Submitted:
16 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
- patients treated with a 5-week multilayer bandage protocol (BEND)
- patients treated with a 5-week multilayer bandage protocol combined with diet (BEND+DIET)
- patients reevaluated after 5 weeks without receiving any treatment and without dietary modification (CONTR)
2.2. Clinical Assessment
2.3. Symptom Assessment
2.4. Treatments
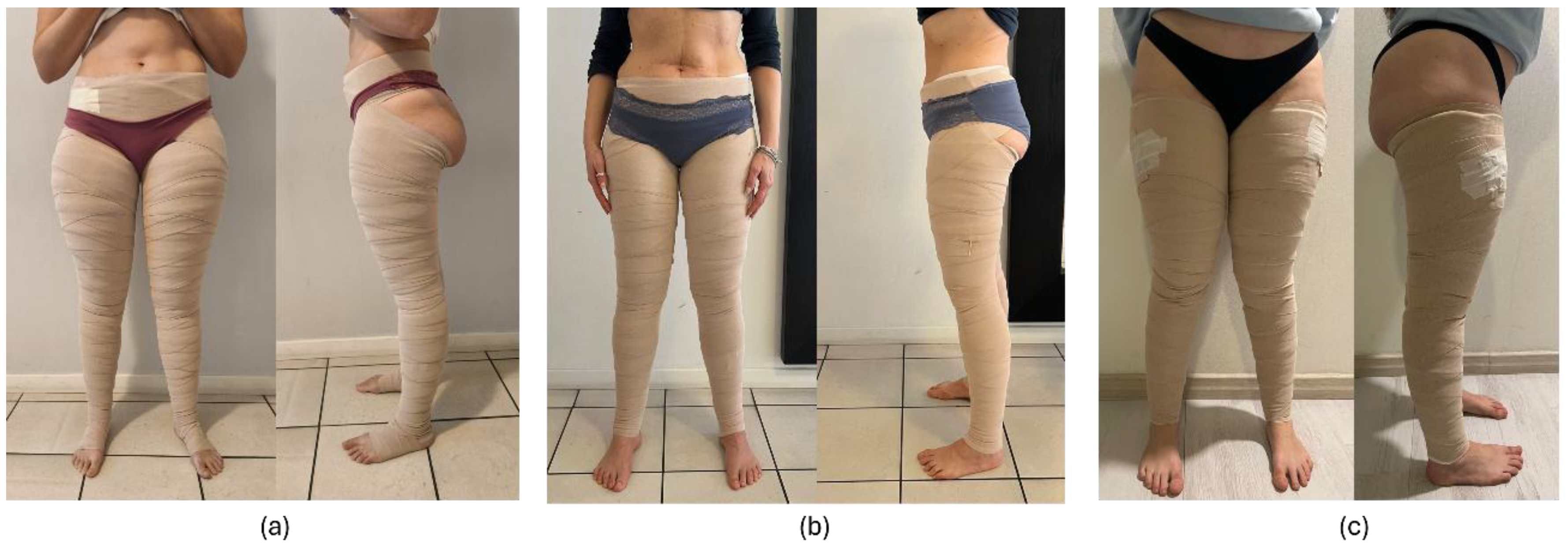
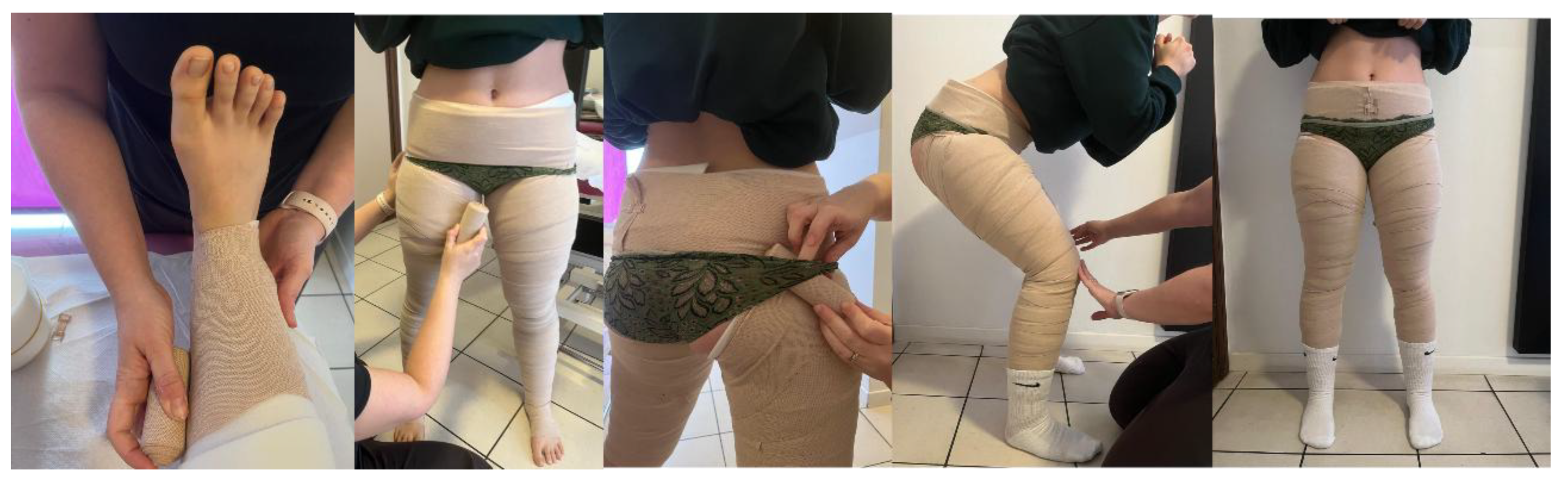
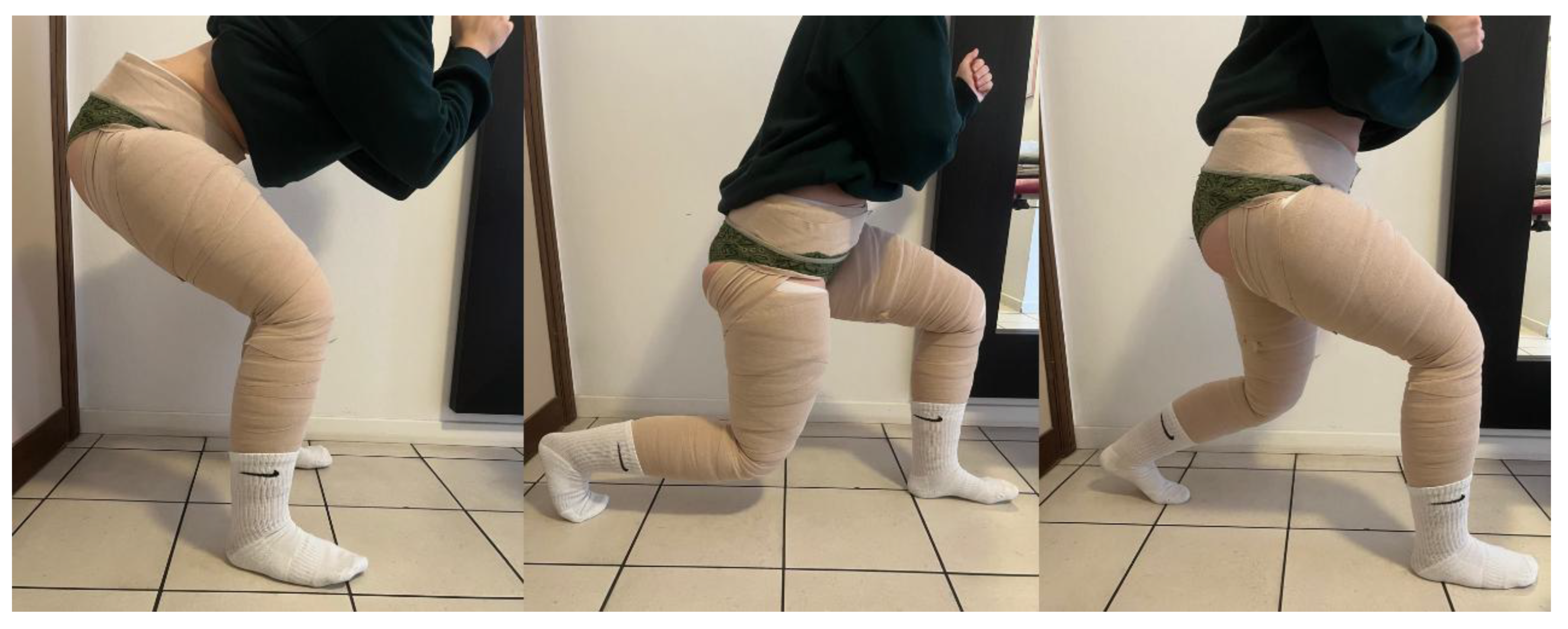
2.4.1. Multilayer Bandage Treatment Protocol
2.4.2. Dietary Intervention
2.5. Statistical Analysis
3. Results
3.1. General Description, Phenotypic, and Staging
- Bandage-only treatment group (BEND): 35 patients
- Bandage-plus-diet treatment group (BEND+DIET): 48 patients
- Untreated control group (CONTR): 31 patients
3.2. Anthropometric Measurements and Volumes
3.2.1. Anthropometric Measurements
3.2.2. Lower Limbs Volume
3.3. Pain
3.4. Symptoms
3.5. Multivariate Analysis
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CTD | Complex decongestive therapy |
| MLD | manual lymphatic drainage |
| MLB | multilayer bandaging |
| IL-6 | interleukin 6 |
| TNF-α | tumor necrosis factor-alpha |
| MCP-1 | monocyte chemoattractant protein 1 |
| PPARγ | peroxisome proliferator-activated receptor gamma |
| C/EBPα | CCAAT/enhancer-binding protein alpha |
| MRI | magnetic resonance imaging |
| BEND | Bandage-only treatment group |
| BEND+DIET | Bandage-plus-diet treatment group |
| CONTR | Untreated control group |
| BMI | Body mass index |
| PPC | Progressive Pain Check |
| RPS | Ricolfi-Patton Score |
| LBPS | Lower Body Pain Score |
| UBPS | Upper Body Pain Score |
References
- Patton, L; Ricolfi, L; Bortolon, M; et al. Observational Study on a Large Italian Population with Lipedema: Biochemical and Hormonal Profile, Anatomical and Clinical Evaluation, Self-Reported History. Int J Mol Sci. 2024, 25(3), 1599. [Google Scholar] [CrossRef]
- Herbst, KL; Kahn, LA; Iker, E; et al. Standard of care for lipedema in the United States. Phlebology 2021, 36(10), 779–796. [Google Scholar] [CrossRef] [PubMed]
- Halk, AB; Damstra, RJ. First Dutch guidelines on lipedema using the international classification of functioning, disability and health. Phlebology 2017, 32(3), 152–159. [Google Scholar] [CrossRef] [PubMed]
- Wounds, UK; Best. Practice Guidelines: The Management of Lipoedema. 2017. Available online: https://wounds-uk.com/bestpractice-statements/management-lipoedema/ (accessed on 28 August 2017).
- Alcolea, J.M.; Alonso, A.B.; Arroyo, B.A.; Domingo, P.; Garcia, G.A.; Graells, G.M.; Urraca, I.C.; Nipoti, I.E.; Castillo, M.E.; Alvarez, M., Jr.; et al. Consensus Document on Lipedema 2018. In cludes Current Status of Lipedema 2019; LITOGAMA SL: Barcelona, Spain, 2018. [Google Scholar]
- Bertsch, T; Erbacher, G; Elwell, R. Lipoedema: a paradigm shift and consensus. J Wound Care 2020, 29(Sup11b), 1–51. [Google Scholar] [CrossRef]
- Faerber, G; Cornely, M; Daubert, C; et al. S2k guideline lipedema. J Dtsch Dermatol Ges. 2024, 22(9), 1303–1315. [Google Scholar] [CrossRef] [PubMed]
- Amato, ACM; Peclat, APRM; Kikuchi, R; et al. Brazilian Consensus Statement on Lipedema using the Delphi methodology. J Vasc Bras. 2025, 24, e20230183. [Google Scholar] [CrossRef]
- O’Donnell, TF, Jr.; Allison, GM; Iafrati, MD. A systematic review of guidelines for lymphedema and the need for contemporary intersocietal guidelines for the management of lymphedema. J Vasc Surg Venous Lymphat Disord. 2020, 8(4), 676–684. [Google Scholar] [CrossRef]
- Li, G; Fu, N; Yang, X; et al. Mechanical compressive force inhibits adipogenesis of adipose stem cells. Cell Prolif. 2013, 46(5), 586–594. [Google Scholar] [CrossRef]
- Szolnoky, G; Nagy, N; Kovács, RK; et al. Complex decongestive physiotherapy decreases capillary fragility in lipedema. Lymphology 2008, 41(4), 161–166. [Google Scholar]
- Saitou, K; Tokunaga, M; Yoshino, D; et al. Local cyclical compression modulates macrophage function in situ and alleviates immobilization-induced muscle atrophy. Clin Sci (Lond) 2018, 132(19), 2147–2161. [Google Scholar] [CrossRef]
- Rabe, E; Partsch, H; Morrison, N; et al. Risks and contraindications of medical compression treatment - A critical reappraisal. An international consensus statement. Phlebology 2020, 35(7), 447–460. [Google Scholar] [CrossRef] [PubMed]
- Ligi, D; Croce, L; Mannello, F. Inflammation and compression: the state of art. Veins & Lymphatics 2016, 5(1). [Google Scholar] [CrossRef]
- Partsch, H. Assessing the effectiveness of multilayer inelastic bandaging. Journal of Lymphoedema 2007, 2(2), 55. [Google Scholar]
- Beidler, SK; Douillet, CD; Berndt, DF; Keagy, BA; Rich, PB; Marston, WA. Inflammatory cytokine levels in chronic venous insufficiency ulcer tissue before and after compression therapy. J Vasc Surg. 2009, 49(4), 1013–1020. [Google Scholar] [CrossRef]
- Murphy, MA; Joyce, WP; Condron, C; Bouchier-Hayes, D. A reduction in serum cytokine levels parallels healing of venous ulcers in patients undergoing compression therapy. Eur J Vasc Endovasc Surg. 2002, 23(4), 349–352. [Google Scholar] [CrossRef]
- Partsch, H. Compression therapy: clinical and experimental evidence. Ann Vasc Dis. 2012, 5(4), 416–422. [Google Scholar] [CrossRef]
- Hossain, G; Iwata, T; Mizusawa, N; Shima, SWN; Okutsu, T; Ishimoto, K; et al. Compressive force inhibits adipogenesis through COX-2-mediated down-regulation of PPAR-γ2 and C/EBP α. J. Biosci. Bioeng. 2010, 109, 297–303. [Google Scholar] [CrossRef]
- Wright, T; Scarfino, CD; O’Malley, EM. Effect of pneumatic compression device and stocking use on symptoms and quality of life in women with lipedema: A proof-in-principle randomized trial. Phlebology 2023, 38(1), 51–61. [Google Scholar] [CrossRef]
- Szolnoky, G; Varga, E; Varga, M; Tuczai, M; Dósa-Rácz, E; Kemény, L. Lymphedema treatment decreases pain intensity in lipedema. Lymphology 2011, 44(4), 178–182. [Google Scholar]
- Szolnoky, G; Borsos, B; Bársony, K; Balogh, M; Kemény, L. Complete decongestive physiotherapy with and without pneumatic compression for treatment of lipedema: a pilot study. Lymphology 2008, 41(1), 40–44. [Google Scholar]
- Ricolfi, L; Reverdito, V; Gabriele, G; et al. Micromassage Compression Leggings Associated with Physical Exercise: Pilot Study and Example of Evaluation of the Clinical and Instrumental Effectiveness of Conservative Treatment in Lipedema. Life (Basel) 2024, 14(7), 854. [Google Scholar] [CrossRef] [PubMed]
- Czerwińska, M; Gruszecki, M; Rumiński, J; Hansdorfer-Korzon, R. Evaluation of the Effectiveness of Compression Therapy Combined with Exercises Versus Exercises Only Among Lipedema Patients Using Various Outcome Measures. Life (Basel) 2024, 14(11), 1346. [Google Scholar] [CrossRef] [PubMed]
- Collier, M; Barbul, A; Campbell, RF; Filori, P; Haughey, L; Hazledine, M; Hellgren, M; Hopkins, A; Isoherranen, K; Lagus, H; Ryan, H; Moffatt, C. Chronic Oedema of the Lower Limb. Practical guidance on diagnosis, effective treatment and ongoing management. J Wound Management 2025, 26(3 Sup1), S1–S88. [Google Scholar] [CrossRef]
- Chakraborty, A; Crescenzi, R; Usman, TA; et al. Indications of Peripheral Pain, Dermal Hypersensitivity, and Neurogenic Inflammation in Patients with Lipedema. Int J Mol Sci. 2022, 23(18), 10313. [Google Scholar] [CrossRef]
- Kruppa, P; Georgiou, I; Biermann, N; Prantl, L; Klein-Weigel, P; Ghods, M. Lipedema-Pathogenesis, Diagnosis, and Treatment Options. Dtsch Arztebl Int. 2020, 117(22-23), 396–403. [Google Scholar] [CrossRef]
- de Oliveira, J; de Paula, ACP; Guimarães, VHD. Clinical or cultural? Dietary interventions for lipedema: a systematic review. Maturitas 2025, 202, 108716. [Google Scholar] [CrossRef]
- Atabilen Pınar, B; Çelik, MN; Altıntaş Başar, HB; Ağagündüz, D; Karaca, OB. Current Evidence-Based Clinical Nutritional Approaches in Lipedema: A Scoping Review. Nutr Rev 2025. [Google Scholar] [CrossRef]
- Aydin, A; Bilgic, P. Nutritional Approaches and Supplementation in Lipedema Management: A Narrative Review of Current Evidence. Curr Nutr Rep. Published. 2025, 14(1), 113. [Google Scholar] [CrossRef]
- Amato, ACM; Amato, JLS; Benitti, DA. The Efficacy of Ketogenic Diets (Low Carbohydrate; High Fat) as a Potential Nutritional Intervention for Lipedema: A Systematic Review and Meta-Analysis. Nutrients 2024, 16(19), 3276. [Google Scholar] [CrossRef]
- Tel Adıgüzel, K; Yaman, A; Kürklü, NS; Adıgüzel, E. Dietary Inflammatory Index and Mediterranean Diet Score are associated with systemic inflammation in women with lipedema. Int J Obes (Lond). 27 November 2025. [CrossRef]
- Jeziorek, M; Chachaj, A; Sowicz, M; et al. The Benefits of Low-Carbohydrate, High-Fat (LCHF) Diet on Body Composition, Leg Volume, and Pain in Women with Lipedema. J Obes. 2023, 2023, 5826630. [Google Scholar] [CrossRef]
- Di Renzo, L; Gualtieri, P; Zomparelli, S; et al. Modified Mediterranean-Ketogenic Diet and Carboxytherapy as Personalized Therapeutic Strategies in Lipedema: A Pilot Study. Nutrients 2023, 15(16), 3654. [Google Scholar] [CrossRef] [PubMed]
- Sørlie, V; De Soysa, AK; Hyldmo, ÅA; Retterstøl, K; Martins, C; Nymo, S. Effect of a ketogenic diet on pain and quality of life in patients with lipedema: The LIPODIET pilot study. Obes Sci Pract. 2022, 8(4), 483–493. [Google Scholar] [CrossRef] [PubMed]
- Jeziorek, M; Szuba, A; Kujawa, K; Regulska-Ilow, B. The Effect of a Low-Carbohydrate, High-Fat Diet versus Moderate-Carbohydrate and Fat Diet on Body Composition in Patients with Lipedema. Diabetes Metab Syndr Obes. 2022, 15, 2545–2561. [Google Scholar] [CrossRef]
- Jeziorek, M; Chachaj, A; Szuba, A; Różańska, D; Prescha, A. Exploring the Anti-Inflammatory Potential of a Mediterranean-Style Ketogenic Diet in Women with Lipedema. Nutrients 2025, 17(18), 3014. [Google Scholar] [CrossRef] [PubMed]
- Lundanes, J; Sandnes, F; Gjeilo, KH; et al. Effect of a low-carbohydrate diet on pain and quality of life in female patients with lipedema: a randomized controlled trial. Obesity (Silver Spring) 2024, 32(6), 1071–1082. [Google Scholar] [CrossRef]
- Lundanes, J; Gårseth, M; Taylor, S; et al. The effect of a low-carbohydrate diet on subcutaneous adipose tissue in females with lipedema. Front Nutr. 2024, 11, 1484612. [Google Scholar] [CrossRef]
- Di Renzo, L; Cinelli, G; Romano, L; et al. Potential Effects of a Modified Mediterranean Diet on Body Composition in Lipoedema. Nutrients Published. 2021, 13(2), 358. [Google Scholar] [CrossRef]
- Fedre, B; Dessalvi, S; Boccardo, F. A Case Series on Combining Modified Mediterranean Diet and Ketogenic Diet in a “Sandwich” Approach for Patients with Lipedema and Comorbidities. Lymphology 2025, 58(3), 108–118. [Google Scholar] [CrossRef]
- Tsigalou, C; Konstantinidis, T; Paraschaki, A; Stavropoulou, E; Voidarou, C; Bezirtzoglou, E. Mediterranean Diet as a Tool to Combat Inflammation and Chronic Diseases. An Overview. Biomedicines 2020, 8(7), 201. [Google Scholar] [CrossRef]
- Bruna-Mejias, A; San Martin, J; Arciniegas-Diaz, D; et al. Comparison of the Mediterranean Diet and Other Therapeutic Strategies in Metabolic Syndrome: A Systematic Review and Meta-Analysis. Int J Mol Sci. 2025, 26(12), 5887. [Google Scholar] [CrossRef]
- Ferraris, C; Guglielmetti, M; Neri, LCL; et al. A Review of Ketogenic Dietary Therapies for Epilepsy and Neurological Diseases: A Proposal to Implement an Adapted Model to Include Healthy Mediterranean Products. Foods 2023, 12(9), 1743. [Google Scholar] [CrossRef] [PubMed]
- Bailey, MA; Holscher, HD. Microbiome-Mediated Effects of the Mediterranean Diet on Inflammation. Adv Nutr. 2018, 9(3), 193–206. [Google Scholar] [CrossRef] [PubMed]
- Patton, L; Reverdito, V; Bellucci, A; Bortolon, M; Macrelli, A; Ricolfi, L. A Case Series on the Efficacy of the Pharmacological Treatment of Lipedema: The Italian Experience with Exenatide. Clin Pract. 2025, 15(7), 128. [Google Scholar] [CrossRef] [PubMed]
- Kuhnke, E. Volumbestimmung aus Umfangmessungen. Folia Angiologica 1976, 24, 228–232. [Google Scholar]
- Atan, T; Bahar-Özdemir, Y. The Effects of Complete Decongestive Therapy or Intermittent Pneumatic Compression Therapy or Exercise Only in the Treatment of Severe Lipedema: A Randomized Controlled Trial. Lymphat Res Biol. 2021, 19(1), 86–95. [Google Scholar] [CrossRef]
- Esmer, M; Schingale, FJ. Effect of Physical Therapy on Circumference Measurement and Extremity Volume in Patients Suffering from Lipedema with Secondary Lymphedema. Lymphat Res Biol. 2024, 22(1), 8–11. [Google Scholar] [CrossRef]
- Földi, E. Therapie des Lymphödems [Therapy of lymphedema]. Hautarzt 2012, 63(8), 627–633. [Google Scholar] [CrossRef]
- Partsch, H; Mortimer, P. Compression for leg wounds. Br J Dermatol 2015, 173(2), 359–369. [Google Scholar] [CrossRef]
- Al-Ghadban, S; Cromer, W; Allen, M; et al. Dilated Blood and Lymphatic Microvessels, Angiogenesis, Increased Macrophages, and Adipocyte Hypertrophy in Lipedema Thigh Skin and Fat Tissue. J Obes. 2019, 2019, 8747461. [Google Scholar] [CrossRef]
- Trayhurn, P; Wood, IS. Adipokines: inflammation and the pleiotropic role of white adipose tissue. Br J Nutr. 2004, 92(3), 347–355. [Google Scholar] [CrossRef]
- Eidler, SK; Douillet, CD; Berndt, DF; et al. Compression Therapy in the Management of Cellulitis: A Comparative Study. The Journal of Surgery 2018, 6, 68. Available online: https://api.semanticscholar.org/CorpusID:55884619.
- Pasquali, R; Vicennati, V; Gambineri, A; Pagotto, U. Hormones and pathophysiology of obesity. Eat Weight Disord. 2001, 6((3) Suppl 3, 9–20. [Google Scholar]
- Repaci, A; Gambineri, A; Pasquali, R. The role of low-grade inflammation in the polycystic ovary syndrome. Mol Cell Endocrinol. 2011, 335(1), 30–41. [Google Scholar] [CrossRef] [PubMed]
| Variable | ALL (114) |
BEND (35) |
BEND+DIET (48) |
CONTR (31) |
P value ALL |
P value TREAT |
|---|---|---|---|---|---|---|
| Age (yrs) - mean (sd) | 43.2 (11.1) | 41.2 (10.3) | 45.2 (12.9) | 42.6 (8.65) | 0.261 | 0.125 |
| Body weight (kg) - mean (sd) | 79.6 (18.5) | 79.6 (19.7) | 81.5 (17.8) | 76.5 (18.3) | 0.376‡ | 0.546† |
| Body mass index (kg/m2) - mean (sd) | 30.0 (6.43) | 30.2 (6.62) | 30.5 (6.54) | 29.0 (6.2) | 0.573 | 0.830 |
| Waist circumference (cm) - mean (sd) | 91.7 (14.6) | 89.7 (13.2) | 92.7 (16.5) | 92.5 (13.1) | 0.617 | 0.358 |
| Hip circumference (cm) - mean (sd) | 116 (12.9) | 116 (13.0) | 116 (13.4) | 114 (12.2) | 0.796 | 0.978 |
| Waist-to-hip ratio - mean (sd) | 0.79 (0.84) | 0.07 (0.01) | 0.79 (0.09) | 0.81 (0.08) | 0.214 | 0.341 |
| Pretibial fovea – n (%) | 34 (33.0) | 11 (36.7) | 16 (35.6) | 7 (25.0) | 0.992 | |
| Telangiectasias – n (%) | 81 (81.8) | 21 (77.8) | 35 (79.5) | 25 (89.3) | 0.438 | 0.859 |
| Distribution of clinical stages | 0.233 | 0.532 | ||||
| Stage 1 – n (%) | 39 (34.2) | 11 (31.4) | 13 (27.1) | 15 (48.8) | ||
| Stage 2 – n (%) | 48 (42.1) | 14 (40.0) | 25 (52.1) | 9 (29.0) | ||
| Stage 3 – n (%) | 27 (23.7 | 10 (28.6) | 10 (20.8) | 7 (22.6) | ||
| Upper limbs involvement - n (%) | 97 (85.1) | 30 (85.7) | 38 (79.2) | 29 (93.0) | 0.234* | 0.634 |
| Lower limb volume – average (mL) - mean (sd) | - | 15538 (4429) | 15841 (3982) | 0.750 | ||
| Lower limb volume - right (mL) - mean (sd) | - | 15532 (4361) | 15945 (4120) | 0.664 | ||
| Lower limb volume – left (mL) - mean (sd) | - | 15545 (4517) | 15736 (3923) | 0.723† | ||
| Total Body Pain Score - mean (sd) | 31.6 (6.30) | 31.2 (6.85) | 30.8 (6.75) | 33.3 (4.59) | 0.320‡ | 0.705† |
| Lower Body Pain Score - mean (sd) | 26.7 (4.56) | 26.1 (4.55) | 26.3 (5.35) | 28.0 (2.77 | 0.195‡ | 0.584† |
| Upper Body Pain Score - mean (sd) | 4.87 (2.81) | 5.11 (3.17) | 4.44 (2.81) | 5.26 (2.37) | 0.445‡ | 0.352† |
| Total Symptoms score – mean (sd) | 43.7 (13.7) | 45.2 (12.8) | 40.6 (13.7) | 46.6 (14.2) | 0.115 | 0.117 |
| Time of use of garments (hours) - mean (sd) | 24.7 (11.5) | 22.3 (9.02) | 26.5 (12.8) | - | 0.177† |
| BEND | BEND+DIET | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Variable | T0 | T1 | delta | P T0-T1 |
T0 | T1 | delta | P T0-T1 |
P DELTA |
| Body weight (kg) | 79,63 (19.65) | 79.1 (19.4) | -0.50 (1.09) | 0.011 | 81,48 (17.81) | 78.0 (17.7) | -3.45 (1.84) | <0.001 | <0.001 |
| Body weight loss (%) | 0.590 (1.34) | 4.38 (2.56) | <0.001 | ||||||
| Waist circumference (cm) | 88.8 (13.3) | 87.4 (13.0) | 1.37 (2.51) | <0.001† | 90.1 (14.3) | 87.7 (13.9) | 3.86 (3.27) | <0.001† | <0.001† |
| Hip circumference (cm) | 116 (14.1) | 114 (14.4) | 2.04 (2.30) | <0.001† | 115 (12.9) | 112 (12.3) | 3.78 (2.49) | <0.001† | <0.001† |
| Lower limbs volume – average (mL) | 15538 (4429) | 14807 (4360) | 732 (242) | <0.001 | 15841 (3982) | 14418 (3644) | 1423 (662) | <0.001† | <0.001† |
| Volume reduction in the lower limbs – average (%) | 4.96 (1.84) | 9.02 (3.21) | <0.001 | ||||||
| Lower limb volume - right (mL) | 15532 (4361) | 14847 (4316) | 685 (321) | <0.001 | 15945 (4120) | 14498 (3789) | 1447 (650) | <0.001† | <0.001† |
| Lower limb volume – left (mL) | 15545 (4517) | 14847 (4316) | 698 (701) | <0.001 | 15736 (3223) | 14498 (3789) | 1239 (1322) | <0.001† | <0.001 |
| Total Body Pain Score or RPS | 31.2 (6.85) | 14.1 (8.63) | 17.1 (6.88) | <0.001 | 30.8 (6.75) | 13.3 (7.98) | 14.4 (7.99) | <0.001 | NS |
| Lower Body Pain Score | 26.1 (4.56) | 10.3 (6.91) | 15.8 (6.61) | <0.001 | 26.3 (5.35) | 10.1 (6.24) | 16.2 (678) | <0.001 | NS |
| Upper Body Pain Score | 5.11 (3.17) | 3.83 (3.01) | 1.29 (2.02) | <0.001† | 4.44 (2.81) | 3.21 (2.98) | 1.23 (2.40) | <0.001† | NS |
| Total Symptoms score | 44.3 (12.8) | 16.7 (11.7) | 28.5 (10.9) | <0.001 | 40.6 (13.7) | 17.8 (11.7) | 25.9 (10.2) | <0.001 | NS |
| BEND | BEND+DIET | CONTR | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pain assessment site | T0 | T1 | P t0-t1 | T0 | T1 | P t0-t1 | T0 | T1 | P t0-t1 | P DELTA TREAT | P DELTA CB | P DELTA CBD |
| Medial lower third of the leg Mediana(IQR) |
4(1) | 1(2) | <0.001† | 4(0) | 1(1) | <0.001† | 4(0) | 4(0) | NS† | NS† | <0.001† | <0.001† |
| Middle third of the back of the leg Mediana(IQR) |
4(1) | 1(1.5) | <0.001† | 4(0) | 1(1) | <0.001† | 4(0) | 4(0) | NS† | NS† | <0.001† | <0.001† |
| Medial upper third of the leg Mediana(IQR) |
4(0) | 1(1) | <0.001† | 4(0) | 2(1) | <0.001† | 4(0) | 4(0) | NS† | NS† | <0.001† | <0.001† |
| Lateral upper third of the thigh Mediana(IQR) |
3(2) | 1(1) | <0.001† | 4(2) | 1(2) | <0.001† | 4(1) | 4(1) | NS† | NS† | <0.001† | <0.001† |
| later lower third of the thigh Mediana(IQR) |
4(1) | 1(2) | <0.001† | 4(1) | 1(2) | <0.001† | 4(1) | 4(1) | NS† | NS† | <0.001† | <0.001† |
| Medial upper third of the thigh Mediana(IQR) |
4(1) | 1(2) | <0.001† | 4(1) | 1(1) | <0.001† | 4(0) | 4(0) | NS† | NS† | <0.001† | <0.001† |
| Medial lower third of the thigh Mediana(IQR) |
4(1) | 2(1.5) | <0.001† | 4(1) | 1(1) | <0.001† | 4(0) | 4(0) | NS† | NS† | <0.001† | <0.001† |
| Lower abdomen Mediana(IQR) |
2(3) | 1(2) | <0.001† | 2(1.25) | 0(1) | <0.001† | 2(3) | 2(3) | NS† | NS† | <0.001† | <0.001† |
| Lateral edge of the tissue covering the teres major muscle Mediana(IQR) |
2(2.5) | 1(2) | <0.001† | 2(2) | 1(2) | <0.001† | 2(2) | 2(2) | NS† | NS† | <0.001† | 0.005† |
| Arm Mediana(IQR) |
3(2) | 2(2) | <0.001† | 3(2) | 2(2) | <0.001† | 3(2) | 3(2) | NS† | NS† | <0.001† | <0.001† |
| Forearm Mediana(IQR) |
0(0.5) | 0(1) | NS† | 0(0) | 0(0) | NS † | 0(0) | 0(0) | NS† | NS† | NS† | NS† |
| Total Body Pain Score or RPS | 32(10) | 13(14) | <0.001 | 31(8) | 12.5(10.75) | 12.5(10.75) | 34(7) | 34(7) | NS† | NS | <0.001† | <0.001† |
| Lower Body Pain Score | 27(7) | 9(12) | <0.001 | 28(3) | 10(9.5) | 10(9.25) | 28(6) | 28(6) | NS† | NS | <0.001† | <0.001† |
| Upper Body Pain Score | 6(4) | 4(5) | <0.001† | 4.5(4.75) | 2(3.75) | 2(3.75) | 5(3) | 5(3) | NS† | NS† | <0.001† | 0.001† |
| BEND | BEND+DIET | CONTR | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Item | T0 | T1 | P T0-T1 |
T0 | T1 | P T0-T1 |
T0 | T1 | P T0-T1 |
P DELTA TREAT |
P DELTA CB | P DELTA CBD |
| Spontaneous pain in the lower limbs | 4(1) | 1(2) | <0.001† | 3(2) | 1(2) | <0.001† | 3(2) | 3(2) | NS† | NS† | 0.001† | <0.001† |
| Feeling of swelling in the lower limbs | 3(1) | 1(1) | <0.001† | 3(1) | 1(1) | <0.001† | 3(1) | 3(1) | NS† | NS† | <0.001† | <0.001† |
| feeling of heaviness in the lower limbs | 4(1) | 1(1) | <0.001† | 4(1) | 1(1) | <0.001† | 4(1) | 4(1) | NS† | NS† | <0.001† | <0.001† |
| Sensation of swelling in the ankles | 2(3) | 0(1) | <0.001† | 3(3) | 0(1) | <0.001† | 3(2) | 3(2) | NS† | NS† | <0.001† | <0.001† |
| Sensation of swelling in the foot (back and toes) | 0(0) | 0(0) | 0.013† | 0(2) | 0(0) | <0.001† | 1(3) | 1(3) | NS† | NS† | 0.005† | 0.003† |
| Sensation of swelling elsewhere (on the hands, around the eyes) | 0(1) | 0(0) | 0.040† | 0(1) | 0(0) | 0.006† | 2(3) | 2(3) | NS† | NS† | 0.036† | 0.013† |
| Skin cold to the touch on the lower limbs | 3(2) | 1(1.5) | <0.001† | 2(4) | 0.5(1.25) | <0.001† | 3(2) | 3(2) | NS† | NS† | <0.001† | <0.001† |
| Hypersensitivity to touch in the lower limbs | 3(2.5) | 1(1.5) | <0.001† | 2(4) | 0(1.25) | <0.001† | 3(2) | 3(2) | NS† | NS† | <0.001† | <0.001† |
| Pressure pain in the lower limbs | 4(1) | 2(1) | <0.001† | 4(1) | 1(2) | <0.001† | 4(1) | 4(1) | NS† | NS† | <0.001† | <0.001† |
| Paresthesia and tingling in the lower limbs | 1(2) | 0(0.5) | 0.003† | 0.5(2) | 0(0) | <0.001† | 1(3) | 1(3) | NS† | NS† | <0.001† | 0.002† |
| Burning sensation in the lower limbs | 1(2) | 0(1) | 0.008† | 0(1) | 0(0) | 0.003† | 1(2) | 1(2) | NS† | NS† | 0.015† | 0.005† |
| Easy bruising | 3(2.5) | 1(2) | <0.001† | 3(2.25) | 1(2) | <0.001† | 3(2) | 3(2) | NS† | NS† | <0.001† | <0.001† |
| Fatigue in the lower limbs when doing movement or physical activity | 3.63(1.5) | 1.54(1.27) | <0.001 | 3.19(1.57) | 1.38(1.2) | <0.001 | 4(1) | 3(1) | NS† | NS | <0.001† | <0.001† |
| Pain in the lower limbs during or after movement/physical activity | 4(1) | 1(2) | <0.001† | 3(2) | 1(1.25) | <0.001† | 3(2) | 3(1) | NS† | NS† | <0.001† | <0.001† |
| Swelling or discomfort in the lower limbs following physical activity | 3(2) | 1(2) | <0.001† | 2(3) | 0(1) | <0.001† | 3(2) | 3(2) | NS† | NS† | <0.001† | <0.001† |
| Pain in the lower limbs when standing for a long time | 4(1) | 1(2) | <0.001† | 3(2.25) | 1(2.25) | <0.001† | 3(1) | 3(1) | NS† | NS† | <0.001† | <0.001† |
| Swelling/discomfort in the lower limbs when standing for a long time | 4(1) | 1(2) | <0.001† | 3(2) | 1(2) | <0.001† | 4(1) | 3(1) | NS† | NS† | <0.001† | <0.001† |
| Total Symptom Score | 44(18) | 16(17) | <0.001 | 40.5(16) | 10.5(16) | <0.001 | 44(22) | 45(20) | NS† | NS | <0.001† | <0.001† |
| DEPENDENT VARIABLE: | Lower Body Pain Score T1 Model 1 |
Total Symptom Score T1 Model 2 |
volume of the lower limbs T1 Model 3 |
% change in lower limb volume. Model 4 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Coefficient | [95% conf. interval] | P VALUE | Coefficient | [95% conf. interval] | P VALUE | Coefficient | [95% conf. interval] | P VALUE | Coefficient | [95% conf. interval] | P VALUE | |
| Dependent variable T0 | 0.3422 | [ -0.311, 0.996] | 0.289 | 0.5348 | [0.224, 0.845] |
0.0017 | 0.97850 | [0.914 , 1.043] |
<0.001 | -0.0001 | [-0.001, 0.000] | 0.583 |
| Age | 0.1387 | [ -0.131, 0.408] |
0.297 | 0.1952 | [-0.205, 0.595] |
0.3222 | -3.36479 | [-14.333 , 7.603] |
0.531 | 0.0223 |
[-0.054, 0.0999] |
0.553 |
| BMI | -0.2420 | [-0.876 , 0.392] | 0.437 | 0.2990 | [-0.666, 1.264] | 0.5272 | -7.69172 | [ -49.612, 34.229] |
0.707 | 0.0006 |
[-0.293, 0.294] |
0.997 |
| Stage 2 | 2.6319 | [-4.367, 9.631] |
0.444 | 4.4791 | [-5.266, 14.225] | 0.3509 | -17.09741 | [-275.288, 241.093] | 0.892 | -0.1452 |
[-1.951, 1.660] |
0.869 |
| Stage 3 | 7.2967 | [-3.426, 18.019] | 0.172 | -1.9058 | [-18.475, 14.663] |
0.8137 | 121.33824 | [-344.057, 586.733] | 0.594 | -0.6534 |
[-3.908, 2.601] |
0.681 |
| Bandage wearing time | -0.1650 | [-0.480, 0.150] |
0.290 | 0.1809 | [-0.271, 0.632] | 0.4152 | 0.97684 | [ -11.705 , 13.659] |
0.875 | 0.0007 | [-0.088, 0.089] | 0.987 |
| Pretibial fovea | -1.6227 | [-8.220, 4.975] |
0.615 | -0.6552 | [-10.49, 9.187] | 0.8914 | 118.74814 | [-155.659 , 393.155] | 0.379 | -0.8380 | [-2.757, 1.081] |
0.375 |
| DEPENDENT VARIABLE: | Lower Body Pain Score T1 Model 5 |
Total Symptom Score T1 Model 6 |
volume of the lower limbs T1 Model 7 |
% change in lower limb volume. Model 8 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Coefficient | [95% conf. interval] | P VALUE | Coefficient | [95% conf. interval] | P VALUE | Coefficient | [95% conf. interval] | P VALUE | Coefficient | [95% conf. interval] | P VALUE | |
| Dependent variable T0 | 0.25593 | [-0.170, 0.682] |
0.231 | 0.62701 | [0.411, 0.843] |
<0.001 | 0.89824 | [ 0.793, 1.004] |
<0.001 | 0.0000 | [-0.001, 0.001] | 0.945 |
| Age | 0.04621 | [-0.108, 0.201] |
0.548 | -0.15138 | [-0.367, 0.065] |
0.164 | -0.90579 | [ -16.069, 14.257] |
0.904 | -0.0019 |
[-0.090, 0.086 |
0.966 |
| BMI | 0.31379 | [ -0.064,. 691] |
0.101 | 0.17045 | [-0.366, 0.707] | 0.524 | -5.65610 | [-60.934, 49.622] |
0.837 | 0.0334 | [-0.288, 0.355] | 0.835 |
| Stage 2 | -1.69530 | [-6.650, 3.260] |
0.492 | -2.07086 | [-9.374, 5.232] | 0.569 | -42.96491 | [-526.747, 440.817] | 0.858 | 0.9193 | [-1.897, 3.735] | 0.512 |
| Stage 3 | -0.54301 | [-7.065, 5.979] |
0.867 | 1.73793 | [-8.082, 11.557] | 0.722 | -200.14047 | [-894.567, 494.286] | 0.563 | 1.5080 | [-2.534, 5.550] | 0.454 |
| Time of use of garments | -0.02747 | [ -0.190, 0.135] |
0.734 | 0.01468 | [-0.210 , 0.240] | 0.895 | -11.55433 | [-26.515, 3.406] |
0.126 | 0.0919 |
[0.005, 0.179] |
0.036 |
| Pretibial fovea | -0.30457 | [ -4.731, 4.122] |
0.890 | -0.45878 | [-6.583, 5.665] | 0.880 | 174.29107 | [-245.813, 594.395] |
0.406 | -0.4766 | [-2.299, 1.969] | 0.700 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




