Submitted:
14 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Ethical Approval and Compliance
2.2. Study Design and Experimental Units
2.3. Animals, Housing, and Husbandry
2.4. Sample Size Calculation
2.5. Randomization and Allocation Concealment
2.6. Dietary Treatments
2.7. Clinical Monitoring and Animal Welfare
2.8. Sampling Procedures
2.8.1. Feed and Fecal Sampling and Mycotoxin Analysis
2.8.2. Blood Sampling and Biochemical Analyses
2.8.3. Histopathology
2.9. Statistical Analysis
3. Results
3.1. Clinical Observations and General Health Status
3.2. Mycotoxin Contamination of the Total Mixed Ration
3.3. Biochemical Parameters
3.3. Oxidative Stress Biomarkers
3.5. Fecal Mycotoxin Concentrations
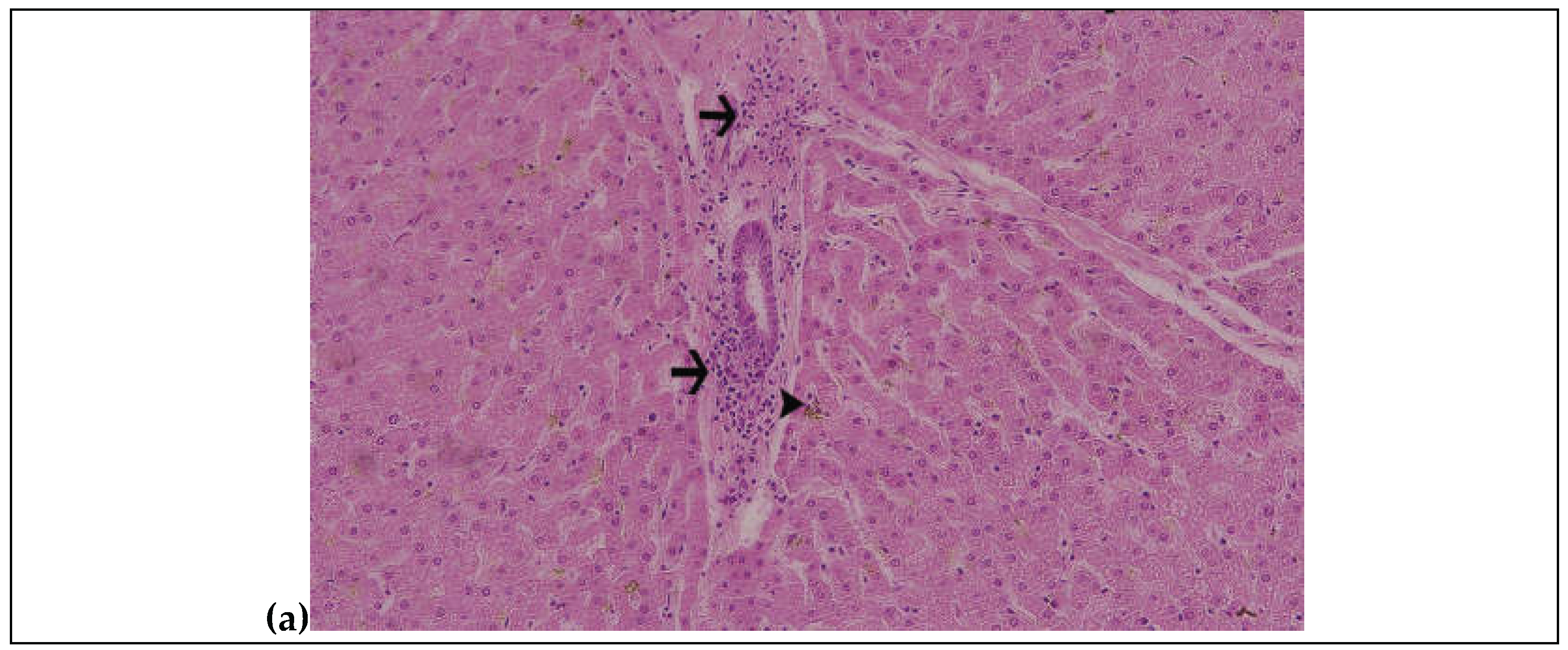
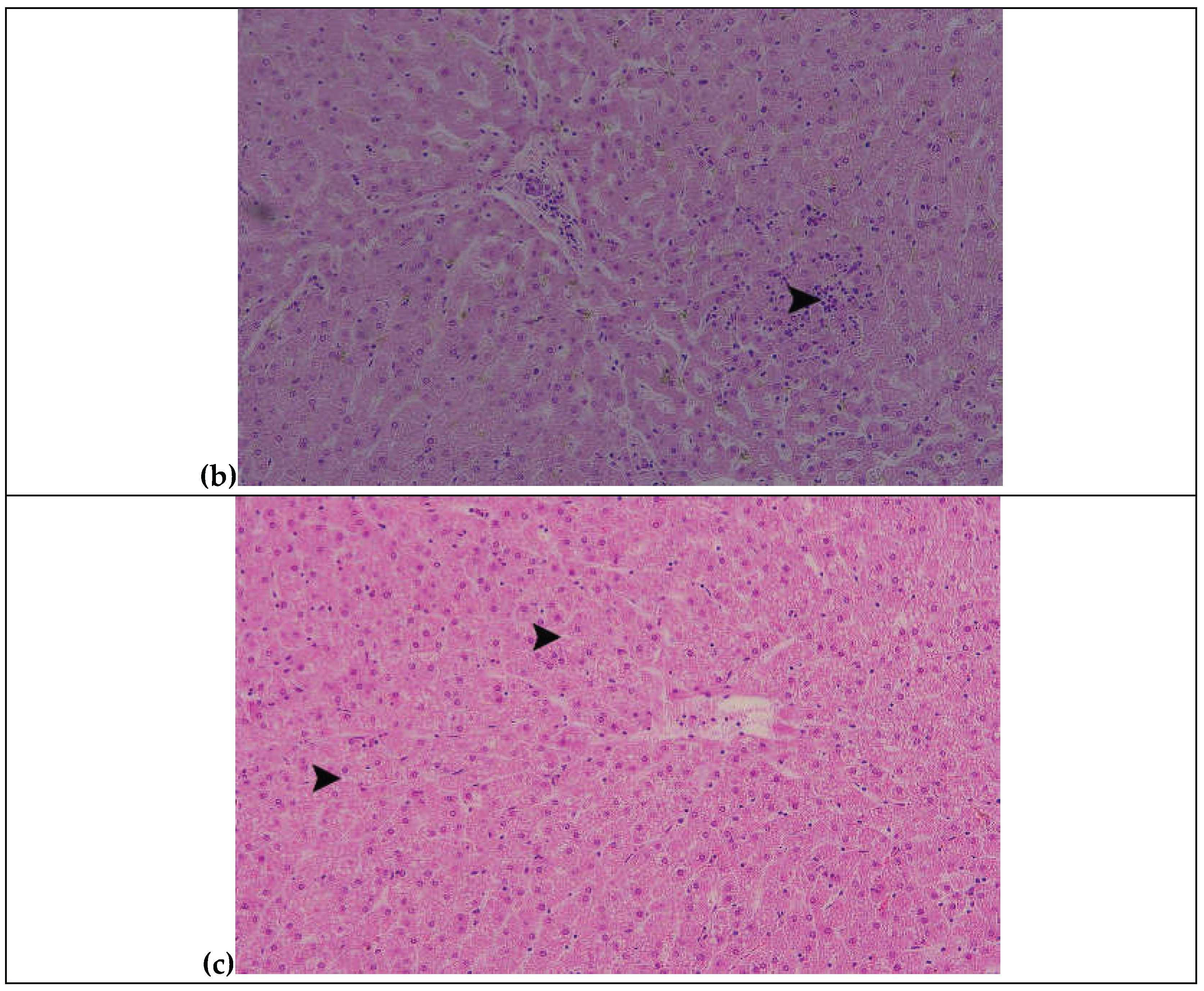
3.6. Histopathological Findings
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Abbreviation | Definition |
| γ-GT | Gamma-glutamyl transferase |
| AF | Aflatoxins |
| AFB1 | Aflatoxin B1 |
| ALT | Alanine aminotransferase |
| aNDFom | Amylase-treated neutral detergent fiber, expressed exclusive of residual ash |
| AST | Aspartate aminotransferase |
| BFH12 | Bovine fetal hepatocyte cell line |
| CARBs | Protein carbonyls |
| CP | Crude protein |
| CYP1A1 | Cytochrome P450 1A1 |
| CYP3A74 | Cytochrome P450 3A74 |
| DM | Dry matter |
| DON | Deoxynivalenol |
| E.R.D.C. | Ethics and Research Deontology Committee |
| EE | Ether extract |
| EU | European Union |
| FASS | Federation of Animal Science Societies |
| FUMs | Fumonisins |
| FUMB1 | Fumonisin B1 |
| H&E | Hematoxylin and eosin |
| HT-2 | HT-2 toxin |
| LC–MS/MS | Liquid chromatography–tandem mass spectrometry |
| LOD | Limit of detection |
| LOQ | Limit of quantification |
| MDA | Malondialdehyde |
| nd | Not detected |
| ppb | parts per billion |
| OTA | Ochratoxin A |
| SD | Standard deviation |
| SPSS | Statistical Package for the Social Sciences |
| T-2 | T-2 toxin |
| TAC | Total antioxidant capacity |
| TBARS | Thiobarbituric acid reactive substances |
| TMR | Total mixed ration |
| ZEN | Zearalenone |
References
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef]
- Zain, M.E. Impact of mycotoxins on humans and animals. J. Saudi Chem. Soc. 2011, 15, 129–144. [Google Scholar] [CrossRef]
- Fink-Gremmels, J. The role of mycotoxins in the health and performance of dairy cows. Vet. J. 2008, 176, 84–92. [Google Scholar] [CrossRef]
- Magnoli, A.P.; Poloni, V.L.; Cavaglieri, L. Impact of mycotoxin contamination in the animal feed industry. Curr. Opin. Food Sci. 2019, 29, 99–108. [Google Scholar] [CrossRef]
- Zinedine, A.; El Akhdari, S. Food Safety and Climate Change: Case of Mycotoxins. In Handbook of Research on Global Environmental Changes and Human Health; IGI Global: Hershey, PA, USA, 2019; pp. 39–62. [Google Scholar]
- Rodrigues, I. A Review on the Effects of Mycotoxins in Dairy Ruminants. Anim. Prod. Sci. 2014, 54, 1155–1165. [Google Scholar] [CrossRef]
- Streit, E.; Schwab, C.; Sulyok, M.; Naehrer, K.; Krska, R.; Schatzmayr, G. Multi-Mycotoxin Screening Reveals the Occurrence of 139 Different Secondary Metabolites in Feed and Feed Ingredients. Toxins 2013, 5, 504–523. [Google Scholar] [CrossRef] [PubMed]
- Gallo, A.; Giuberti, G.; Frisvad, J.C.; Bertuzzi, T.; Nielsen, K.F. Review on mycotoxin issues in ruminants: Occurrence in forages, effects on health and animal performance and practical strategies to counteract their negative effects. Toxins 2015, 7, 3057–3111. [Google Scholar] [CrossRef]
- Dänicke, S.; Brezina, U. Kinetics and metabolism of the Fusarium toxin deoxynivalenol in farm animals. Food Chem. Toxicol 2013, 60, 58–75. [Google Scholar] [CrossRef]
- Bryden, W.L. Mycotoxin contamination of the feed supply chain: Implications for animal productivity and feed security. Anim. Feed Sci. Technol. 2012, 173, 134–158. [Google Scholar] [CrossRef]
- Marroquin-Cardona, A.G.; Johnson, N.M.; Phillips, T.D.; Hayes, A.W. Mycotoxins in a changing global environment: A review. Food Chem. Toxicol. 2014, 69, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Iori, S.; Lahtela-Kakkonen, M.; D'Onofrio, C.; Maietti, F.; Mucignat, G.; Bardhi, A.; Barbarossa, A.; Zaghini, A.; Pauletto, M.; Dacasto, M.; Giantin, M. New insights into aflatoxin B1 mechanistic toxicology in cattle liver: an integrated approach using molecular docking and biological evaluation in CYP1A1 and CYP3A74 knockout BFH12 cell lines. Arch Toxicol. 2024, 98, 3097–3108. [Google Scholar] [CrossRef] [PubMed]
- Patsoukis, N.; Georgiou, C. D. Determination of the thiol redox state of organisms: new oxidative stress indicators. Anal. Bioanal. Chem. 2004, 378(7), 1783–1792. [Google Scholar] [CrossRef]
- Celi, P. Biomarkers of oxidative stress in ruminant medicine. Immunopharmacol Immunotoxicol. 2011, 33, 233–240. [Google Scholar] [CrossRef]
- Jouany, J.P. Methods for preventing, decontaminating and minimizing the toxicity of mycotoxins in feeds. Anim. Feed Sci. Technol. 2007, 137, 342–362. [Google Scholar] [CrossRef]
- Kolosova, A.; Stroka, J. Substances for reduction of the contamination of feed by mycotoxins: A review. World Mycotoxin J. 2011, 4, 225–256. [Google Scholar] [CrossRef]
- Fiorbelli, E.; Lapris, M.; Errico, M.; Della Badia, A.; Riahi, I.; Rocchetti, G.; Gallo, A. Mycotoxin Challenge in Dairy Cows: Assessment of the Efficacy of an Anti-Mycotoxin Agent by Adopting an In Vitro Rumen Simulation Method. Toxins. 2024, 16, 490. [Google Scholar] [CrossRef]
- Papatsiros, V.G.; Eliopoulos, C.; Voulgarakis, N.; Arapoglou, D.; Riahi, I.; Sadurní, M. Effects of a multi-component mycotoxin-detoxifying agent on oxidative stress, health and performance of sows. Toxins 2023, 15, 580. [Google Scholar] [CrossRef]
- Papatsiros, V.G.; Papakonstantinou, G.I.; Voulgarakis, N.; Eliopoulos, C.; Marouda, C.; Meletis, E. Effects of a curcumin/silymarin/yeast-based mycotoxin detoxifier on redox status and growth performance of weaned piglets. Toxins 2024, 16, 168. [Google Scholar] [CrossRef]
- Armanini, E.H.; Boiago, M.M.; de Oliveira, P.V.; Roscamp, E.; Strapazzon, J.V.; de Lima, A.G.; Copetti, P.M.; Morsch, V.M.; de Oliveira, F.C.; Wagner, R.; et al. Inclusion of a phytogenic bend in broiler diet as a performance enhancer and anti-aflatoxin agent: Impacts on health, performance, and meat quality. Res. Vet. Sci. 2021, 137, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.; He, J.; Ahmad, H.; Shen, M.; Zhao, Y.; Gan, Z.; Zhang, L.; Zhong, X.; Wang, C.; Wang, T. Dietary Curcumin Supplementation Increases Antioxidant Capacity, Upregulates Nrf2 and Hmox1 Levels in the Liver of Piglet Model with Intrauterine Growth Retardation. Nutrients. 2019, 11, 2978. [Google Scholar] [CrossRef] [PubMed]
- Ledur, P.C.; Santurio, J.M. Cytoprotective Effects of Curcumin and Silymarin on PK-15 Cells Exposed to Ochratoxin A, Fumonisin B1 and Deoxynivalenol. Toxicon. 2020, 185, 97–103. [Google Scholar] [CrossRef]
- Tedesco, D.; Steidler, S.; Galletti, S.; Tameni, M.; Sonzogni, O.; Ravarotto, L. Efficacy of Silymarin-Phospholipid Complex in Reducing the Toxicity of Aflatoxin B1 in Broiler Chicks. Poult. Sci. 2004, 83, 1839–1843. [Google Scholar] [CrossRef]
- Tuong, D. T. C.; Moniruzzaman, M.; Smirnova, E.; Chin, S.; Sureshbabu, A.; Karthikeyan, A.; Min, T. Curcumin as a Potential Antioxidant in Stress Regulation of Terrestrial, Avian, and Aquatic Animals: A Review. Antioxidants 2023, 12, 1700. [Google Scholar] [CrossRef]
- Tedesco, D.E.A.; Guerrini, A. Use of milk thistle in farm and companion animals: A review. Planta Med. 2023, 89, 584–607. [Google Scholar] [CrossRef] [PubMed]
- Wellington, K.; Jarvis, B. Silymarin: A review of its clinical properties in the management of hepatic disorders. BioDrugs 2001, 15, 465–489. [Google Scholar] [CrossRef] [PubMed]
- Catellani, A.; Ghilardelli, F.; Trevisi, E.; Cecchinato, A.; Bisutti, V.; Fumagalli, F.; Gallo, A. Effects of supplementation of a mycotoxin mitigation feed additive in lactating dairy cows. Toxins 2023, 15, 546. [Google Scholar] [CrossRef] [PubMed]
- Helferich, W.G.; Baldwin, R.L.; Hinders, R.G. Effects of aflatoxin B1 on feed intake, growth, and liver function in dairy cattle. J. Dairy Sci. 1986, 69, 2237–2245. [Google Scholar]
- European Commission. European Commission Directive 2002/32/EC of The European Parliament and of The Council of 7 May 2002 on Undesirable Substances in Animal Feed. Off. J. Eur. Union 2019, L289, 32–36. [Google Scholar]
- European Commission. Commission Recommendation of 17 August 2006 on the presence of deoxynivalenol; zearalenone; ochratoxin A; T-2 and HT-2 and fumonisins in products intended for animal feeding. Off. J. Eur. Union 2006, L229, 7–9. [Google Scholar]
- European Commission. Commission Recommendation of 27 March 2013 on the presence of T-2 and HT-2 toxin in cereals and cereal products. Off. J. Eur. Union 2013, L91, 12–15. [Google Scholar]
- European Commission. Commission Regulation (EU) No 574/2011 of 16 June 2011 amending Annex I to Directive 2002/32/EC of the European Parliament and of the Council as regards maximum levels for nitrite; melamine; Ambrosia spp. and car-ry-over of certain coccidiostats and histomonostats and consolidating Annexes I and II thereto. Off. J. Eur. Union 2011, L159, 7–24. [Google Scholar]
- Gatellier, P.; Mercier, Y.; Renerre, M. Effect of Diet Finishing Mode (Pasture or Mixed Diet) on Antioxidant Status of Charolais Bovine Meat. Meat Sci. 2004, 67, 385–394. [Google Scholar] [CrossRef]
- Bozakova, N.; Ivanov, V. Opportunities for sheep welfare improvement by silymarin additive–a review article. Bulg. J. Agric. Sci. 2022, 28, 502–509. [Google Scholar]
- Jaguezeski, A.M.; Gündel, S.S.; Favarin, F.R.; Gündel, A.; Souza, C.F.; Baldissera, M.D.; Cazarotto, C.C.; Volpato, A.; Fortuoso, B.F.; Ourique, A.F.; Da Silva, A.S. Low-dose curcumin-loaded Eudragit L100-nanocapsules in the diet of dairy sheep increases antioxidant levels and reduces lipid peroxidation in milk. J. Food Biochem. 2019, 43(8). [Google Scholar] [CrossRef]
- Celi, P.; Gabai, G. Oxidant/Antioxidant Balance in Animal Nutrition and Health: The Role of Protein Oxidation. Front. Vet. Sci. 2015, 2. [Google Scholar] [CrossRef] [PubMed]
- Di Trana, A.; Bonanno, A.; Cecchini, S.; Giorgio, D.; Di Grigoli, A.; Claps, S. Effects of Sulla Forage (Sulla coronariumL.) on the Oxidative Status and Milk Polyphenol Content in Goats. J. Dairy Sci. 2015, 98, 37–46. [Google Scholar] [CrossRef]
- Bacou, E.; Walk, C.; Rider, S.; Litta., G.; Perez-Calvo, E. Dietary Oxidative Distress: A Review of Nutritional Challenges as Models for Poultry, Swine and Fish. Antioxidants. 2021, 10(4), 525. [Google Scholar] [CrossRef]
- Bernabucci, U.; Colavecchia, L.; Danieli, P.P.; Basiricò, L.; Lacetera, N.; Nardone, A.; Ronchi, B. Aflatoxin B1 and fumonisin B1 affect the oxidative status of bovine peripheral blood mononuclear cells. Toxicol. In Vitro 2011, 25, 684–691. [Google Scholar] [CrossRef]
- Papakonstantinou, G. I.; Eliopoulos, C.; Meletis, E.; Riahi, I.; Stampinas, E. G.; Arapoglou, D.; Gougoulis, D.; Dimoveli, K.; Filippou, D.; Manouras, A.; Tsekouras, N.; Fotos, L.; Kostoulas, P.; Christodoulopoulos, G.; Papatsiros, V. G. Effects of a phytogenic mycotoxin detoxifier on oxidative status, health, and performance in dairy sheep. Toxins. 2025, 17, 425. [Google Scholar] [CrossRef] [PubMed]
- Vila-Donat, P.; Marín, S.; Sanchis, V.; Ramos, A.J. A review of the mycotoxin adsorbing agents, with an emphasis on multi-binding capacity. Food Chem. Toxicol. 2018, 114, 246–259. [Google Scholar] [CrossRef]
- Upadhaya, S.D.; Sung, H.G.; Lee, C.H. Comparative study on the aflatoxin B1 degradation ability of rumen fluid from Holstein steers and Korean native goats. J. Vet. Sci. 2009, 10, 29–34. [Google Scholar] [CrossRef]
- Gruber-Dorninger, C.; Faas, J.; Doupovec, B. Metabolism of zearalenone in the rumen of dairy cows with and without application of a zearalenone-degrading enzyme. Toxins 2021, 13, 84. [Google Scholar] [CrossRef] [PubMed]
- Bandyk, C.A. Review: Mycotoxins in ruminant livestock production. Appl. Anim. Sci. 2024, 40, 802–817. [Google Scholar] [CrossRef]
- Karlovsky, P. Biological detoxification of the mycotoxin deoxynivalenol and its use in genetically engineered crops and feed additives. Appl Microbiol Biotechnol. 2011, 91(3), 491–504. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Contaminants in the Food Chain (CONTAM). Risks to animal and human health related to deoxynivalenol. EFSA Journal. 2017, 15(9), 4718.
- Pestka, J.J. Deoxynivalenol: mechanisms of action, human exposure, and toxicological relevance. Arch Toxicol. 2010, 84, 663–79. [Google Scholar] [CrossRef]
- Grenier, B.; Applegate, T.J. Modulation of intestinal functions following mycotoxin ingestion: meta-analysis of published experiments in animals. Toxins 2013, 5, 396–430. [Google Scholar] [CrossRef]
- Surai, P.F. Silymarin as a Natural Antioxidant: An Overview of the Current Evidence and Perspectives. Antioxidants 2015, 4, 204–247. [Google Scholar] [CrossRef]
- Riahi, I.1; Sadurní, M.1; Codina, R.1; Ferrús, D.1; Escrivá, L.2; Meca, G. The in vitro efficacy of a detoxifying agent to mitigate don-induced oxidative stress in hepatic cells. Book of abstracts of 1st International Workshop of the Spanish network on mycotoxins, toxigenic fungi and their decontamination processes & MycoTWIN Workshops 5-6, Valencia, June 28 to 30, 2023; p. 44. [Google Scholar]
- Mavrommatis, A.; Giamouri, E.; Tavrizelou, S.; Zacharioudaki, M.; Danezis, G.; Simitzis, P. E.; Zoidis, E.; Tsiplakou, E.; Pappas, A. C.; Georgiou, C. A.; Feggeros, K. Impact of Mycotoxins on Animals' Oxidative Status. Antioxidants 2021, 10, 214. [Google Scholar] [CrossRef]
- Riahi, I.; Ramos, A.J.; Raj, J.; Jakovˇcevi´c, Z.; Farkaš, H.; Vasiljevi´c, M.; Pérez-Vendrell, A.M. Effect of a Mycotoxin Binder (MMDA) on the Growth Performance, Blood and Carcass Characteristics of Broilers Fed Ochratoxin A and T-2 Mycotoxin Contaminated Diets. Animals 2021, 11, 3205. [Google Scholar] [CrossRef]
- Damiano, S.; Longobardi, C.; Andretta, E.; Prisco, F.; Piegari, G.; Squillacioti, C.; Montagnaro, S.; Pagnini, F.; Badino, P.; Florio, S.; et al. Antioxidative Effects of Curcumin on the Hepatotoxicity Induced by Ochratoxin A in Rats. Antioxidants. 2021, 10, 125. [Google Scholar] [CrossRef] [PubMed]
- Tedesco, D.; Domeneghini, C.; Sciannimanico, D.; Tameni, M.; Steidler, S.; Galletti, S. Silymarin, a possible hepatoprotector in dairy cows: biochemical and histological observations. J Vet Med A Physiol Pathol Clin Med. 2004, 51(2), 85–90. [Google Scholar] [CrossRef] [PubMed]
- Gibson-Corley, K. N.; Olivier, A. K.; Meyerholz, D. K. Principles for valid histopathologic scoring in research. Vet. Pathol 2013, 50(6), 1007–1015. [Google Scholar] [CrossRef] [PubMed]
| Ingredients | Kg as fed | Chemicals | |||||
|---|---|---|---|---|---|---|---|
| DM | CP | EE | Starch | Ash | aNDFom | ||
| Oat Hay | 5.00 | 88.0 ± 1.0 | 8.0 ± 0.5 | 1.5 ± 0.2 | 2.0 ± 0.3 | 8.5 ± 0.5 | 50.0 ± 1.0 |
| Corn meal | 4.3 | 88.02 ± 0.28 | 10.73 ± 0.14 | 4.34 ± 0.15 | 69.47 ± 1.73 | 1.53 ± 0.11 | - |
| Soymeal (CP: 47%) | 0.550 | 88.83 ± 0.04 | 48.41 ± 0.71 | 3.70 ± 0.14 | - | 7.17 ± 0.48 | - |
| Barley | 3.75 | 88.0 ± 1.0 | 11.0 ± 0.5 | 2.0 ± 0.2 | 60.0 ± 1.5 | 2.5 ± 0.3 | 19.0 ± 1.0 |
| Wheat bran | 0.93 | 89.0 ± 1.0 | 15.5 ± 0.5 | 4.0 ± 0.2 | 18.0 ± 1.0 | 5.0 ± 0.3 | 42.0 ± 1.5 |
| Cotton seed (kg/DM) | 0.93 | 92.0 ± 1.0 | 23.0 ± 1.0 | 19.0 ± 0.5 | 0.5 ± 0.1 | 4.0 ± 0.2 | 30.0 ± 1.0 |
| Mineral and vitamin premix kg/DM* | 0.04 | 95.0 ± 1.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 90.0 ± 2.0 | 0.0 ± 0.0 |
| Salt | 0.04 | 95.0 ± 1.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 95.0 ± 2.0 | 0.0 ± 0.0 |
| CaCO3 | 0.06 | 95.0 ± 1.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 95.0 ± 2.0 | 0.0 ± 0.0 |
| *Chemical Analysis %: Sodium (Na): 7.70% Calcium (Ca): 22.13% Phosphorus (P): 5.15% Magnesium (Mg): 0.72% Additives per kg: Vitamins: Vitamin A (3a672a) 480,000 I.U. Vitamin D3 (3a671) 96,000 I.U. Vitamin E (3a700) 4,000 mg, Vitamin B1 (3a821) 200 mg, Niacin (3a314) 4,000 mg, Biotin (3a880) 200 mg, Choline (3a890) 2,000 mg Trace Elements: Manganese (Oxide-3b502) 2,080 mg, Iron (Oxide-E172) 2,000 mg, Zinc (Oxide-3b603) 4,800 mg, Iodine (Calcium Iodate-3b202) 80 mg, Selenium (Sodium Selenite-3b801) 20 mg DM: Dry Matter, CP: Crude Protein, EE: Ether Extract, aNDFom: amylase-treated Neutral Detergent Fiber, expressed exclusive of residual ash | |||||||
| Type of mycotoxin | TMR (ppb) |
EU regulatory maximum levels (p.p.b.) | Guidance values (p.p.b.) |
|---|---|---|---|
| AFB1 | 37 | 10 | 5 |
| FUMs | 8402 | 20,000 | 1,500 |
| DON | 4077 | 2,000 | 300 |
| ZEN | 342.3 | 500 | 100 |
| T-2 / HT-2 toxins | <40 | – | 50 |
| OTA | 1.75 | 100 | 50 |
| * European Commission [29,30,31,32] | |||
| Parameter | T1 (Control group) | T2 (Experimental group) |
|---|---|---|
| Urea (mg/dL) | 22.17 ± 2.79 | 18.17 ± 8.28 |
| Creatinine (mg/dL) | 1.91 ± 0.18 | 1.89 ± 0.17 |
| SGOT (AST) (U/L) | 111.5 ± 17.2 | 112.5 ± 15.33 |
| SGPT (ALT) (U/L) | 50.5 ± 6.02 | 53.17 ± 6.77 |
| γ-GT (U/L) | 20.0 ± 2.45* | 16.67 ± 2.70* |
| Total Proteins (g/dL) | 8.52 ± 0.60 | 8.62 ± 0.56 |
| Albumin (g/dL) | 4.42 ± 0.13 | 4.65 ± 0.15 |
| * A statistically significant decrease (P < 0.05) in γ-glutamyl transferase (γ-GT) activity was observed in the treatment group compared with the control group. | ||
| Biomarkers | T1 (control group) |
T2 (experimental group) |
P value |
|---|---|---|---|
| CARBs (nmol/mg protein) | 1.14 ± 0.2 | 0.87 ± 0.3 | <0.05 |
| TBARS (μM MDA) | 6.27 ± 0.2 | 5.45 ± 0.3 | 0.24 |
| TAC (mM Trolox Equivalent) | 0.83 ± 0.1 | 0.83 ± 0.01 | 0.11 |
| Mycotoxin | T1 (control group) |
T2 (experimental group) |
|---|---|---|
| AFB1 | nd | 19.0 ± 3.0 |
| FUMB1 | nd | nd |
| DON | 90.6 ± 11.1 | nd |
| ZEN | 23.0 ± 3.0 | nd |
| T2/HT-2 toxins | nd | nd |
| OTA | nd | nd |
| nd = not detected. | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




