Submitted:
14 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Bioprinting Technologies and Techniques
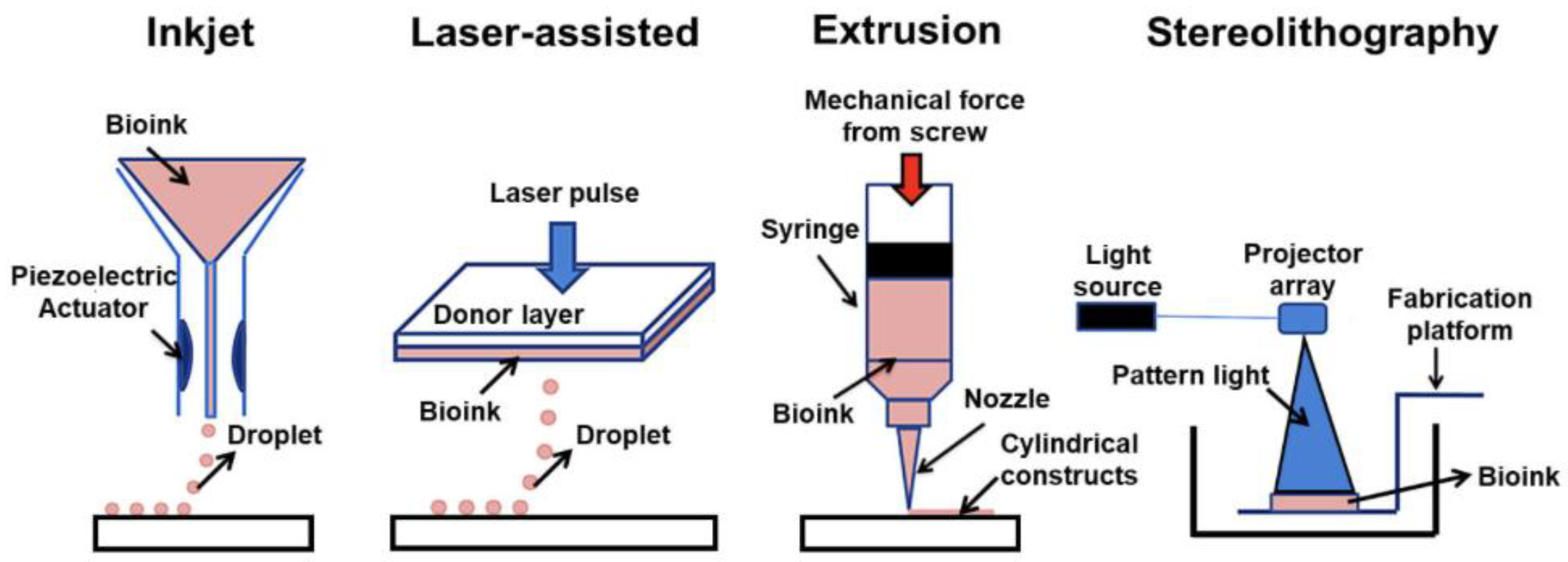
2.1. Extrusion-Based and Inkjet Bioprinting
2.2. Stereolithography Bioprinting
2.3. Laser-Assisted Bioprinting
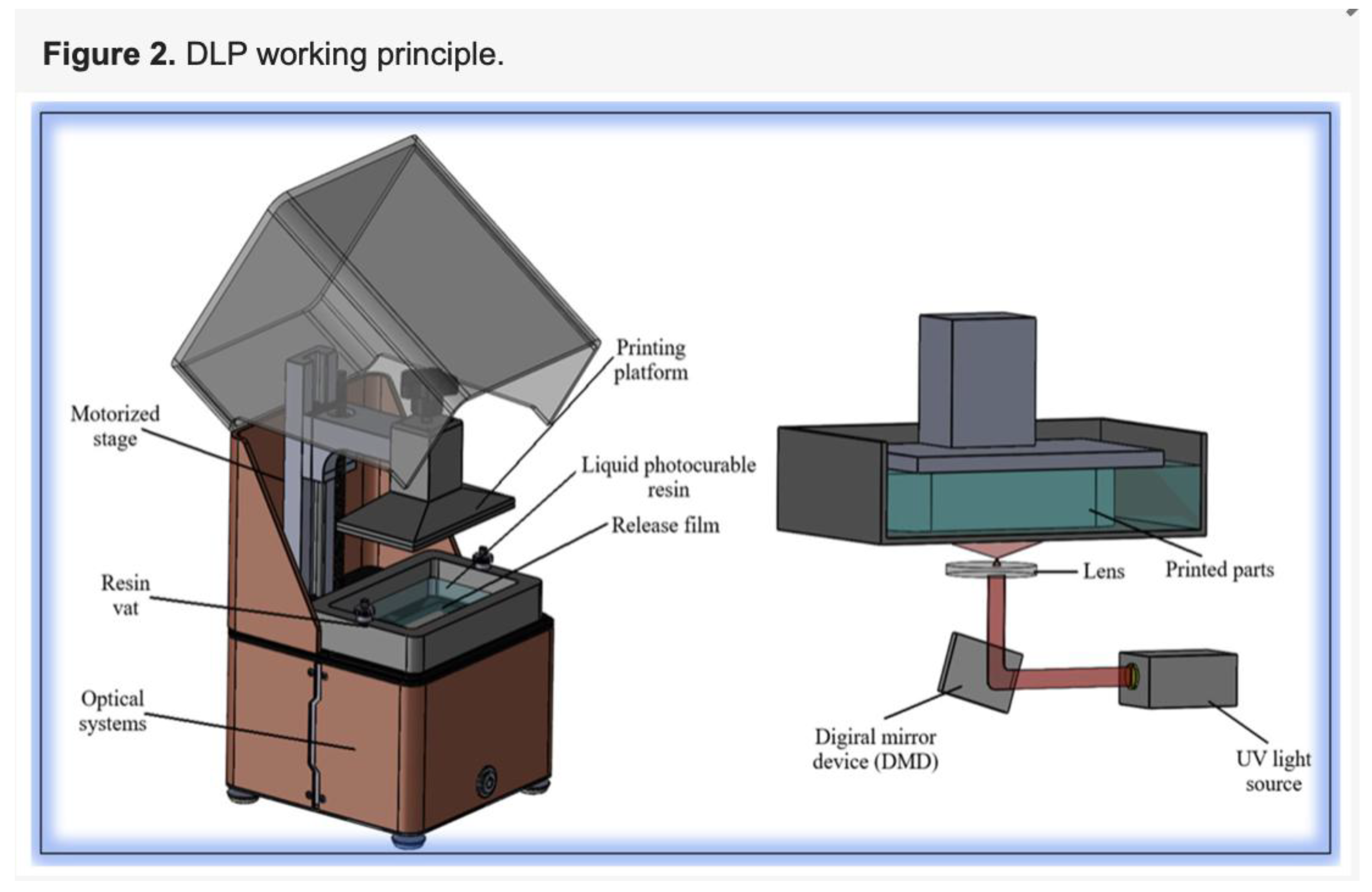
2.4. Digital Light Processing (Dlp) Bioprinting
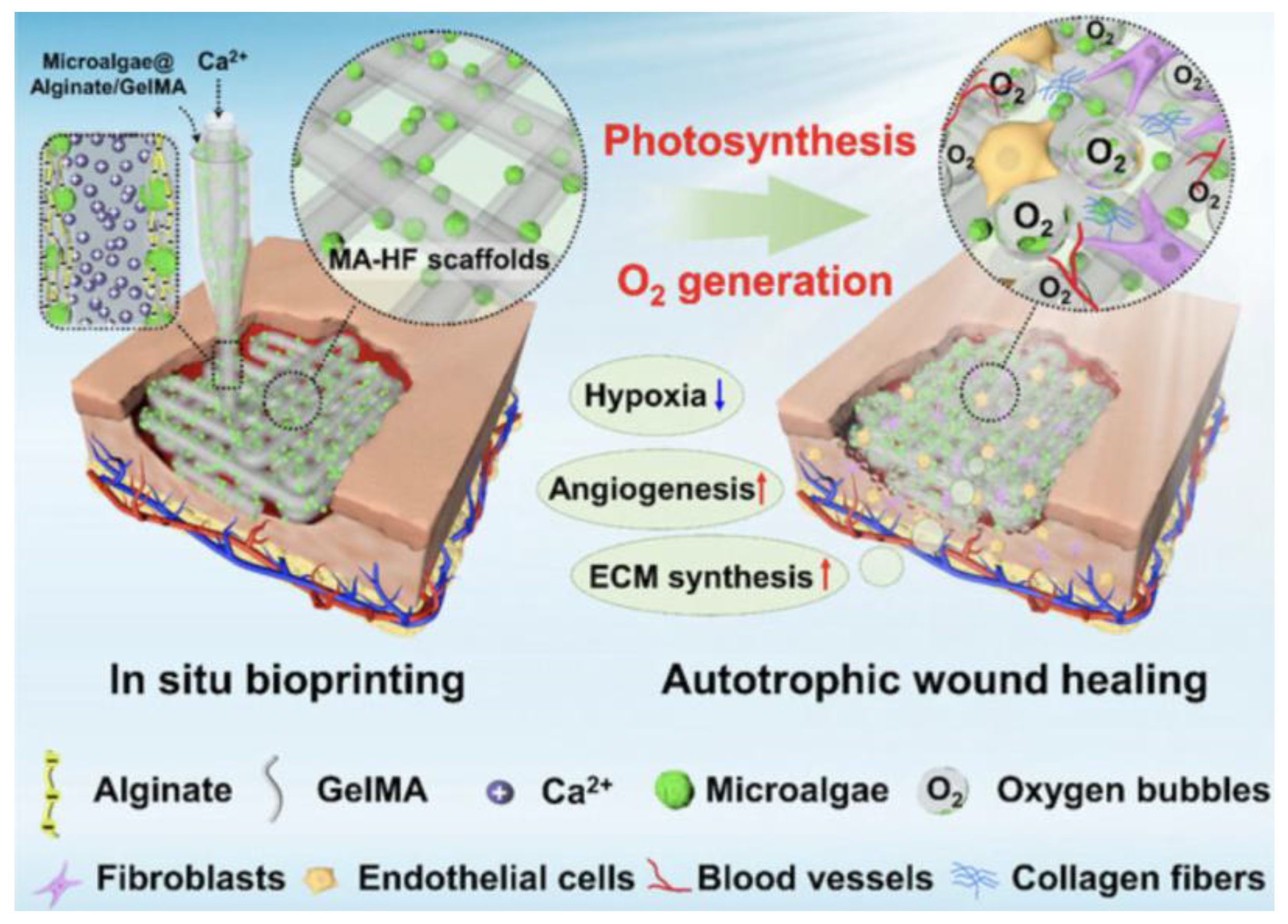
2.5. In Situ Bioprinting
2.6. Coaxial and Microfluidic Bioprinting
3. Biomaterials for Wound Healing
3.1. Natural Polymers
3.2. Synthetic Polymers
3.3. Composite and Hybrid Bioinks
3.4. Bioactive Components
4. Mechanisms of Chronic Wound Healing
4.1. Pathophysiology of Chronic Wounds
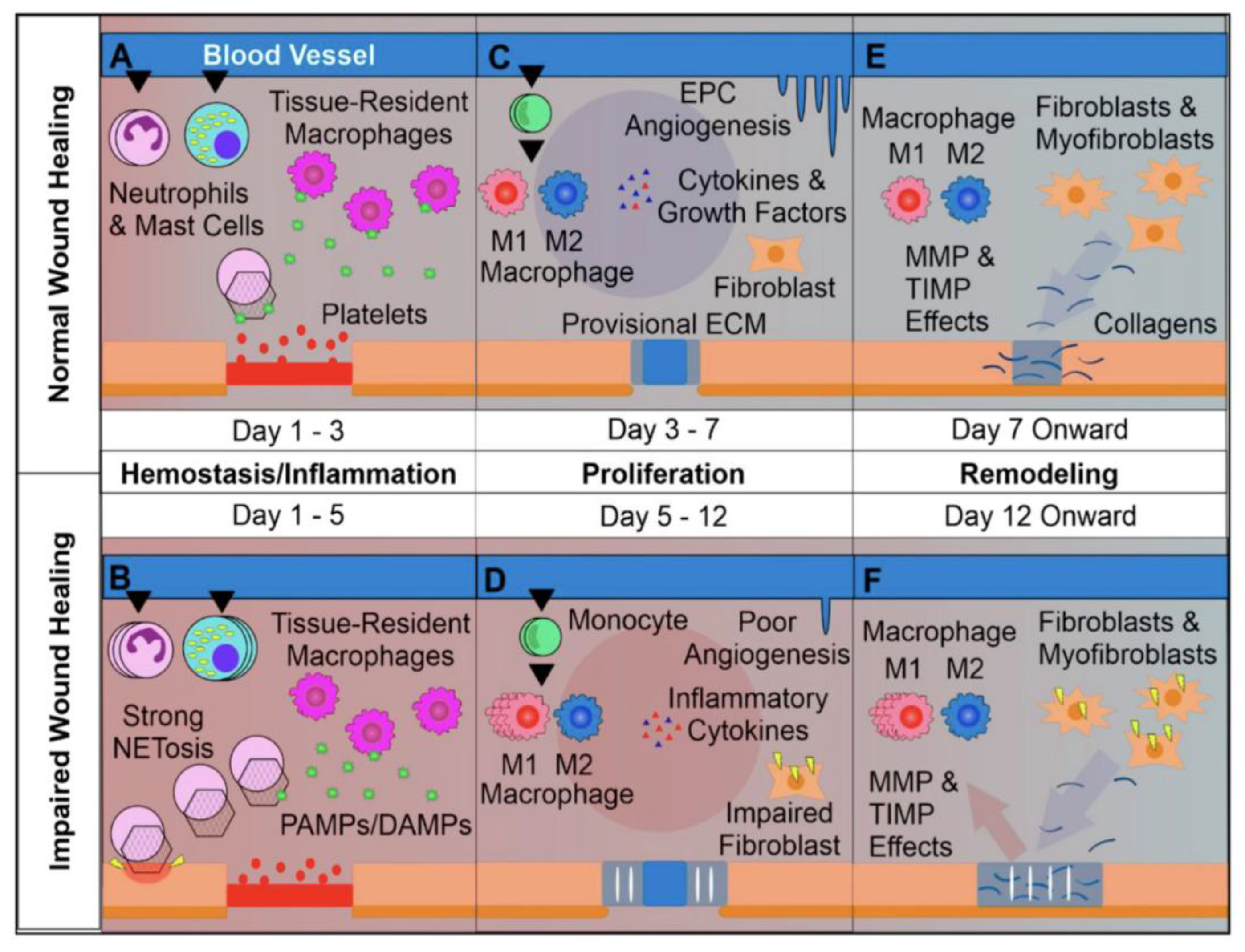
4.2. Angiogenesis and Vascularization
4.3. Immunomodulation and Inflammation Control
4.4. Extracellular Matrix Remodeling
5. Clinical Applications and Outcomes
5.1. Diabetic Wound Healing
5.2. Full-Thickness Skin Defects
5.3. Comparative Analysis of Bioprinted Constructs
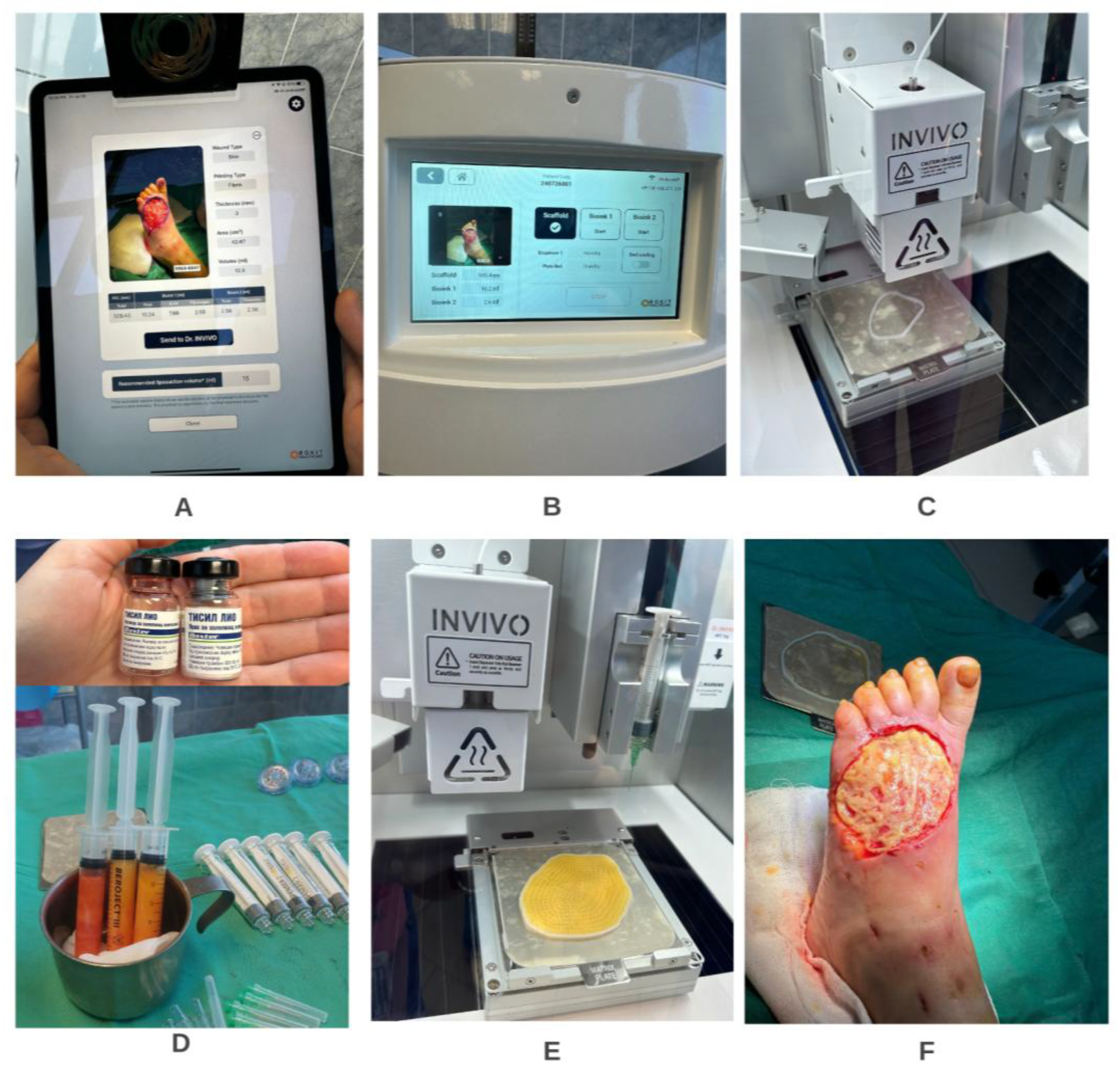
5.4. Clinical Case Series: 3D Dr. Invivo Bioprinter Experience
5.4.1. Overview
5.4.2. Patient Population and Study Design
- Wound duration: >4 years of failed treatment
- Comorbidities: Stage 2 arterial hypertension (well-controlled) in all patients (100%); diabetes mellitus type 2 (well-controlled) in one patient (25%)
- Infection status: Microbiological examination confirmed absence of active infection in all cases
- Previous treatments: Multiple debridements, advanced wound dressings, negative pressure wound therapy, and failed surgical interventions including skin grafting and local flap reconstruction
5.4.3. Technology and Treatment Protocol
5.4.4. Clinical Outcomes
| Patient Information | Preoperative (Baseline) | Week 2 Postoperative | Week 5 Postoperative | Week 8 Postoperative | Week 11 Postoperative |
| PATIENT 1 67-year-old Male Hypertension (controlled) Wound Duration: 4.5 years |
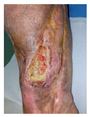 Chronic wound (4.5 years) Failed multiple debridements Failed NPWT Non-healing, stalled epithelialization Infection: Negative Minimal granulation tissue |
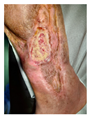 Post-bioprinting (2 weeks) Initial material integration Early cellular migration No infection/rejection Standard wound care maintained |
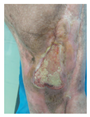 Mid-treatment (5 weeks) Progressive epithelialization Increased granulation tissue Wound contraction beginning Scaffold integration ongoing No adverse events |
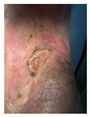 Advanced healing (8 weeks) Significant size reduction Robust epithelialization Healthy granulation tissue Continued remodeling Patient tolerating well |
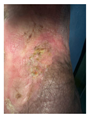 COMPLETE EPITHELIALIZATION 100% wound closure achieved Mature epithelium present No residual defect Successful outcome No complications |
| PATIENT 2 62-year-old Female Hypertension (controlled) Wound Duration: 4.2 years |
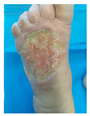 Chronic wound (4.2 years) Failed conservative management Failed surgical closure attempt Persistent non-healing defect Infection: Negative Previous surgery unsuccessful |
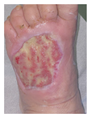 Post-bioprinting (2 weeks) Material adherence confirmed Initial wound bed response Early cellular infiltration No material rejection Well-tolerated treatment |
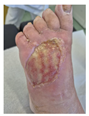 Mid-treatment (5 weeks) Progressive healing response Peripheral epithelialization Improved wound bed quality Bioink scaffold supporting regeneration Continued positive response |
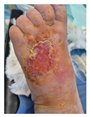 Advanced healing (8 weeks) Marked wound contraction Accelerated epithelialization Healthy vascular tissue formation Success where previous surgery failed No complications |
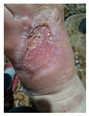 COMPLETE EPITHELIALIZATION 100% wound closure achieved Successful where surgery failed Mature epithelial layer No residual defect |
| PATIENT 3 78-year-old Male Hypertension (controlled) Wound Duration: 5.1 years (Longest duration) (Oldest patient) |
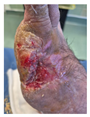 Chronic wound (5.1 years) LONGEST DURATION IN SERIES Extended conservative mgmt failed Failed flap coverage attempt Severely chronic, non-healing Infection: Negative Oldest patient (78 years) |
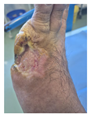 Post-bioprinting (2 weeks) Post-bioprinting (2 weeks)Successful material integration Initial healing in elderly patient Wound bed improvement noted No age-related complications Good tolerance despite age |
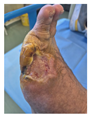 Mid-treatment (5 weeks) Mid-treatment (5 weeks)Encouraging healing progression Epithelialization advancing despite age Granulation tissue formation Positive geriatric response No adverse events |
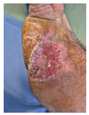 Advanced healing (8 weeks) Continued wound size reduction Strong epithelialization despite prolonged duration Healthy tissue regeneration Efficacy in elderly demonstrated |
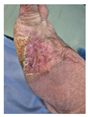 COMPLETE EPITHELIALIZATION 100% wound closure achieved Successful in 78-year-old patient Longest duration (5.1yr) treated successfully Demonstrates efficacy across age spectrum No complications |
| PATIENT 4 74-year-old Male Hypertension (controlled) + DIABETES MELLITUS Type 2 (HbA1c <7.0%, controlled) Wound Duration: 4.8 years (ONLY DIABETIC PATIENT) |
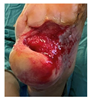 Chronic diabetic wound (4.8 years) Failed comprehensive diabetic care Failed advanced biologics treatment Chronic diabetic wound characteristics Infection: Negative Good glycemic control maintained |
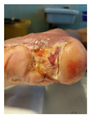 Post-bioprinting (2 weeks) Initial integration in diabetic wound bed Early response observed Diabetic healing challenges evident No infection/rejection Slower initial response vs non-diabetic |
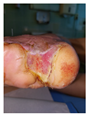 Mid-treatment (5 weeks) Partial healing response Wound size reduction observed Epithelialization slower than non-diabetic Diabetes impacting healing kinetics Bioprinted material facilitating partial closure |
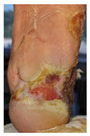 Advanced healing (8 weeks) Continued partial improvement Significant wound size reduction Incomplete epithelialization Residual defect remaining Diabetes limiting complete closure despite good control |
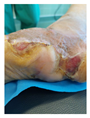 INCOMPLETE EPITHELIALIZATION Partial wound closure achieved Residual defect present REQUIRED SKIN GRAFTING Bioprinting reduced defect size significantly Facilitated subsequent surgical intervention Demonstrates diabetes impact despite control |
5.4.5. Safety Profile
5.4.6. Comparison with Conventional Approaches
5.4.7. Mechanisms of Action
- Scaffold function: Three-dimensional matrix for cell migration and proliferation
- Biochemical signaling: Delivery of growth factors/cytokines, immunomodulation
- Cellular therapy: Direct contribution to tissue regeneration and paracrine affect
- Moisture management: Maintaining optimal wound environment
- Protection: Physical barrier against external contaminants
5.4.8. Extrusion-Based Bioprinting Advantages
- High cell density: Can incorporate high concentrations of viable cells
- Viscous materials: Compatible with wide range of bioink formulations
- Scalability: Treats wounds of various sizes and geometries
- Cost-effectiveness: Relatively economical compared to other bioprinting modalities
- Clinical translation: Suitable for bedside or operating room use
- In situ application: Capability for direct printing onto wound beds
5.4.9. Clinical Implications and Patient Selection
- Ensuring adequate infection control
- Optimizing comorbid conditions (particularly diabetes)
- Managing patient expectations, especially in diabetic populations
- Appropriate wound bed preparation
- Consideration of wound duration and previous treatment failures
5.4.10. Limitations and Future Research Needs
5.4.11. Key Takeaways From Clinical Experience
- High success rate: 75% complete healing in refractory cases represents significant clinical benefit
- Safety profile: No serious adverse events across diverse patient population
- Diabetes challenge: Continued difficulty achieving complete healing in diabetic patients despite advanced therapy
- Age independence: Successful outcomes across age spectrum (62-78 years)
- Refractory wound efficacy: Success in patients who failed >4 years of conventional treatment
- Partial response value: Even incomplete healing reduced defect size, facilitating subsequent grafting
6. Challenges and Future Directions
6.1. Technical Challenges
6.2. Clinical Translation
6.3. Emerging Innovations
7. Conclusions
References
- Abuhamad, S.; et al. Application of 3D-Printed Bioinks in Chronic Wound Healing: A Scoping Review. Polymers 2024, 16(17), 2456. [Google Scholar] [CrossRef]
- Guptha, R.; et al. A comprehensive review of the application of 3D-bioprinting in chronic wound management. Expert Opinion on Drug Delivery 2024, 21(6), 789–810. [Google Scholar] [CrossRef]
- Naser, A. Z.; et al. Updates of Diabetic Foot Ulcer (DFU) Management Critical Review. International Journal of Pharmaceutical Sciences and Technology 2021, 29(1), 3634. [Google Scholar] [CrossRef]
- Tabriz, A. G.; et al. 3D printing technologies for skin wound healing applications. In Advanced 3D Printing Technologies; Elsevier, 2024; pp. 89–124. [Google Scholar] [CrossRef]
- Puri, A.; et al. Biomaterials-Based Additive Manufactured Products for Diabetic Wound Healing. In Advances in Biomaterials; Springer, 2025; pp. 245–278. [Google Scholar] [CrossRef]
- Luo, Z.; et al. 3D bioprinted mesenchymal stromal cells in skin wound repair. Frontiers in Surgery 2022, 9, 988843. [Google Scholar] [CrossRef]
- Dandoulakis, G. Next-generation 3D-printed bioengineered skin grafts: From experimental validation to clinical implementation. World Journal of Biology Pharmacy and Health Sciences 2025, 23(1), 0694. [Google Scholar] [CrossRef]
- Kammona, O.; Kiparissides, C. Recent Developments in 3D-(Bio)printed Hydrogels as Wound Dressings. Gels 2024, 10(2), 147. [Google Scholar] [CrossRef]
- Liao, W.; et al. 3D-bioprinted double-crosslinked angiogenic alginate/chondroitin sulfate patch for diabetic wound healing. International Journal of Biological Macromolecules 2023, 242, 123952. [Google Scholar] [CrossRef]
- Glover, K.; et al. 3D bioprinted scaffolds for diabetic wound-healing applications. Drug Delivery and Translational Research 2022, 12(5), 1133–1147. [Google Scholar] [CrossRef]
- Amo, C.; et al. Wound-Microenvironment Engineering through Advanced-Dressing Bioprinting. International Journal of Molecular Sciences 2022, 23(5), 2836. [Google Scholar] [CrossRef]
- Zhang, Y.; et al. 3-D bioprinted human-derived skin organoids accelerate full-thickness skin defects repair. Bioactive Materials 2024, 40, 234–248. [Google Scholar] [CrossRef]
- Xue, D.; et al. 3D printing of cell-delivery scaffolds for tissue regeneration. Regenerative Biomaterials 2023, 10, rbad032. [Google Scholar] [CrossRef]
- Fu, Z.; et al. Pro-angiogenic dual-crosslinked collagen bioinks for precise cell-laden DLP 3D printing and rapid vascularized diabetic wound repair. Advanced Functional Materials. in press. [CrossRef]
- Kammona, O.; Kiparissides, C. Recent Developments in 3D-(Bio)printed Hydrogels as Wound Dressings. Gels 2024, 10(2), 147. [Google Scholar] [CrossRef]
- Tabriz, A. G.; et al. 3D printing technologies for skin wound healing applications. In Advanced 3D Printing Technologies; Elsevier, 2024; pp. 89–124. [Google Scholar] [CrossRef]
- Xue, D.; et al. 3D printing of cell-delivery scaffolds for tissue regeneration. Regenerative Biomaterials 2023, 10, rbad032. [Google Scholar] [CrossRef]
- Kesavan, R.; et al. Management of diabetic foot ulcer with MA–ECM (minimally manipulated autologous extracellular matrix) using 3D bioprinting technology–an innovative approach. Journal of Tissue Engineering and Regenerative Medicine. in press. [CrossRef]
- Wang, S.; et al. In Situ 3D Bioprinting Living Photosynthetic Scaffolds for Autotrophic Wound Healing. Research 2022, 2022, 9794745. [Google Scholar] [CrossRef]
- Xue, D.; et al. 3D printing of cell-delivery scaffolds for tissue regeneration. Regenerative Biomaterials 2023, 10, rbad032. [Google Scholar] [CrossRef]
- Weaver, E.; et al. Combining microfluidics and coaxial 3D-bioprinting for the manufacturing of diabetic wound healing dressings. Biomaterials Advances 2023, 152, 213557. [Google Scholar] [CrossRef]
- Wang, X.; et al. Three-Dimensional Bioprinting of Biphasic Nanobioink for Enhanced Diabetic Wound Healing. ACS Nano 2025, 19(3), 2847–2861. [Google Scholar] [CrossRef]
- Weaver, E.; et al. Combining microfluidics and coaxial 3D-bioprinting for the manufacturing of diabetic wound healing dressings. Biomaterials Advances 2023, 152, 213557. [Google Scholar] [CrossRef]
- Kammona, O.; Kiparissides, C. Recent Developments in 3D-(Bio)printed Hydrogels as Wound Dressings. Gels 2024, 10(2), 147. [Google Scholar] [CrossRef]
- Abuhamad, S.; et al. Application of 3D-Printed Bioinks in Chronic Wound Healing: A Scoping Review. Polymers 2024, 16(17), 2456. [Google Scholar] [CrossRef]
- Liao, W.; et al. 3D-bioprinted double-crosslinked angiogenic alginate/chondroitin sulfate patch for diabetic wound healing. International Journal of Biological Macromolecules 2023, 242, 123952. [Google Scholar] [CrossRef]
- Wang, S.; et al. In Situ 3D Bioprinting Living Photosynthetic Scaffolds for Autotrophic Wound Healing. Research 2022, 2022, 9794745. [Google Scholar] [CrossRef]
- Shi, L.; et al. Three-dimensional bioprinted cell-adaptive hydrogel with anisotropic micropores for enhancing skin wound healing. International Journal of Biological Macromolecules 2024, 278, 136106. [Google Scholar] [CrossRef]
- Fu, Z.; et al. Pro-angiogenic dual-crosslinked collagen bioinks for precise cell-laden DLP 3D printing and rapid vascularized diabetic wound repair. Advanced Functional Materials. in press. [CrossRef]
- Schmitt, T.; et al. A Feasibility Study on 3D Bioprinting of Microfat Constructs Towards Wound Healing Applications. Frontiers in Bioengineering and Biotechnology 2021, 9, 707098. [Google Scholar] [CrossRef]
- 3D-bioprinted peptide coupling patches for wound healing. Materials Today Bio 2022, 13, 100188. [CrossRef]
- 3D-bioprinted peptide coupling patches for wound healing. Materials Today Bio 2022, 13, 100188. [CrossRef]
- Glover, K.; et al. 3D bioprinted scaffolds for diabetic wound-healing applications. Drug Delivery and Translational Research 2022, 12(5), 1133–1147. [Google Scholar] [CrossRef]
- Glover, K.; et al. 3D bioprinted scaffolds for diabetic wound-healing applications. Drug Delivery and Translational Research 2022, 12(5), 1133–1147. [Google Scholar] [CrossRef]
- Kammona, O.; Kiparissides, C. Recent Developments in 3D-(Bio)printed Hydrogels as Wound Dressings. Gels 2024, 10(2), 147. [Google Scholar] [CrossRef]
- Ma, K.; et al. 3D Printing of Strontium Silicate Microcylinder-Containing Multicellular Biomaterial Inks for Vascularized Skin Regeneration. Advanced Healthcare Materials 2021, 10(15), 2100523. [Google Scholar] [CrossRef]
- Metwally, S.; et al. Bioinspired 3D-printed scaffold embedding DDAB-nano ZnO/nanofibrous microspheres for regenerative diabetic wound healing. Biofabrication 2023, 15(4), 045021. [Google Scholar] [CrossRef]
- Amo, C.; et al. Wound-Microenvironment Engineering through Advanced-Dressing Bioprinting. International Journal of Molecular Sciences 2022, 23(5), 2836. [Google Scholar] [CrossRef]
- Jorgensen, A. M.; et al. Multicellular bioprinted skin facilitates human-like skin architecture in vivo. Science Translational Medicine 2023, 15(710), eadf7547. [Google Scholar] [CrossRef]
- Lihao, C.; et al. 3D bioprinting of Salvianolic acid B-sodium alginate-gelatin skin scaffolds promotes diabetic wound repair via antioxidant, anti-inflammatory mechanisms. In Biomaterials Science; in press.
- Liu, Y.; et al. Bioprinted Symbiotic Dressings: A Lichen-Inspired Approach to Diabetic Wound Healing with Enhanced Bioactivity and Structural Integrity. Small 2024, 20(45), 2407105. [Google Scholar] [CrossRef]
- Guptha, R.; et al. A comprehensive review of the application of 3D-bioprinting in chronic wound management. Expert Opinion on Drug Delivery 2024, 21(6), 789–810. [Google Scholar] [CrossRef]
- Abuhamad, S.; et al. Application of 3D-Printed Bioinks in Chronic Wound Healing: A Scoping Review. Polymers 2024, 16(17), 2456. [Google Scholar] [CrossRef]
- Naser, A. Z.; et al. Updates of Diabetic Foot Ulcer (DFU) Management Critical Review. International Journal of Pharmaceutical Sciences and Technology 2021, 29(1), 3634. [Google Scholar] [CrossRef]
- Guptha, R.; et al. A comprehensive review of the application of 3D-bioprinting in chronic wound management. Expert Opinion on Drug Delivery 2024, 21(6), 789–810. [Google Scholar] [CrossRef]
- Naser, A. Z.; et al. Updates of Diabetic Foot Ulcer (DFU) Management Critical Review. International Journal of Pharmaceutical Sciences and Technology 2021, 29(1), 3634. [Google Scholar] [CrossRef]
- Wang, S.; et al. In Situ 3D Bioprinting Living Photosynthetic Scaffolds for Autotrophic Wound Healing. Research 2022, 2022, 9794745. [Google Scholar] [CrossRef]
- Ma, K.; et al. 3D Printing of Strontium Silicate Microcylinder-Containing Multicellular Biomaterial Inks for Vascularized Skin Regeneration. Advanced Healthcare Materials 2021, 10(15), 2100523. [Google Scholar] [CrossRef]
- Fu, Z.; et al. Pro-angiogenic dual-crosslinked collagen bioinks for precise cell-laden DLP 3D printing and rapid vascularized diabetic wound repair. Advanced Functional Materials. in press. [CrossRef]
- Liu, Y.; et al. Bioprinted Symbiotic Dressings: A Lichen-Inspired Approach to Diabetic Wound Healing with Enhanced Bioactivity and Structural Integrity. Small 2024, 20(45), 2407105. [Google Scholar] [CrossRef]
- Amo, C.; et al. Wound-Microenvironment Engineering through Advanced-Dressing Bioprinting. International Journal of Molecular Sciences 2022, 23(5), 2836. [Google Scholar] [CrossRef]
- Liao, W.; et al. 3D-bioprinted double-crosslinked angiogenic alginate/chondroitin sulfate patch for diabetic wound healing. International Journal of Biological Macromolecules 2023, 242, 123952. [Google Scholar] [CrossRef]
- Zhang, X.; et al. 3D-bioprinted biomimetic multilayer implants comprising microfragmented adipose extracellular matrix and cells improve wound healing in a murine model of full-thickness wounds. In Tissue Engineering Part A; in press.
- Naser, A. Z.; et al. Updates of Diabetic Foot Ulcer (DFU) Management Critical Review. International Journal of Pharmaceutical Sciences and Technology 2021, 29(1), 3634. [Google Scholar] [CrossRef]
- Zhang, Y.; et al. 3-D bioprinted human-derived skin organoids accelerate full-thickness skin defects repair. Bioactive Materials 2024, 40, 234–248. [Google Scholar] [CrossRef]
- Ma, K.; et al. 3D Printing of Strontium Silicate Microcylinder-Containing Multicellular Biomaterial Inks for Vascularized Skin Regeneration. Advanced Healthcare Materials 2021, 10(15), 2100523. [Google Scholar] [CrossRef]
- Fu, Z.; et al. Pro-angiogenic dual-crosslinked collagen bioinks for precise cell-laden DLP 3D printing and rapid vascularized diabetic wound repair. Advanced Functional Materials. in press. [CrossRef]
- Kammona, O.; Kiparissides, C. Recent Developments in 3D-(Bio)printed Hydrogels as Wound Dressings. Gels 2024, 10(2), 147. [Google Scholar] [CrossRef]
- Abuhamad, S.; et al. Application of 3D-Printed Bioinks in Chronic Wound Healing: A Scoping Review. Polymers 2024, 16(17), 2456. [Google Scholar] [CrossRef]
- Tabriz, A. G.; et al. 3D printing technologies for skin wound healing applications. In Advanced 3D Printing Technologies; Elsevier, 2024; pp. 89–124. [Google Scholar] [CrossRef]
- Glover, K.; et al. 3D bioprinted scaffolds for diabetic wound-healing applications. Drug Delivery and Translational Research 2022, 12(5), 1133–1147. [Google Scholar] [CrossRef]
- Glover, K.; et al. 3D bioprinted scaffolds for diabetic wound-healing applications. Drug Delivery and Translational Research 2022, 12(5), 1133–1147. [Google Scholar] [CrossRef]
- Puri, A.; et al. Biomaterials-Based Additive Manufactured Products for Diabetic Wound Healing. In Advances in Biomaterials; Springer, 2025; pp. 245–278. [Google Scholar] [CrossRef]
- Dandoulakis, G. Next-generation 3D-printed bioengineered skin grafts: From experimental validation to clinical implementation. World Journal of Biology Pharmacy and Health Sciences 2025, 23(1), 0694. [Google Scholar] [CrossRef]
- Dandoulakis, G. Next-generation 3D-printed bioengineered skin grafts: From experimental validation to clinical implementation. World Journal of Biology Pharmacy and Health Sciences 2025, 23(1), 0694. [Google Scholar] [CrossRef]
- Guptha, R.; et al. A comprehensive review of the application of 3D-bioprinting in chronic wound management. Expert Opinion on Drug Delivery 2024, 21(6), 789–810. [Google Scholar] [CrossRef]
- Dandoulakis, G. Next-generation 3D-printed bioengineered skin grafts: From experimental validation to clinical implementation. World Journal of Biology Pharmacy and Health Sciences 2025, 23(1), 0694. [Google Scholar] [CrossRef]
- Abuhamad, S.; et al. Application of 3D-Printed Bioinks in Chronic Wound Healing: A Scoping Review. Polymers 2024, 16(17), 2456. [Google Scholar] [CrossRef]
- Puri, A.; et al. Biomaterials-Based Additive Manufactured Products for Diabetic Wound Healing. In Advances in Biomaterials; Springer, 2025; pp. 245–278. [Google Scholar] [CrossRef]
- Guptha, R.; et al. A comprehensive review of the application of 3D-bioprinting in chronic wound management. Expert Opinion on Drug Delivery 2024, 21(6), 789–810. [Google Scholar] [CrossRef]
- Abuhamad, S.; et al. Application of 3D-Printed Bioinks in Chronic Wound Healing: A Scoping Review. Polymers 2024, 16(17), 2456. [Google Scholar] [CrossRef]
- Guptha, R.; et al. A comprehensive review of the application of 3D-bioprinting in chronic wound management. Expert Opinion on Drug Delivery 2024, 21(6), 789–810. [Google Scholar] [CrossRef]
- Dandoulakis, G. Next-generation 3D-printed bioengineered skin grafts: From experimental validation to clinical implementation. World Journal of Biology Pharmacy and Health Sciences 2025, 23(1), 0694. [Google Scholar] [CrossRef]
- Tabriz, A. G.; et al. 3D printing technologies for skin wound healing applications. In Advanced 3D Printing Technologies; Elsevier, 2024; pp. 89–124. [Google Scholar] [CrossRef]
- Kammona, O.; Kiparissides, C. Recent Developments in 3D-(Bio)printed Hydrogels as Wound Dressings. Gels 2024, 10(2), 147. [Google Scholar] [CrossRef]
- Glover, K.; et al. 3D bioprinted scaffolds for diabetic wound-healing applications. Drug Delivery and Translational Research 2022, 12(5), 1133–1147. [Google Scholar] [CrossRef]
- Luo, Z.; et al. 3D bioprinted mesenchymal stromal cells in skin wound repair. Frontiers in Surgery 2022, 9, 988843. [Google Scholar] [CrossRef]
- Dandoulakis, G. Next-generation 3D-printed bioengineered skin grafts: From experimental validation to clinical implementation. World Journal of Biology Pharmacy and Health Sciences 2025, 23(1), 0694. [Google Scholar] [CrossRef]
- Weaver, E.; et al. Combining microfluidics and coaxial 3D-bioprinting for the manufacturing of diabetic wound healing dressings. Biomaterials Advances 2023, 152, 213557. [Google Scholar] [CrossRef]
- Wang, S.; et al. In Situ 3D Bioprinting Living Photosynthetic Scaffolds for Autotrophic Wound Healing. Research 2022, 2022, 9794745. [Google Scholar] [CrossRef]
- Xue, D.; et al. 3D printing of cell-delivery scaffolds for tissue regeneration. Regenerative Biomaterials 2023, 10, rbad032. [Google Scholar] [CrossRef]
- Lim, K. S.; et al. Bio-resin for high resolution lithography-based biofabrication of complex cell-laden constructs. Biofabrication 2020, 12(2), 025014. [Google Scholar] [CrossRef]
- Chen, Z.; et al. Visible light-cured high-strength hydrogels for stereolithography-based 3D bioprinting. Virtual and Physical Prototyping 2022, 17(1), 1–14. [Google Scholar]
- Grigoryan, B.; et al. Multivascular networks and functional intravascular topologies within biocompatible hydrogels. Science 2021, 364(6439), 458–464. [Google Scholar] [CrossRef]
- Skoog, S. A.; et al. Stereolithography-based bioprinting of multi-material tissue constructs. Trends in Biotechnology 2023, 41(5), 678–692. [Google Scholar]
- Dou, C.; et al. Laser-assisted bioprinting of cells and biomaterials for tissue engineering applications. Journal of Materials Science & Technology 2021, 88, 145–159. [Google Scholar]
- Riester, O. A.; et al. Advances in Laser-Assisted Bioprinting for Complex Tissue Fabrication. Annual Review of Biomedical Engineering 2022, 24, 231–252. [Google Scholar]
- Kérourédan, O.; et al. Laser-assisted bioprinting for bone repair in a mouse calvarial defect model. Scientific Reports 2020, 10(1), 1–11. [Google Scholar]
- Zhang, Y.; et al. High-precision laser-assisted bioprinting: Mechanisms, applications, and future perspectives. Bioprinting 2023, 31, e00268. [Google Scholar]
- https://facellitate.com/what-are-the-types-of-3d-bioprinting-technologies/.
- Alparslan, C.; Bayraktar, Ş. Advances in Digital Light Processing (DLP) Bioprinting: A Review of Biomaterials and Its Applications, Innovations, Challenges, and Future Perspectives. Polymers 2025, 17, 1287. [Google Scholar] [CrossRef]
- Holl, J.; Kowalewski, C.; Zimek, Z.; Fiedor, P.; Kaminski, A.; Oldak, T.; Moniuszko, M.; Eljaszewicz, A. Chronic DiabeticWounds and Their Treatment with Skin Substitutes. Cells 2021. [Google Scholar] [CrossRef]
- Inkjet-based bioprinting for tissue engineering. Organoid Published online. 2023, 3, e12. [CrossRef]
- Van Damme, L.; Blondeel, P.; Van Vlierberghe, S. Reconstructing Curves: A Bottom-Up Approach toward Adipose Tissue Regeneration with Recombinant Biomaterials. Macromol. Biosci. 2024, 24, 2300466. [Google Scholar] [CrossRef]
- Bajuri, M. Y.; Kim, J.; Yu, Y.; Shahul Hameed, M. S. New Paradigm in Diabetic Foot Ulcer Grafting Techniques Using 3D-Bioprinted Autologous Minimally Manipulated Homologous Adipose Tissue (3D-AMHAT) with Fibrin Gel Acting as a Biodegradable Scaffold. Gels 2023, 9, 66. [Google Scholar] [CrossRef]
| Study | Bioprinting Technique | Key Biomaterials | Wound Model | Primary Outcomes |
| Wang et al. (2022) [19] | In situ microfluidic-assisted | Alginate, GelMA, C. pyrenoidosa | Diabetic mouse | 92% wound closure by day 15; 3-fold increase in microvessel density |
| Ma et al. (2021) [36] | Cell-writing extrusion | Strontium silicate microcylinders | Acute/chronic wounds (3 models) | Accelerated healing; enhanced vascularization and graft integration |
| Liao et al. (2023) [9] | Extrusion-based | Alginate, chondroitin sulfate | Diabetic wound | Enhanced angiogenesis and ECM remodeling |
| Glover et al. (2022) [10] | Extrusion-based | PCL, levofloxacin | In vitro (antibacterial) | 4-week sustained drug release; antibacterial efficacy |
| Zhang et al. (2024) [12] | Extrusion with dual-photo crosslinking | Human cells (keratinocytes, fibroblasts, endothelial cells) | Full-thickness defect (immunodeficient mice) | Accelerated healing via in situ regeneration and vascularization |
| Fu et al. [14] | DLP bioprinting | Pro-angiogenic dual-crosslinked collagen | Diabetic wound | Enhanced vascular regeneration in angiogenesis-impaired wounds |
| Liu et al. (2024) [41] | Extrusion-compatible | Hydrogel with microalgae and probiotics | Diabetic wound | 90% skin structure restoration within 12 days; reduced inflammation |
| Shi et al. (2024) [28] | Extrusion with thermo/ion/photo-crosslinking | GelMA, sodium alginate, PEO | Full-thickness wound | Accelerated closure via inflammation mitigation and angiogenesis |
| Amo et al. (2022) [11] | Extrusion-based | Decellularized adipose matrix, plasma, fibroblasts | In vitro | 70-fold increase in MCP-1; sustained cytokine release |
| Schmitt et al. (2021) [30] | Extrusion with FRESH technique | Methacrylated collagen, microfat | In vitro | Maintained cell viability for 10 days; temporal cytokine expression |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




