Submitted:
16 February 2026
Posted:
24 February 2026
You are already at the latest version
Abstract
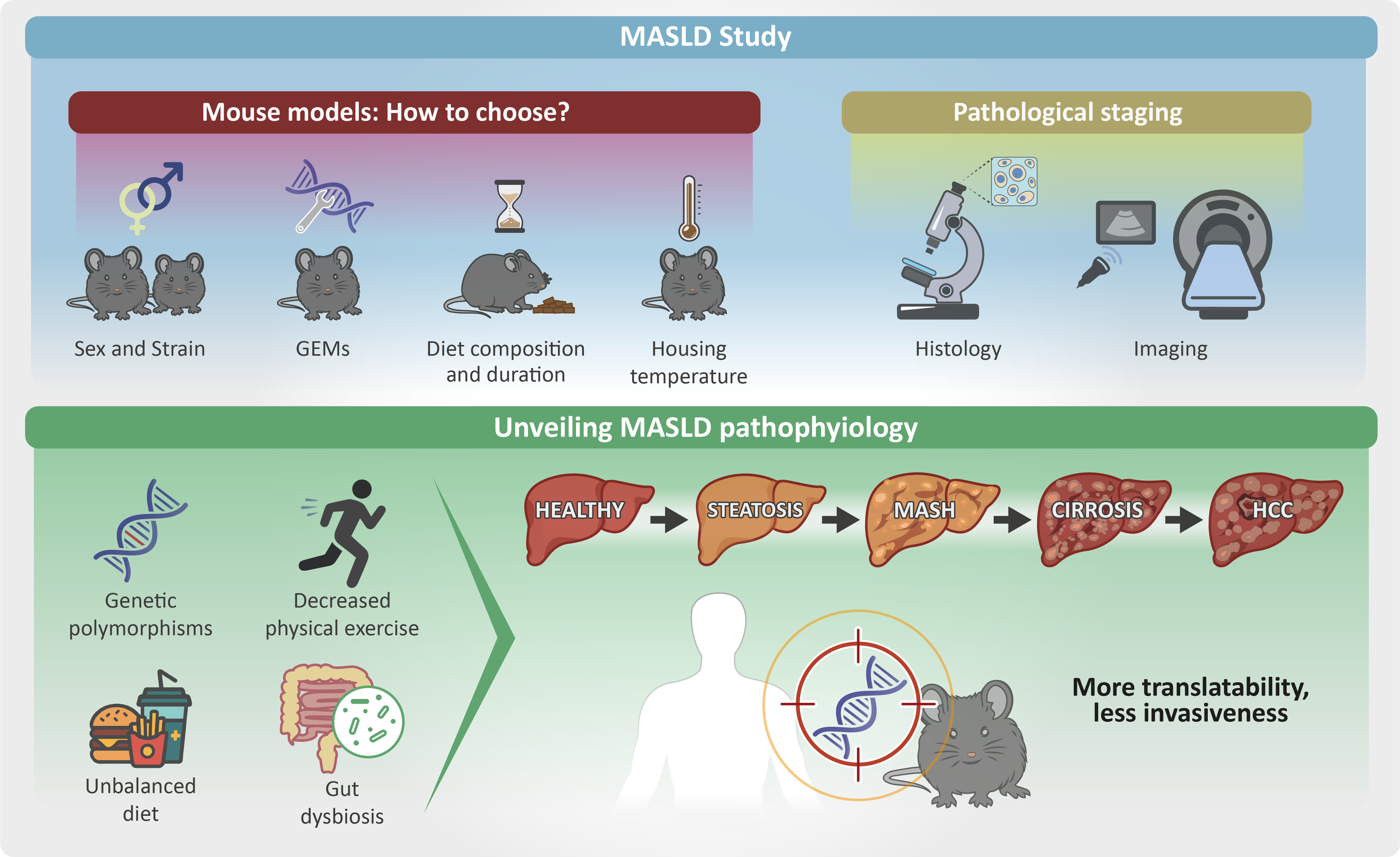
Keywords:
1. Hepatic Steatosis and Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD): Overview
2. Mouse Models of MASLD: Strength and Pitfalls in Comparative Pathology
3. Histopathological Findings in MASLD
4. MASLD: Clinical and Experimental Imaging Assessment
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| IR | Insulin Resistance |
| MASLD | Metabolic Dysfunction-Associated Steatotic Liver Disease |
| NAFLD | Non-Alcoholic Fatty Liver Disease |
| MASH | Metabolic Dysfunction-Associated Steatohepatitis |
| HCC | Hepatocellular carcinoma |
| BMI | Body Mass Index |
| PNPLA3 | Patatin-like Phospholipase domain-containing 3 |
| HSD17B13 | 17β-hydroxysteroid dehydrogenase-13 |
| GCRK | Glucokinase regulatory protein gene |
| MBOAT7 | Membrane-Bound O-Acyltransferase domain–containing 7 |
| GLP-1 | Glucagon-Like Peptide-1 |
| PPAR | Peroxisome Proliferator–Activated Receptor |
| HFD | High Fat Diet |
| MCD | Methionine and/or Choline Deficient |
| WD | Western diet |
| GEM | Genetically Engineered Mouse |
| KO | Knock-Out |
| GAN | Gubra Amylin diet for non-alcoholic steatohepatitis |
| TN | Thermoneutrality |
| NZO | New Zealand Obese |
| SD | Standard Diet |
| MAPK15 | Mitogen-Activated Protein Kinase 15 |
| Atg7 | Autophagy-related gene 7 |
| AKAP1 | A-Kinase Anchoring Protein 1 |
| ACSL1 | Acyl-CoA Synthetase Long-chain family member 1 |
| GPAT1 | Glycerol-3-Phosphate Acyltransferase 1 |
| Ay | Agouti Yellow |
| DKO | Cyp2a12/Cyp2c70 knock-out |
| BW | Body Weight |
| ER | Endoplasmic Reticulum |
| HSC | Hepatic Stellate Cells |
| NAS | NAFLD Activity Score |
| H&E | Hematoxylin & Eosin |
| US | UltraSound |
| CT | Computed Tomography |
| MRI | Magnetic Resonance Imaging |
| HVPG | Hepatic Venous Pressure Gradient |
| EUS-LB | Endoscopic UltraSound-guided Liver Biopsy |
| FNA | Fine-Needle Aspiration |
| FNB | Fine-Needle Biopsy |
| NITs | Non-Invasive Tests |
| AI | Artificial Intelligence |
| WFUMB | World Federation for Ultrasound in Medicine and Biology |
| MRI-PDFF | Magnetic Resonance Imaging Proton Density Fat Fraction |
| MRS | Magnetic Resonance Spectroscopy |
| QUS | Quantitative UltraSound |
| STEAM | Stimulated Echo Acquisition Mode |
| DCE | Dynamic Contrast Enhanced |
| VAPOR | VAriable Power radiofrequency pulses with Optimized Relaxation delays |
| GC-MS | Gas Chromatography-Mass Spectrometry |
| PEG-FGF21v | Polyethylene glycol fibroblast growth factor 21 variant |
References
- Aron-Wisnewsky, J.; Vigliotti, C.; Witjes, J.; Le, P.; Holleboom, A.G.; Verheij, J.; Nieuwdorp, M.; Clément, K. Gut Microbiota and Human NAFLD: Disentangling Microbial Signatures from Metabolic Disorders. Nat Rev Gastroenterol Hepatol 2020, 17, 279–297. [Google Scholar] [CrossRef]
- Rinella, M.E.; Lazarus, J.V.; Ratziu, V.; Francque, S.M.; Sanyal, A.J.; Kanwal, F.; Romero, D.; Abdelmalek, M.F.; Anstee, Q.M.; Arab, J.P.; et al. A Multisociety Delphi Consensus Statement on New Fatty Liver Disease Nomenclature. Hepatology 2023, 78, 1966–1986. [Google Scholar] [CrossRef] [PubMed]
- Stevanović-Silva, J.; Beleza, J.; Coxito, P.; Costa, R.C.; Ascensão, A.; Magalhães, J. Fit Mothers for a Healthy Future: Breaking the Intergenerational Cycle of Non-alcoholic Fatty Liver Disease with Maternal Exercise. Eur J Clin Investigation 2022, 52, e13596. [Google Scholar] [CrossRef]
- Maliken, B.D.; Nelson, J.E.; Klintworth, H.M.; Beauchamp, M.; Yeh, M.M.; Kowdley, K.V. Hepatic Reticuloendothelial System Cell Iron Deposition Is Associated with Increased Apoptosis in Nonalcoholic Fatty Liver Disease. Hepatology 2013, 57, 1806–1813. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Golabi, P.; Paik, J.M.; Henry, A.; Van Dongen, C.; Henry, L. The Global Epidemiology of Nonalcoholic Fatty Liver Disease (NAFLD) and Nonalcoholic Steatohepatitis (NASH): A Systematic Review. Hepatology 2023, 77, 1335–1347. [Google Scholar] [CrossRef]
- Huo, Z.; Chen, Y.; Huang, Y.; Yang, Z.; Long, Y.; Zhang, Q.; Chen, S.; Wang, G.; Zhu, S.; Sun, D.; et al. Long-Term Prognosis of Lean MASLD: Evidence from Three Population-Based Prospective Cohorts. Gut 2025, gutjnl-2025-336127. [Google Scholar] [CrossRef]
- Ha, S.; Wong, V.W.-S.; Zhang, X.; Yu, J. Interplay between Gut Microbiome, Host Genetic and Epigenetic Modifications in MASLD and MASLD-Related Hepatocellular Carcinoma. Gut 2024, 74, 141–152. [Google Scholar] [CrossRef]
- Jegodzinski, L.; Rudolph, L.; Castven, D.; Sayk, F.; Rout, A.K.; Föh, B.; Hölzen, L.; Meyhöfer, S.; Schenk, A.; Weber, S.N.; et al. PNPLA3 I148M Variant Links to Adverse Metabolic Traits in MASLD during Fasting and Feeding. JHEP Rep 2025, 7, 101450. [Google Scholar] [CrossRef]
- Su, W.; Wang, Y.; Jia, X.; Wu, W.; Li, L.; Tian, X.; Li, S.; Wang, C.; Xu, H.; Cao, J.; et al. Comparative Proteomic Study Reveals 17β-HSD13 as a Pathogenic Protein in Nonalcoholic Fatty Liver Disease. Proc Natl Acad Sci U S A 2014, 111, 11437–11442. [Google Scholar] [CrossRef] [PubMed]
- Orho-Melander, M.; Melander, O.; Guiducci, C.; Perez-Martinez, P.; Corella, D.; Roos, C.; Tewhey, R.; Rieder, M.J.; Hall, J.; Abecasis, G.; et al. Common Missense Variant in the Glucokinase Regulatory Protein Gene Is Associated With Increased Plasma Triglyceride and C-Reactive Protein but Lower Fasting Glucose Concentrations. Diabetes 2008, 57, 3112–3121. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekaran, P.; Weiskirchen, R. The Pivotal Role of the Membrane-Bound O-Acyltransferase Domain Containing 7 in Non-Alcoholic Fatty Liver Disease. Livers 2023, 4, 1–14. [Google Scholar] [CrossRef]
- O’Hare, E.A.; Yang, R.; Yerges-Armstrong, L.M.; Sreenivasan, U.; McFarland, R.; Leitch, C.C.; Wilson, M.H.; Narina, S.; Gorden, A.; Ryan, K.A.; et al. TM6SF2 Rs58542926 Impacts Lipid Processing in Liver and Small Intestine. Hepatology 2017, 65, 1526–1542. [Google Scholar] [CrossRef] [PubMed]
- Nemer, M.; Osman, F.; Said, A. Dietary Macro and Micronutrients Associated with MASLD: Analysis of a National US Cohort Database. Annals of Hepatology 2024, 29, 101491. [Google Scholar] [CrossRef] [PubMed]
- Hong, F.; Radaeva, S.; Pan, H.; Tian, Z.; Veech, R.; Gao, B. Interleukin 6 Alleviates Hepatic Steatosis and Ischemia/Reperfusion Injury in Mice with Fatty Liver Disease. Hepatology 2004, 40, 933–941. [Google Scholar] [CrossRef]
- Zheng, Y.; Huang, C.; Zhao, L.; Chen, Y.; Liu, F. Regulation of Decorin by Ursolic Acid Protects against Non-Alcoholic Steatohepatitis. Biomedicine & Pharmacotherapy 2021, 143, 112166. [Google Scholar] [CrossRef]
- Shen, F.; Zheng, R.-D.; Sun, X.-Q.; Ding, W.-J.; Wang, X.-Y.; Fan, J.-G. Gut Microbiota Dysbiosis in Patients with Non-Alcoholic Fatty Liver Disease. Hepatobiliary & Pancreatic Diseases International 2017, 16, 375–381. [Google Scholar] [CrossRef]
- Drygalski, K. Pharmacological Treatment of MASLD: Contemporary Treatment and Future Perspectives. IJMS 2025, 26, 6518. [Google Scholar] [CrossRef]
- Hudson, D.; Afzaal, T.; Bualbanat, H.; AlRamdan, R.; Howarth, N.; Parthasarathy, P.; AlDarwish, A.; Stephenson, E.; Almahanna, Y.; Hussain, M.; et al. Modernizing Metabolic Dysfunction-Associated Steatotic Liver Disease Diagnostics: The Progressive Shift from Liver Biopsy to Noninvasive Techniques. Therap Adv Gastroenterol 2024, 17, 17562848241276334. [Google Scholar] [CrossRef]
- Tacke, F.; Horn, P.; Wai-Sun Wong, V.; Ratziu, V.; Bugianesi, E.; Francque, S.; Zelber-Sagi, S.; Valenti, L.; Roden, M.; Schick, F.; et al. EASL–EASD–EASO Clinical Practice Guidelines on the Management of Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD). Journal of Hepatology 2024, 81, 492–542. [Google Scholar] [CrossRef]
- Gargiulo, S.; Gramanzini, M.; Bonente, D.; Tamborrino, T.; Inzalaco, G.; Gherardini, L.; Franci, L.; Bertelli, E.; Barone, V.; Chiariello, M. Preclinical Application of Computer-Aided High-Frequency Ultrasound (HFUS) Imaging: A Preliminary Report on the In Vivo Characterization of Hepatic Steatosis Progression in Mouse Models. J. Imaging 2025, 11, 369. [Google Scholar] [CrossRef] [PubMed]
- Pelechá, M.; Villanueva-Bádenas, E.; Timor-López, E.; Donato, M.T.; Tolosa, L. Cell Models and Omics Techniques for the Study of Nonalcoholic Fatty Liver Disease: Focusing on Stem Cell-Derived Cell Models. Antioxidants 2021, 11, 86. [Google Scholar] [CrossRef]
- Caddeo, A.; Maurotti, S.; Kovooru, L.; Romeo, S. 3D Culture Models to Study Pathophysiology of Steatotic Liver Disease. Atherosclerosis 2024, 393, 117544. [Google Scholar] [CrossRef] [PubMed]
- Youhanna, S.; Taebnia, N.; Liang, Y.; Cheng, N.; Wang, Y.; Michel, M.; Lauschke, V.M. Primary Human Tissue Models for Metabolic Dysfunction-Associated Liver Disease - toward Streamlining Drug Discovery with Patient-Derived Assays. Advanced Biology 2025, 9, e00337. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.; Gottmann, P.; Wang, S.; Tissink, J.; Motzler, K.; Sekar, R.; Albrecht, W.; Cadenas, C.; Hengstler, J.G.; Schürmann, A.; et al. Induction of Steatosis in Primary Human Hepatocytes Recapitulates Key Pathophysiological Aspects of Metabolic Dysfunction-Associated Steatotic Liver Disease. Journal of Hepatology 2025, 82, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Kruepunga, N.; Hakvoort, T.B.M.; Hikspoors, J.P.J.M.; Köhler, S.E.; Lamers, W.H. Anatomy of Rodent and Human Livers: What Are the Differences? Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 2019, 1865, 869–878. [Google Scholar] [CrossRef]
- Luo, Y.; Lu, H.; Peng, D.; Ruan, X.; Eugene Chen, Y.; Guo, Y. Liver-humanized Mice: A Translational Strategy to Study Metabolic Disorders. Journal Cellular Physiology 2022, 237, 489–506. [Google Scholar] [CrossRef]
- Nagarajan, P. Genetically Modified Mouse Models for the Study of Nonalcoholic Fatty Liver Disease. WJG 2012, 18, 1141. [Google Scholar] [CrossRef]
- Jahn, D.; Kircher, S.; Hermanns, H.M.; Geier, A. Animal Models of NAFLD from a Hepatologist’s Point of View. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 2019, 1865, 943–953. [Google Scholar] [CrossRef]
- Flessa, C.-M.; Nasiri-Ansari, N.; Kyrou, I.; Leca, B.M.; Lianou, M.; Chatzigeorgiou, A.; Kaltsas, G.; Kassi, E.; Randeva, H.S. Genetic and Diet-Induced Animal Models for Non-Alcoholic Fatty Liver Disease (NAFLD) Research. IJMS 2022, 23, 15791. [Google Scholar] [CrossRef]
- Fu, Y.; Hua, Y.; Alam, N.; Liu, E. Progress in the Study of Animal Models of Metabolic Dysfunction-Associated Steatotic Liver Disease. Nutrients 2024, 16, 3120. [Google Scholar] [CrossRef]
- Cui, X.; Li, H.; Li, L.; Xie, C.; Gao, J.; Chen, Y.; Zhang, H.; Hao, W.; Fu, J.; Guo, H. Rodent Model of Metabolic Dysfunction-associated Fatty Liver Disease: A Systematic Review. J of Gastro and Hepatol 2025, 40, 48–66. [Google Scholar] [CrossRef]
- Vacca, M.; Kamzolas, I.; Harder, L.M.; Oakley, F.; Trautwein, C.; Hatting, M.; Ross, T.; Bernardo, B.; Oldenburger, A.; Hjuler, S.T.; et al. An Unbiased Ranking of Murine Dietary Models Based on Their Proximity to Human Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD). Nat Metab 2024, 6, 1178–1196. [Google Scholar] [CrossRef]
- Chen, J.; Jiang, X. A High-Fructose Diet Leads to Osteoporosis by Suppressing the Expression of Thrb and Facilitating the Accumulation of Cholesterol. Cell Death Discov. 2025, 11, 159. [Google Scholar] [CrossRef] [PubMed]
- Estévez-Vázquez, O.; Benedé-Ubieto, R.; Guo, F.; Gómez-Santos, B.; Aspichueta, P.; Reissing, J.; Bruns, T.; Sanz-García, C.; Sydor, S.; Bechmann, L.P.; et al. Fat: Quality, or Quantity? What Matters Most for the Progression of Metabolic Associated Fatty Liver Disease (MAFLD). Biomedicines 2021, 9, 1289. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.H.; Nøhr-Meldgaard, J.; Møllerhøj, M.B.; Oró, D.; Pors, S.E.; Andersen, M.W.; Kamzolas, I.; Petsalaki, E.; Vacca, M.; Harder, L.M.; et al. Characterization of Six Clinical Drugs and Dietary Intervention in the Nonobese CDAA-HFD Mouse Model of MASH and Progressive Fibrosis. American Journal of Physiology-Gastrointestinal and Liver Physiology 2025, 328, G51–G71. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Toth, E.; Cherrington, N.J. Asking the Right Questions With Animal Models: Methionine- and Choline-Deficient Model in Predicting Adverse Drug Reactions in Human NASH. Toxicological Sciences 2018, 161, 23–33. [Google Scholar] [CrossRef]
- Fang, T.; Wang, H.; Pan, X.; Little, P.J.; Xu, S.; Weng, J. Mouse Models of Nonalcoholic Fatty Liver Disease (NAFLD): Pathomechanisms and Pharmacotherapies. Int. J. Biol. Sci. 2022, 18, 5681–5697. [Google Scholar] [CrossRef]
- Pompili, S.; Vetuschi, A.; Gaudio, E.; Tessitore, A.; Capelli, R.; Alesse, E.; Latella, G.; Sferra, R.; Onori, P. Long-Term Abuse of a High-Carbohydrate Diet Is as Harmful as a High-Fat Diet for Development and Progression of Liver Injury in a Mouse Model of NAFLD/NASH. Nutrition 2020, 75–76, 110782. [Google Scholar] [CrossRef]
- Maddie, N.; Chacko, N.; Matatov, D.; Carrillo-Sepulveda, M.A. Western Diet Promotes the Progression of Metabolic Dysfunction-associated Steatotic Liver Disease in Association with Ferroptosis in Male Mice. Physiological Reports 2024, 12, e70139. [Google Scholar] [CrossRef]
- Meijnikman, A.S.; Fondevila, M.F.; Arrese, M.; Kisseleva, T.; Bataller, R.; Schnabl, B. Towards More Consistent Models and Consensual Terminology in Preclinical Research for Steatotic Liver Disease. Journal of Hepatology 2025, 82, 760–766. [Google Scholar] [CrossRef]
- Svobodová, G.; Horní, M.; Velecká, E.; Boušová, I. Metabolic Dysfunction-Associated Steatotic Liver Disease-Induced Changes in the Antioxidant System: A Review. Arch Toxicol 2025, 99, 1–22. [Google Scholar] [CrossRef]
- Ma, X.; Bian, W.; Song, W.; Lu, Y.; Wang, Z.; Yao, Z.; Xuan, Q. Metabolome Profiling across Liver Lobes and Metabolic Shifts of the MASLD Mice. Genes Nutr 2025, 20, 9. [Google Scholar] [CrossRef]
- Li, Y.-Q.; Huang, C.; Chen, J.; Yang, S.; Cheng, J.; Chen, H.; Zhou, Y. Identification of the Role of Sugar-Sweetened Beverages in the Progression of a Murine Metabolic Dysfunction-Associated Steatotic Liver Disease Model. Front. Nutr. 2025, 12, 1710267. [Google Scholar] [CrossRef]
- Jeon, H.J.; Rou, W.S.; Kim, S.H.; Lee, B.S.; Kim, H.N.; Choi, H.-G.; Seo, J.; Eun, H.S.; Jung, S. Sugar-Sweetened Beverage Consumption and Metabolic Dysfunction-Associated Steatotic Liver Disease: A Beverage Type-Specific Analysis Using Korea National Health and Nutrition Examination Survey. Epidemiol Health 2025, 47, e2025038. [Google Scholar] [CrossRef]
- Hsu, W.-F.; Lee, M.-H.; Lii, C.-K.; Peng, C.-Y. No Difference in Liver Damage Induced by Isocaloric Fructose or Glucose in Mice with a High-Fat Diet. Nutrients 2024, 16, 3571. [Google Scholar] [CrossRef]
- Zhang, Z.; Qin, X.; Yi, T.; Li, Y.; Li, C.; Zeng, M.; Luo, H.; Lin, X.; Xie, J.; Xia, B.; et al. Gubra Amylin-NASH Diet Induced Nonalcoholic Fatty Liver Disease Associated with Histological Damage, Oxidative Stress, Immune Disorders, Gut Microbiota, and Its Metabolic Dysbiosis in Colon. Molecular Nutrition Food Res 2024, 68, 2300845. [Google Scholar] [CrossRef]
- Green, C.D.; Weigel, C.; Brown, R.D.R.; Bedossa, P.; Dozmorov, M.; Sanyal, A.J.; Spiegel, S. A New Preclinical Model of Western Diet-induced Progression of Non-alcoholic Steatohepatitis to Hepatocellular Carcinoma. The FASEB Journal 2022, 36, e22372. [Google Scholar] [CrossRef]
- Makri, E.S.; Xanthopoulos, K.; Mavrommatis Parasidis, P.; Makri, E.; Pettas, S.; Tsingotjidou, A.; Cheva, A.; Ballaouri, I.; Gerou, S.; Goulas, A.; et al. Partial Validation of a Six-Month High-Fat Diet and Fructose-Glucose Drink Combination as a Mouse Model of Nonalcoholic Fatty Liver Disease. Endocrine 2024, 85, 704–716. [Google Scholar] [CrossRef] [PubMed]
- Khoj, D.; Huang, R.; Altvater, E.; Ishfaq, Z.N.; Jiang, X.; Axen, K.V.; Caviglia, J.M. Mouse Model of Metabolic Dysfunction-Associated Steatotic Liver Disease with Fibrosis. JoVE 2025, 68294. [Google Scholar] [CrossRef] [PubMed]
- Hansen, H.H.; Ægidius, H.M.; Oró, D.; Evers, S.S.; Heebøll, S.; Eriksen, P.L.; Thomsen, K.L.; Bengtsson, A.; Veidal, S.S.; Feigh, M.; et al. Human Translatability of the GAN Diet-Induced Obese Mouse Model of Non-Alcoholic Steatohepatitis. BMC Gastroenterol 2020, 20, 210. [Google Scholar] [CrossRef] [PubMed]
- Dutta, S.; Sengupta, P. Men and Mice: Relating Their Ages. Life Sciences 2016, 152, 244–248. [Google Scholar] [CrossRef]
- Jackson, S.J.; Andrews, N.; Ball, D.; Bellantuono, I.; Gray, J.; Hachoumi, L.; Holmes, A.; Latcham, J.; Petrie, A.; Potter, P.; et al. Does Age Matter? The Impact of Rodent Age on Study Outcomes. Lab Anim 2017, 51, 160–169. [Google Scholar] [CrossRef]
- Li, X.; Lu, Y.; Liang, X.; Zhou, X.; Li, D.; Zhang, Z.; Niu, Y.; Liu, S.; Ye, L.; Zhang, R. A New NASH Model in Aged Mice with Rapid Progression of Steatohepatitis and Fibrosis. PLoS ONE 2023, 18, e0286257. [Google Scholar] [CrossRef]
- Hansen, H.H.; Pors, S.; Andersen, M.W.; Vyberg, M.; Nøhr-Meldgaard, J.; Nielsen, M.H.; Oró, D.; Madsen, M.R.; Lewinska, M.; Møllerhøj, M.B.; et al. Semaglutide Reduces Tumor Burden in the GAN Diet-Induced Obese and Biopsy-Confirmed Mouse Model of NASH-HCC with Advanced Fibrosis. Sci Rep 2023, 13, 23056. [Google Scholar] [CrossRef]
- Benegiamo, G.; Von Alvensleben, G.V.G.; Rodríguez-López, S.; Goeminne, L.J.E.; Bachmann, A.M.; Morel, J.-D.; Broeckx, E.; Ma, J.Y.; Carreira, V.; Youssef, S.A.; et al. The Genetic Background Shapes the Susceptibility to Mitochondrial Dysfunction and NASH Progression. Journal of Experimental Medicine 2023, 220, e20221738. [Google Scholar] [CrossRef]
- Hupa-Breier, K.L.; Schenk, H.; Campos-Murguia, A.; Wellhöner, F.; Heidrich, B.; Dywicki, J.; Hartleben, B.; Böker, C.; Mall, J.; Terkamp, C.; et al. Novel Translational Mouse Models of Metabolic Dysfunction-Associated Steatotic Liver Disease Comparable to Human MASLD with Severe Obesity. Molecular Metabolism 2025, 93, 102104. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-C.; Lee, H.S.; Jeon, S.; Lee, Y.-J.; Kwon, Y.-J.; Lee, J.-W. Assessing Nutritional Factors for Metabolic Dysfunction-Associated Steatotic Liver Disease via Diverse Statistical Tools. Diabetes Metab J 2026, 50, 178–189. [Google Scholar] [CrossRef]
- Mann, J.P.; Semple, R.K.; Armstrong, M.J. How Useful Are Monogenic Rodent Models for the Study of Human Non-Alcoholic Fatty Liver Disease? Front. Endocrinol. 2016, 7. [Google Scholar] [CrossRef]
- Hintze, K.J.; Benninghoff, A.D.; Cho, C.E.; Ward, R.E. Modeling the Western Diet for Preclinical Investigations. Advances in Nutrition 2018, 9, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.M.P.; Polizzi, A.; Alquier-Bacquié, V.; Huillet, M.; Rives, C.; Dauriat, C.J.G.; Bruse, J.; Melin, V.; Naylies, C.; Lippi, Y.; et al. Thermoneutral Housing Worsens MASLD and Reveals Defective Brown Adipose Tissue Response to Β3-Adrenergic Stimulation. iScience 2025, 28, 113221. [Google Scholar] [CrossRef]
- Blok, N.B.; Myronovych, A.; McMahon, G.; Bozadjieva-Kramer, N.; Seeley, R.J. The Evolution of Steatosis and Fibrosis in Mice on a MASH-Inducing Diet and the Effects of Housing Temperature. American Journal of Physiology-Endocrinology and Metabolism 2025, 328, E513–E523. [Google Scholar] [CrossRef]
- Jancova, P.; Ismail, K.; Vistejnova, L. Relationship between MASLD and Women’s Health: A Review. Womens Health (Lond Engl) 2025, 21, 17455057251376883. [Google Scholar] [CrossRef]
- Alves, E.S.; Santos, J.D.M.; Cruz, A.G.; Camargo, F.N.; Talarico, C.H.Z.; Santos, A.R.M.; Silva, C.A.A.; Morgan, H.J.N.; Matos, S.L.; Araujo, L.C.C.; et al. Hepatic Estrogen Receptor Alpha Overexpression Protects Against Hepatic Insulin Resistance and MASLD. Pathophysiology 2025, 32, 1. [Google Scholar] [CrossRef]
- Korovila, I.; Höhn, A.; Jung, T.; Grune, T.; Ott, C. Reduced Liver Autophagy in High-Fat Diet Induced Liver Steatosis in New Zealand Obese Mice. Antioxidants 2021, 10, 501. [Google Scholar] [CrossRef] [PubMed]
- Inzalaco, G.; Gargiulo, S.; Bonente, D.; Gherardini, L.; Franci, L.; Lorito, N.; Del Turco, S.; Tatoni, D.; Tamborrino, T.; Galvagni, F.; et al. MAPK15 Controls Intracellular Lipid Uptake and Protects Mammalian Liver from Steatotic Disease. Hepatology Communications 2026, 10. [Google Scholar] [CrossRef]
- Sakane, S.; Hikita, H.; Shirai, K.; Myojin, Y.; Sasaki, Y.; Kudo, S.; Fukumoto, K.; Mizutani, N.; Tahata, Y.; Makino, Y.; et al. White Adipose Tissue Autophagy and Adipose-Liver Crosstalk Exacerbate Nonalcoholic Fatty Liver Disease in Mice. Cellular and Molecular Gastroenterology and Hepatology 2021, 12, 1683–1699. [Google Scholar] [CrossRef]
- Ji, L.; Zhao, Y.; He, L.; Zhao, J.; Gao, T.; Liu, F.; Qi, B.; Kang, F.; Wang, G.; Zhao, Y.; et al. X1AKAP1 Deficiency Attenuates Diet-Induced Obesity and Insulin Resistance by Promoting Fatty Acid Oxidation and Thermogenesis in Brown Adipocytes. Advanced Science 2022, 9, 2204669. [Google Scholar] [CrossRef]
- He, L.; She, X.; Guo, L.; Gao, M.; Wang, S.; Lu, Z.; Guo, H.; Li, R.; Nie, Y.; Xing, J.; et al. Hepatic AKAP1 Deficiency Exacerbates Diet-Induced MASLD by Enhancing GPAT1-Mediated Lysophosphatidic Acid Synthesis. Nat Commun 2025, 16, 4286. [Google Scholar] [CrossRef] [PubMed]
- Makri, E.S.; Mouskeftara, T.; Gika, H.; Xanthopoulos, K.; Makri, E.; Mavrommatis-Parasidis, P.; Tsingotjidou, A.; Cheva, A.; Goulas, A.; Polyzos, S.A. Serum and Liver Lipidome Following Empagliflozin Administration for Six Months in a Fast Food Diet Mouse Model. IJMS 2025, 26, 9273. [Google Scholar] [CrossRef]
- Gargiulo, S.; Barone, V.; Bonente, D.; Tamborrino, T.; Inzalaco, G.; Gherardini, L.; Bertelli, E.; Chiariello, M. Integrated Ultrasound Characterization of the Diet-Induced Obesity (DIO) Model in Young Adult C57bl/6j Mice: Assessment of Cardiovascular, Renal and Hepatic Changes. J. Imaging 2024, 10, 217. [Google Scholar] [CrossRef] [PubMed]
- Burelle, C.; Clapatiuc, V.; Deschênes, S.; Cuillerier, A.; De Loof, M.; Higgins, M.-È.; Boël, H.; Daneault, C.; Chouinard, B.; Clavet, M.-É.; et al. A Genetic Mouse Model of Lean-NAFLD Unveils Sexual Dimorphism in the Liver-Heart Axis. Commun Biol 2024, 7, 356. [Google Scholar] [CrossRef]
- Zhang, X.; Lau, H.C.-H.; Ha, S.; Liu, C.; Liang, C.; Lee, H.W.; Ng, Q.W.-Y.; Zhao, Y.; Ji, F.; Zhou, Y.; et al. Intestinal TM6SF2 Protects against Metabolic Dysfunction-Associated Steatohepatitis through the Gut–Liver Axis. Nat Metab 2025, 7, 102–119. [Google Scholar] [CrossRef] [PubMed]
- Fisher-Wellman, K.H.; Ryan, T.E.; Smith, C.D.; Gilliam, L.A.A.; Lin, C.-T.; Reese, L.R.; Torres, M.J.; Neufer, P.D. A Direct Comparison of Metabolic Responses to High-Fat Diet in C57BL/6J and C57BL/6NJ Mice. Diabetes 2016, 65, 3249–3261. [Google Scholar] [CrossRef] [PubMed]
- Ueda, H.; Honda, A.; Miyazaki, T.; Morishita, Y.; Hirayama, T.; Iwamoto, J.; Ikegami, T. High-Fat/High-Sucrose Diet Results in a High Rate of MASH with HCC in a Mouse Model of Human-like Bile Acid Composition. Hepatology Communications 2025, 9. [Google Scholar] [CrossRef]
- Takahashi, Y. Histopathology of Nonalcoholic Fatty Liver Disease/Nonalcoholic Steatohepatitis. WJG 2014, 20, 15539. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.T.; Kleiner, D.E. Histopathology of Nonalcoholic Fatty Liver Disease and Nonalcoholic Steatohepatitis. Metabolism 2016, 65, 1080–1086. [Google Scholar] [CrossRef]
- Goodman, Z.D. Role of Liver Biopsy in Clinical Trials and Clinical Management of Nonalcoholic Fatty Liver Disease. Clinics in Liver Disease 2023, 27, 353–362. [Google Scholar] [CrossRef]
- Kim, H.Y.; Rosenthal, S.B.; Liu, X.; Miciano, C.; Hou, X.; Miller, M.; Buchanan, J.; Poirion, O.B.; Chilin-Fuentes, D.; Han, C.; et al. Multi-Modal Analysis of Human Hepatic Stellate Cells Identifies Novel Therapeutic Targets for Metabolic Dysfunction-Associated Steatotic Liver Disease. Journal of Hepatology 2025, 82, 882–897. [Google Scholar] [CrossRef]
- Cobelo-Gómez, S.; García-Formoso, L.; Fernández-Pombo, A.; Lázare-Iglesias, H.; Díaz-López, E.; Prado-Moraña, T.; Rodríguez-Sobrino, L.; Senra, A.; Araújo-Vilar, D.; Sánchez-Iglesias, S. Metabolic-Associated Steatotic Liver Disease and FGF21 Dysregulation in Seipin-Deficient and BSCL2-Associated Celia’s Encephalopathy Murine Models. IJMS 2025, 26, 12037. [Google Scholar] [CrossRef]
- Jeong, B.-K.; Choi, W.-I.; Choi, W.; Moon, J.; Lee, W.H.; Choi, C.; Choi, I.Y.; Lee, S.-H.; Kim, J.K.; Ju, Y.S.; et al. A Male Mouse Model for Metabolic Dysfunction-Associated Steatotic Liver Disease and Hepatocellular Carcinoma. Nat Commun 2024, 15, 6506. [Google Scholar] [CrossRef]
- Bedossa, P. [Presentation of a grid for computer analysis for compilation of histopathologic lesions in chronic viral hepatitis C. Cooperative study of the METAVIR group]. Ann Pathol 1993, 13, 260–265. [Google Scholar]
- Kleiner, D.E.; Brunt, E.M.; Van Natta, M.; Behling, C.; Contos, M.J.; Cummings, O.W.; Ferrell, L.D.; Liu, Y.; Torbenson, M.S.; Unalp-Arida, A.; et al. Design and Validation of a Histological Scoring System for Nonalcoholic Fatty Liver Disease†. Hepatology 2005, 41, 1313–1321. [Google Scholar] [CrossRef] [PubMed]
- Brunt, E.M.; Janney, C.G.; Di Bisceglie, A.M.; Neuschwander-Tetri, B.A.; Bacon, B.R. Nonalcoholic Steatohepatitis: A Proposal for Grading and Staging The Histological Lesions. American Journal of Gastroenterology 1999, 94, 2467–2474. [Google Scholar] [CrossRef]
- Brunt, E.M.; Kleiner, D.E.; Wilson, L.A.; Belt, P.; Neuschwander-Tetri, B.A. Nonalcoholic Fatty Liver Disease (NAFLD) Activity Score and the Histopathologic Diagnosis in NAFLD: Distinct Clinicopathologic Meanings §Δ. Hepatology 2011, 53, 810–820. [Google Scholar] [CrossRef] [PubMed]
- Bedossa, P.; Poitou, C.; Veyrie, N.; Bouillot, J.-L.; Basdevant, A.; Paradis, V.; Tordjman, J.; Clement, K. Histopathological Algorithm and Scoring System for Evaluation of Liver Lesions in Morbidly Obese Patients. Hepatology 2012, 56, 1751–1759. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Menke, A.L.; Driessen, A.; Koek, G.H.; Lindeman, J.H.; Stoop, R.; Havekes, L.M.; Kleemann, R.; Van Den Hoek, A.M. Establishment of a General NAFLD Scoring System for Rodent Models and Comparison to Human Liver Pathology. PLoS ONE 2014, 9, e115922. [Google Scholar] [CrossRef]
- Liss, K.H.H.; McCommis, K.S.; Chambers, K.T.; Pietka, T.A.; Schweitzer, G.G.; Park, S.L.; Nalbantoglu, I.; Weinheimer, C.J.; Hall, A.M.; Finck, B.N. The Impact of Diet-induced Hepatic Steatosis in a Murine Model of Hepatic Ischemia/Reperfusion Injury. Liver Transpl 2018, 24, 908–921. [Google Scholar] [CrossRef]
- Yan, M.; Cui, Y.; Xiang, Q. Metabolism of Hepatic Stellate Cells in Chronic Liver Diseases: Emerging Molecular and Therapeutic Interventions. Theranostics 2025, 15, 1715–1740. [Google Scholar] [CrossRef]
- Yashaswini, C.N.; Qin, T.; Bhattacharya, D.; Amor, C.; Lowe, S.; Lujambio, A.; Wang, S.; Friedman, S.L. Phenotypes and Ontogeny of Senescent Hepatic Stellate Cells in Metabolic Dysfunction-Associated Steatohepatitis. Journal of Hepatology 2024, 81, 207–217. [Google Scholar] [CrossRef]
- Ramachandran, P.; Brice, M.; Sutherland, E.F.; Hoy, A.M.; Papachristoforou, E.; Jia, L.; Turner, F.; Kendall, T.J.; Marwick, J.A.; Carragher, N.O.; et al. Aberrant Basement Membrane Production by HSCs in MASLD Is Attenuated by the Bile Acid Analog INT-767. Hepatology Communications 2024, 8. [Google Scholar] [CrossRef]
- Wattacheril, J.J.; Abdelmalek, M.F.; Lim, J.K.; Sanyal, A.J. AGA Clinical Practice Update on the Role of Noninvasive Biomarkers in the Evaluation and Management of Nonalcoholic Fatty Liver Disease: Expert Review. Gastroenterology 2023, 165, 1080–1088. [Google Scholar] [CrossRef] [PubMed]
- Neuberger, J.; Patel, J.; Caldwell, H.; Davies, S.; Hebditch, V.; Hollywood, C.; Hubscher, S.; Karkhanis, S.; Lester, W.; Roslund, N.; et al. Guidelines on the Use of Liver Biopsy in Clinical Practice from the British Society of Gastroenterology, the Royal College of Radiologists and the Royal College of Pathology. Gut 2020, 69, 1382–1403. [Google Scholar] [CrossRef] [PubMed]
- Bassegoda, O.; Olivas, P.; Turco, L.; Mandorfer, M.; Serra-Burriel, M.; Tellez, L.; Kwanten, W.; Laroyenne, A.; Farcau, O.; Alvarado, E.; et al. Decompensation in Advanced Nonalcoholic Fatty Liver Disease May Occur at Lower Hepatic Venous Pressure Gradient Levels Than in Patients With Viral Disease. Clin Gastroenterol Hepatol 2022, 20, 2276–2286.e6. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, S.N.; Magdaleno, T.; Klocksieben, F.; MacFarlan, J.E.; Goonewardene, S.; Zator, Z.; Shah, S.; Shah, H.N. A Prospective, Head-to-Head Comparison of 2 EUS-Guided Liver Biopsy Needles in Vivo. Gastrointestinal Endoscopy 2021, 93, 1133–1138. [Google Scholar] [CrossRef]
- Cassidy, F.H.; Yokoo, T.; Aganovic, L.; Hanna, R.F.; Bydder, M.; Middleton, M.S.; Hamilton, G.; Chavez, A.D.; Schwimmer, J.B.; Sirlin, C.B. Fatty Liver Disease: MR Imaging Techniques for the Detection and Quantification of Liver Steatosis. Radiographics 2009, 29, 231–260. [Google Scholar] [CrossRef]
- Pineda, N.; Sharma, P.; Xu, Q.; Hu, X.; Vos, M.; Martin, D.R. Measurement of Hepatic Lipid: High-Speed T2-Corrected Multiecho Acquisition at1 H MR Spectroscopy—A Rapid and Accurate Technique. Radiology 2009, 252, 568–576. [Google Scholar] [CrossRef]
- Heinemann, F.; Gross, P.; Zeveleva, S.; Qian, H.S.; Hill, J.; Höfer, A.; Jonigk, D.; Diehl, A.M.; Abdelmalek, M.; Lenter, M.C.; et al. Deep Learning-Based Quantification of NAFLD/NASH Progression in Human Liver Biopsies. Sci Rep 2022, 12, 19236. [Google Scholar] [CrossRef]
- Huang, Q.; Qadri, S.F.; Bian, H.; Yi, X.; Lin, C.; Yang, X.; Zhu, X.; Lin, H.; Yan, H.; Chang, X.; et al. A Metabolome-Derived Score Predicts Metabolic Dysfunction-Associated Steatohepatitis and Mortality from Liver Disease. Journal of Hepatology 2025, 82, 781–793. [Google Scholar] [CrossRef]
- Frączek, J.; Sowa, A.; Agopsowicz, P.; Migacz, M.; Dylińska-Kala, K.; Holecki, M. Non-Invasive Tests as a Replacement for Liver Biopsy in the Assessment of MASLD. Medicina 2025, 61, 736. [Google Scholar] [CrossRef]
- Aggarwal, P.; Alkhouri, N. Artificial Intelligence in Nonalcoholic Fatty Liver Disease: A New Frontier in Diagnosis and Treatment. Clinical Liver Disease 2021, 17, 392–397. [Google Scholar] [CrossRef]
- Goh, G.B.-B.; Leow, W.Q.; Liang, S.; Wan, W.K.; Lim, T.K.H.; Tan, C.K.; Chang, P.E. Quantification of Hepatic Steatosis in Chronic Liver Disease Using Novel Automated Method of Second Harmonic Generation and Two-Photon Excited Fluorescence. Sci Rep 2019, 9, 2975. [Google Scholar] [CrossRef] [PubMed]
- Ferraioli, G.; Barr, R.G.; Berzigotti, A.; Sporea, I.; Wong, V.W.-S.; Reiberger, T.; Karlas, T.; Thiele, M.; Cardoso, A.C.; Ayonrinde, O.T.; et al. WFUMB Guidelines/Guidance on Liver Multiparametric Ultrasound. Part 2: Guidance on Liver Fat Quantification. Ultrasound Med Biol 2024, 50, 1088–1098. [Google Scholar] [CrossRef]
- Yin, H.; Xiong, B.; Yu, J.; Fan, Y.; Zhou, B.; Sun, Y.; Wang, L.; Xu, H.; Zhu, Y. Interoperator Reproducibility of Quantitative Ultrasound Analysis of Hepatic Steatosis in Participants with Suspected MASLD: A Prospective Study. European Journal of Radiology 2024, 175, 111427. [Google Scholar] [CrossRef]
- Ferraioli, G.; Monteiro, L.B.S. Ultrasound-Based Techniques for the Diagnosis of Liver Steatosis. WJG 2019, 25, 6053–6062. [Google Scholar] [CrossRef]
- Pirmoazen, A.M.; Khurana, A.; El Kaffas, A.; Kamaya, A. Quantitative Ultrasound Approaches for Diagnosis and Monitoring Hepatic Steatosis in Nonalcoholic Fatty Liver Disease. Theranostics 2020, 10, 4277–4289. [Google Scholar] [CrossRef]
- Starekova, J.; Hernando, D.; Pickhardt, P.J.; Reeder, S.B. Quantification of Liver Fat Content with CT and MRI: State of the Art. Radiology 2021, 301, 250–262. [Google Scholar] [CrossRef] [PubMed]
- Khalid, W.B.; Farhat, N.; Lavery, L.; Jarnagin, J.; Delany, J.P.; Kim, K. Non-Invasive Assessment of Liver Fat in Ob/Ob Mice Using Ultrasound-Induced Thermal Strain Imaging and Its Correlation with Hepatic Triglyceride Content. Ultrasound in Medicine & Biology 2021, 47, 1067–1076. [Google Scholar] [CrossRef]
- Sha, T.; You, Y.; Miao, X.; Deng, H.; Zhang, W.; Ye, H.; Wang, P.; Zheng, R.; Ren, J.; Yin, T. Sequential Ultrasound Molecular Imaging for Noninvasive Identification and Assessment of Non-Alcoholic Steatohepatitis in Mouse Models. Liver Research 2023, 7, 342–351. [Google Scholar] [CrossRef]
- Czernuszewicz, T.J.; Wang, Y.; Jiang, L.; Kim, K.; Mikulski, Z.; Aji, A.M.; Rojas, J.D.; Gessner, R.C.; Schnabl, B. Noninvasive Monitoring of Steatotic Liver Disease in Western Diet-Fed Obese Mice Using Automated Ultrasound and Shear Wave Elastography. Liver International 2025, 45, e16141. [Google Scholar] [CrossRef]
- Amin, M.N.; Rushdi, M.A.; Marzaban, R.N.; Yosry, A.; Kim, K.; Mahmoud, A.M. Wavelet-Based Computationally-Efficient Computer-Aided Characterization of Liver Steatosis Using Conventional B-Mode Ultrasound Images. Biomedical Signal Processing and Control 2019, 52, 84–96. [Google Scholar] [CrossRef]
- De Rosa, L.; L’Abbate, S.; Kusmic, C.; Faita, F. Applications of Deep Learning Algorithms to Ultrasound Imaging Analysis in Preclinical Studies on In Vivo Animals. Life 2023, 13, 1759. [Google Scholar] [CrossRef]
- Wibulpolprasert, P.; Subpinyo, B.; Chirnaksorn, S.; Shantavasinkul, P.C.; Putadechakum, S.; Phongkitkarun, S.; Sritara, C.; Angkathunyakul, N.; Sumritpradit, P. Correlation between Magnetic Resonance Imaging Proton Density Fat Fraction (MRI-PDFF) and Liver Biopsy to Assess Hepatic Steatosis in Obesity. Sci Rep 2024, 14, 6895. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Washington, M.K.; Izzy, M.J.; Piantek, G.; Lu, M.; Yan, X.; Gore, J.C.; Xu, J. Noninvasive Assessment of Liver Inflammation in Metabolic Dysfunction Associated Steatohepatitis Using MR Cytometry. npj Imaging 2025, 3, 17. [Google Scholar] [CrossRef] [PubMed]
- Bohte, A.E.; Koot, B.G.P.; Van Der Baan-Slootweg, O.H.; Rijcken, T.H.P.; Van Werven, J.R.; Bipat, S.; Nederveen, A.J.; Jansen, P.L.M.; Benninga, M.A.; Stoker, J. US Cannot Be Used to Predict the Presence or Severity of Hepatic Steatosis in Severely Obese Adolescents. Radiology 2012, 262, 327–334. [Google Scholar] [CrossRef]
- Lee, S.S.; Park, S.H.; Kim, H.J.; Kim, S.Y.; Kim, M.-Y.; Kim, D.Y.; Suh, D.J.; Kim, K.M.; Bae, M.H.; Lee, J.Y.; et al. Non-Invasive Assessment of Hepatic Steatosis: Prospective Comparison of the Accuracy of Imaging Examinations. Journal of Hepatology 2010, 52, 579–585. [Google Scholar] [CrossRef]
- Van Werven, J.R.; Marsman, H.A.; Nederveen, A.J.; Smits, N.J.; Ten Kate, F.J.; Van Gulik, T.M.; Stoker, J. Assessment of Hepatic Steatosis in Patients Undergoing Liver Resection: Comparison of US, CT, T1-Weighted Dual-Echo MR Imaging, and Point-Resolved1 H MR Spectroscopy. Radiology 2010, 256, 159–168. [Google Scholar] [CrossRef]
- Xavier, A.; Zacconi, F.; Santana-Romo, F.; Eykyn, T.R.; Lavin, B.; Phinikaridou, A.; Botnar, R.; Uribe, S.; Oyarzún, J.E.; Cabrera, D.; et al. Assessment of Hepatic Fatty Acids during Non-Alcoholic Steatohepatitis Progression Using Magnetic Resonance Spectroscopy. Annals of Hepatology 2021, 25, 100358. [Google Scholar] [CrossRef]
- Waghorn, P.A.; Ferreira, D.S.; Erstad, D.J.; Rotile, N.J.; Masia, R.; Jones, C.M.; Tu, C.; Sojoodi, M.; Chen, Y.I.; Schlerman, F.; et al. Quantitative, Noninvasive MRI Characterization of Disease Progression in a Mouse Model of Non-Alcoholic Steatohepatitis. Sci Rep 2021, 11, 6105. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Li, J.; Zinker, B.; Boehm, S.; Mauer, A.; Rex-Rabe, S.; Glaser, K.J.; Fronheiser, M.; Bradstreet, T.; Nakao, Y.; et al. Evaluation of a PEGylated Fibroblast Growth Factor 21 Variant Using Novel Preclinical Magnetic Resonance Imaging and Magnetic Resonance Elastography in a Mouse Model of Nonalcoholic Steatohepatitis. Magnetic Resonance Imaging 2022, 56, 712–724. [Google Scholar] [CrossRef]
- Xia, H.; Min, Y.; Wang, Y.; Gao, S.; Wang, H.; Yan, F.; Liu, R.; Wang, J.; Gu, X.; Bo, T. Multiparametric MRI Evaluation of Liver Fat and Iron after Glucagon-like Peptide-1 Receptor and Glucagon Receptor Dual-Agonist Treatment in a High-Fat Diet–Induced Mouse Model. Radiology 2025, 316, e243780. [Google Scholar] [CrossRef] [PubMed]
- Lavin, B.; Eykyn, T.R.; Phinikaridou, A.; Xavier, A.; Kumar, S.; Buqué, X.; Aspichueta, P.; Sing-Long, C.; Arrese, M.; Botnar, R.M.; et al. Characterization of Hepatic Fatty Acids Using Magnetic Resonance Spectroscopy for the Assessment of Treatment Response to Metformin in an eNOS−/− Mouse Model of Metabolic Nonalcoholic Fatty Liver Disease/Nonalcoholic Steatohepatitis. NMR in Biomedicine 2023, 36, e4932. [Google Scholar] [CrossRef] [PubMed]
- Lister, D.; Blizard, G.; Hosseini, M.; Messer, K.; Wellen, J.; Sirlin, C.B.; Ahrens, E.T. Imaging Non-Alcoholic Fatty Liver Disease Model Using H-1 and F-19 MRI. Mol Imaging Biol 2023, 25, 443–449. [Google Scholar] [CrossRef] [PubMed]
- McLeod, M.; Chang, M.C.; Rushin, A.; Ragavan, M.; Mahar, R.; Sharma, G.; Badar, A.; Giacalone, A.; Glanz, M.E.; Malut, V.R.; et al. Detecting Altered Hepatic Lipid Oxidation by MRI in an Animal Model of MASLD. Cell Reports Medicine 2024, 5, 101714. [Google Scholar] [CrossRef] [PubMed]
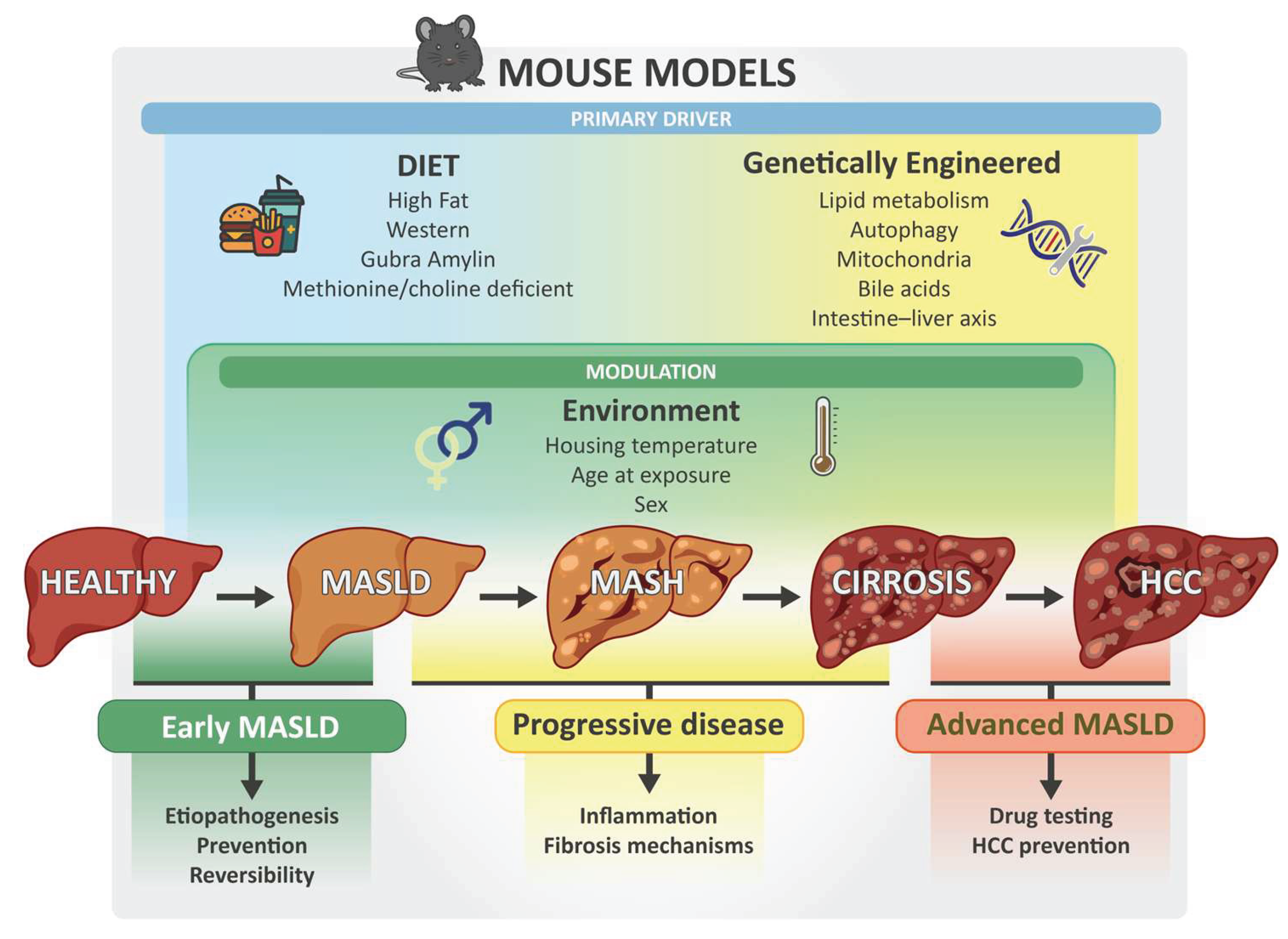
| Mouse age and model | Sex |
MASLD induction |
Key characteristics for MASLD research | Ref. | |
| Liver | Systemic | ||||
| 10-weeks-old C57BL/6J | Male | CD 20 weeks | Hepatomegaly Steatosis Inflammation Fibrosis HCC |
↓ BW ↓ plasmatic transaminases, insulin and glucose |
[35] |
| 4-weeks-old C57BL/6J | Male | HFD 17 months |
Hepatomegaly Steatosis |
↑ BW ↑ IR Transcriptome changes in collagen and lipid regulatory genes |
[38] |
| 7 weeks-old NZO C57BL/6J |
Male | HFD 32 weeks |
Hepatomegaly Steatosis |
↑ BW ↑ autophagy-related proteins |
[64] |
| 8-weeks-old C57BL/6J | Male and female | WD 17 weeks |
Hepatomegaly Steatosis |
↑ BW sexual dimorphism ↑ plasmatic transaminases, cholesterol, insulin and glucose In vivo heart, kidney, liver US alterations Kidney alterations |
[70] |
| 8-weeks-old C57BL/6J | Male | WD 16 weeks |
Metabolic changes comparable to humans | Changes in lipidome and metabolome profiles comparable to those in humans | [42] |
| 8-weeks-old C57BL/6N | Male | WD + fructose 10 weeks |
Hepatomegaly Steatosis |
↑ BW IR ↑ plasmatic transaminases, cholesterol, insulin and glucose autophagy-related transcriptome changes |
[53] |
| 6 weeks-old C57BL/6J | Male | WD + fructose/glucose 24 weeks |
Hepatomegaly Steatosis Inflammation Fibrosis |
↑ BW IR ↑ plasmatic transaminases, cholesterol, insulin and glucose ↑ lipogenic enzymes ↑ oxidative stress markers ↑ lipid peroxidation |
[45] |
| 8 weeks-old C57BL/6J | Male | WD + fructose/glucose 25 weeks | Hepatomegaly Steatosis |
↑ BW IR ↑ plasmatic transaminases, cholesterol, insulin and glucose |
[48] |
| 10-weeks-old C57BL/6J | Male | WD + TN 13 weeks |
Hepatomegaly Steatosis Inflammation Fibrosis |
↑ BW IR ↑ plasmatic insulin and glucose Transcriptome changes in response to β3-adrenergic stimulation |
[60] |
| 8 weeks-old C57BL/6J | Male | GAN + TN 7 months |
Steatosis Inflammation |
↑ BW ↑ plasmatic transaminases |
[61] |
| 8 weeks-old C57BL/6J | Male | GAN 44 weeks |
Hepatomegaly Steatosis Inflammation Fibrosis |
↑ BW IR ↑ plasmatic transaminases, cholesterol, insulin and glucose MASH-related transcriptomic alterations |
[50] |
| 6 weeks-old C57BL/6J | Male | GAN 10 weeks |
Hepatomegaly Steatosis Mild inflammation |
↑ BW ↑ plasmatic cholesterol Microbiota alterations |
[46] |
| 14 months-old C57BL/6J | Male and female | GAN 10 weeks |
Hepatomegaly Accelerated steatosis and inflammation |
↑ BW Sexual dimorphism Hepatomegaly ↑ plasmatic transaminases, cholesterol, insulin and glucose |
[53] |
| 6 weeks-old C57BL/6J | Male | GAN 72 weeks |
Hepatomegaly Steatosis Inflammation Fibrosis HCC |
↑ BW ↑ plasmatic transaminases ↑ fibrosis markers |
[54] |
| 8 weeks-old C57BL/6NJ | Male | WD + fructose/glucose 54 weeks | Hepatomegaly Steatosis Inflammation Fibrosis HCC |
↑ BW ↑ plasmatic transaminases, cholesterol, insulin and glucose |
[47] |
| 7 weeks-old PWK/PhJ C57BL/6J |
Male | WD + TN 18 weeks |
Hepatomegaly Steatosis Inflammation Fibrosis |
↑ BW IR ↑ plasmatic transaminases, cholesterol, insulin and glucose Transcriptomic and mitochondrial alterations |
[55] |
| AKAP1 KO C57BL/6N |
Male | HFD 24 weeks |
Steatosis | ↓ BW Hyperlipidemia ↑ thermogenesis |
[67] |
| 8-weeks-old liver specific- AKAP1 KO C57BL/6J |
Male | HFD/ WD + fructose/glucose 24 weeks |
Hepatomegaly Steatosis Inflammation Fibrosis |
↑ BW IR ↑ plasma lipids ↑ mitochondrial GPAT activity |
[68] |
| 8 weeks-old Atg7 KO C57BL/6 |
Male | HFD 8 months |
Hepatomegaly Steatosis Inflammation Fibrosis |
↓ BW ↑ plasmatic transaminases ↓ expression of autophagy-related proteins |
[66] |
| 8-weeks-old MAPK15 KO C57BL/6J |
Male and female | WD 17 weeks |
Hepatomegaly Steatosis Mild inflammation Fibrosis |
↑ BW Sexual dimorphism ↑ plasmatic transaminases ↑ cholesterol ↑ insulin and glucose in vivo US alterations |
[65] |
| 8-weeks-old Ay C57BL/6J |
Male | WD + fructose/glucose 12 months |
Hepatomegaly Steatosis Inflammation Fibrosis |
↑ BW IR ↑ plasma lipids |
[49] |
| Liver specific-Lrpprc KO C57BL/6N | Male and female | SD 14 weeks |
↓ Liver weight Steatosis Inflammation Fibrosis Mitochondrial disfunction ↑ ER stress markers |
↓ BW Sexual dimorphism ↓ weight heart, adipose tissue, soleus ↓ insulin and glucose ↑ plasma lipids Cardiometabolic impairment |
[71] |
| 7 weeks-old intestine-specific Tm6sf2 KO C57BL/6 | Male | CD/WD 8/14 weeks |
Hepatomegaly Steatosis Mild inflammation |
↑ BW ↑ plasmatic transaminases ↑ cholesterol Microbiota alterations |
[72] |
| 4 weeks-old TALLYHO/JngJ and NONcNZO10/LtJ | Female | HFD + fructose/glucose 16 weeks | Hepatomegaly Steatosis Inflammation Fibrosis Ballooning |
↑ BW IR ↑ plasmatic transaminases ↑ cholesterol ↑ insulin and glucose Kidney injury |
[56] |
| 11 weeks-old Cyp2a12/Cyp2c70 KO C57BL/6J | Male | WD + fructose/glucose 36 weeks | Hepatomegaly Steatosis Inflammation Fibrosis HCC |
↑ BW ↑ plasmatic transaminases ↑ cholesterol ↑ insulin and glucose |
[74] |
|
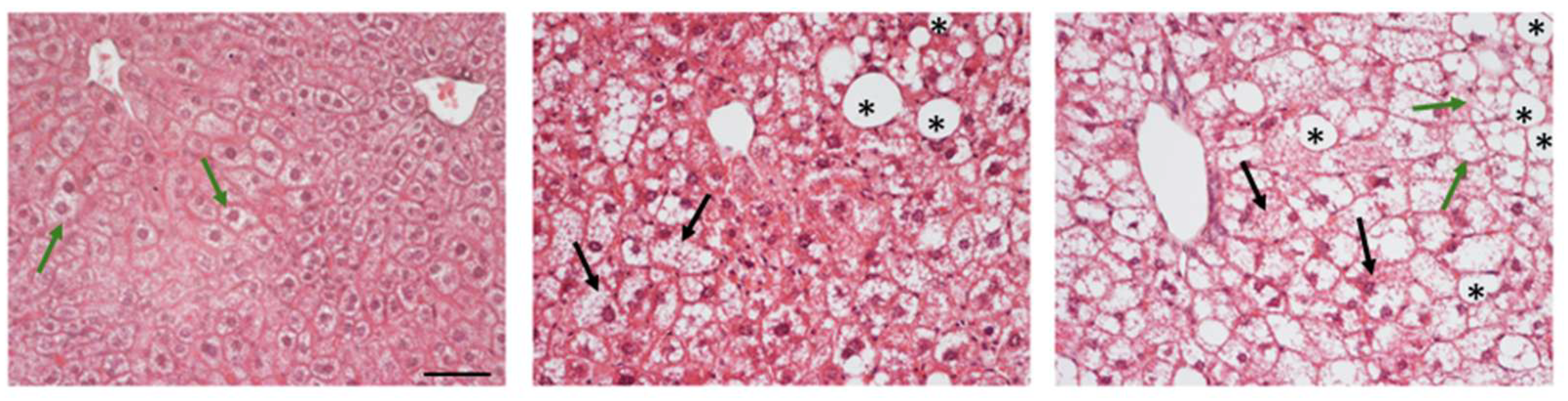
Histological feature |
Human MASLD/MASH | Murine MASLD/MASH models | Ref. |
| Steatosis | Diffuse macrovesicular steatosis, often centrilobular | Model dependent, sometimes prevalently microvesicular or patchy | [75,76,77,87] |
| Hepatocyte ballooning |
Common in MASH | Often less pronounced than in humans | [56,75,76,77] |
| Lobular inflammation |
Disseminated inflammatory infiltrates, mainly mononuclear cells | Inflammatory infiltrates milder; certain models show a predominance of intrahepatic T cells | [56,75,76,77] |
| Perisinusoidal / pericellular fibrosis | Progression to pericellular and periportal fibrosis in advanced MASH | Variable, some models display mild to moderate fibrosis with slow and less severe progression; others display rapid progression | [36,53,56,75,76,77] |
| Hepatic stellate cell (HSC) activation | HSC could have a role in exacerbating MASLD into MASH and inducing fibrosis | HSC activation occurs in response to injury, but with variable kinetics | [78,88,89,90] |
| Lobular zonation | Disease initially affects zone 3 and then spreads | Segmental or lobar heterogeneity | [42,75] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




