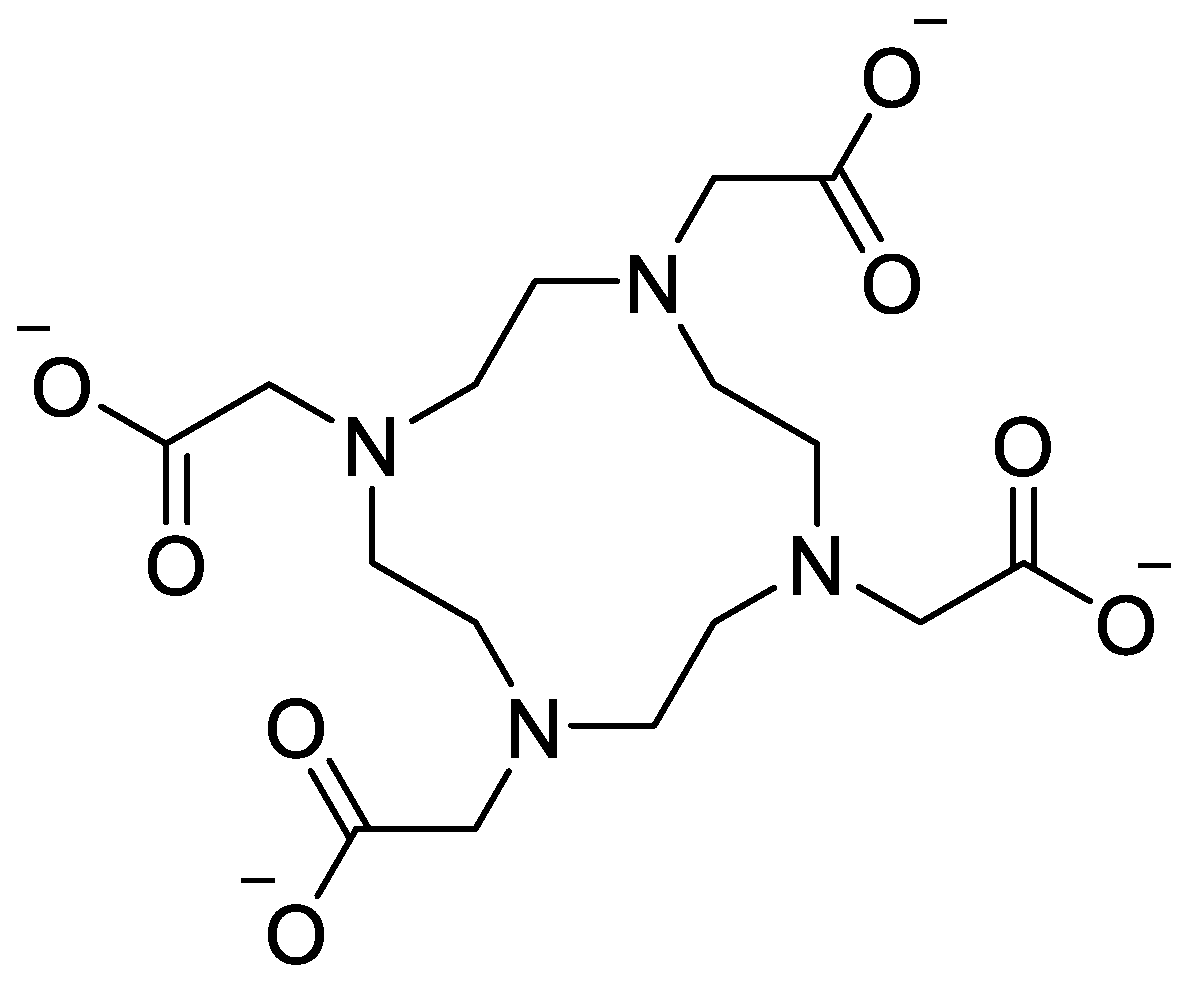
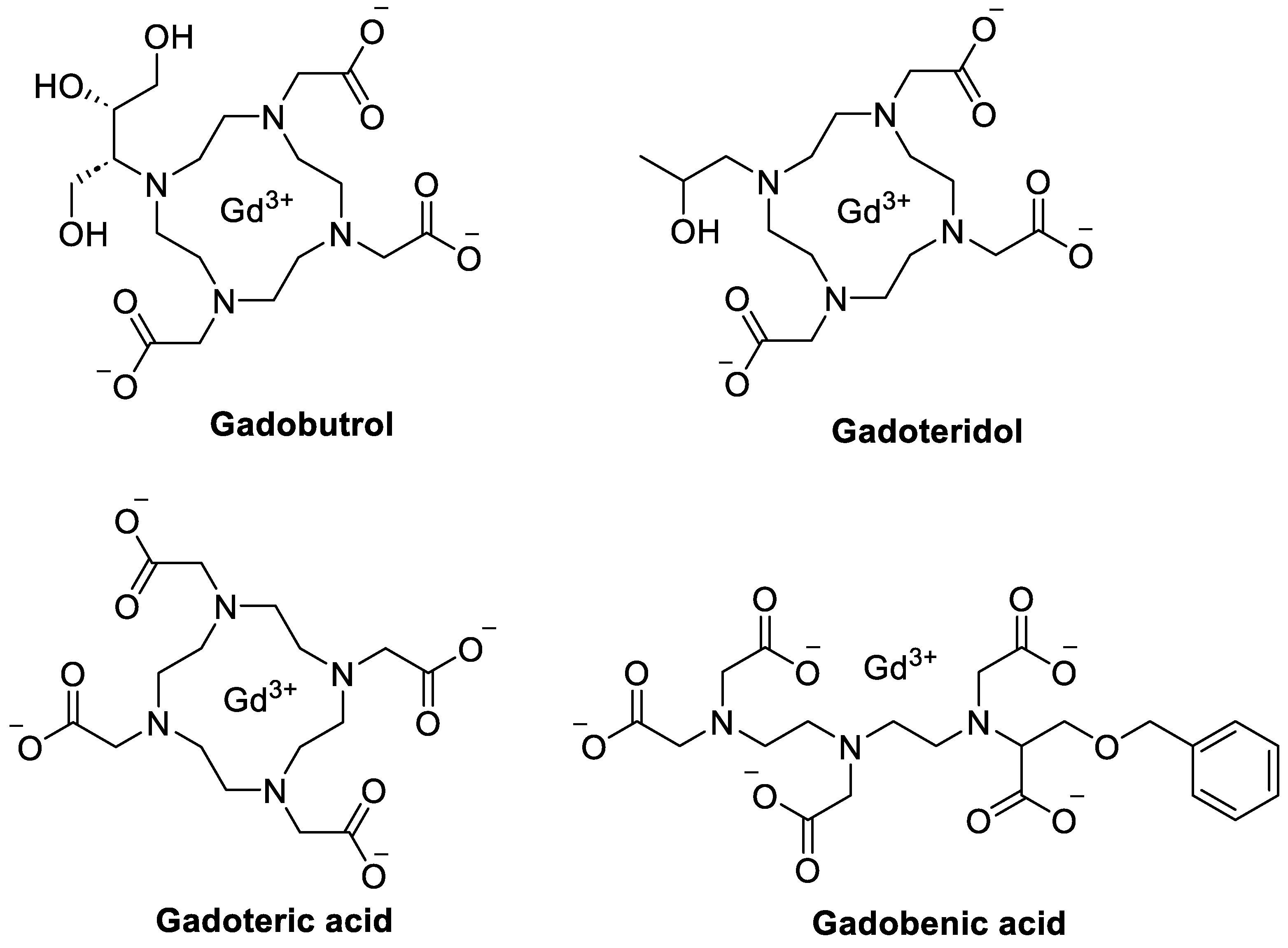
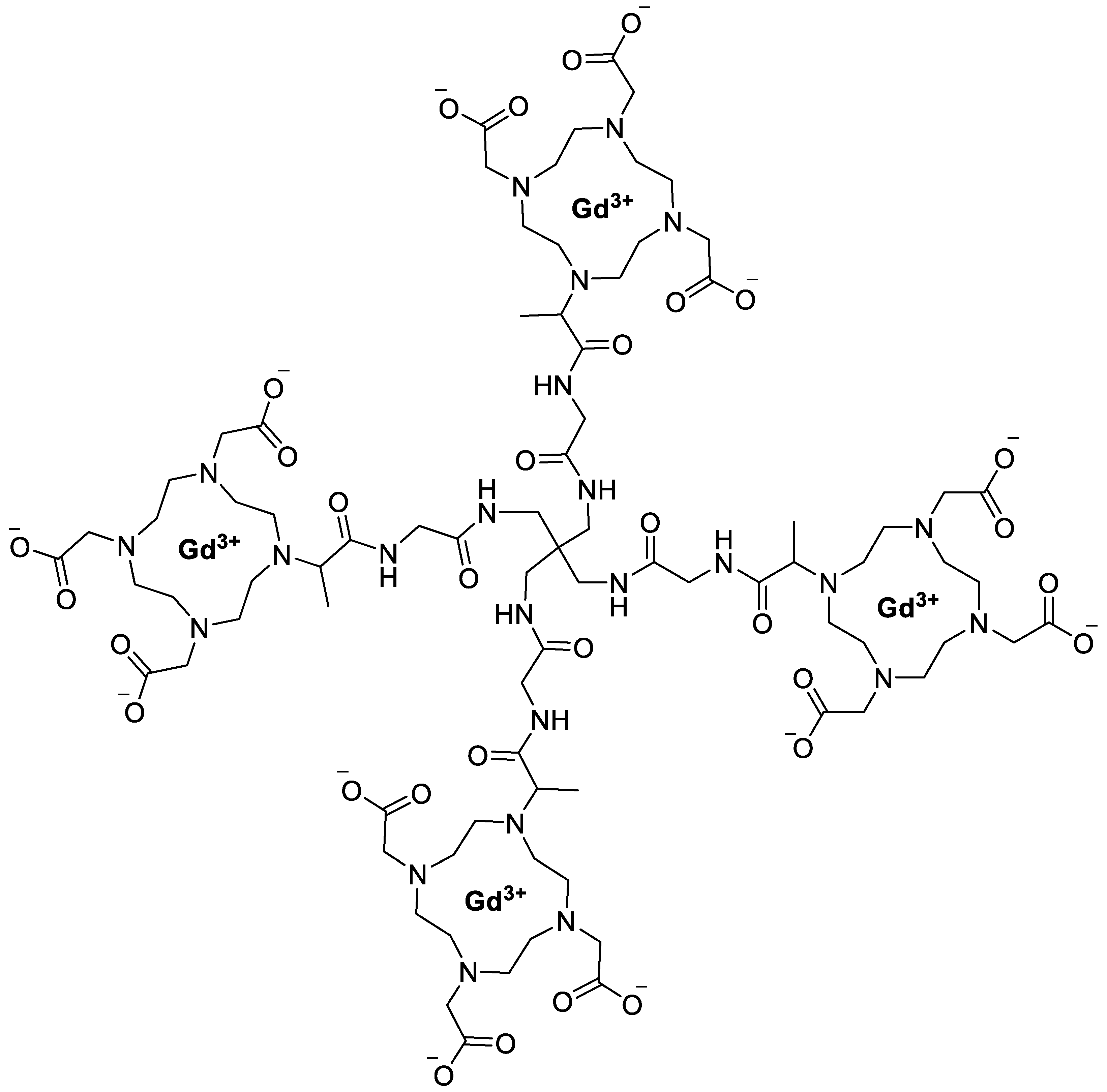
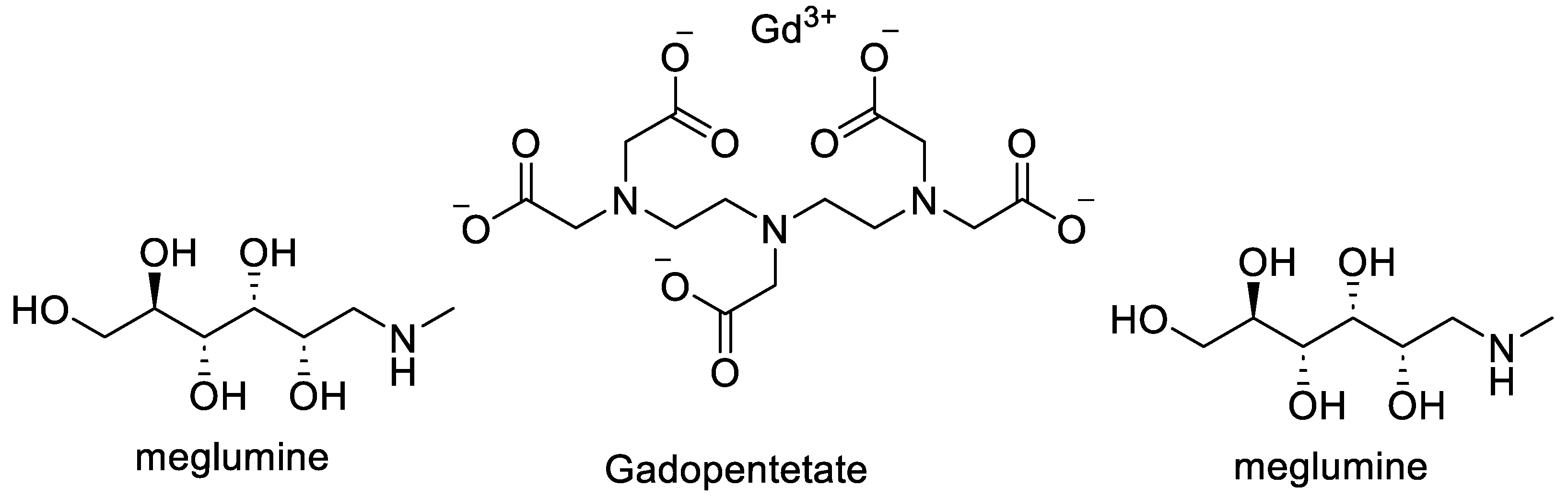
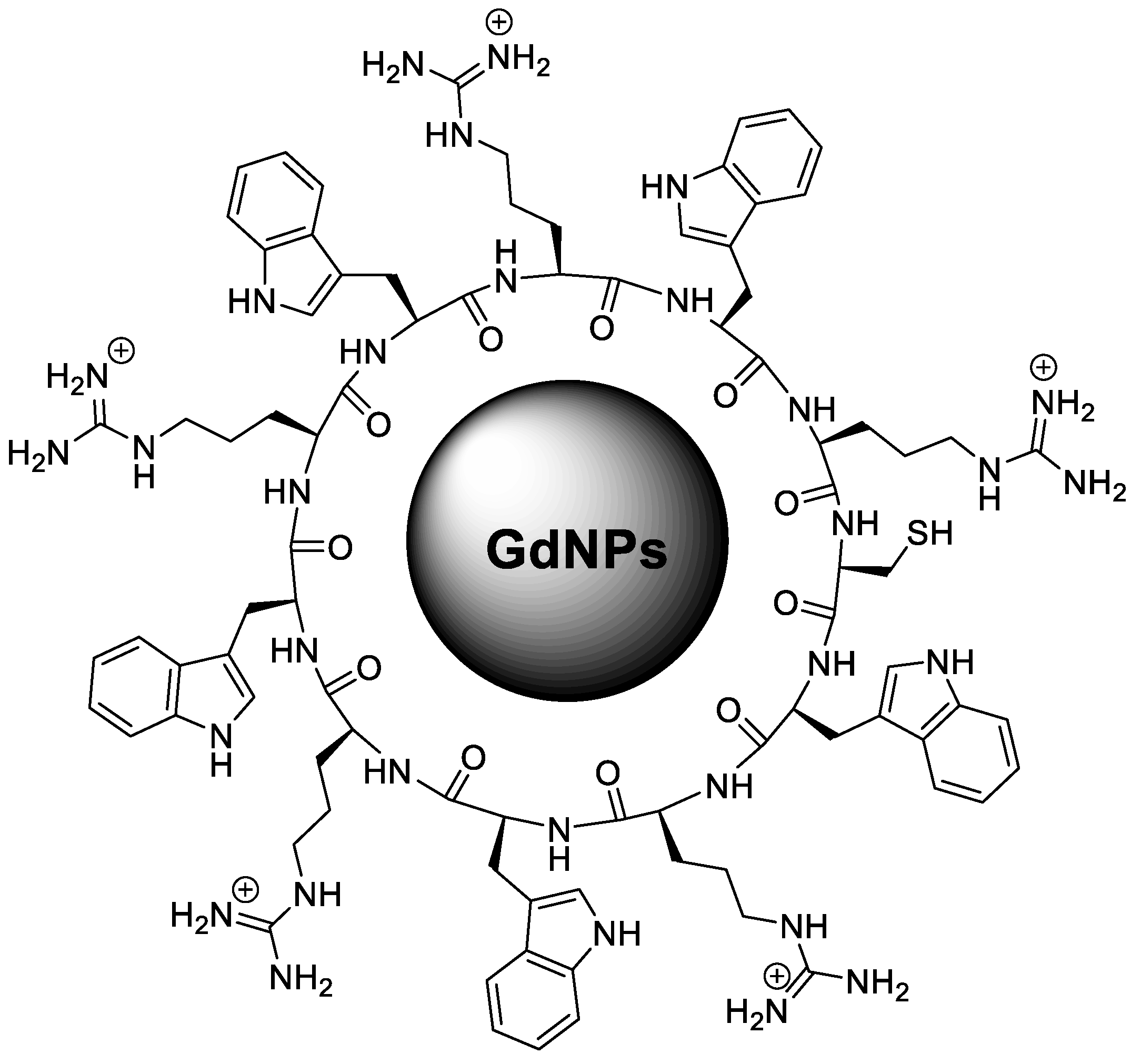
| Nanoparticle/System | Energy Modality | Major Results | Key Drug-Delivery / Theranostic Implications | Reference |
|---|---|---|---|---|
| Gd₂O₃ nanoparticles; Gd-doped hybrid nanostructures | Radiofrequency (RF), NIR light | Efficient non-radiative relaxation → localized heat generation | Hyperthermia | [128] |
| GdPO₄/CS/Fe₃O₄ scaffold | 808 nm NIR laser | Temp ↑ 20 → 47.7 °C in 10 min; tumor apoptosis; BV/TV bone regeneration ↑ to ~61% | Dual-functional platform for tumor ablation + bone regeneration drug delivery | [129] |
| Microwave-responsive Gd-MOF (Gd/MPC) + anti-PD-1 antibody | Microwave irradiation | Temp ↑ >45 °C in 5 min; tumor inhibition ↑ ~26% vs free drug | Temperature-triggered immunotherapy drug release | [130] |
| Gd@Carbon dots loaded with Dox + IR825 | 808 nm NIR laser | Temp ↑ to ~57 °C; >97% tumor cell kill; strong MRI contrast | Controlled drug release + imaging-guided therapy | [131] |
| GdNP System / Functionalization | Therapeutic Payload | Key Structural Features | Drug Delivery / Biological Outcome | Reference |
|---|---|---|---|---|
| Chitosan-coated ZnO/TiO₂/Gd₂O₃ nano-composite (RNC) | Rapamycin | Crystalline ZnO, TiO₂, Gd₂O₃; chitosan coating | 100% drug loading; sustained release; ROS amplification; apoptosis; G0/G1 arrest in A549 cells | [139] |
| Solid vs hollow Gd₂O₃ nanospheres with ZnO coating | Doxorubicin | Hollow spheres (~120 nm) with high BET surface area | pH-responsive release; theranostic imaging capability | [140] |
| Cyclic peptide-GdNPs [(WR)₅C-Gd] | Small-molecule drugs; siRNA | Star-shaped nanoparticles (240–260 nm); cationic peptide surface | 6-fold increased intracellular delivery; enhanced platinum drug efficacy; >10-fold siRNA uptake and STAT-3 knockdown | [25] |
| Gd-doped hollow CeO₂–ZrO₂ nanoplatform (PEGylated) | Doxorubicin | Mesoporous hollow structure; high surface area; PEG coating | pH-responsive release; dual MRI/CT imaging; strong tumor growth inhibition in vivo | [141] |
| Albumin-coated Gd₂O₃ nanoparticles (Gd₂O₃@BSA) | Curcumin | Core <10 nm; hydrodynamic size 17–26 nm; stable colloidal surface | pH-responsive release; high biocompatibility; enhanced cytotoxicity toward nasal carcinoma cells | [142] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




