Submitted:
14 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Case Report
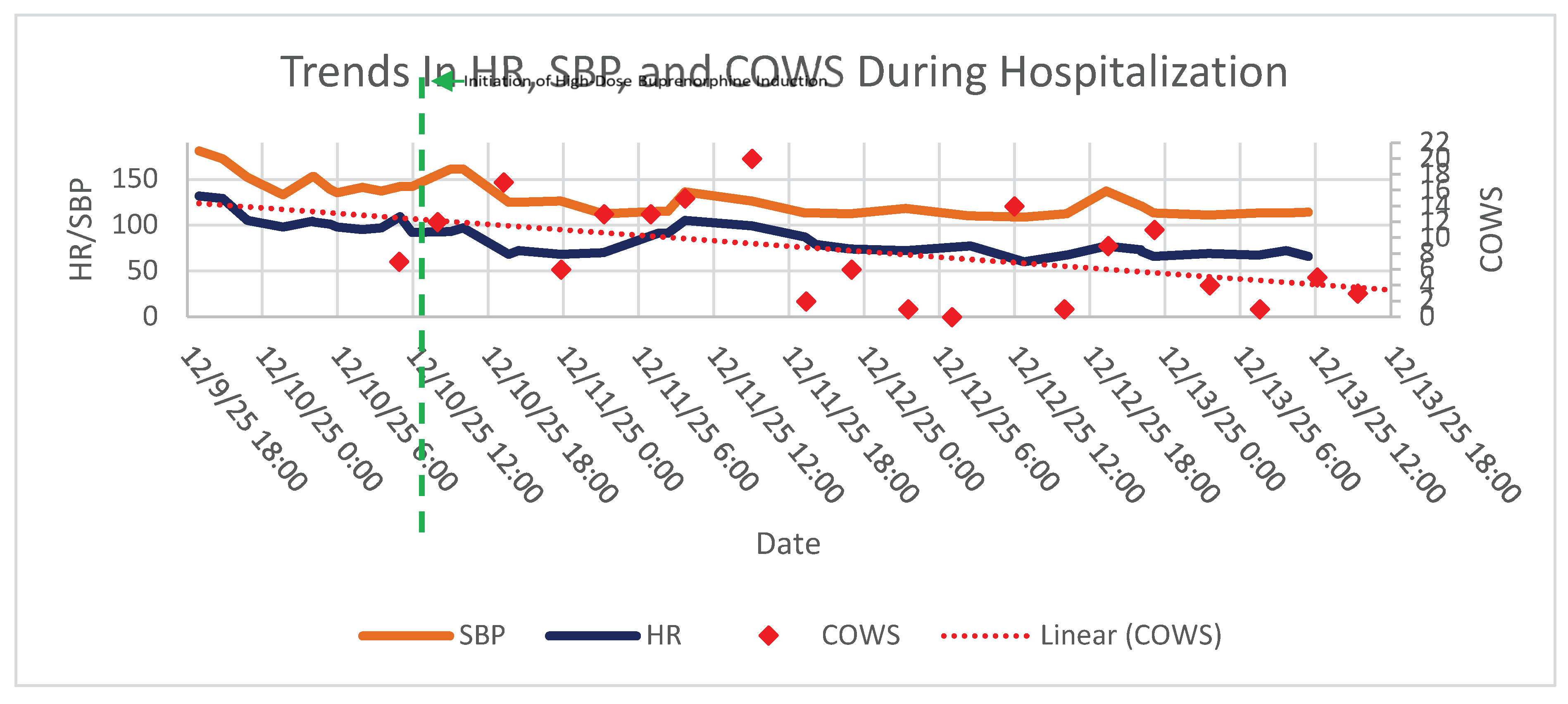
3. Results
4. Discussion
4.1. Kratom Misuse and Withdrawal
4.2. Macro-Induction Buprenorphine: Safety Profile and Clinical Indications
4.3. Synthetic Opioid and Adulterant Withdrawal and Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 7-HMC | 7-hydroxymitragynine |
| MP | mitragynine pseudoindoxyl |
| AWS | Alcohol Withdrawal Scale |
| HR | Heart Rate |
| SBP | Systolic Blood Pressure |
| COWS | Clinical Opiate Withdrawal Scale |
| LAIB | Long Acting Injectable Buprenorphine |
References
- Chakraborty, S.; Uprety, R.; Slocum, S. T.; Irie, T.; Le Rouzic, V.; Li, X.; Wilson, L. L.; Scouller, B.; Alder, A. F.; Kruegel, A. C.; Ansonoff, M.; Váradi, A.; Eans, S. O.; Hunkele, A.; Allaoa, A.; Kalra, S.; Xu, J.; Pan, Y.-X.; Pintar, J.; Kivell, B. M.; Pasternak, G. W.; Cameron, M. D.; McLaughlin, J. P.; Sames, D.; Majumdar, S. Oxidative metabolism as a modulator of kratom’s biological actions. Journal of Medicinal Chemistry 2021, 64(22), 16553–16572. [Google Scholar] [CrossRef]
- Wilson, L. L.; Chakraborty, S.; Eans, S. O.; et al. Kratom alkaloids, natural and semi-synthetic, show less physical dependence and ameliorate opioid withdrawal. Cellular and Molecular Neurobiology 2021, 41, 1131–1143. [Google Scholar] [CrossRef] [PubMed]
- Chang-Chien, G. C.; Odonkor, C. A.; Amorapanth, P. Is kratom the new “legal high” on the block? The case of an emerging opioid receptor agonist with substance abuse potential. Pain Physician 2017, 20(1), E195–E198. [Google Scholar] [CrossRef] [PubMed]
- Buresh, M. Treatment of kratom dependence with buprenorphine-naloxone maintenance. Journal of Addiction Medicine 2018, 12(6), 481–483. [Google Scholar] [CrossRef] [PubMed]
- Ellis, C. R.; Racz, R.; Kruhlak, N. L.; Kim, M. T.; Zakharov, A. V.; Southall, N.; Hawkins, E. G.; Burkhart, K.; Strauss, D. G.; Stavitskaya, L. Evaluating kratom alkaloids using PHASE. PLoS ONE 2020, 15(3), e0229646. [Google Scholar] [CrossRef]
- Weiss, S. T.; Douglas, H. E. Treatment of kratom withdrawal and dependence with buprenorphine/naloxone: A case series and systematic literature review. Journal of Addiction Medicine 2021, 15(2), 167–172. [Google Scholar] [CrossRef]
- Sivakumar, D.; Pascual, J.; Kennedy, M.; Clark, K. T.; Mason, J. M.; Walfield, A. L.; Maldonado, G. The successful use of buprenorphine to manage kratom withdrawal secondary to self-treatment of opioid withdrawal. Clinical Toxicology 2025, 63(11), 1018–1019. [Google Scholar] [CrossRef]
- Khazaeli, A.; Jerry, J. M.; Vazirian, M. Treatment of kratom withdrawal and addiction with buprenorphine. Journal of Addiction Medicine 2018, 12(6), 493–495. [Google Scholar] [CrossRef]
- Lei, J.; Butz, A.; Valentino, N. Management of kratom dependence with buprenorphine/naloxone in a veteran population. Substance Abuse 2021, 42(4), 497–502. [Google Scholar] [CrossRef]
- Hill, K.; Boyer, E. W.; Grundmann, O.; Smith, K. E. De facto opioids: Characterization of novel 7-hydroxymitragynine and mitragynine pseudoindoxyl product marketing. Drug and alcohol dependence 2025, 272, 112701. [Google Scholar] [CrossRef]
- Henningfield, J. E.; Grundmann, O.; Huestis, M. A.; Smith, K. E. Kratom safety and toxicology in the public health context: Research needs to better inform regulation. Frontiers in Pharmacology 2024, 15, 1403140. [Google Scholar] [CrossRef] [PubMed]
- Wightman, Rachel S.; Hu, David. A case of 7-OH mitragynine use requiring inpatient medically managed withdrawal. Journal of Addiction Medicine 2024, 10–1097. [Google Scholar] [CrossRef] [PubMed]
- Weiss, ST; Douglas, HE. Treatment of kratom withdrawal and dependence with buprenorphine/naloxone: a case series and systematic literature review. Journal of addiction medicine 2021, 15(2), 167–72. [Google Scholar] [CrossRef] [PubMed]
- Snyder, H.; Chau, B.; Kalmin, M.M.; et al. High-Dose Buprenorphine Initiation in the Emergency Department Among Patients Using Fentanyl and Other Opioids. JAMA Netw. Open 2023, 6, e231572. [Google Scholar] [CrossRef]
- Herring, A.A.; Vosooghi, A.A.; Luftig, J.; Anderson, E.S.; Zhao, X.; Dziura, J.; Hawk, K.F.; McCormack, R.P.; Saxon, A.; D'Onofrio, G. High-Dose Buprenorphine Induction in the Emergency Department for Treatment of Opioid Use Disorder. JAMA Netw. Open 2021, 4, e2117128. [Google Scholar] [CrossRef]
- Wu, S.A.; Archer, G.N.; Schnipke, B.D. High-dose buprenorphine inductions in hospital settings. Am. J. Med. Open 2025, 14, 100118. [Google Scholar] [CrossRef]
- Berry, M.; Kiefer, M.K.; Hinely, K.A.; Bowden, H.; Jordan, A.; Vilensky, M.; Rood, K.M. High-Dose Buprenorphine Initiation in the Management of Opioid Use Disorder in Pregnancy. Obstet. Gynecol. 2024, 143, 815–818. [Google Scholar] [CrossRef]
- Tsui, J.I.; Bhatraju, E.P.; Hallgren, K.A.; Rhew, I.C.; Samet, J.H. High-Dose Buprenorphine Treatment in the Setting of Fentanyl. JAMA Netw. Open 2025, 8, e2528119. [Google Scholar] [CrossRef]
- Danilewitz, M.; McLean, M. High-dose buprenorphine for treatment of high potency opioid use disorder. Drug Alcohol Rev. 2020, 39, 135–137. [Google Scholar] [CrossRef]
- Kahan, M.; Marion-Bellemare, L.; Samson, J.; et al. Macrodosing" sublingual buprenorphine and extended-release buprenorphine in a hospital setting: 2 case reports. J. Addict. Med. 2023, 17, 485–487. [Google Scholar] [CrossRef]
- Bormann, N.L.; Gout, A.; Kijewski, V.; Lynch, A. Case Report: Buprenorphine-precipitated fentanyl withdrawal treated with high-dose buprenorphine [version 2; peer review: 2 approved]. F1000Research 2023, 11, 487. [Google Scholar] [CrossRef]
- Quattlebaum, T.H.N.; Kiyokawa, M.; Murata, K.A. A case of buprenorphine-precipitated withdrawal managed with high-dose buprenorphine. Fam. Pract. 2022, 39, 292–294. [Google Scholar] [CrossRef]
- Ahmadi, J.; Jahromi, M.S.; Ghahremani, D.; et al. Single high-dose buprenorphine for opioid craving during withdrawal. Trials 2018, 19, 675. [Google Scholar] [CrossRef]
- Skolnick, P; Paavola, J; Heidbreder, C. Synthetic opioids have disrupted conventional wisdom for treating opioid overdose. Drug Alcohol Depend Rep. 2024, 12, 100268. [Google Scholar] [CrossRef]
- London, KS; Li, Y; Kahoud, JL; et al. Tranq Dope: Characterization of an ED cohort treated with a novel opioid withdrawal protocol in the era of fentanyl/xylazine. Am J Emerg Med. 2024, 85, 130–139. [Google Scholar] [CrossRef]
- D’Orazio, J; Nelson, L; Perrone, J; Wightman, R; Haroz, R. Xylazine adulteration of the heroin–fentanyl drug supply: a narrative review. Annals of Internal Medicine 2023, 176(10), 1370–6. [Google Scholar] [CrossRef] [PubMed]
- Durney, P; Paquin, E; Fitzpatrick, G; et al. Profound opioid and medetomidine withdrawal: a case series and narrative review of available literature. Psychoactives 2025, 4(4), 37. [Google Scholar] [CrossRef]
- London, KS; Durney, P; Warrick-Stone, T; Alexander, K; Kahoud, JL. Decreased effectiveness of a novel opioid withdrawal protocol following the emergence of medetomidine as a fentanyl adulterant. BioMed. 2025, 5(2), 13. [Google Scholar] [CrossRef]
- London, KS; Huo, S; Murphy, L; et al. Severe fentanyl withdrawal associated with medetomidine adulteration: a multicenter study from Philadelphia, PA. J Addict Med. 2025. [Google Scholar] [CrossRef]
- Durney, P; Kahoud, JL; Warrick-Stone, T; et al. Biochemical identification and clinical description of medetomidine exposure in people who use fentanyl in Philadelphia, PA. Int J Mol Sci. 2025, 26(14), 6715. [Google Scholar] [CrossRef]
| Hospitalization Day | Maximum COWS Score | Buprenorphine Dosage Given (mg/day in dose) | AWS Score Range (24h) | Benzodiazepines given (for alcohol withdrawal) |
|---|---|---|---|---|
| 1 | 26 | 32 mg buprenorphine (2x 16 mg) | 6-9 | 15 mg diazepam (including 5 mg for buprenorphine macro-induction) + 2 mg lorazepam |
| 2 | 20 | 36 mg buprenorphine (24mg [8mg TID] + 8mg and 2x2 mg PRN) |
3-11 | 1 mg lorazepam |
| 3 | 14 | 32 mg buprenorphine (24 mg + 4x2 mg PRN) | 0-9 | 30 mg diazepam |
| 4 | 5 | 24 mg buprenorphine (8 mg TID) | 2-3 | 20 mg diazepam (5 mg QID) |
| 5 | 5 | 24 mg buprenorphine (8 mg TID) | 2-3 | 10 mg diazepam (5 mg BID) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




