Submitted:
14 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
- Study Design and Population
- Procedural Management and Follow-Up
- Data Collection
- External Validation – AUTHEARTVISIT Study
- Structured Literature search
- Endpoints
- Statistical Analysis
Results
- Baseline Clinical and Demographic Characteristics
- Procedural data
- Pacemaker implantation after TAVI
- Predictors of PM implantation within 30 days of TAVI
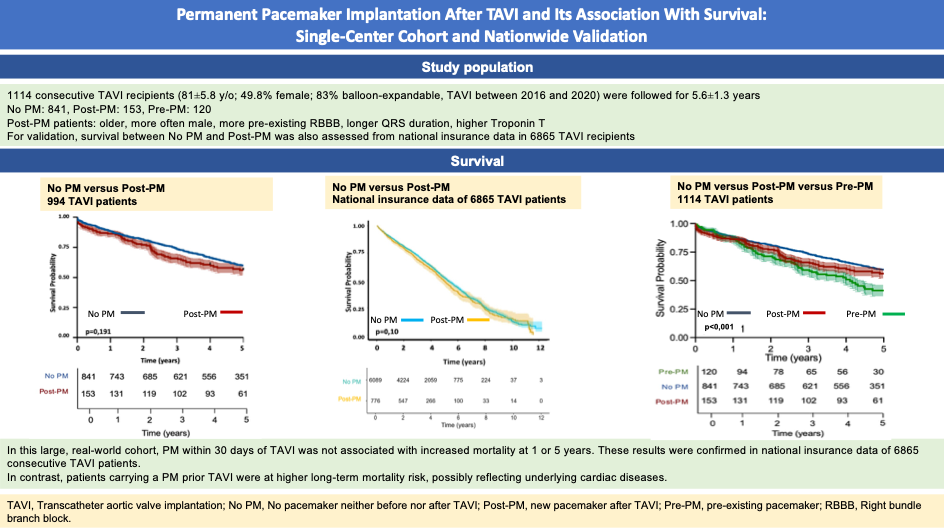
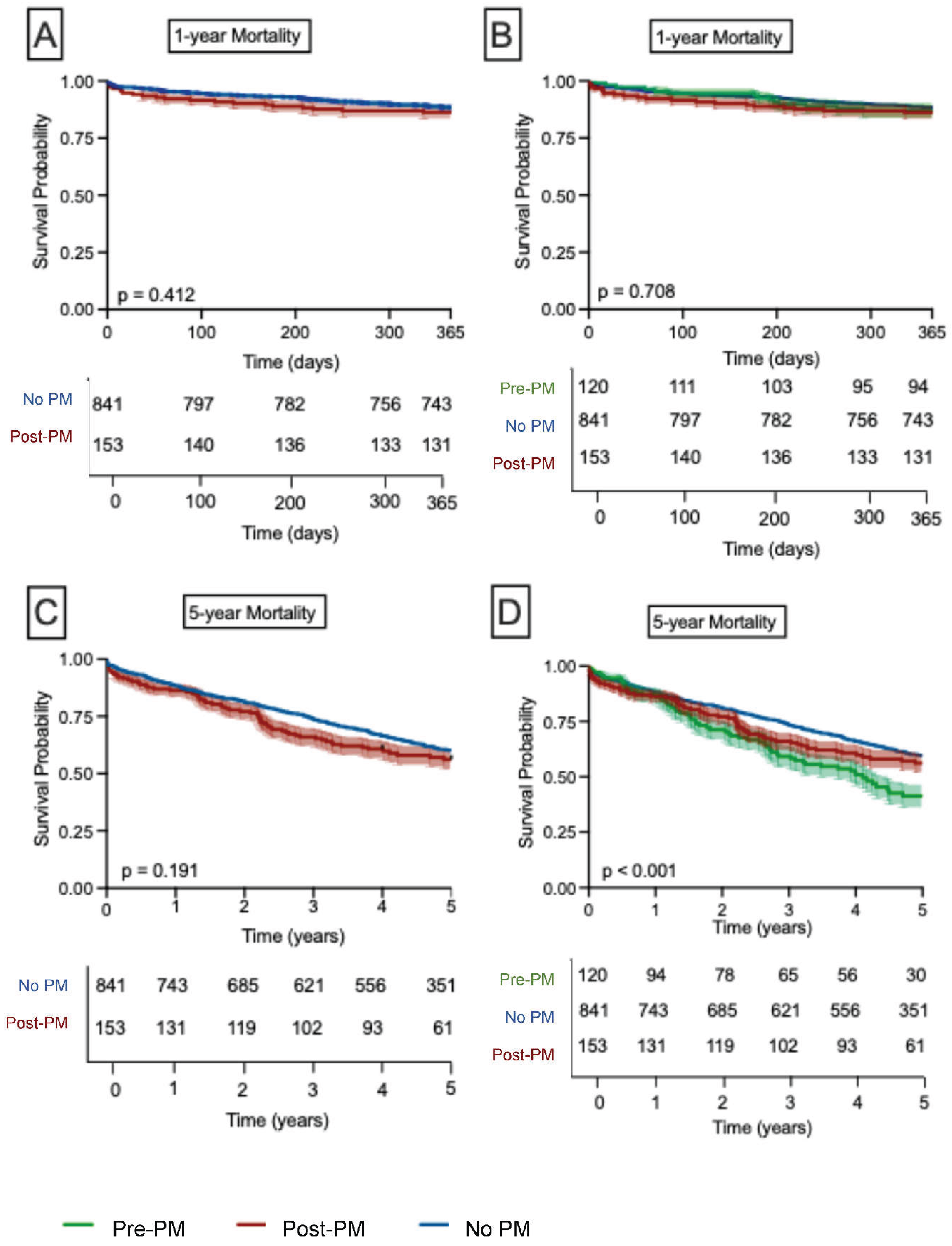
- Outcome at one and five years
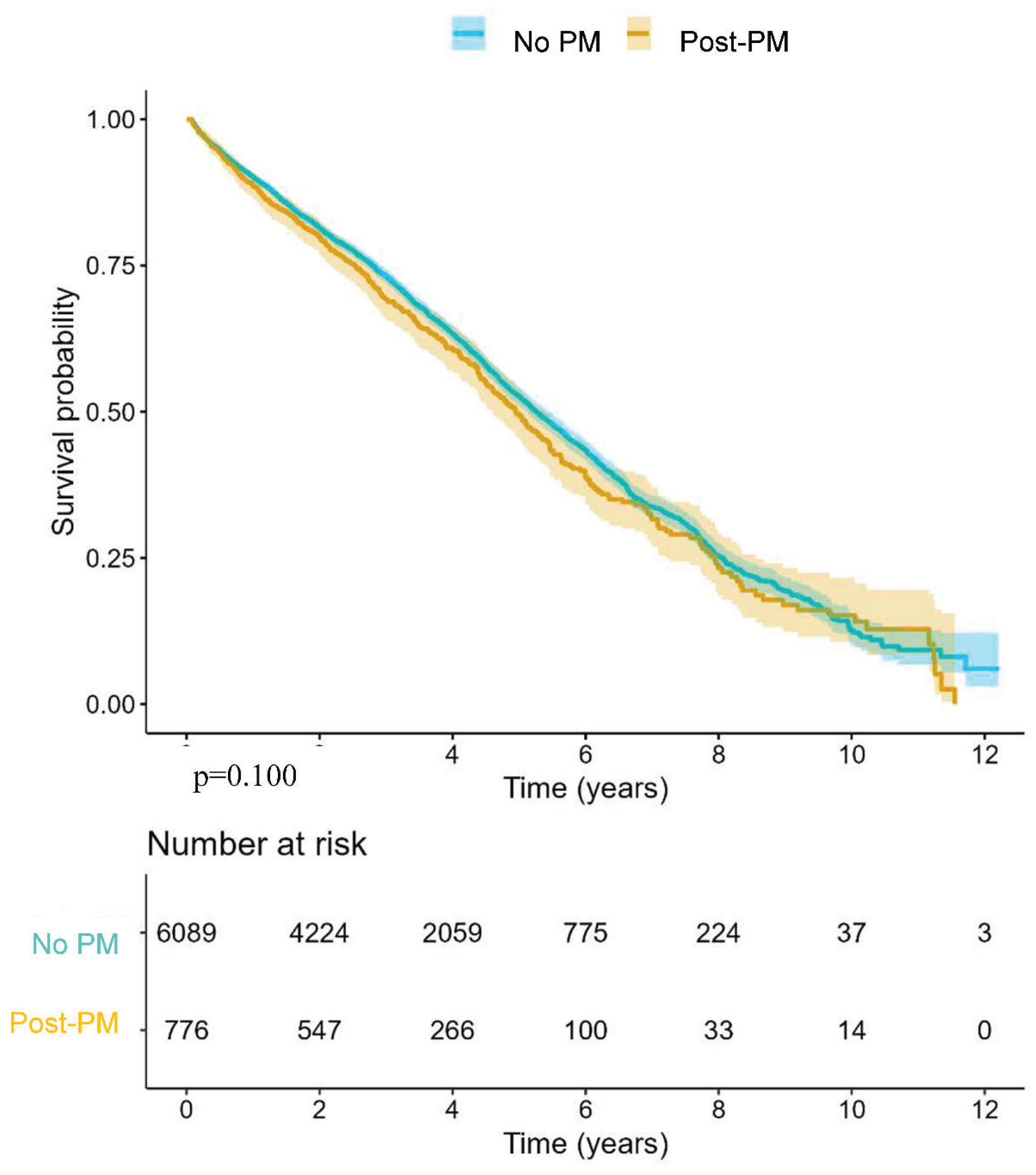
- AUTHEARTVISIT National Dataset
- Overview of key studies on post-TAVI permanent PM and clinical outcomes
Discussion
| Study | Design & Population | Study Period | Valve Types | Post-PM Rate | Follow-up | Mean Age | Main Findings | Limitations |
|---|---|---|---|---|---|---|---|---|
| Studies reporting no significant effect of post-TAVI PM on mortality | ||||||||
| Present Study (Lamm et al., 2025) | Single-center prospective cohort, n = 1,114+ validation in AUTHEARTVISIT (n ≈ 8,000) |
2011–2022 | 84% balloon-expandable, 12% self-expanding, 4% | 14% | Median 4.2 y | 81 ± 6 | Post-PM not associated with increased 1- or 5-year mortality; confirmed in national dataset; Pre-PM significantly associated with worse outcomes | No pacing-burden or CT-based depth data; no cause-specific mortality or HF hospitalization analysis |
| SWEDEHEART [12] | Nationwide registry, n = 3,420 | 2008–2018 | 38% ballon expandable, Self-expandable (not reported) |
14% | Median 2.7 y Up to 10 y |
82 ± 7 | No significant differences in mortality, HF hospitalization, or endocarditis between Post-PM and No-PM groups | No pacing-burden data; older-generation devices |
| Hochstadt et al. [13] | Single-center study, n = 1,489 | 2009–2019 | Ballon expandable (not reported) 62% self-expandable |
19% | Up to 6 y | 80 ± 7 | No significant association between Post-PM and long-term mortality, even at high pacing burden; Pre-PM significantly associated with higher mortality | Retrospective design, no CT-based implantation-depth assessment |
| Chen (2024) PARTNER 2 S3 [18] |
Multicenter registry, USA, n = 857 | 2014–2017 | Balloon-expandable (SAPIEN 3 only) | 12.5% | 60 Months | PM 83 ± 5 No PM 81 ± 7 |
No increased long-term mortality with post-TAVI PM; outcomes mainly driven by comorbidities | Limited sample size, device-specific, retrospective design |
| Wasim (2025) TAVI-NOR [19] |
Single-center n = 548 |
2012–2019 | ≈70% self-expanding, 18% mechanically expandable |
31.5% | 84 Months | 80 ± 7 | No significant difference in 7-year mortality between post-PM and no-PM groups | Small cohort, high SEV share, retrospective |
| Myat (2021) UK-TAVI [17] |
Nationwide registry, n = 6,815 |
2007-2015 | ≈60% balloon-expandable, ≈40% self-expanding | 19.2% | 84 Months | PM 82 ±7 No PM 81 ±7 |
No independent association of post-PM with long-term mortality after adjustment | Retrospective design, limited granularity of procedural data |
| Studies reporting increased risk associated with post-TAVI PM | ||||||||
| Swiss TAVI [9] | Nationwide registry, n = 13,360 | 2011–2022 | 49% balloon-expandable, 47% self-expanding, 2,5% mechanically expandable | 20% | Median 4.9 y 120 Months |
81 ± 6 | Post-PM associated with higher all-cause mortality; absolute survival differences small; Kaplan–Meier curves cross multiple times | No valve-specific subgroup analyses, no pacing-burden data, potential non-proportional hazards |
| Danish Study [10] | Single-center study, n =816 | 2007–2017 | 9% balloon-expandable, 83% self-expanding, 8% mechanically expandable | 16% | Median 3.5 y | 81 ± 7 | Post-PM associated with increased long-term mortality, HF hospitalization, and reduced LVEF | High SEV share, limited generalizability, retrospective Design |
| Auffret (2024) FRANCE-TAVI [20] |
Nationwide retrospective registry, n = 34,717 | 2013–2019 | ≈60% balloon-expandable, ≈40% self-expanding | 20.1% | 60 Months |
PM 83± 6 No PM 82 ± 7 |
Post-PM associated with higher long-term mortality; absolute effect size modest | Retrospective design, no pacing-burden or CT-depth data |
- Strengths and limitations
Conclusions
- Clinical perspective
Author Contributions
Funding
Clinical Trial Registration
Ethics Approval
Data Availability Statement
Permission to Reproduce Material from Other Sources
Acknowledgments
Conflicts of Interest
Disclosures
Abbreviations
| CI | confidence interval |
| HR | hazard ratio |
| LBBB | left bundle branch block |
| LVEF | left ventricular ejection fraction |
| PM | pacemaker |
| Pre-PM | pre-existing pacemaker |
| Post-PM | new pacemaker within 30 days of TAVI |
| No PM | no pacemaker neither before nor after TAVI |
| RBBB | right bundle branch block |
| TAVI | transcatheter aortic valve implantation |
References
- Sharma T, Krishnan AM, Lahoud R, Polomsky M, Dauerman HL. National Trends in TAVR and SAVR for Patients With Severe Isolated Aortic Stenosis. J Am Coll Cardiol. 2022;80(21):2054-2056. [CrossRef]
- Cilingiroglu M, Marmagkiolis K. Paravalvular leak versus need for permanent pacemaker after TAVR: Sailing between Scylla and Charybdis. Catheter Cardiovasc Interv. 2017:155-156. [CrossRef]
- Van Rosendael PJ,Delgado V, Bax JJ. Conduction disturbances after transcatheter aortic valve implantation. Eur Heart J 2018; 39(21):2003-2013.
- Faroux L, Chen S, Muntane-Carol G, et al. Clinical impact of conduction disturbances in transcatheter aortic valve replacement recipients: A systematic review and meta-analysis. Eur Heart J. 2020;41:2771–2781. [CrossRef]
- Ravaux JM, Di Mauro M, Vernooy K et al. Permanent pacemaker implantation following transcatheter aortic valve implantation using self-expandable, balloon- expandable or mechanically expandalbe devices: a network meta-analysis. Europace. 2021;23(12):1998-2009. [CrossRef]
- Steinberg JS, Fischer A, Wang P et al. The clinical implications of cumulative right ventricular pacing in the Multicenter Automatic Defibrillator Trial II. J Cardiovasc Electrophysiol. 2005;16: 359 –365. [CrossRef]
- Gupta R, Mahajan S, Behnoush AH et al.Short- and Long-Term Clinical Outcomes Following Permanent Pacemaker Insertion Post-TAVR: A Systematic Review and Meta-Analysis. JACC Cardiovascular Interventions 2022;15:1683-92.
- Shen L, Jhund PS, Docherty KF et al. Prior Pacemaker Implantation and Clinical Outcomes in Patients With Heart Failure and Preserved Ejection Fraction. J Am Coll Cardiol HF 2019;7:418–27. [CrossRef]
- Badertscher P, Stortecky S, Serban T et al. SwissTAVI Investigators. Long-term outcomes of patients requiring pacemaker implantation after transcatheter aortiv valve replacement: The SwissTAVI registry. JACC Cardiovasc Interv. 2025 May 12;18(9):1163-1171.
- Jorgensen TH, De Backer O, Gerds TA, Bieliauskas G, Svendsen JH, Sondergaard L. Mortality and heart failure hospitalization in patients with conduction abnormalities after transcatheter aortic valve replacement. J Am Coll Cardiol Intv. 2019;12:52–61. [CrossRef]
- Zito A, Princi G, Lombardi M et al. Long-term clinical impact of permanent pacemaker implantation in patients undergoing transcatheter aortic valve implantation: a systematic review and meta-analysis. Europace. 2022 Jul 21;24(7):1127-1136. [CrossRef]
- Rück A, Saleh N, Glaser N. Outcomes Following Permanent Pacemaker Implantation After Transcatheter Aortic Valve Replacement: SWEDEHEART Observational Study. JACC Cardiovasc Interv. 2021;14(19):2173-2181.
- Hochstadt A, Merdler I, Meridor Y, et al. Effect of pacemaker implantation after transcatheter aortic valve replacement on long- and mid-term mortality. Heart Rhythm. 2021;18:199–206. [CrossRef]
- Auer J, Krotka P, Reichardt B, et al. Selection for transcatheter versus surgical aortic valve replacement and mitdterm survival: results of the AUTHEARTVISIT study. Eur J Cardiothorac Surg. 2024;66(1):ezae214. [CrossRef]
- The Task Force on cardiac pacing and resynchronization therapy of the European Society of Cardiology (ESC). Developed in collaboration with the European Heart Rhythm Association (EHRA). Brignole M, Auricchio A, Baron-Esquivias G, et al. 2013 ESC Guidelines on cardiac pacing and cardiac resynchronization therapy. European Heart Journal (2013) 34, 2281–2329.
- Kusumoto FM, Schoenfeld MH, Barrett C et al. 2018 ACC/AHA/HRS Guideline on the Evaluation and Management of Patients With Bradycardia and Cardiac Conduction Delay: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2019;74(7): 932-987.
- Myat A, Mouy F, Buchner L et al. Survival relative to pacemaker status after transcatheter aortic valve implantation. Cath Cardiovasc Intv 2021;98(3): E444-452. [CrossRef]
- Chen S, Dizon J, Hahn R et al. Predictors and 5-year clinical outcomes of pacemaker after TAVR. J Am Coll Cardiol Intv 2024;17:1325–1336. [CrossRef]
- Wasim D, Ali AM, Bleie O et al. Prevalence and predictors of permanent pacemaker implantation in patients with aortic stenosis undergoing transcatheter aortic valve implantation: a prospective cohort study. BMJ Open 2025;15:e093073. [CrossRef]
- Auffret V, Boulmier D, Didier R et al. Clinical effects of permanent pacemaker implantation after transcatheter aortic valve implantation: Insights from the nationwideFRANCE-TAVI registry. Arch Cardiovasc Dis. 2024;117(3):213-223. [CrossRef]
- Sweeney MO, Hellkamp AS, Ellenbogen KA et al. Adverse effect of ventricular pacing on heart failure and atrial fibrillation among patients with normal baseline QRS duration in a clinical trial of pacemaker therapy for sinus node dysfunction. Circulation2003;107:2932–2937.
- Yu CM, Chan JY, Zhang Q, et al. Biventricular pacing in patients with bradycardia and normal ejection fraction. N Engl J Med. 2009;361: 2123–2134. [CrossRef]
- Costa G, Zappulla P, Barbanti M, et al. Pacemaker dependency after transcatheter aortic valve implantation: incidence, predictors and long-term outcomes. EuroIntervention. 2019;15:875–883. [CrossRef]
- Ueyama HA, Miyamoto Y, Hashimoto K, et al. Comparison of Patient Outcomes Between Leadless vs Transvenous Pacemakers Following Transcatheter Aortic Valve Replacement. JACC Cardiovasc Interv 2024;17:1779–1791. [CrossRef]
| No PM n=841 |
Post-PM n=153 |
Pre-PM N=120 |
p-value | |
|---|---|---|---|---|
| Demographic data | ||||
| Age (years) ## | 805.7 | 81.95.5 | 82.55.8 | 0.006 |
| Gender (female) # | 445 (52.9) | 71 (46.4) | 39 (32.5) | <0.001 |
| BMI (kg/m2) ## | 28.07.5 | 28.512.7 | 28.5 10.7 | 0.246 |
| Comorbidities and biomarkers: | ||||
| DM # | 282 (33.5) | 40 (33.3) | 43 (28.1) | 0.417 |
| CAD # | 420 (49.9) | 71 (46.4) | 67 (55.8) | 0.298 |
| PAD # | 53 (6.3) | 16 (10.5) | 15 (12.5) | 0.019 |
| AF # | 311 (37.0) | 55 (35.9) | 64 (53.3) | 0.002 |
| Euroscore II ## | 4.5.5 | 5.45.9 | 5.5.3 | 0.246 |
| Troponin T (µg/l) * | 23 (16.0; 37.0) | 28.3 (18.0; 32.1) | 30.4 (20.1; 41.0) | 0.002 |
| NT-proBNP (ng/l)* | 1508 (553; 3755) | 1273 (468; 2779) | 2593 (1081; 5055) | <0.001 |
| CRP (mg/dl) * | 0.3 (0.1; 0.7) | 0.2 (0.1; 0.4) | 0.3 (0.1; 0.8) | 0.208 |
| Creatinine (mg/dl) * | 1.0 (0.9; 1.3) | 1.1 (0.9; 1.4) | 1.1 (0.9; 1.4) | 0.003 |
| ECG-parameters (prior to intervention): | ||||
| RBBB # | 27 (3.2) | 43(28.1) | 3 (2.5) | <0.001 |
| LBBB # | 85 (10.1) | 11 (7.2) | 2 (1.7) | 0.007 |
| LAH # | 166 (21.3) | 52 (36.1) | 23 (20) | <0.001 |
| QRS duration * | 100 (80; 110) | 110 (90; 140) | 130 (100; 150) | <0.001 |
| PQ interval * | 170 (150; 200) | 180 (150; 220) | 190 (162; 217) | 0.116 |
| Resting heart rate (bpm) * | 71 (63; 81) | 70 (61; 78) | 60 (60, 70) | 0.047 |
| Echo-parameters (prior to intervention): | ||||
| LVEF (%)* | 60 (50, 60) | 60 (60, 60) | 55 (35, 60) | <0.001 |
| AVmean Gradient (mmHg) * | 44 (36; 55) | 44 (37; 52) | 40 (30; 51) | 0.064 |
| AVA (cm2) * | 0.7 (0.6; 0.8) | 0.7 (0.6; 0.8) | 0.7 (0.6; 0.9) | 0.017 |
| AV Vmax (m/s)* | 4.2 (3.8; 4.6) | 4.2 (3.8; 4.5) | 4.0 (3.5; 4.5) | 0.060 |
| Procedural parameters | ||||
| Valve type | ||||
| Edwards SAPIEN 3 THV- Model 9600 TFX # | 440(52.4) | 83 (54.2) | 73 (60.8) | |
| Edwards Sapien 3 Ultra THV- Model 9750 TFX # | 259 (30.8) | 36 (23.5) | 27 (22.5) | |
| Edwards SAPIEN XT THV-Model 9300 TFX # | 4 (0.5) | 2 (1.7) | 2 (1.3) | |
| Portico™ Transcatheter Aortic Valve # | 76 (9.0) | 17 (11.1) | 13 (10.8) | |
| Evolut™ R system # | 23 (2.7) | 5 (3.3) | 2 (1.7) | |
| Evolut™ PRO+ system # | 5 (0.6) | 1 (0.7) | 0 (0.0) | |
| LOTUS Edge™ Aortic Valve System # | 31 (3.7) | 9 (5.9) | 3 (2.5) | |
| ALLEGRA™ TAVI System TF # | 2 (0.2) | 0 (0.0) | 0 (0.0) | 0.467 |
| Aortic valve size (mm) # | 26.2 2.2 | 26.92.2 | 27.22.0 | <0.001 |
| Type of expansion | ||||
| Balloon-expandable # | 704 (83.7) | 121 (79.1) | 102 (85.0) | |
| Self-expanding # | 106 (12.6) | 23 (15.0) | 15 (12.5) | |
| Mechanically expandable # | 31 (3.7) | 9 (5.9) | 3 (2.5) | 0.516 |
| Balloon pre-dilatation # | 20 (2.4) | 2 (1.3) | 3 (2.5) | 0.699 |
| Balloon post-dilatation # | 86 (10.2) | 15 (9.8) | 10 (8.3) | 0.809 |
| Complications | ||||
| Annular rupture # | 2 (0.2) | 1 (0.7) | 0 (0.0) | 0.550 |
| Cardiac arrest # | 2 (0.2) | 1 (0.7) | 0 (0.0) | 0.550 |
| Aortic dissection # | 1 (0.1) | 0 (0.0) | 0 (0.0) | 0.850 |
| Conversion to open surgery # | 2 (0.2) | 0 (0.0) | 1 (0.8) | 0.393 |
| No PM n = 6089 |
Post-PM n = 776 |
p-value |
|
|---|---|---|---|
| Demographic data | |||
| Age (years)* | 81 (78 - 85) | 82 (78 - 85) | 0.107 |
| Gender (female)# | 3400 (55.8) | 403 (51.9) | 0.043 |
| Comorbidities | |||
| Heart failure # | 1422 (23.3) | 190 (24.4) | 0.512 |
| ACS# | 122 (2.0) | 15 (1.9) | 1 |
| Stroke# | 113 (1.8) | 14 (1.8) | 1 |
| Diabetes mellitus# | 1206 (19.8) | 154 (19.8) | 1 |
| Obesity# | 347 (5.7) | 53 (6.8) | 0.236 |
| Hyperlipidemia# | 1494 (24.5) | 218 (28.0) | 0.035 |
| Hyperuricemia# | 224 (3.6) | 38 (4.9) | 0.117 |
| Valvular CMP# | 5571 (91.4) | 718 (92.5) | 0.364 |
| Ischemic CMP# | 3213 (52.7) | 408 (52.5) | 0.951 |
| Artherosclerosis# | 506 (8.3) | 64 (8.2) | 1 |
| Pulmonary diseases# | 382 (6.2) | 52 (6.7) | 0.702 |
| Kidney diseases Yes# | 1235 (20.2) | 176 (22.6) | 0.131 |
| Malignant diseases# | 385 (6.32%) | 49 (6.31%) | 1 |
| Univariate | Multivariate | ||||||
|---|---|---|---|---|---|---|---|
| OR | CI 95% | p-value | OR | CI 95% | p-value | ||
| Age | ≥ 82 years | 1.4 | 1.0-2.0 | 0.040 | 1.1 | 0.7-1.7 | 0.441 |
| Sex | Male | 1.2 | 0.9-1.7 | 0.172 | |||
| DM | 0.7 | 0.5-1.1 | 0.171 | ||||
| CAD | 0.8 | 0.6-1.2 | 0.362 | ||||
| PAD | 1.6 | 0.9-3.0 | 0.077 | ||||
| AF | 0.9 | 0.6-1.3 | 0.817 | ||||
| Euroscore II | ≥5 points | 1.1 | 0.7-1.6 | 0.465 | |||
| Troponin T (µg/l) | ≥24 pg/ml | 1.2 | 0.8-1.8 | 0.201 | |||
| NT-proBNP (ng/l) | ≥1532 ng/L | 0.8 | 0.5-1.1 | 0.273 | |||
| Creatinine (mg/dl) | ≥1.3mg/dl | 1.2 | 0.9-1.8 | 0.148 | |||
| RBBB | 11.9 | 7.1-20.1 | <0.001 | 12.1 | 6.8-21.4 | <0.001 | |
| LBBB | 0.6 | 0.3-1.3 | 0.267 | ||||
| LAH | 2.0 | 1.4-3.0 | <0.001 | 1.6 | 1.0-2.5 | 0.020 | |
| LVEF | <60% | 1.6 | 1.0.-2.4 | 0.020 | 1.9 | 1.1-3.0 | 0.007 |
| Prosthesis size | >26 mm | 1.7 | 1.2-2.4 | 0.002 | 1.8 | 1.2-2.7 | 0.002 |
| Balloon pre-dilatation | 0.5 | 0.1-2.3 | 0.426 | ||||
| Balloon post-dilatation | 0.9 | 0.5-1.7 | 0.916 | ||||
| Univariate | Multivariate | ||||||
|---|---|---|---|---|---|---|---|
| HR | CI 95% | p-value | HR | CI 95% | p-value | ||
| 1-year all-cause mortality | |||||||
| PM | No PM | 1.0 | |||||
| Post-PM | 1.1 | 0.7-1.9 | 0.455 | ||||
| Pre-PM | 0.9 | 0.5-1.7 | 0.975 | ||||
| Age | ≥ 82 years | 1.1 | 0.8.1.6 | 0.319 | |||
| Gender | Male | 0.9 | 0.6-1.3 | 0.775 | |||
| DM | 1.0 | 0.7-1.4 | 0.976 | ||||
| CAD | 1.1 | 0.8-1.6 | 0.375 | ||||
| PAD | 0.9 | 0.7-1.3 | 0.956 | ||||
| AF | 0.7 | 0.2-2.0 | 0.590 | ||||
| CKD | 2.6 | 1.4-4.9 | 0.002 | 1.4 | 0.5-3.8 | 0.511 | |
| Euroscore II | ≥5 points | 1.7 | 1.2-2.4 | 0.002 | 1.1 | 0.7-1.8 | 0.515 |
| Troponin T | ≥24 pg/ml | 3.3 | 2.1-5.3 | <0.001 | 2.2 | 1.3-3.8 | 0.002 |
| NT-proBNP | ≥1532 ng/L | 2.8 | 1.8-4.4 | <0.001 | 2.2 | 1.3-3.8 | 0.002 |
| Creatinine | ≥1.3mg/dl | 1.6 | 1.2-2.3 | 0.003 | |||
| RBBB | 1.3 | 07-2.4 | 0.372 | ||||
| LBBB | 0.8 | 0.4.1.5 | 0.546 | ||||
| LAH | 1.1 | 0.7-1.7 | 0.403 | ||||
| LVEF | <60% | 1.7 | 1.2-2-4 | <0.001 | 1.1 | 0.7-1.7 | 0.629 |
| Univariate | Multivariate | |||||||
|---|---|---|---|---|---|---|---|---|
| HR | CI 95% | p-value | HR | CI 95% | p-value | |||
| 5 year all-cause mortality | ||||||||
| PM | No PM | 1.0 | ||||||
| Post-PM | 1.2 | 0.9-1.5 | 0.181 | 1.2 | 0.8-1.6 | 0.220 | ||
| Pre-PM | 1.6 | 1.2-2.1 | <0.001 | 1.4 | 1.0-1.9 | 0.030 | ||
| Age | ≥ 82 years | 1.3 | 1.1.-1.6 | <0.001 | 1.3 | 1.1-1.7 | 0.004 | |
| Sex | Female | 1.0 | 0.8-1.2 | 0.556 | ||||
| DM | 1.1 | 0.9-1.3 | 0.170 | |||||
| CAD | 1.1 | 0.9-1.3 | 0.286 | |||||
| PM | 1.1 | 0.9-1.3 | 0.083 | |||||
| AF | 1.5 | 1.3-1.9 | <0.001 | 1.3 | 1.0-1.7 | 0.006 | ||
| CKD | 2.2 | 1.4-3.2 | <0.001 | 2.0 | 1.1-3.8 | 0.020 | ||
| Euroscore II | ≥5 points | 1.5 | 1.2-1.8 | <0.001 | 1.2 | 0.9-1.5 | 0.159 | |
| Troponin T | ≥24 pg/ml | 2.0 | 1.6-2.4 | <0.001 | 1.6 | 1.2-2.0 | <0.001 | |
| NTproBNP | ≥1532 ng/L | 1.8 | 1.5-2.3 | <0.001 | 1.4 | 1.1-1.8 | 0.006 | |
| Creatinine | ≥1.3mg/dl | 1.5 | 1.3-1.9 | <0.001 | ||||
| RBBB | Yes | 1.3 | 0.9-1.9 | 0.071 | ||||
| LBBB | Yes | 1.3 | 0.9-1.5 | 0.072 | ||||
| LAH | Yes | 1.2 | 0.7-1.7 | 0.096 | ||||
| LVEF | <60% | 1.5 | 1.2-2.8 | <0.001 | 1.0 | 0.8-1.3 | 0.656 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




