Submitted:
16 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
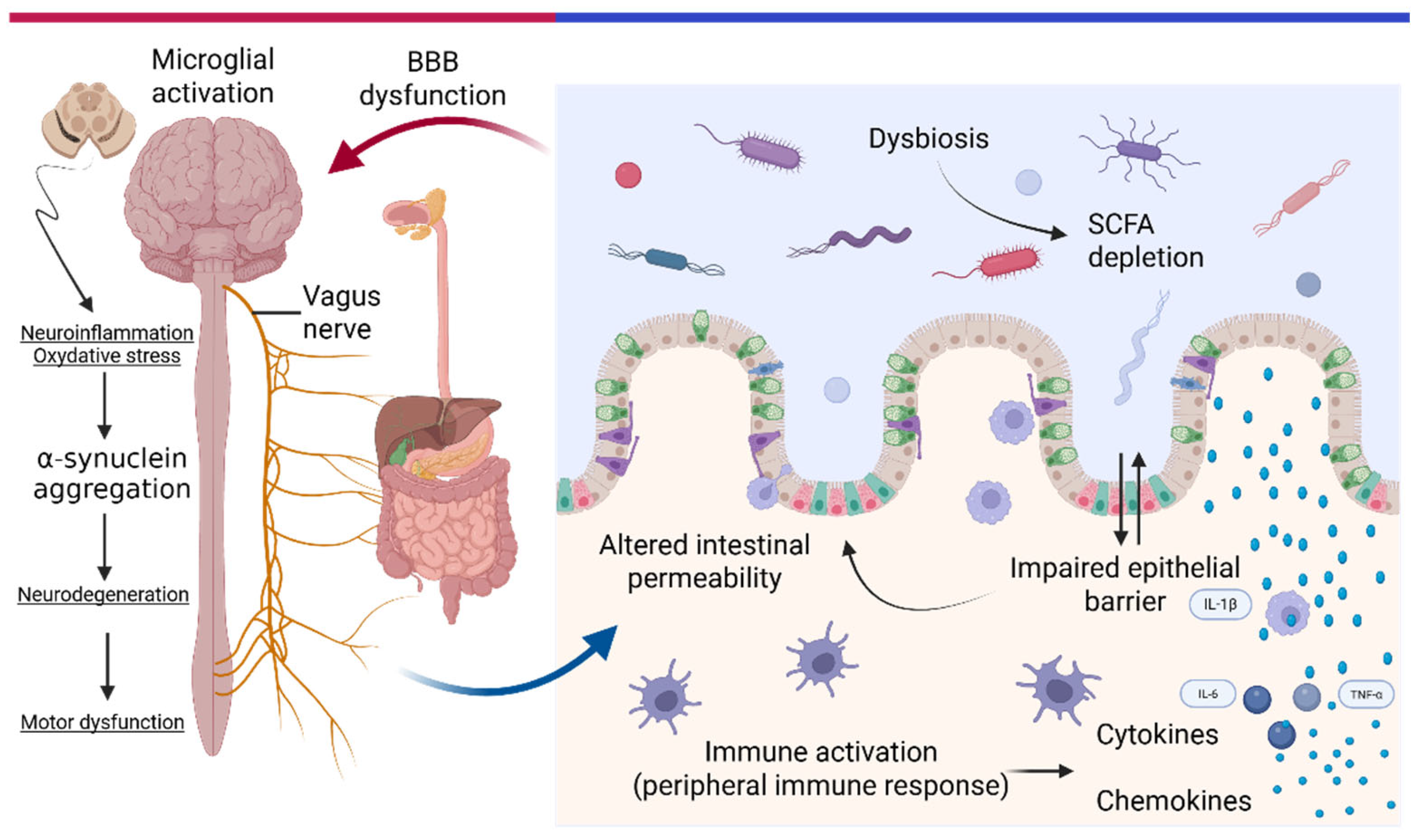
1. Introduction
2. Materials and Methods
2.1. Review Design and Literature Search Strategy
2.2. Data Categorization and Thematic Integration
3. Results
3.1. Reproducible Taxonomic Signatures
3.2. Loss of Microbial Functional Diversity
3.3. Microbial Metabolites and Dopaminergic Vulnerability
3.3.1. Short-Chain Fatty Acids (SCFAs)
3.3.2. Branched-Chain Amino Acids (BCAAs)
3.3.3. Gamma-Aminobutyric Acid (GABA)
3.3.4. Xenobiotic and Environmental Pathways
3.4. Intestinal Barrier Dysfunction and Immune Activation
3.5. α-Synuclein Propagation and Basal Ganglia Dysfunction
3.6. Therapeutic Implications
3.6.1. Diet-Based Interventions
3.6.2. Probiotics and Prebiotics
3.6.3. Fecal Microbiota Transplantation (FMT)
3.6.4. Pharmacomicromodulation
4. Limitations and Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bai, H.; Ma, W.; Zhu, L.; Lu, Y.; Fan, J.; Chen, M.; Huang, C. Updates on Parkinson’s Disease. Neuropsychiatr. Dis. Treat. 2025, Volume 21, 1945–1953. [Google Scholar] [CrossRef]
- Cannon, T.; Gruenheid, S. Microbes and Parkinson’s Disease: From Associations to Mechanisms. Trends Microbiol. 2022, 30, 749–760. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.; Abbas, K.; Mustafa, M.; Usmani, N.; Habib, S. Microbiome-Based Therapies for Parkinson’s Disease. Front. Nutr. 2024, 11, 1496616. [Google Scholar] [CrossRef]
- Benvenuti, L.; Di Salvo, C.; Bellini, G.; Seguella, L.; Rettura, F.; Esposito, G.; Antonioli, L.; Ceravolo, R.; Bernardini, N.; Pellegrini, C.; et al. Gut-Directed Therapy in Parkinson’s Disease. Front. Pharmacol. 2024, 15, 1407925. [Google Scholar] [CrossRef] [PubMed]
- Iordache, M.P.; Buliman, A.; Costea-Firan, C.; Gligore, T.C.I.; Cazacu, I.S.; Stoian, M.; Teoibaș-Şerban, D.; Blendea, C.-D.; Protosevici, M.G.-I.; Tanase, C.; et al. Immunological and Inflammatory Biomarkers in the Prognosis, Prevention, and Treatment of Ischemic Stroke: A Review of a Decade of Advancement. Int. J. Mol. Sci. 2025, 26, 7928. [Google Scholar] [CrossRef] [PubMed]
- Ai, P.; Xu, S.; Yuan, Y.; Xu, Z.; He, X.; Mo, C.; Zhang, Y.; Yang, X.; Xiao, Q. Targeted Gut Microbiota Modulation Enhances Levodopa Bioavailability and Motor Recovery in MPTP Parkinson’s Disease Models. Int. J. Mol. Sci. 2025, 26, 5282. [Google Scholar] [CrossRef]
- Alfonsetti, M.; Castelli, V.; d’Angelo, M. Are We What We Eat? Impact of Diet on the Gut–Brain Axis in Parkinson’s Disease. Nutrients 2022, 14, 380. [Google Scholar] [CrossRef]
- Beisembayeva, M.; Shayakhmetova, Y.; Muratbekova, S.; Li, I.; Yakupov, E.; Kaiyrzhanov, R.; Grigolashvili, M. A Comparative Analysis of the Gut Microbiota in Patients With Parkinson’s Disease Worldwide: A Systematic Review. Int. J. Clin. Pract. 2025, 2025, 5511146. [Google Scholar] [CrossRef]
- Camacho, M.; Greenland, J.C.; Daruwalla, C.; Scott, K.M.; Patel, B.; Apostolopoulos, D.; Ribeiro, J.; O’Reilly, M.; Hu, M.T.; Williams-Gray, C.H. The Profile of Gastrointestinal Dysfunction in Prodromal to Late-Stage Parkinson’s Disease. Npj Park. Dis. 2025, 11, 123. [Google Scholar] [CrossRef]
- Lee, S.; Kim, J.; Baek, J.W.; Jung, K.-Y.; Lee, Y.; Koh, A.; Kim, H.-J. Enrichment of Gut-Derived Metabolites in a Parkinson’s Disease Subtype with REM Sleep Behavior Disorder. Npj Park. Dis. 2025, 11, 189. [Google Scholar] [CrossRef]
- Wang, J.-Y.; Xie, R.; Feng, Y.; Zhang, M.-N.; He, L.; Yang, B.; Wang, H.-G.; Yang, X.-Z. Gut Microbiota Helps Identify Clinical Subtypes of Parkinson’s Disease. Mil. Med. Res. 2024, 11, 42. [Google Scholar] [CrossRef]
- Abou Izzeddine, N.; Ahmad, K.; Bacha, C.; Jabbour, M.; Najjar, M.; Salhab, S.; Ghadieh, H.E.; Kanaan, A.; Azar, S.; Khattar, Z.A.; et al. The Microbial Guardians: Unveiling the Role of Gut Microbiota in Shaping Neurodegenerative Disease. IBRO Neurosci. Rep. 2025, 19, 17–37. [Google Scholar] [CrossRef]
- Agostini, D.; Bartolacci, A.; Rotondo, R.; De Pandis, M.F.; Battistelli, M.; Micucci, M.; Potenza, L.; Polidori, E.; Ferrini, F.; Sisti, D.; et al. Homocysteine, Nutrition, and Gut Microbiota: A Comprehensive Review of Current Evidence and Insights. Nutrients 2025, 17, 1325. [Google Scholar] [CrossRef] [PubMed]
- Lima, I.S.; Pêgo, A.C.; Martins, A.C.; Prada, A.R.; Barros, J.T.; Martins, G.; Gozzelino, R. Gut Dysbiosis: A Target for Protective Interventions against Parkinson’s Disease. Microorganisms 2023, 11, 880. [Google Scholar] [CrossRef]
- Pfaffinger, J.M.; Hays, K.E.; Seeley, J.; Ramesh Babu, P.; Ryznar, R. Gut Dysbiosis as a Potential Driver of Parkinson’s and Alzheimer’s Disease Pathogenesis. Front. Neurosci. 2025, 19, 1600148. [Google Scholar] [CrossRef]
- Ayten, Ş.; Bilici, S. Modulation of Gut Microbiota Through Dietary Intervention in Neuroinflammation and Alzheimer’s and Parkinson’s Diseases. Curr. Nutr. Rep. 2024, 13, 82–96. [Google Scholar] [CrossRef]
- Nishiwaki, H.; Ueyama, J.; Ito, M.; Hamaguchi, T.; Takimoto, K.; Maeda, T.; Kashihara, K.; Tsuboi, Y.; Mori, H.; Kurokawa, K.; et al. Meta-Analysis of Shotgun Sequencing of Gut Microbiota in Parkinson’s Disease. Npj Park. Dis. 2024, 10, 106. [Google Scholar] [CrossRef]
- Shalash, A.; Ezzeldin, S.; Hashish, S.; Salah, Y.; Dawood, N.L.; Moustafa, A.; Salama, M. Gut Microbial Shifts toward Inflammation in Parkinson’s Disease: Insights from Pilot Shotgun Metagenomics Egyptian Cohort. J. Park. Dis. 2025, 15, 1540–1543. [Google Scholar] [CrossRef]
- Nakhal, M.M.; Yassin, L.K.; Alyaqoubi, R.; Saeed, S.; Alderei, A.; Alhammadi, A.; Alshehhi, M.; Almehairbi, A.; Al Houqani, S.; BaniYas, S.; et al. The Microbiota–Gut–Brain Axis and Neurological Disorders: A Comprehensive Review. Life 2024, 14, 1234. [Google Scholar] [CrossRef]
- Bedarf, J.R.; Romano, S.; Heinzmann, S.S.; Duncan, A.; Traka, M.H.; Ng, D.; Segovia-Lizano, D.; Simon, M.-C.; Narbad, A.; Wüllner, U.; et al. A Prebiotic Dietary Pilot Intervention Restores Faecal Metabolites and May Be Neuroprotective in Parkinson’s Disease. Npj Park. Dis. 2025, 11, 66. [Google Scholar] [CrossRef]
- Department of Nutrition and Dietetics, Faculty of Health Sciences, Üsküdar University, Istanbul, Turkey.; Atak, E.S.; Yıldız, D.; Department of Nutrition and Dietetics, Faculty of Health Sciences, Üsküdar University, Istanbul, Turkey.; Kocatürk, R.R.; Department of Nutrition and Dietetics, Faculty of Health Sciences, Üsküdar University, Istanbul, Turkey.; Temizyürek, A.; Department of Physiology, School of Medicine, Koç University, Istanbul, Turkey.; Özcan, Ö.Ö.; Department of Molecular Neuroscience, Health Sciences Institute, Üsküdar University, Istanbul, Turkey.; et al. Therapeutic Targets of Probiotics in Parkinson Disease: A Systematic Review of Randomized Controlled Trials. Basic Clin. Neurosci. J. 2024, 15, 165–174. [CrossRef]
- Xiromerisiou, G.; Marogianni, C.; Androutsopoulou, A.; Ntavaroukas, P.; Mysiris, D.; Papoutsopoulou, S. Parkinson’s Disease, It Takes Guts: The Correlation between Intestinal Microbiome and Cytokine Network with Neurodegeneration. Biology 2023, 12, 93. [Google Scholar] [CrossRef] [PubMed]
- Romano, S.; Wirbel, J.; Ansorge, R.; Schudoma, C.; Ducarmon, Q.R.; Narbad, A.; Zeller, G. Machine Learning-Based Meta-Analysis Reveals Gut Microbiome Alterations Associated with Parkinson’s Disease. Nat. Commun. 2025, 16, 4227. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, S.A.; Chaudhary, S.; Rawat, S. Understanding Parkinson’s Disease: Current Trends and Its Multifaceted Complications. Front. Aging Neurosci. 2025, 17, 1617106. [Google Scholar] [CrossRef]
- Choe, U. Role of Dietary Fiber and Short-Chain Fatty Acids in Preventing Neurodegenerative Diseases through the Gut-Brain Axis. J. Funct. Foods 2025, 129, 106870. [Google Scholar] [CrossRef]
- Hegelmaier, T.; Duscha, A.; Desel, C.; Fuchs, S.; Shapira, M.; Amidror, S.; Shan, Q.; Stangl, G.I.; Hirche, F.; Kempa, S.; et al. Supplementation with Short-Chain Fatty Acids and a Prebiotic Improves Clinical Outcome in Parkinson’s Disease: A Randomized Double-Blind Prospective Study. Sci. Rep. 2025, 16, 315. [Google Scholar] [CrossRef]
- Kalyanaraman, B.; Cheng, G.; Hardy, M. Gut Microbiome, Short-Chain Fatty Acids, Alpha-Synuclein, Neuroinflammation, and ROS/RNS: Relevance to Parkinson’s Disease and Therapeutic Implications. Redox Biol. 2024, 71, 103092. [Google Scholar] [CrossRef]
- Rusu, E.; Necula, L.G.; Neagu, A.I.; Alecu, M.; Stan, C.; Albulescu, R.; Tanase, C.P. Current Status of Stem Cell Therapy: Opportunities and Limitations. Turk. J. Biol. 2016, 40, 955–967. [Google Scholar] [CrossRef]
- Eslami, M.; Adampour, Z.; Fadaee Dowlat, B.; Yaghmayee, S.; Motallebi Tabaei, F.; Oksenych, V.; Naderian, R. A Novel Frontier in Gut–Brain Axis Research: The Transplantation of Fecal Microbiota in Neurodegenerative Disorders. Biomedicines 2025, 13, 915. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Gao, H.; Wang, Y.; Xiang, Y. Exploring the Role of Gut Microbiota in Parkinson’s Disease: Insights from Fecal Microbiota Transplantation. Front. Neurosci. 2025, 19, 1574512. [Google Scholar] [CrossRef]
- Nabil, Y.; Helal, M.M.; Qutob, I.A.; Dawoud, A.I.A.; Allam, S.; Haddad, R.; Manasrah, G.M.; AlEdani, E.M.; Sleibi, W.; Faris, A.; et al. Efficacy and Safety of Fecal Microbiota Transplantation in the Management of Parkinson’s Disease: A Systematic Review. BMC Neurol. 2025, 25, 291. [Google Scholar] [CrossRef]
- Denman, C.R.; Park, S.M.; Jo, J. Gut-Brain Axis: Gut Dysbiosis and Psychiatric Disorders in Alzheimer’s and Parkinson’s Disease. Front. Neurosci. 2023, 17, 1268419. [Google Scholar] [CrossRef]
- Blendea, C.-D.; Khan, M.T.; Stoian, M.; Gligore, T.C.I.; Cuculici, Ștefan; Stanciu, I.L.; Protosevici, M.G.-I.; Iordache, M.; Buliman, A.; Costea-Firan, C.; et al. Advances in Minimally Invasive Treatments for Prostate Cancer: A Review of the Role of Ultrasound Therapy and Laser Therapy. Balneo PRM Res. J. 2025, 16, 827–827. [Google Scholar] [CrossRef]
- Jin, Y.; Wang, L.; Lin, R.; He, J.; Liu, D.; Liu, Y.; Deng, Y. Synergistic Effects of Plant Polysaccharides and Probiotics: A Novel Dietary Approach for Parkinson’s Disease Intervention. Pharmaceuticals 2026, 19, 157. [Google Scholar] [CrossRef]
- Miyaue, N.; Yamamoto, H.; Liu, S.; Ito, Y.; Yamanishi, Y.; Ando, R.; Suzuki, Y.; Mogi, M.; Nagai, M. Association of Enterococcus Faecalis and Tyrosine Decarboxylase Gene Levels with Levodopa Pharmacokinetics in Parkinson’s Disease. Npj Park. Dis. 2025, 11, 49. [Google Scholar] [CrossRef]
- De Sciscio, M.; Bryant, R.V.; Haylock-Jacobs, S.; Day, A.S.; Pitchers, W.; Iansek, R.; Costello, S.P.; Kimber, T.E. Faecal Microbiota Transplant in Parkinson’s Disease: Pilot Study to Establish Safety & Tolerability. Npj Park. Dis. 2025, 11, 203. [Google Scholar] [CrossRef]
- Chui, Z.S.W.; Chan, L.M.L.; Zhang, E.W.H.; Liang, S.; Choi, E.P.H.; Lok, K.Y.W.; Tun, H.M.; Kwok, J.Y.Y. Effects of Microbiome-Based Interventions on Neurodegenerative Diseases: A Systematic Review and Meta-Analysis. Sci. Rep. 2024, 14, 9558. [Google Scholar] [CrossRef]
- Christopher, C.; Morgan, K.; Tolleson, C.; Trudell, R.; Fernandez-Romero, R.; Rice, L.; Abiodun, B.; Vickery, Z.; Jones, K.; Woodall, B.; et al. Specific Bacterial Taxa and Their Metabolite, DHPS, May Be Linked to Gut Dyshomeostasis in Patients with Alzheimer’s Disease, Parkinson’s Disease, and Amyotrophic Lateral Sclerosis. Nutrients 2025, 17, 1597. [Google Scholar] [CrossRef] [PubMed]
- Forero-Rodríguez, J.; Zimmermann, J.; Taubenheim, J.; Arias-Rodríguez, N.; Caicedo-Narvaez, J.D.; Best, L.; Mendieta, C.V.; López-Castiblanco, J.; Gómez-Muñoz, L.A.; Gonzalez-Santos, J.; et al. Changes in Bacterial Gut Composition in Parkinson’s Disease and Their Metabolic Contribution to Disease Development: A Gut Community Reconstruction Approach. Microorganisms 2024, 12, 325. [Google Scholar] [CrossRef]
- Li, Z.; Liang, H.; Hu, Y.; Lu, L.; Zheng, C.; Fan, Y.; Wu, B.; Zou, T.; Luo, X.; Zhang, X.; et al. Gut Bacterial Profiles in Parkinson’s Disease: A Systematic Review. CNS Neurosci. Ther. 2023, 29, 140–157. [Google Scholar] [CrossRef]
- Soto-Avellaneda, A.; Prigent, A.; Meyerdirk, L.; Schautz, N.; Pospisilik, J.A.; Brundin, L.; Henderson, M.X. Helicobacter Pylori Infection and α-Synuclein Pathology Drive Parallel Neurodegenerative Pathways in the Substantia Nigra. J. Neuroinflammation 2025, 22, 293. [Google Scholar] [CrossRef]
- Rust, C.; Van Den Heuvel, L.L.; Bardien, S.; Carr, J.; Pretorius, E.; Seedat, S.; Hemmings, S.M.J. Association between the Relative Abundance of Butyrate-Producing and Mucin-Degrading Taxa and Parkinson’s Disease. Neuroscience 2025, 576, 149–154. [Google Scholar] [CrossRef]
- Balsamo, J.M.; Yan, Y.; Thai, D.; Cologna, S.M.; Bess, E.N. Multiomic Analysis Reveals Molecular Pathways Associated with Intestinal Aggregation of α-Synuclein. ACS Chem. Biol. 2026, 21, 83–95. [Google Scholar] [CrossRef]
- Jia, Y.; Zhang, Y.; Tai, X.; Zhao, T.; Zhang, H.; Zhou, H. Research Progress on Natural Products in Regulating the Gut Microbiota in Parkinson’s Disease. Front. Pharmacol. 2025, 16, 1667694. [Google Scholar] [CrossRef]
- Bai, F.; You, L.; Lei, H.; Li, X. Association between Increased and Decreased Gut Microbiota Abundance and Parkinson’s Disease: A Systematic Review and Subgroup Meta-Analysis. Exp. Gerontol. 2024, 191, 112444. [Google Scholar] [CrossRef] [PubMed]
- Clasen, F.; Yildirim, S.; Arıkan, M.; Garcia-Guevara, F.; Hanoğlu, L.; Yılmaz, N.H.; Şen, A.; Celik, H.K.; Neslihan, A.A.; Demir, T.K.; et al. Microbiome Signatures of Virulence in the Oral-Gut-Brain Axis Influence Parkinson’s Disease and Cognitive Decline Pathophysiology. Gut Microbes 2025, 17, 2506843. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-X.; Sun, N.-Q.; Mo, S.-J. Rhapontin Activating Nuclear Factor Erythroid 2-Related Factor 2 to Ameliorate Parkinson’s Disease-Associated Gastrointestinal Dysfunction. World J. Gastroenterol. 2026, 32. [Google Scholar] [CrossRef]
- Barbu, L.A.; Vasile, L.; Cercelaru, L.; Șurlin, V.; Mogoantă, S.-Ștefaniță; Mogoș, G.F.R.; Țenea Cojan, T.S.; Mărgăritescu, N.-D.; Iordache, M.P.; Buliman, A. Aggressiveness in Well-Differentiated Small Intestinal Neuroendocrine Tumors: A Rare Case and Narrative Literature Review. J. Clin. Med. 2025, 14, 5821. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xue, L.; Zhang, M.; Shen, P.; Zhao, W.; Tong, Q.; Wu, S.; Dai, W.; Yang, X.; Wang, H. Colonoscopic Fecal Microbiota Transplantation for Mild-to-Moderate Parkinson’s Disease: A Randomized Controlled Trial. Brain. Behav. Immun. 2025, 130, 106086. [Google Scholar] [CrossRef]
- Xie, L.; Chen, D.; Zhu, X.; Cheng, C. Efficacy and Safety of Probiotics in Parkinson’s Constipation: A Systematic Review and Meta-Analysis. Front. Pharmacol. 2023, 13, 1007654. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Pang, D.; Shang, H. Akkermansia Muciniphila: A Double-Edged Sword in Life-Stage-Specific Nutritional Modulation of Parkinson’s Disease via the Gut-Brain Axis. Microbiol. Res. 2026, 305, 128436. [Google Scholar] [CrossRef]
- Buliman, A.; Iordache, M.P.; Protosevici, M.I.; Tanase, C. Therapeutic Strategies Targeting Anti- CD47 Therapies in Glioblastoma Multiforme: Lead or Dead End? J. Cell. Mol. Med. 2025, 29, e70889. [Google Scholar] [CrossRef]
- Albani, G.; Chellamuthu, V.R.; Morlacchi, L.; Zirone, F.; Youssefi, M.; Giardini, M.; Chao, Y.-X.; Tan, E.-K.; Albani, S. Gut Microbiota and Dopamine: Producers, Consumers, Enzymatic Mechanisms, and In Vivo Insights. Bioengineering 2025, 13, 55. [Google Scholar] [CrossRef] [PubMed]
- Marzouk, N.H.; Rashwan, H.H.; El-Hadidi, M.; Ramadan, R.; Mysara, M. Proinflammatory and GABA Eating Bacteria in Parkinson’s Disease Gut Microbiome from a Meta-Analysis Perspective. Npj Park. Dis. 2025, 11, 145. [Google Scholar] [CrossRef] [PubMed]
- Camberos-Barraza, J.; Guadrón-Llanos, A.M.; De La Herrán-Arita, A.K. The Gut Microbiome-Neuroglia Axis: Implications for Brain Health, Inflammation, and Disease. Neuroglia 2024, 5, 254–273. [Google Scholar] [CrossRef]
- Buliman, A.; Iordache, M.P.; Protosevici, M.G.-I.; Coroescu, M.-M.; Oncioiu, I.; Popa, M.-L.; Bondar, A.-C. What’s Left When All Is Gone? Limitations of the ABCD2 Score in Transient Ischemic Attacks. Balneo PRM Res. J. 2025, No. 4. [Google Scholar] [CrossRef]
- Cannas, F.; Kopeć, K.K.; Zuddas, N.; Cesare Marincola, F.; Arcara, G.; Loi, M.; Mussap, M.; Fanos, V. Parkinson’s Disease Through the Lens of Metabolomics: A Targeted Systematic Review on Human Studies (2019–2024). J. Clin. Med. 2025, 14, 6277. [Google Scholar] [CrossRef]
- Buliman, A.; Chiotoroiu, A.L.; Panchici, T.-A.; Cintacioiu, D.; Parasca, S.V.; Boiangiu, I.C.; Iordache, M.P. Modified Meek Technique Using Pre-Folded Polyamide Gauzes:A 10-Patient Case Series with Extensive Burns. Ind. Textila 2025, 76, 731–736. [Google Scholar] [CrossRef]
- Bolen, M.L.; Buendia, M.; Shi, J.; Staley, H.; Kachergus, J.M.; Efron, P.A.; Park, G.; Nagpal, R.; Alvarez, S.D.; Xue, Q.-S.; et al. Spatial Single-Cell Multiomics Reveals Peripheral Immune Dysfunction in Parkinson’s and Inflammatory Bowel Disease. Npj Park. Dis. 2026, 12, 25. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Chen, Q.; Zhang, Y.; Asakawa, T. Multidirectional Associations between the Gut Microbiota and Parkinson’s Disease, Updated Information from the Perspectives of Humoral Pathway, Cellular Immune Pathway and Neuronal Pathway. Front. Cell. Infect. Microbiol. 2023, 13, 1296713. [Google Scholar] [CrossRef]
- Lin, Z.; Li, Y.; Liu, Y.; Yang, B.; Yin, P.; Guan, C.; Fang, Y.; Yang, L.; Zan, K.; Cui, G.; et al. Alterations in Gut Microbiota and Plasma Metabolites: A Multi-Omics Study of Mild Cognitive Impairment in Parkinson’s Disease. Front. Neurosci. 2025, 19, 1667331. [Google Scholar] [CrossRef]
- Roy, S.; Chattopadhyay, D.; Choudhury, L.; Ghosh, S.; Mahajan, A.A. Harnessing the Power of Faecal Microbiota Transplantation: Optimizing Neuroimmune Function for Improved Treatment of Parkinson’s, Alzheimer’s, and Multiple Sclerosis. Discov. Neurosci. 2025, 20, 21. [Google Scholar] [CrossRef]
- Shafieinouri, M.; Hong, S.; Lee, P.S.; Grant, S.M.; Khani, M.; Dadu, A.; Schumacher Schuh, A.F.; Makarious, M.B.; Sandon, R.; Simmonds, E.; et al. Gut-Brain Nexus: Mapping Multimodal Links to Neurodegeneration at Biobank Scale. Sci. Adv. 2025, 11, eadu2937. [Google Scholar] [CrossRef]
- Villette, R.; Ortís Sunyer, J.; Novikova, P.V.; Aho, V.T.E.; Petrov, V.A.; Hickl, O.; Busi, S.B.; De Rudder, C.; Kunath, B.J.; Heintz-Buschart, A.; et al. Integrated Multi-Omics Highlights Alterations of Gut Microbiome Functions in Prodromal and Idiopathic Parkinson’s Disease. Microbiome 2025, 13, 200. [Google Scholar] [CrossRef] [PubMed]
- Constantinoiu, S.; Cochior, D. Severe Acute Pancreatitis - Determinant Factors and Current Therapeutic Conduct. Chirurgia (Bucur.) 2018, 113, 385. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Lai, Y.; Darweesh, S.K.L.; Bloem, B.R.; Forsgren, L.; Hansen, J.; Katzke, V.A.; Masala, G.; Sieri, S.; Sacerdote, C.; et al. Gut Microbial Metabolites and Future Risk of Parkinson’s Disease: A Metabolome-Wide Association Study. Mov. Disord. 2025, 40, 556–560. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Wang, H.; Song, J. Gut-Brain Axis Modulation in Remote Rehabilitation of Parkinson’s Disease: Reconstructing the Fecal Metabolome and Nigral Network Connectivity. Front. Neurol. 2025, 16, 1644490. [Google Scholar] [CrossRef] [PubMed]
- Villette, R.; Novikova, P.V.; Laczny, C.C.; Mollenhauer, B.; May, P.; Wilmes, P. Human Gut Microbiome Gene Co-Expression Network Reveals a Loss in Taxonomic and Functional Diversity in Parkinson’s Disease. Npj Biofilms Microbiomes 2025, 11, 142. [Google Scholar] [CrossRef] [PubMed]
- Buliman, A.; Iordache, M.P.; Bondar, A.-C.; Protosevici, M.G.-I.; Oncioiu, I.; Popa, M.-L. From Basic Blood Counts to Functional Outcomes: Neutrophil– Lymphocyte Ratio as a Predictor of Disability in Acute and Subacute Ischemic Stroke. Balneo PRM Res. J. 2025, No. 4. [Google Scholar] [CrossRef]
- Fan, W.; Li, K.; Wang, R.; Chen, R.; Liu, Y.; Yang, Z.; Zhao, N.; Yan, J. Vasoactive Intestinal Peptide: A Neuropeptide That Plays an ImportantRole in Parkinson’s Disease. Curr. Neuropharmacol. 2025, 23. [Google Scholar] [CrossRef]
- Bondar, A.-C.; Iordache, M.P.; Coroescu, M.; Buliman, A.; Rusu, E.; Budișteanu, M.; Tanase, C. Unlocking the Sugar Code: Implications and Consequences of Glycosylation in Alzheimer’s Disease and Other Tauopathies. Biomedicines 2025, 13, 2884. [Google Scholar] [CrossRef]
- Caradonna, E.; Nemni, R.; Bifone, A.; Gandolfo, P.; Costantino, L.; Giordano, L.; Mormone, E.; Macula, A.; Cuomo, M.; Difruscolo, R.; et al. The Brain–Gut Axis, an Important Player in Alzheimer and Parkinson Disease: A Narrative Review. J. Clin. Med. 2024, 13, 4130. [Google Scholar] [CrossRef]
- Moldovan, C.; Cochior, D.; Gorecki, G.; Rusu, E.; Ungureanu, F.-D. Clinical and Surgical Algorithm for Managing Iatrogenic Bile Duct Injuries during Laparoscopic Cholecystectomy: A Multicenter Study. Exp. Ther. Med. 2021, 22, 1385. [Google Scholar] [CrossRef]
- Tanase, C.; Cruceru, M.L.; Enciu, A.-M.; Popa, A.C.; Albulescu, R.; Neagu, M.; Constantinescu, S.N. Signal Transduction Molecule Patterns Indicating Potential Glioblastoma Therapy Approaches. OncoTargets Ther. 2013, 1737. [Google Scholar] [CrossRef]
- Mendonça, I.P.; Peixoto, C.A. The Double-Edged Sword: The Complex Function of Enteric Glial Cells in Neurodegenerative Diseases. J. Neurochem. 2025, 169, e70069. [Google Scholar] [CrossRef]
- Oliver, P.J.; Civitelli, L.; Hu, M.T. The Gut–Brain Axis in Early Parkinson’s Disease: From Prodrome to Prevention. J. Neurol. 2025, 272, 413. [Google Scholar] [CrossRef] [PubMed]
- Templeton, H.N.; Tobet, S.A.; Schwerdtfeger, L.A. Gut Neuropeptide Involvement in Parkinson’s Disease. Am. J. Physiol.-Gastrointest. Liver Physiol. 2025, 328, G716–G733. [Google Scholar] [CrossRef] [PubMed]
- Simionescu, O.; Tudorache, S.I. Autoimmune Pemphigus: Difficulties in Diagnosis and the Molecular Mechanisms Underlying the Disease. Front. Immunol. 2025, 16, 1481093. [Google Scholar] [CrossRef] [PubMed]
- Cord, D.; Rîmbu, M.C.; Iordache, M.P.; Albulescu, R.; Pop, S.; Tanase, C.; Popa, M.-L. Phytochemicals as Epigenetic Modulators in Chronic Diseases: Molecular Mechanisms. Molecules 2025, 30, 4317. [Google Scholar] [CrossRef]
- Adhikary, K.; Ganguly, K.; Sarkar, R.; Abubakar, Md.; Banerjee, P.; Karak, P. Phytonutrients and Their Neuroprotective Role in Brain Disorders. Front. Mol. Biosci. 2025, 12, 1607330. [Google Scholar] [CrossRef]
- Jaberi, K.R.; Alamdari-palangi, V.; Savardashtaki, A.; Vatankhah, P.; Jamialahmadi, T.; Tajbakhsh, A.; Sahebkar, A. Modulatory Effects of Phytochemicals on Gut–Brain Axis: Therapeutic Implication. Curr. Dev. Nutr. 2024, 8, 103785. [Google Scholar] [CrossRef] [PubMed]
- Lupu, M.; Gradisteanu Pircalabioru, G.; Chifiriuc, M.; Albulescu, R.; Tanase, C. Beneficial Effects of Food Supplements Based on Hydrolyzed Collagen for Skin Care (Review). Exp. Ther. Med. 2019. [Google Scholar] [CrossRef]
- Mocanu, A.-I.; Mocanu, H.; Moldovan, C.; Soare, I.; Niculet, E.; Tatu, A.L.; Vasile, C.I.; Diculencu, D.; Postolache, P.A.; Nechifor, A. Some Manifestations of Tuberculosis in Otorhinolaryngology – Case Series and a Short Review of Related Data from South-Eastern Europe. Infect. Drug Resist. 2022, Volume 15, 2753–2762. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Sun, L.; Liu, Y.; Chen, R. Safety and Efficacy of Fecal Microbiota Transplantation in the Treatment of Parkinson’s Disease: A Systematic Review of Clinical Trials. Front. Neurosci. 2025, 19, 1639911. [Google Scholar] [CrossRef]
- Cheng, Y.; Tan, G.; Zhu, Q.; Wang, C.; Ruan, G.; Ying, S.; Qie, J.; Hu, X.; Xiao, Z.; Xu, F.; et al. Efficacy of Fecal Microbiota Transplantation in Patients with Parkinson’s Disease: Clinical Trial Results from a Randomized, Placebo-Controlled Design. Gut Microbes 2023, 15, 2284247. [Google Scholar] [CrossRef] [PubMed]
- Duru, I.C.; Lecomte, A.; Shishido, T.K.; Laine, P.; Suppula, J.; Paulin, L.; Scheperjans, F.; Pereira, P.A.B.; Auvinen, P. Metagenome-Assembled Microbial Genomes from Parkinson’s Disease Fecal Samples. Sci. Rep. 2024, 14, 18906. [Google Scholar] [CrossRef]
- Jabbari Shiadeh, S.M.; Chan, W.K.; Rasmusson, S.; Hassan, N.; Joca, S.; Westberg, L.; Elfvin, A.; Mallard, C.; Ardalan, M. Bidirectional Crosstalk between the Gut Microbiota and Cellular Compartments of Brain: Implications for Neurodevelopmental and Neuropsychiatric Disorders. Transl. Psychiatry 2025, 15, 278. [Google Scholar] [CrossRef]
- Chai, Z.; Ouyang, Y.; Debebe, A.; Picker, M.; Lee, W.-J.; Fenton, S.; Becker-Dorison, A.; Augustin-Emmerichs, K.; Schwiertz, A.; Weber, S.N.; et al. Intestinal Biomarkers, Microbiota Composition, and Genetic Predisposition to Inflammatory Bowel Disease as Predictors of Parkinson’s Disease Manifestation. J. Park. Dis. 2025, 15, 766–779. [Google Scholar] [CrossRef]
- Chtioui, N.; Duval, C.; St-Pierre, D.H. The Impact of an Active Lifestyle on Markers of Intestinal Inflammation in Parkinson’s Disease: Preliminary Findings. Clin. Park. Relat. Disord. 2025, 12, 100301. [Google Scholar] [CrossRef]
- Fan, T.; Peng, J.; Liang, H.; Chen, W.; Wang, J.; Xu, R. Potential Common Pathogenesis of Several Neurodegenerative Diseases. Neural Regen. Res. 2026, 21, 972–988. [Google Scholar] [CrossRef]
- Terenzi, D.; Muth, A.-K.; Park, S.Q. Nutrition and Gut–Brain Pathways Impacting the Onset of Parkinson’s Disease. Nutrients 2022, 14, 2781. [Google Scholar] [CrossRef]
- Yan, Z.; Zhao, G. The Associations Among Gut Microbiota, Branched Chain Amino Acids, and Parkinson’s Disease: Mendelian Randomization Study. J. Park. Dis. 2024, 14, 1129–1138. [Google Scholar] [CrossRef] [PubMed]
- Valle, J. Biofilm-Associated Proteins: From the Gut Biofilms to Neurodegeneration. Gut Microbes 2025, 17, 2461721. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, Q.; Su, R. Interplay of Human Gastrointestinal Microbiota Metabolites: Short-Chain Fatty Acids and Their Correlation with Parkinson’s Disease. Medicine (Baltimore) 2024, 103, e37960. [Google Scholar] [CrossRef] [PubMed]
- Buliman, A.; Calin, M.A.; Iordache, M.P. Targeting Anxiety with Light: Mechanistic and Clinical Insights into Photobiomodulation Therapy: A Mini Narrative Review. Balneo PRM Res. J. 2025, 16, 846–846. [Google Scholar] [CrossRef]
- Derkinderen, P.; Cossais, F.; Kulcsárová, K.; Škorvánek, M.; Sellier-Montaigne, L.; Coron, E.; Leclair-Visonneau, L.; Cerri, S.; Pellegrini, C.; Rolli-Derkinderen, M. How Leaky Is the Gut in Parkinson’s Disease? eBioMedicine 2025, 117, 105796. [Google Scholar] [CrossRef]
- Yue, M.; Chen, T.; Chen, W.; Wei, J.; Liao, B.; Zhang, J.; Li, F.; Hong, D.; Fang, X. The Engineered Probiotic Strain Lactococcus Lactis MG1363-pMG36e-GLP-1 Regulates Microglial Polarization and Gut Dysbiosis in a Transgenic Mouse Model of Parkinson’s Disease. Neural Regen. Res. 2026, 21, 1211–1221. [Google Scholar] [CrossRef]
- Cervantes-Arriaga, A.; Sarabia-Tapia, C.; Esquivel-Zapata, O.; López-Alamillo, S.; Reséndiz-Henriquez, E.; Corona, T.; Rodríguez-Violante, M. Pitfalls and Caveats in the Diagnostic Pathway of People with ParkinsonÓ?S Disease. Rev. Mex. Neurocienc. 2022, 23, 7436. [Google Scholar] [CrossRef]
- Faysal, Md.; Zehravi, M.; Sutradhar, B.; Al Amin, M.; Shanmugarajan, T.S.; Arjun, U.V.N.V.; Ethiraj, S.; Durairaj, A.; Dayalan, G.; Ahamad, S.K.; et al. The Microbiota-Gut-Brain Connection: A New Horizon in Neurological and Neuropsychiatric Disorders. CNS Neurosci. Ther. 2025, 31, e70593. [Google Scholar] [CrossRef]
- Ramos, H.; Araújo, A.M.; Ferreira, I.M.; Faria, M.A. The Neurotoxic Impact of Food Chemical Contaminants: A Growing Concern? Curr. Opin. Food Sci. 2026, 67, 101369. [Google Scholar] [CrossRef]
- Xie, Y.-A.; Kong, J.-D.; Li, S.; Wei, D.-F. What Is the Impact of Dopamine D2 Receptor in the Brain-Gut Axis? A Narrative Review of the Mechanism Based on Gut Microbiota in Modulating Emotion and Behavior. Alpha Psychiatry 2025, 26, 39226. [Google Scholar] [CrossRef]
- Feix, J.B.; Cheng, G.; Hardy, M.; Kalyanaraman, B. Microbial Metabolism of Levodopa as an Adjunct Therapeutic Target in Parkinson’s Disease. Antioxidants 2026, 15, 120. [Google Scholar] [CrossRef] [PubMed]
- Bi, M.; Liu, C.; Wang, Y.; Liu, S.-J. Therapeutic Prospect of New Probiotics in Neurodegenerative Diseases. Microorganisms 2023, 11, 1527. [Google Scholar] [CrossRef] [PubMed]
- Hey, G.; Nair, N.; Klann, E.; Gurrala, A.; Safarpour, D.; Mai, V.; Ramirez-Zamora, A.; Vedam-Mai, V. Therapies for Parkinson’s Disease and the Gut Microbiome: Evidence for Bidirectional Connection. Front. Aging Neurosci. 2023, 15, 1151850. [Google Scholar] [CrossRef]
- Patel, N.J.; Hajoori, M.; Desai, P. Evaluating Probiotic Properties of Gut Microflora for Gut Modulation as an Adjuvant Therapy for Parkinson’s Disease. J. Future Foods 2025, 5, 304–316. [Google Scholar] [CrossRef]
- Takahashi, R.; Yamakado, H.; Uemura, N.; Taguchi, T.; Ueda, J. The Gut–Brain Axis Based on α-Synuclein Propagation—Clinical, Neuropathological, and Experimental Evidence. Int. J. Mol. Sci. 2025, 26, 3994. [Google Scholar] [CrossRef]
- Domínguez Rojo, N.; Blanco Benítez, M.; Cava, R.; Fuentes, J.M.; Canales Cortés, S.; González Polo, R.A. Convergence of Neuroinflammation, Microbiota, and Parkinson’s Disease: Therapeutic Insights and Prospects. Int. J. Mol. Sci. 2024, 25, 11629. [Google Scholar] [CrossRef]
- Zheng, Y.; Bonfili, L.; Wei, T.; Eleuteri, A.M. Understanding the Gut–Brain Axis and Its Therapeutic Implications for Neurodegenerative Disorders. Nutrients 2023, 15, 4631. [Google Scholar] [CrossRef]
- Iyengar, A.R.S.; Dunkley, P.R.; Dickson, P.W. Immunity in Parkinson’s Disease - the Role of Adaptive and Auto-Immune Responses and Gut-Microbiome Axis. J. Neuroimmunol. 2025, 409, 578755. [Google Scholar] [CrossRef]
- Kearns, R. Gut–Brain Axis and Neuroinflammation: The Role of Gut Permeability and the Kynurenine Pathway in Neurological Disorders. Cell. Mol. Neurobiol. 2024, 44, 64. [Google Scholar] [CrossRef]
- Liang, Y.; Zhao, Y.; Fasano, A.; Su, C.-W. Gut Permeability and Microbiota in Parkinson’s Disease: Mechanistic Insights and Experimental Therapeutic Strategies. Int. J. Mol. Sci. 2025, 26, 9593. [Google Scholar] [CrossRef]
- Padhi, P.; Abdalla, A.; Schneider, B.; Backes, N.; Otto, A.A.; Scheibe, I.J.; Thomas, J.P.; Khadse, G.; Samidurai, M.; Jochmans, A.K.; et al. Bioengineered Gut Bacterium Synthesizing Levodopa Alleviates Motor Deficits in Models of Parkinson’s Disease. Cell Host Microbe 2025, 33, 1837–1854.e13. [Google Scholar] [CrossRef]
- Swain, D.; Panigrahi, L.L.; Pradhan, S.K.; Arakha, M.; Jadhao, K.R.; Rout, G.R. Elucidation of Dual Antimicrobial and Anti-Parkinsonian Activities through an In-Silico Approach of Ipomoea Mauritiana Jacq. in the Context of the Gut–Brain Axis. ACS Omega 2026, 11, 4021–4036. [Google Scholar] [CrossRef]
- Parrella, E.; Gennari, M.M.; Abate, G.; Pucci, M.; Schioppa, T.; Bosisio, D.; Tirelli, E.; Benarese, M.; Vegezzi, G.; Silletti, M.G.; et al. Early Alpha-Synuclein Accumulation, Oxidative Stress and Inflammation in the Proximal Colon of c-Rel-/- Mouse Model of Parkinson’s Disease. Neurobiol. Dis. 2025, 217, 107182. [Google Scholar] [CrossRef]
- Rust, C.; Tonge, D.; Van Den Heuvel, L.L.; Asmal, L.; Carr, J.; Pretorius, E.; Seedat, S.; Hemmings, S.M.J. Investigating the Blood Microbiome in Parkinson’s Disease, Schizophrenia, and Posttraumatic Stress Disorder. Brain Behav. 2025, 15, e70629. [Google Scholar] [CrossRef]
- Borrego-Ruiz, A.; Borrego, J.J. Influence of Human Gut Microbiome on the Healthy and the Neurodegenerative Aging. Exp. Gerontol. 2024, 194, 112497. [Google Scholar] [CrossRef] [PubMed]
- Leta, V.; Zinzalias, P.; Batzu, L.; Mandal, G.; Staunton, J.; Jernstedt, F.; Rosqvist, K.; Timpka, J.; Van Vliet, T.; Trivedi, D.; et al. Effects of a Four-Strain Probiotic on Gut Microbiota, Inflammation, and Symptoms in Parkinson’s Disease: A Randomized Clinical Trial. Mov. Disord. 2025, 40, 2710–2721. [Google Scholar] [CrossRef] [PubMed]
- Panaitescu, P.-Ștefan; Răzniceanu, V.; Mocrei-Rebrean, Ștefania-M.; Neculicioiu, V.S.; Dragoș, H.-M.; Costache, C.; Filip, G.A. The Effect of Gut Microbiota-Targeted Interventions on Neuroinflammation and Motor Function in Parkinson’s Disease Animal Models—A Systematic Review. Curr. Issues Mol. Biol. 2024, 46, 3946–3974. [Google Scholar] [CrossRef]
- Han, M.N.; Di Natale, M.R.; Lei, E.; Furness, J.B.; Finkelstein, D.I.; Hao, M.M.; Diwakarla, S.; McQuade, R.M. Assessment of Gastrointestinal Function and Enteric Nervous System Changes over Time in the A53T Mouse Model of Parkinson’s Disease. Acta Neuropathol. Commun. 2025, 13, 58. [Google Scholar] [CrossRef]
- Morais, L.H.; Stiles, L.; Freeman, M.; Oguienko, A.D.; Hoang, J.D.; Ji, J.; Jones, J.; Quan, B.; Devine, J.; Bois, J.S.; et al. The Gut Microbiome Promotes Mitochondrial Respiration in the Brain of a Parkinson’s Disease Mouse Model. Npj Park. Dis. 2025, 11, 301. [Google Scholar] [CrossRef] [PubMed]
- Claudino Dos Santos, J.C.; Oliveira, L.F.; Noleto, F.M.; Gusmão, C.T.P.; Brito, G.A.D.C.; Viana, G.S.D.B. Gut-Microbiome-Brain Axis: The Crosstalk between the Vagus Nerve, Alpha-Synuclein and the Brain in Parkinson’s Disease. Neural Regen. Res. 2023, 18, 2611–2614. [Google Scholar] [CrossRef]
- Li, T.; Wu, J.; Zhou, S.; Li, M.; Zhao, L.; Wang, A.; Song, Y.; Huang, W.; Tan, L.; Qiao, C.; et al. Neuroprotective Effects of Time-Restricted Feeding Combined With Different Protein Sources in MPTP-Induced Parkinson’s Disease Mice Model and Its Modulatory Impact on Gut Microbiota Metabolism. Adv. Sci. 2026, e16502. [Google Scholar] [CrossRef]
- Carrossa, G.; Misenti, V.; Faggin, S.; Giron, M.C.; Antonini, A. The Small Intestinal Microbiota and the Gut–Brain Axis in Parkinson’s Disease: A Narrative Review. Biomedicines 2025, 13, 1769. [Google Scholar] [CrossRef] [PubMed]
- Hein, Z.M.; Arbain, M.F.F.; Kumar, S.; Mehat, M.Z.; Hamid, H.A.; Che Ramli, M.D.; Che Mohd Nassir, C.M.N. Intermittent Fasting as a Neuroprotective Strategy: Gut–Brain Axis Modulation and Metabolic Reprogramming in Neurodegenerative Disorders. Nutrients 2025, 17, 2266. [Google Scholar] [CrossRef]
- Dubey, S.; Ghosh, R.; Dubey, M.J.; Sengupta, S.; Das, S. From Allegory to Conceptualization, Hypothesis and Finally Evidences: Alzheimer’s Dementia, Parkinson’s Disease “Gut–Brain Axis” and Their Preclinical Phenotype. Egypt. J. Neurol. Psychiatry Neurosurg. 2024, 60, 89. [Google Scholar] [CrossRef]
- Mahbub, N.U.; Islam, M.M.; Hong, S.-T.; Chung, H.-J. Dysbiosis of the Gut Microbiota and Its Effect on α-Synuclein and Prion Protein Misfolding: Consequences for Neurodegeneration. Front. Cell. Infect. Microbiol. 2024, 14, 1348279. [Google Scholar] [CrossRef]
- Przewodowska, D.; Alster, P.; Madetko-Alster, N. Role of the Intestinal Microbiota in the Molecular Pathogenesis of Atypical Parkinsonian Syndromes. Int. J. Mol. Sci. 2025, 26, 3928. [Google Scholar] [CrossRef] [PubMed]
- Kroker Kimber, I.; Tremblay, M.-È. Food for Thought: Probiotic Modulation of Microglial Activity in Parkinson’s Disease. Front. Mol. Neurosci. 2025, 18, 1690507. [Google Scholar] [CrossRef]
- Pokora, B.; Pokora, K.; Binienda, A.; Fichna, J. The Ketogenic Diet in Parkinson’s Disease: A Potential Therapeutic Strategy. Pharmacol. Rep. 2025, 77, 1491–1513. [Google Scholar] [CrossRef] [PubMed]
- Theis, B.F.; Park, J.S.; Kim, J.S.A.; Zeydabadinejad, S.; Vijay-Kumar, M.; Yeoh, B.S.; Saha, P. Gut Feelings: How Microbes, Diet, and Host Immunity Shape Disease. Biomedicines 2025, 13, 1357. [Google Scholar] [CrossRef]
- Kwon, D.; Zhang, K.; Paul, K.C.; Folle, A.D.; Del Rosario, I.; Jacobs, J.P.; Keener, A.M.; Bronstein, J.M.; Ritz, B. Diet and the Gut Microbiome in Patients with Parkinson’s Disease. Npj Park. Dis. 2024, 10, 89. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Wang, K.; Su, N.; Yuan, C.; Zhang, N.; Hu, X.; Fu, Y.; Zhao, F. Microbiota–Gut–Brain Axis in Health and Neurological Disease: Interactions between Gut Microbiota and the Nervous System. J. Cell. Mol. Med. 2024, 28, e70099. [Google Scholar] [CrossRef]
- Yuan, X.-Y.; Chen, Y.-S.; Liu, Z. Relationship among Parkinson’s Disease, Constipation, Microbes, and Microbiological Therapy. World J. Gastroenterol. 2024, 30, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-Y.; Jo, S.; Lee, J.; Choi, M.; Kim, K.; Lee, S.; Kim, H.S.; Bae, J.-W.; Chung, S.J. Distinct Gut Microbiome Characteristics and Dynamics in Patients with Parkinson’s Disease Based on the Presence of Premotor Rapid-Eye Movement Sleep Behavior Disorders. Microbiome 2025, 13, 108. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Li, Y.; Yan, J.; Chang, R.; Xu, M.; Zhou, G.; Meng, J.; Liu, D.; Mao, Z.; Yang, Y. Gut Microbiota from Patients with Parkinson’s Disease Causes Motor Deficits in Honeybees. Front. Microbiol. 2024, 15, 1418857. [Google Scholar] [CrossRef] [PubMed]
- Wei, B.; Zhao, Y.; Cheng, Y.; Huang, C.; Zhang, F. Helicobacter Pylori Infection and Parkinson’s Disease: Etiology, Pathogenesis and Levodopa Bioavailability. Immun. Ageing 2024, 21, 1. [Google Scholar] [CrossRef]
- Feng, Y.; Zhang, R.; Wen, G.; Xie, L.; Chen, T.; Liu, W. The Role of Gut Microbiota Tyrosine Decarboxylases in Levodopa Pharmacokinetics: Insights from a Levodopa Challenge Test. BMC Neurol. 2025, 25, 460. [Google Scholar] [CrossRef]
- Papić, E.; Rački, V.; Hero, M.; Zimani, A.N.; Čižek Sajko, M.; Rožmarić, G.; Starčević Čizmarević, N.; Ostojić, S.; Kapović, M.; Hauser, G.; et al. Microbial Diversity in Drug-Naïve Parkinson’s Disease Patients. PLOS One 2025, 20, e0328761. [Google Scholar] [CrossRef]
- Chambilo, B.; Dhiver, T.; Sahu, K.; Parashar, R. Gut Microbiota Modulation with Probiotics: Potential Therapeutic Avenues for Alzheimer’s and Parkinson’s Diseases. Neurol. Sci. Neurophysiol. 2025, 42, 75–83. [Google Scholar] [CrossRef]
- Du, Y.; Wang, L.; Cui, Y.; Xu, X.; Zhang, M.; Li, Y.; Gao, T.; Gao, D.; Sheng, Z.; Wang, S.; et al. Effect of Probiotics Supplementation on REM Sleep Behavior Disorder and Motor Symptoms in Parkinson’s Disease: A Pilot Study. CNS Neurosci. Ther. 2025, 31, e70541. [Google Scholar] [CrossRef]
- Papić, E.; Rački, V.; Hero, M.; Tomić, Z.; Starčević-Čižmarević, N.; Kovanda, A.; Kapović, M.; Hauser, G.; Peterlin, B.; Vuletić, V. The Effects of Microbiota Abundance on Symptom Severity in Parkinson’s Disease: A Systematic Review. Front. Aging Neurosci. 2022, 14, 1020172. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Pinto, M.F.; Candeias, E.; Melo-Marques, I.; Esteves, A.R.; Maranha, A.; Magalhães, J.D.; Carneiro, D.R.; Sant’Anna, M.; Pereira-Santos, A.R.; Abreu, A.E.; et al. Gut-First Parkinson’s Disease Is Encoded by Gut Dysbiome. Mol. Neurodegener. 2024, 19, 78. [Google Scholar] [CrossRef] [PubMed]
| Taxon |
Direction in PD |
Functional Role |
Mechanistic Implication |
||||
| Faecalibacterium prausnitzii | ↓ Decreased | Butyrate production | Reduced epithelial barrier integrity | [20] | |||
| Roseburia intestinalis | ↓ Decreased | SCFA production | Impaired anti-inflammatory signaling | [47,48] | |||
| Coprococcus spp. | ↓ Decreased | SCFA production; BCAA modulation | Altered metabolic homeostasis | [44,49,50] | |||
| Akkermansia muciniphila | ↑ Increased | Mucin degradation | Thinning of mucus layer | [42,51] | |||
| Bilophila spp. | ↑ Increased | Sulfur metabolism | Pro-inflammatory environment | [46,52] | |||
| Klebsiella variicola | ↑ Increased | LPS production | Immune activation | [13,53] | |||
| GABA-consuming species (Evtepia gabavorous) | ↑ Increased | GABA metabolism | Altered inhibitory neurotransmission | [16,54,55] |
| Pathway | Direction in PD | Associated Consequence | |
| Riboflavin biosynthesis | ↓ Decreased | Reduced antioxidant capacity | [43,71,72] |
| Biotin biosynthesis | ↓ Decreased | Impaired fatty acid metabolism | [27,73] |
| CAZymes | ↓ Decreased | Reduced complex carbohydrate fermentation |
[74,75] |
| Secondary bile acid biosynthesis | ↓ Decreased | Altered gut–liver axis signaling |
[76,77] |
| Xenobiotic metabolism (solvent/pesticide) | ↑ Increased | Potential modulation of toxin exposure | [78,79,80,81] |
| Flagellar assembly genes | ↓ Decreased | Altered microbial motility | [53,68,82] |
| Metabolite | Direction in PD | Potential Impact | |
| Butyrate | ↓ Decreased | Reduced barrier integrity; microglial regulation |
[42] |
| Propionate | ↓ Decreased | Altered immune signaling | [105,106] |
| Polyamines | ↓ Decreased | Mucosal stability impairment | [53,107] |
| Isoleucine (BCAA) | ↓ Lower levels associated with higher PD risk | Dopaminergic metabolic modulation |
[108] |
| GABA | Dysregulated | Basal ganglia circuit imbalance | [9,47,59] |
| Lipopolysaccharide (LPS) | ↑ Increased systemic levels | TLR activation and neuroinflammation | [80,109] |
| Intervention | Mechanism | Reported Effects | Limitations | |
| High-fiber / Mediterranean diet | Increases SCFA-producing taxa | Improved microbial diversity | Long-term adherence variability | [138] |
| Prebiotics | Stimulate SCFA production | Improved constipation | Limited large RCTs | [20,26] |
| Probiotics | Modulate inflammatory taxa | Improved GI symptoms | Strain heterogeneity | [50,103,139] |
| FMT | Restores microbial diversity | Motor and non-motor symptom improvement (preliminary) | Small sample sizes |
[140,141] |
| Bacterial decarboxylase inhibition | Reduces levodopa metabolism | Improved drug bioavailability (experimental) | Early translational stage | [102,121,142] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




