Introduction
The understanding of airway constriction mechanisms has advanced considerably over the past several decades, reflecting progress not only in pulmonary physiology but also in the neurobiological regulation of breathing [
1,
2]. Airway caliber is controlled by a complex interplay between mechanical forces, airway smooth muscle tone, and autonomic neural inputs, with central modulation increasingly recognized as a critical determinant [
3,
4]. Neurotransmitter systems regulate airway resistance through excitatory and inhibitory signals that converge within the brainstem and peripheral autonomic pathways [
5]. Among these systems, the vagus nerve plays a central role due to its dual function in integrating visceral sensory information and mediating parasympathetic efferent output to the lungs [
6,
7]. Functional interactions of vagal pathways within brainstem nuclei are therefore essential for the control of airway tone and reflex bronchoconstriction [
8].
Within the brainstem, the dorsal motor nucleus of the vagus (DMV) represents a key site of autonomic integration [
9]. Located in the medulla oblongata, the DMV contains parasympathetic preganglionic neurons that provide vagal innervation to thoracic and abdominal organs [
10,
11]. It receives afferent sensory input primarily from the nucleus tractus solitarius and projects efferent fibers influencing intrathoracic airway function, thereby playing an important role in the modulation of respiratory activity [
12,
13]. Accumulating evidence indicates that the DMV functions not merely as a relay nucleus, but as an active modulatory center in which multiple neurotransmitter systems interact to regulate airway mechanics [
14]. Dysregulation within this nucleus has been associated with exaggerated airway reflexes in experimental models [
15].
Gamma-aminobutyric acid (GABA), the principal inhibitory neurotransmitter in the central nervous system, plays a critical role in regulating neuronal excitability within the DMV [
16]. Through activation of ionotropic GABAA_AA and metabotropic GABAB_BB receptors, GABA hyperpolarizes neurons, suppresses action potential firing, and modulates synaptic transmission [
17]. Experimental studies indicate that GABAergic modulation within brainstem autonomic nuclei can attenuate excessive reflex responses associated with airway constriction, suggesting an inhibitory influence on vagal efferent output [
18].
In contrast, cholinergic neurotransmission represents a dominant excitatory influence on airway tone [
21]. Acetylcholine released from parasympathetic nerve terminals induces bronchoconstriction via activation of muscarinic receptors on airway smooth muscle [
22]. Pharmacological manipulation of this pathway provides a well-established experimental approach for probing airway physiology [
23]. Physostigmine, a reversible acetylcholinesterase inhibitor, increases synaptic acetylcholine by preventing its enzymatic degradation and reliably evokes bronchoconstriction and increases airway resistance in several animal models, including ferrets [
6]. These properties make physostigmine a useful experimental tool for examining inhibitory counter-regulatory mechanisms within central autonomic circuits [
24].
Interactions between cholinergic and GABAergic signaling within the DMV are of particular interest, as they reflect the balance between excitatory drive and inhibitory control in a nucleus that directly governs vagal efferent output [
25]. Although the DMV contains relatively few intrinsic interneurons, it receives dense GABAergic input from the nucleus tractus solitarius [
26]. While exaggerated airway reflexes following cholinergic stimulation have been well documented, the extent to which localized inhibitory signaling within the DMV modulates these responses remains incompletely defined [
27]. In particular, the effects of targeted GABA microinjection into the DMV on specific physiological parameters such as pressure in the tracheal segment (Ptseg), lung resistance (RL), and dynamic compliance (Cdyn) have not been systematically quantified [
14,
28].
Accordingly, the present study was designed to examine the effects of bilateral GABA microinjection into the DMV on physostigmine-induced airway constriction in ferrets. Using physostigmine-evoked responses as a reproducible cholinergic challenge, the GABA intervention was assessed specifically in this context. By integrating stereotaxic microinjection with physiological measurements of Ptseg, RL, and Cdyn, this study aims to characterize the role of inhibitory neurotransmission within the DMV in modulating cholinergically driven airway responses.
Materials and Methods
Animal and Ethical Approval
Experiments were performed on adult European ferrets (Mustela putorius furo; n = 6), weighing 700–900 g. All experimental procedures were approved by the Institutional Animal Care and Use Committee and conducted in accordance with the Guide for the Care and Use of Laboratory Animals (National Research Council). Only male animals were used to minimize variability related to hormonal fluctuations. Ferrets were selected because they represent a well-established model for studying respiratory neurophysiology and airway control. Animals were obtained from Marshall Farms (North Rose, NY, USA) and housed at the University Animal Resource Center under controlled environmental conditions, with free access to food and water.
Surgical Preparation and Anesthesia
Non-survival experiments were conducted using a within-subject repeated-measures design. General anesthesia was induced and maintained with α-chloralose (70 mg/kg, i.p.), with supplemental doses (5 mg/kg, i.v.) administered as needed. Adequate depth of anesthesia was confirmed by the absence of motor or cardiovascular responses to noxious stimulation.
Polyethylene catheters were placed in the common carotid artery for arterial pressure monitoring and in the jugular vein for intravenous drug administration. A catheter inserted into the femoral artery was used to obtain arterial blood samples (0.2 mL) for blood gas analysis using an ABL™5/BPH™5 analyzer (Radiometer Medical A/S, Copenhagen, Denmark).
Ventilation and Physiological Monitoring
A tracheal window was created in the cervical trachea, and a tracheostomy tube was inserted into the caudal trachea and connected to a mechanical ventilator (Rodent Ventilator Model 683, Harvard Apparatus). Animals were ventilated with a tidal volume of 8 mL/kg at a respiratory rate of 55–60 breaths/min. Ventilator frequency was adjusted to maintain arterial PaCO2 between 32 and 35 mmHg. The rostral portion of the tracheal window was left open to permit subsequent instrumentation.
Neuromuscular blockade was achieved with gallamine triethiodide (4 mg/kg, i.v.; Sigma-Aldrich, Cat. No. G8134), with additional doses administered hourly. To suppress peripheral chemoreceptor drive and minimize baseline cholinergic tone, animals were ventilated with hyperoxic gas (100% O2), as previously described (Neziri et al., 2017). Body temperature was monitored continuously using an esophageal probe and maintained at 38–39 °C with a thermostatically controlled heating pad.
Experimental Design and Pharmacological Interventions
Each animal served as its own control. After establishing a stable basal tracheal tone of approximately 7–9 cm H2O, experiments were conducted in a fixed sequence of three conditions, separated by washout periods of at least 30 min:
baseline following intravenous saline (0.2 mL),
cholinergic stimulation induced by physostigmine (100 µg/kg body weight, i.v.; Sigma-Aldrich, Cat. No. P1911) infused over 1 min, and
bilateral microinjection of GABA into the dorsal motor nucleus of the vagus (DMV).
During surgical exposure of the carotid sheath, care was taken to preserve the integrity of the vagal nerve and its blood supply. Functional integrity of vagal cholinergic outflow was confirmed by comparing tracheal smooth muscle responses to lung deflation and hyperoxic hypocapnia before and after surgical preparation.
Stereotaxic Microinjection and Histological Verification
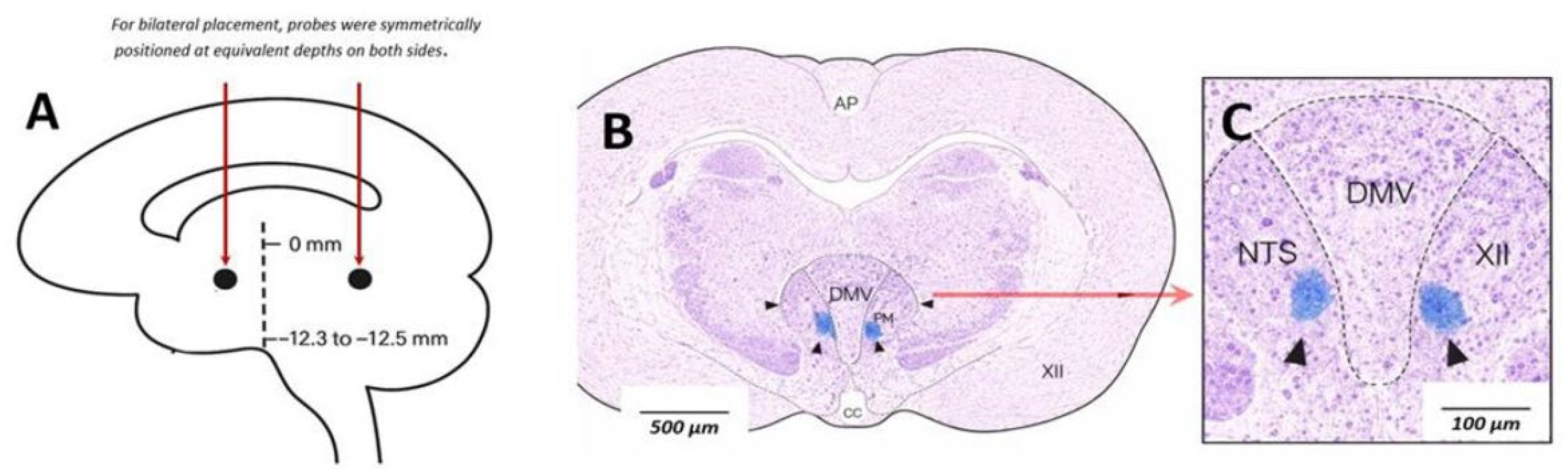
Animals were positioned prone in a stereotaxic frame (Model 900LS; David Kopf Instruments). After skull exposure, a small burr hole (~1 mm) was drilled above the target region, and the dura was carefully opened. Bilateral stereotaxic microinjections of GABA (10 mM; 50–70 nL per side; Sigma-Aldrich, Cat. No. A2129) were targeted to the DMV using established coordinates (anteroposterior: −12.3 to −12.5 mm from bregma; mediolateral: 0 mm; dorsoventral: −7 to −9 mm from the brain surface).
Injection sites were verified post hoc by Pontamine Sky Blue dye deposition and histological examination of coronal brainstem sections. Animals were transcardially perfused with phosphate-buffered saline followed by 4% paraformaldehyde. Brains were removed, post-fixed, and sectioned coronally (100 µm) for microscopic confirmation of bilateral DMV localization (
Figure 1, Panel B and C).
Measurement of Lung Mechanics
A whole-body plethysmograph was sealed using latex collars and coupling gel. Airflow was measured with a pneumotachograph connected to a differential pressure transducer (±1.4 cm H2O; DP250, Validyne Engineering). Inflation pressure was recorded via a second transducer (TRD 5700, Buxco Electronics). The plethysmograph was calibrated by injecting and withdrawing 10 mL of air, and flow calibration was verified using a precision flow meter.
Signals were acquired using an ACQ-7700 interface and Universal XE signal conditioner (Buxco Electronics). Tidal volume and lung mechanics were calculated by integrating airflow and pressure signals using Biosystem XA software (version 2.7 r2, Buxco Electronics). Lung resistance (RL) and dynamic compliance (Cdyn) were computed from these signals.
Measurement of Tracheal Pressure
A balloon-tipped catheter (3 cm length) was inserted into the rostral tracheal segment and inflated with 1.0–1.2 mL saline. Tracheal pressure (Ptseg) was measured using a calibrated pressure transducer (0–45 cm H2O). Basal tracheal tone was established by inducing hyperoxic hypocapnia through gradual increases in ventilatory rate, as previously described (Haxhiu et al., 2003). A basal pressure of 8–10 cm H2O was used as the reference level for all experiments.
Research Protocol
RL, Cdyn, and Ptseg were assessed under baseline conditions and during reflex activation induced by lung deflation and transient asphyxia (30–45 s interruption of ventilation). Following baseline measurements, the experimental protocol proceeded with physostigmine administration and subsequent GABA microinjection, as outlined above. Reflex responses were reproducible before each intervention.
Data Analysis
Five consecutive respiratory cycles were analyzed during control conditions and at the peak of cholinergic responses. Data are presented as mean ± SEM, with individual animal values shown and connected to reflect within-subject comparisons. Statistical analysis was performed using one-way repeated-measures ANOVA followed by Tukey’s post hoc test. Statistical significance was set at P < 0.05.
Results
Localization of Microinjection Sites
Bilateral microinjections of GABA were localized within the dorsal motor nucleus of the vagus (DMV), as illustrated in
Figure 1A. Stereotaxic coordinates of the injection sites were anteroposterior −12.6 mm, mediolateral approximately 0 mm, and dorsoventral −7.6 mm relative to bregma. Blood pressure and arterial blood gas variables were monitored throughout the experiments to ensure physiological stability but were not analyzed as primary outcome measures.
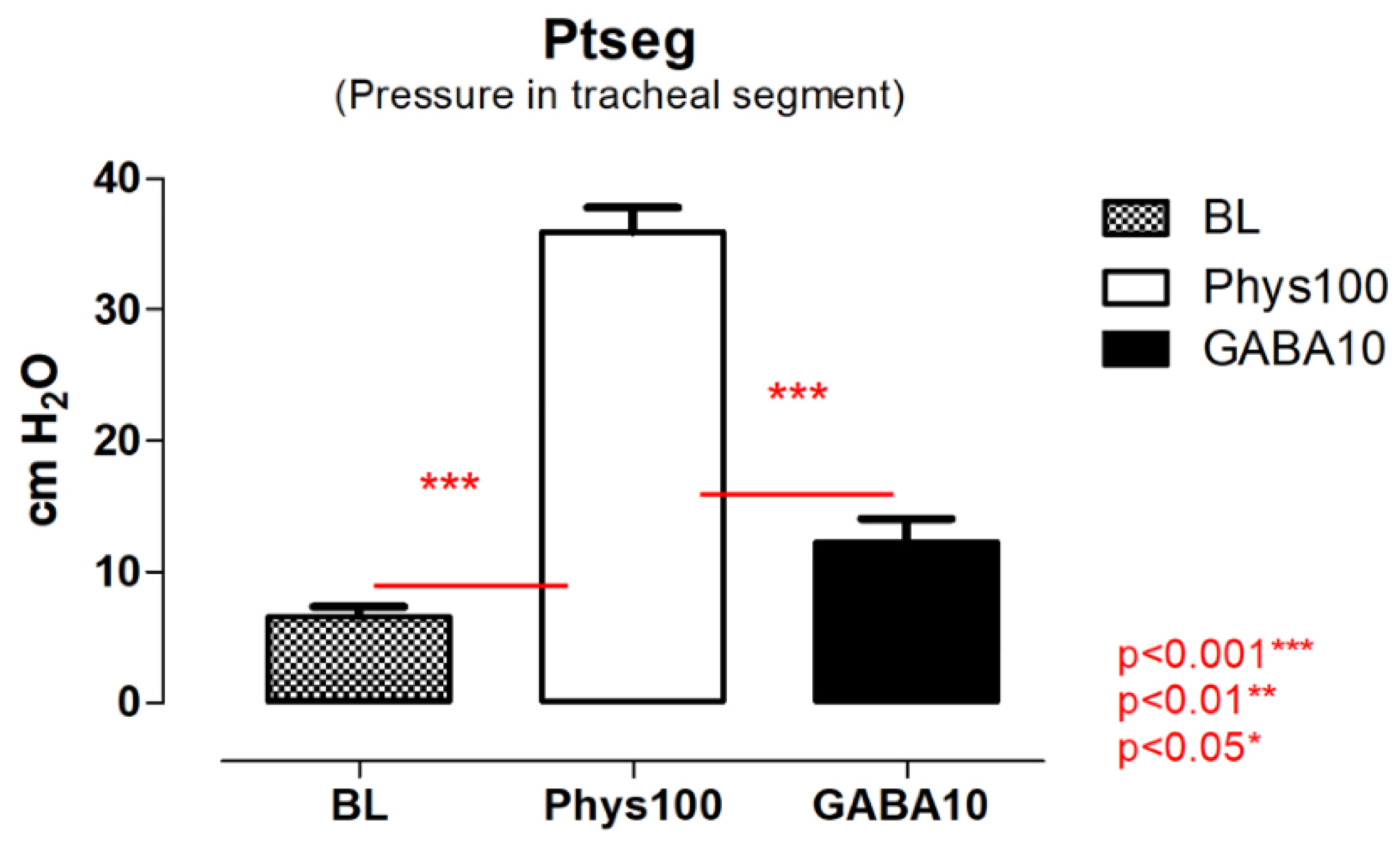
Effects of Physostigmine and GABA on Pressure in the Tracheal Segment (Ptseg)
Intravenous administration of physostigmine (100 µg/kg body weight) produced a marked increase in Ptseg compared with baseline values (
Figure 2). Peak Ptseg values reached 35.94 cm H
2O. Following bilateral microinjection of GABA into the DMV, Ptseg values decreased, with mean values of 12.28 cm H
2O.
Quantitative analysis demonstrated that physostigmine significantly increased Ptseg relative to baseline (P < 0.001). GABA microinjection did not significantly differ from baseline values. A significant difference was observed between physostigmine and GABA conditions (P < 0.001, one-way repeated-measures ANOVA with Tukey’s post hoc test).
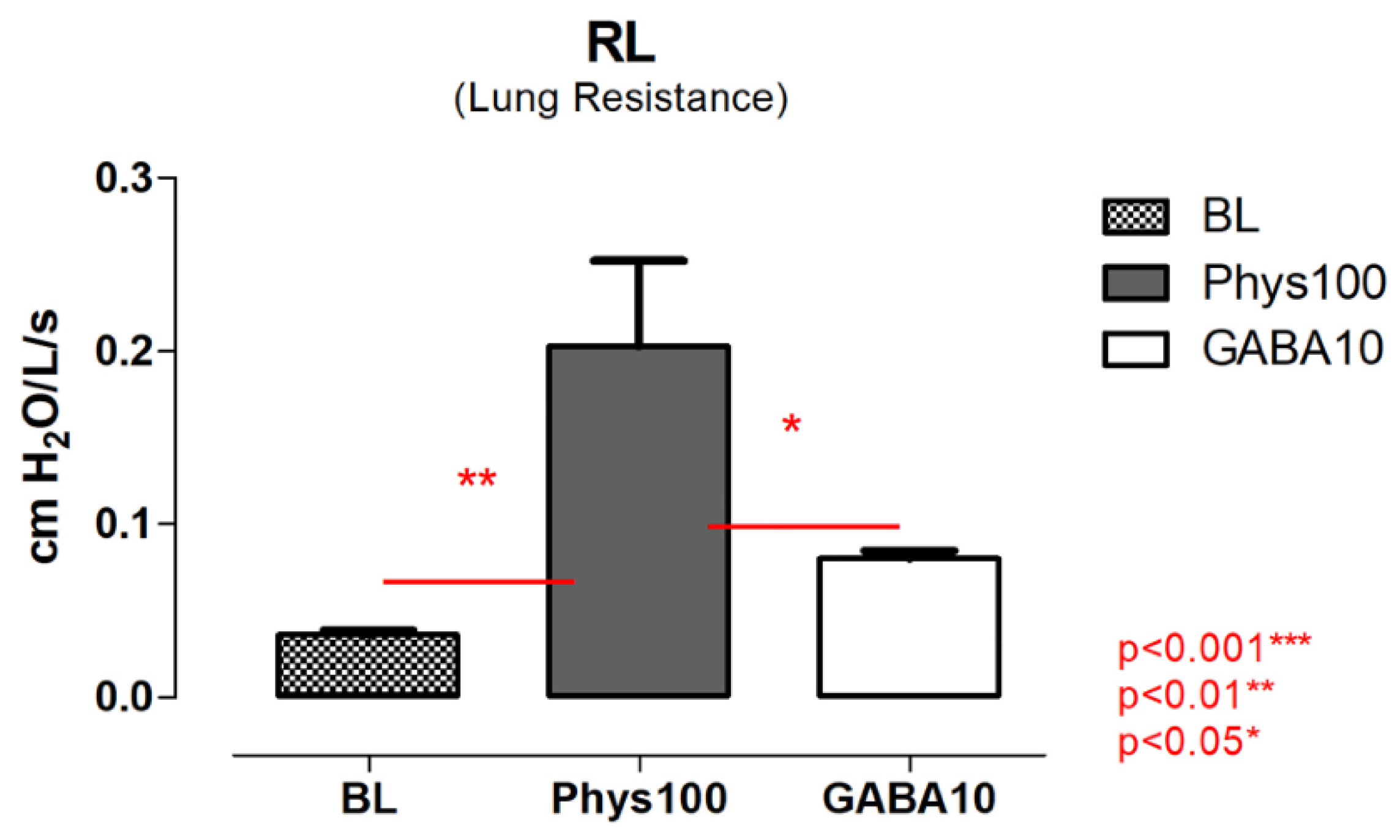
Effects on Lung Resistance (RL)
Changes in lung resistance are shown in
Figure 3. Physostigmine administration resulted in a significant increase in RL compared with baseline (
P < 0.01). Bilateral GABA microinjection into the DMV did not significantly alter RL relative to baseline. Comparison between physostigmine and GABA conditions revealed a significant difference (
P < 0.05).
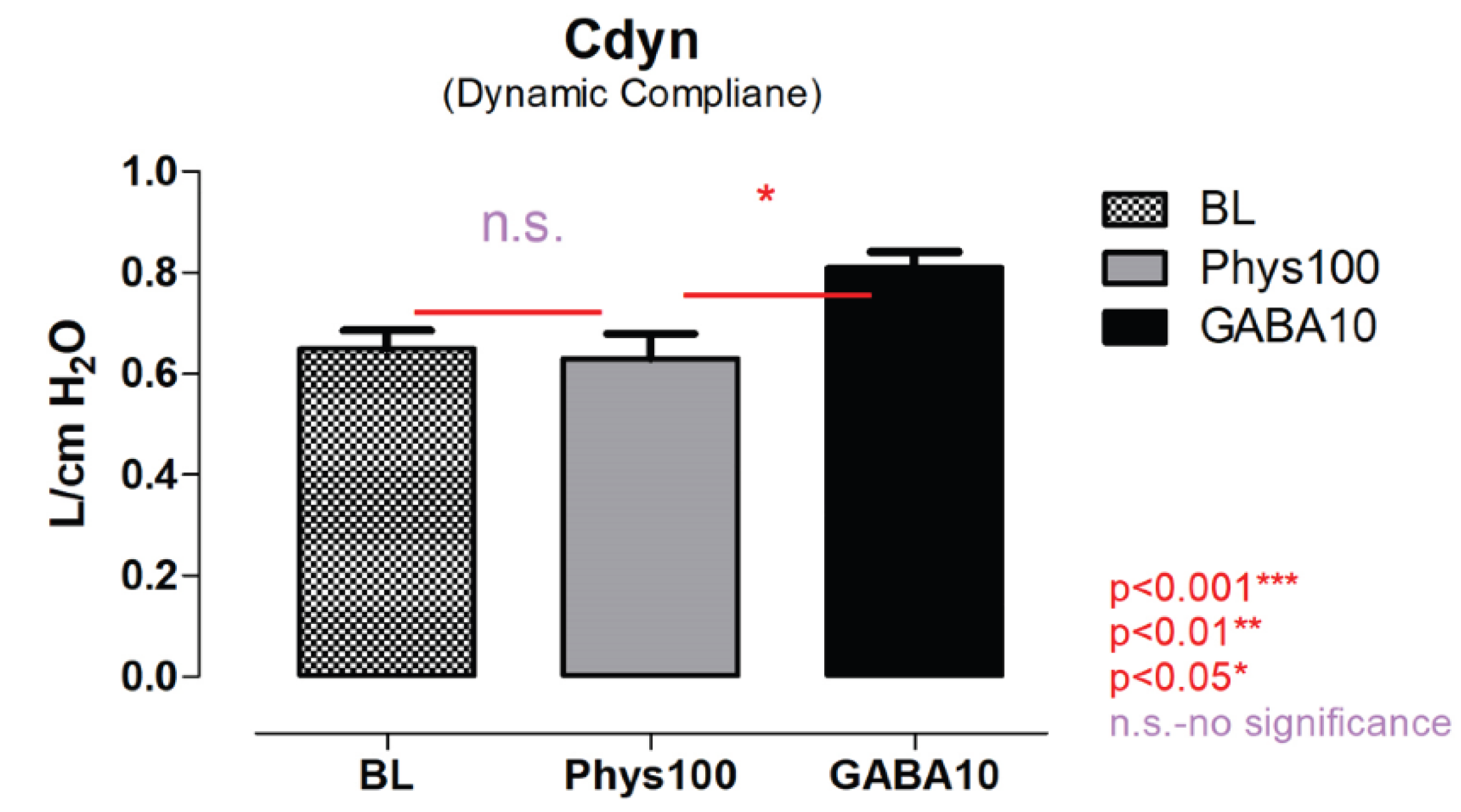
Effects on Dynamic Compliance (Cdyn)
Dynamic compliance values are summarized in
Figure 4. Physostigmine administration did not significantly change Cdyn compared with baseline. In contrast, bilateral GABA microinjection into the DMV was associated with a significant reduction in Cdyn relative to baseline (
P < 0.05). A significant difference was also observed between physostigmine and GABA conditions (
P < 0.05).
Summary of Results
Across all measured parameters, physostigmine and GABA microinjection produced distinct effects on Ptseg, RL, and Cdyn. Physostigmine significantly increased Ptseg and RL, whereas GABA microinjection was associated with lower Ptseg values and altered Cdyn. Statistical comparisons consistently demonstrated significant differences between physostigmine and GABA conditions.
Discussion
The present study examined the effects of targeted GABA microinjection into the dorsal motor nucleus of the vagus (DMV) on airway responses evoked by systemic cholinergic stimulation. The principal finding is that bilateral GABA microinjection into the DMV was associated with attenuation of physostigmine-induced changes in tracheal pressure and lung mechanics, indicating that inhibitory neurotransmission within this nucleus modulates cholinergically driven airway responses. These findings support the concept that excitatory and inhibitory neurotransmitter systems interact within brainstem autonomic nuclei to shape vagal output to the airways.
GABAergic Modulation Within the DMV
GABA is the major inhibitory neurotransmitter in the central nervous system and plays an essential role in regulating neuronal excitability within autonomic circuits. In the present study, bilateral microinjection of GABA into the DMV was associated with reduced Ptseg values and altered dynamic compliance relative to the physostigmine condition. Although the present data do not allow direct assessment of neuronal firing, the observed physiological changes are consistent with reduced activity of vagal preganglionic neurons. Previous studies have demonstrated that activation of GABAa and GABAb receptors in medullary autonomic nuclei hyperpolarizes neurons and limits excitatory synaptic transmission, thereby modulating cardiorespiratory reflexes3,29. The current findings extend this framework by demonstrating that localized GABAergic intervention within the DMV influences airway-related physiological parameters during a defined cholinergic challenge.
Cholinergic Excitation and Physostigmine Response
Systemic administration of physostigmine produced marked increases in tracheal pressure and lung resistance, consistent with enhanced cholinergic signaling. Physostigmine inhibits acetylcholinesterase activity, thereby increasing synaptic acetylcholine availability and amplifying parasympathetic output30,31. The robust elevation in Ptseg observed following physostigmine administration is in agreement with previous reports describing cholinergically mediated airway constriction in the ferret and other experimental models. These observations confirm that physostigmine provides a reliable and reproducible experimental paradigm for studying centrally mediated cholinergic influences on airway mechanics.
Interaction Between Inhibitory and Excitatory Signaling
The contrasting effects of physostigmine and GABA across respiratory parameters highlight the dynamic balance between excitatory cholinergic and inhibitory GABAergic influences within the DMV32. While physostigmine enhanced airway pressure and lung resistance, GABA microinjection produced opposing changes in these measures. The lack of a significant change in lung resistance following GABA microinjection relative to baseline suggests that the primary site of action may be central modulation of vagal motor output rather than direct peripheral effects on airway smooth muscle. Together, these findings are consistent with the view that the DMV functions as an integrative center in which inhibitory inputs regulate the expression of excitatory autonomic drive to the airways.
Although the DMV contributes to parasympathetic efferent control of intrathoracic airways, it operates within a broader brainstem network that includes the nucleus ambiguus, which provides major cholinergic innervation to upper airway and laryngeal musculature and plays a prominent role in respiratory rhythm generation33. The present findings therefore likely reflect modulation within a distributed autonomic network rather than isolated effects confined exclusively to a single nucleus.
Study Limitations
Several limitations should be acknowledged. First, saline-only and GABA-only baseline microinjection controls were not included in the experimental sequence. The primary objective of the study was to assess GABAergic modulation specifically in the context of physostigmine-evoked cholinergic excitation; nevertheless, the absence of these controls limits conclusions regarding baseline effects of microinjection alone. Importantly, previous studies from our laboratory and others have demonstrated that saline microinjection into the DMV does not alter airway mechanics and that GABA application under baseline conditions produces minimal effects on resting airway tone6,34,35,36. These observations suggest that the inhibitory effects observed in the present study primarily reflect modulation of an activated cholinergic state rather than nonspecific procedural effects.
Second, the experiments were conducted under anesthesia and neuromuscular blockade, conditions that are necessary for precise physiological measurements but may influence central respiratory control. Accordingly, extrapolation of these findings to awake or freely behaving conditions should be made with caution.
Physiological Relevance
Within these constraints, the present findings contribute to a growing body of evidence indicating that inhibitory neurotransmission within brainstem autonomic nuclei plays a key role in shaping vagal output to the airways. The reciprocal interaction between cholinergic excitation and GABAergic inhibition observed here underscores the importance of central integration in maintaining stable airway function during physiological and pharmacological perturbations.
Conclusions
In summary, bilateral GABA microinjection into the DMV modulated physostigmine-evoked changes in tracheal pressure and lung mechanics in anesthetized, mechanically ventilated ferrets.
The present findings demonstrate that localized GABA delivery within the DMV modulates airway mechanical responses elicited by cholinergic activation. Because experiments were performed under neuromuscular blockade and controlled ventilation, the data reflect central autonomic modulation of airway mechanics rather than direct measurement of respiratory drive. These results extend current understanding of inhibitory signaling within brainstem vagal circuits.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org.
References
- Dempsey, J. A.; Smith, C. A. Pathophysiology of human ventilatory control. European Respiratory Journal 2014, 44, 495–512. [Google Scholar] [CrossRef]
- Kubin, L. Neural Control of the Upper Airway: Respiratory and State-Dependent Mechanisms. Compr. Physiol. 2016, 6, 1801–1850. [Google Scholar] [CrossRef]
- Canning, B. J.; Fischer, A. Neural regulation of airway smooth muscle tone. Respir. Physiol. 2001, 125, 113–127. [Google Scholar] [CrossRef]
- Lam, M.; Lamanna, E.; Bourke, J. E. Regulation of Airway Smooth Muscle Contraction in Health and Disease. Adv. Exp. Med. Biol. 2019, 1124, 381–422. [Google Scholar] [PubMed]
- Haxhiu, M. A. Brain stem excitatory and inhibitory signaling pathways regulating bronchoconstrictive responses. J. Appl. Physiol. 2005, 98, 1961–1982. [Google Scholar] [CrossRef]
- Neziri, B.; Daci, A.; Krasniqi, S.; Sopi, R.; Haxhiu, M. A. The impact of bilateral vagotomy on the physostigmine-induced airway constriction in ferrets. Respir. Physiol. Neurobiol. 2017, 242, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Ottaviani, M. M.; Macefield, V. G. Structure and Functions of the Vagus Nerve in Mammals. Compr. Physiol. 2022, 12, 3989–4037. [Google Scholar] [CrossRef] [PubMed]
- Bautista, T. G.; Pitts, T. E.; Pilowsky, P. M.; Morris, K. F. The Brainstem Respiratory Network. Neuronal Networks in Brain Function, CNS Disorders, and Therapeutics 2014, 235–245. [Google Scholar] [CrossRef]
- Chen, V.; Wang, Y.; Boychuk, C. Anatomical Distribution of Cardiac Vagal Motor Neurons in Dorsal Motor Nucleus of the Vagus. Physiology 2025, 40. [Google Scholar] [CrossRef]
- Ottaviani, M.; Physiology, V.M.-C. Structure and functions of the vagus nerve in mammals. Wiley Online Library 2022, 12, 3989–4037. [Google Scholar]
- Psychol, J. K.-Biol. The multibranched nerve: vagal function beyond heart rate variability. researchgate.net. Available online: https://www.researchgate.net/profile/John-Karemaker/publication/361155437_The_multibranched_nerve_vagal_function_beyond_heart_rate_variability/links/62bca00f60e77b7db83d29eb/The-multibranched-nerve-vagal-function-beyond-heart-rate-variability.pdf.
- Champagnat, J.; et al. Information processing at the nucleus tractus solitarii and respiratory rhythm generation. Respiratory Research 2001, 2, 1–2. [Google Scholar] [CrossRef]
- Miyazaki, M.; Tanaka, I.; Ezure, K. Excitatory and inhibitory synaptic inputs shape the discharge pattern of pump neurons of the nucleus tractus solitarii in the rat. Exp. Brain Res. 1999, 129, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Kc, P.; Martin, R. J. Role of central neurotransmission and chemoreception on airway control. Respir. Physiol. Neurobiol. 2010, 173, 213–222. [Google Scholar] [CrossRef]
- Brusasco, V.; Pellegrino, R. Invited Review: Complexity of factors modulating airway narrowing in vivo: relevance to assessment of airway hyperresponsiveness. Journal of Applied Physiology 2003, 95, 1305–1313. [Google Scholar] [CrossRef]
- Travagli, R. A.; Gillis, R. A.; Rossiter, C. D.; Vicini, S. Glutamate and GABA-mediated synaptic currents in neurons of the rat dorsal motor nucleus of the vagus. American Journal of Physiology-Gastrointestinal and Liver Physiology 1991, 260. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; et al. Pharmacology of GABA and Its Receptors. Frontiers in Pharmacology of Neurotransmitters 2020, 241–292. [Google Scholar] [CrossRef]
- Guyenet, P. G. Regulation of Breathing and Autonomic Outflows by Chemoreceptors. Compr. Physiol. 2014, 4, 1511–1562. [Google Scholar] [CrossRef]
- Tang, X.; Jaenisch, R.; Sur, M. The role of GABAergic signalling in neurodevelopmental disorders. Nat. Rev. Neurosci. 2021, 22, 290–307. [Google Scholar] [CrossRef]
- Mazzone, S. B.; Canning, B. J. Central nervous system control of the airways: pharmacological implications. Curr. Opin. Pharmacol. 2002, 2, 220–228. [Google Scholar] [CrossRef]
- Racké, K.; Matthiesen, S. The airway cholinergic system: physiology and pharmacology. Pulm. Pharmacol. Ther. 2004, 17, 181–198. [Google Scholar] [CrossRef]
- Gosens, R.; Zaagsma, J.; Grootte Bromhaar, M.; Nelemans, A.; Meurs, H. Acetylcholine: a novel regulator of airway smooth muscle remodelling? Eur. J. Pharmacol. 2004, 500, 193–201. [Google Scholar] [CrossRef]
- Nichols, J. E.; et al. Modeling the lung: Design and development of tissue engineered macro- and micro-physiologic lung models for research use. Exp. Biol. Med. 2014, 239, 1135–1169. [Google Scholar] [CrossRef]
- Falk, S.; Lund, C.; Clemmensen, C. Muscarinic receptors in energy homeostasis: Physiology and pharmacology. Basic Clin. Pharmacol. Toxicol. 2020, 126, 66–76. [Google Scholar] [CrossRef]
- McMenamin, C. A.; Travagli, R. A.; Browning, K. N. Inhibitory neurotransmission regulates vagal efferent activity and gastric motility. Exp. Biol. Med. 2016, 241, 1343–1350. [Google Scholar] [CrossRef] [PubMed]
- Fritschy, J. M.; Panzanelli, P. GABAA receptors and plasticity of inhibitory neurotransmission in the central nervous system. European Journal of Neuroscience 2014, 39, 1845–1865. [Google Scholar] [CrossRef] [PubMed]
- Dehkordi, O.; Kc, P.; Balan, K. V.; Haxhiu, M. A. Airway-related vagal preganglionic neurons express multiple nicotinic acetylcholine receptor subunits. Autonomic Neuroscience 2006, 128, 53–63. [Google Scholar] [CrossRef]
- The Rat Brain in Stereotaxic Coordinates - The New Coronal Set - George Paxinos, Charles Watson - Google Books. Available online: https://books.google.com/books?hl=en&lr=&id=xzNyLkQ7-q0C&oi=fnd&pg=PP1&dq=paxinos+and+watson+1982&ots=qw3rxGbldS&sig=mmkvMQso7-Ka7Oum8sNPYx8IT8c#v=onepage&q=paxinos%20and%20watson%201982&f=false.
- Zoccal, D. B.; Furuya, W. I.; Bassi, M.; Colombari, D. S. A.; Colombari, E. The nucleus of the solitary tract and the coordination of respiratory and sympathetic activities. Front. Physiol. 2014, 5, 97313. [Google Scholar] [CrossRef]
- Kim, W. S.; et al. Protection by a transdermal patch containing physostigmine and procyclidine of soman poisoning in dogs. Eur. J. Pharmacol. 2005, 525, 135–142. [Google Scholar] [CrossRef]
- Arens, A. M.; Kearney, T. Adverse Effects of Physostigmine. Journal of Medical Toxicology 2019, 15, 184. [Google Scholar] [CrossRef] [PubMed]
- Zarrindast, M. R.; Lahiji, P.; Shafaghi, B.; Sadegh, M. Effects of GABAergic drugs on physostigmine-induced improvement in memory acquisition of passive avoidance learning in mice. Gen. Pharmacol. 1998, 31, 81–86. [Google Scholar] [CrossRef]
- González-García, M.; et al. Central Autonomic Mechanisms Involved in the Control of Laryngeal Activity and Vocalization. Biology 2024, 13. [Google Scholar] [CrossRef] [PubMed]
- Haxhiu, M. A.; et al. Brain stem excitatory and inhibitory signaling pathways regulating bronchoconstrictive responses. J Appl Physiol 1961, 98, 1961–1982. [Google Scholar] [CrossRef]
- Chen, Z.; et al. Dorsal vagal complex modulates neurogenic airway inflammation in a Guinea Pig model with esophageal perfusion of HCl. Front. Physiol. 2018, 9, 326757. [Google Scholar] [CrossRef] [PubMed]
- Moore, C. T.; et al. A GABAergic inhibitory microcircuit controlling cholinergic outflow to the airways. J. Appl. Physiol. 2004, 96, 260–270. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |