Submitted:
13 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
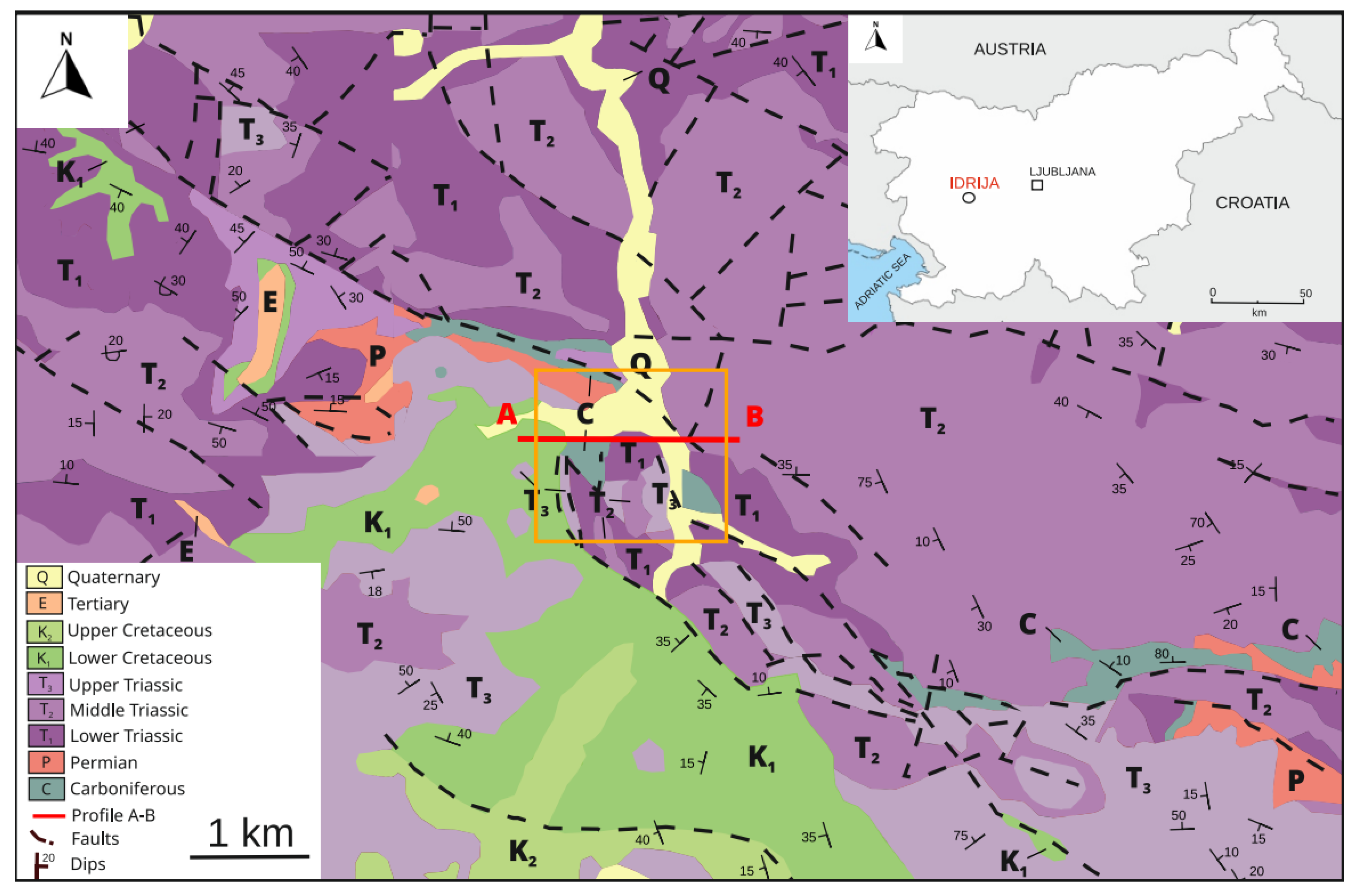
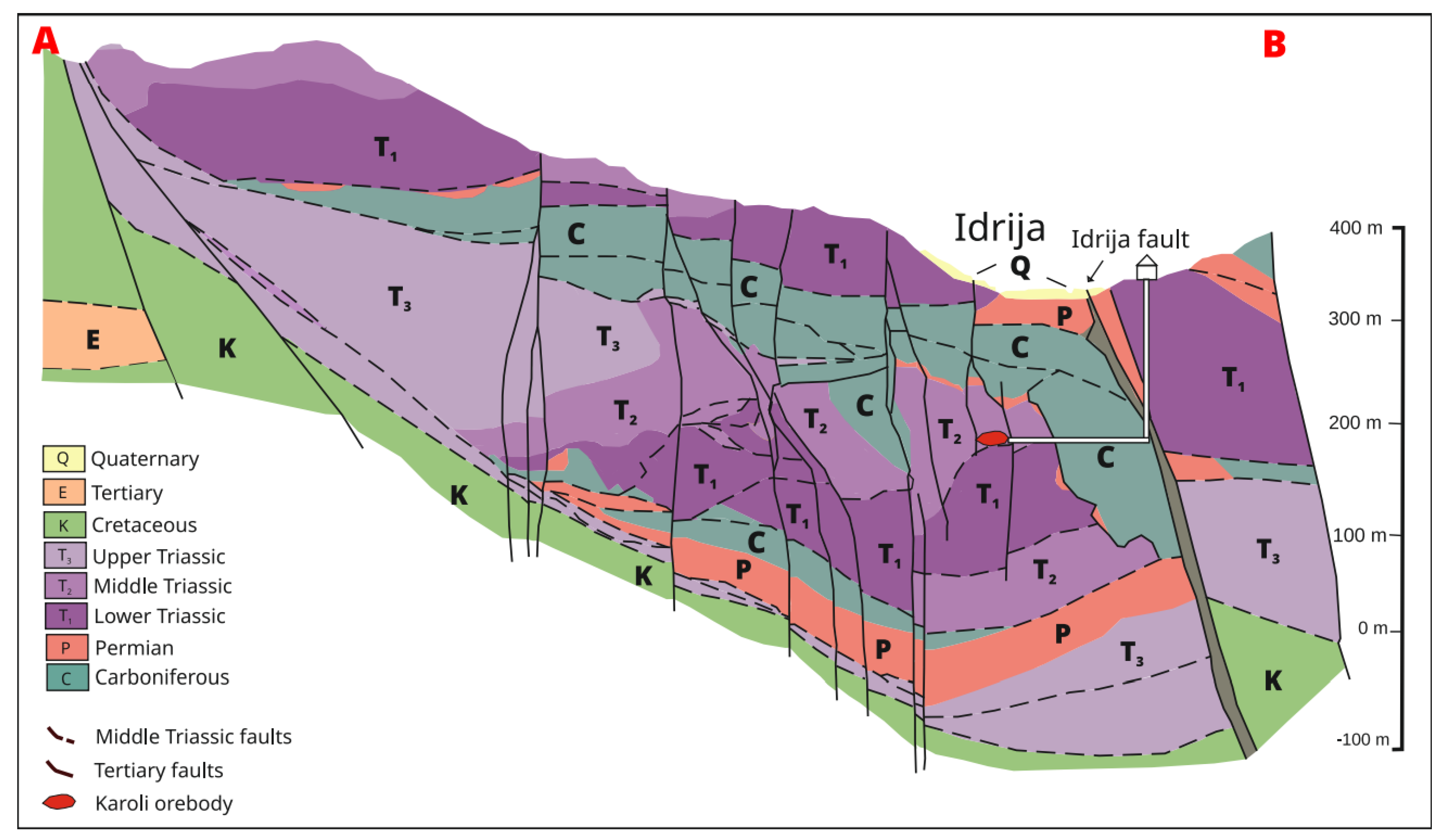
2. Geological Setting
3. Ore Deposit Geology
4. Materials and Methods
4.1. Materials
4.2. Methods
5. Results
5.1. Texture of Pyrite
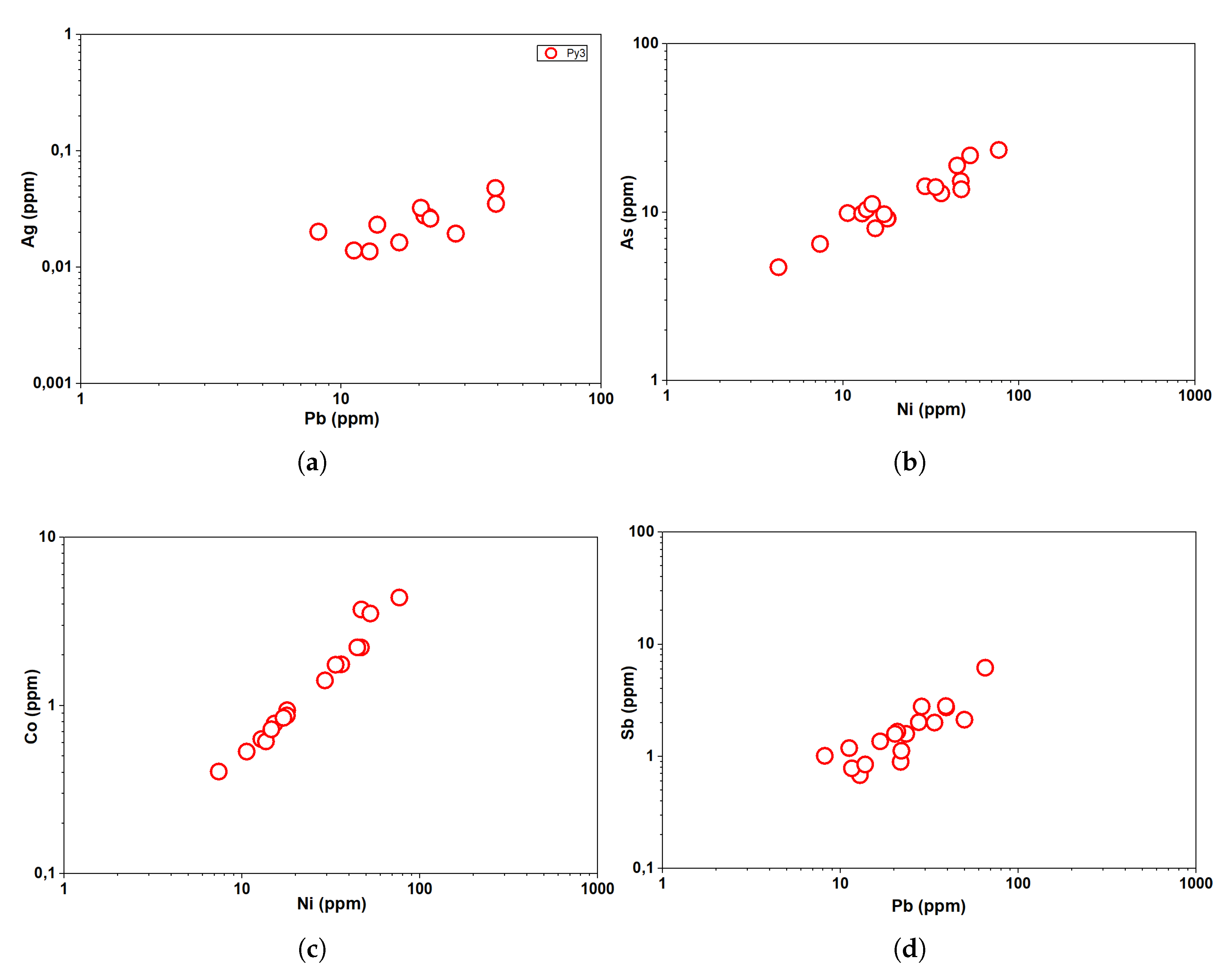
5.2. LA-ICP-MS Results
6. Discussion
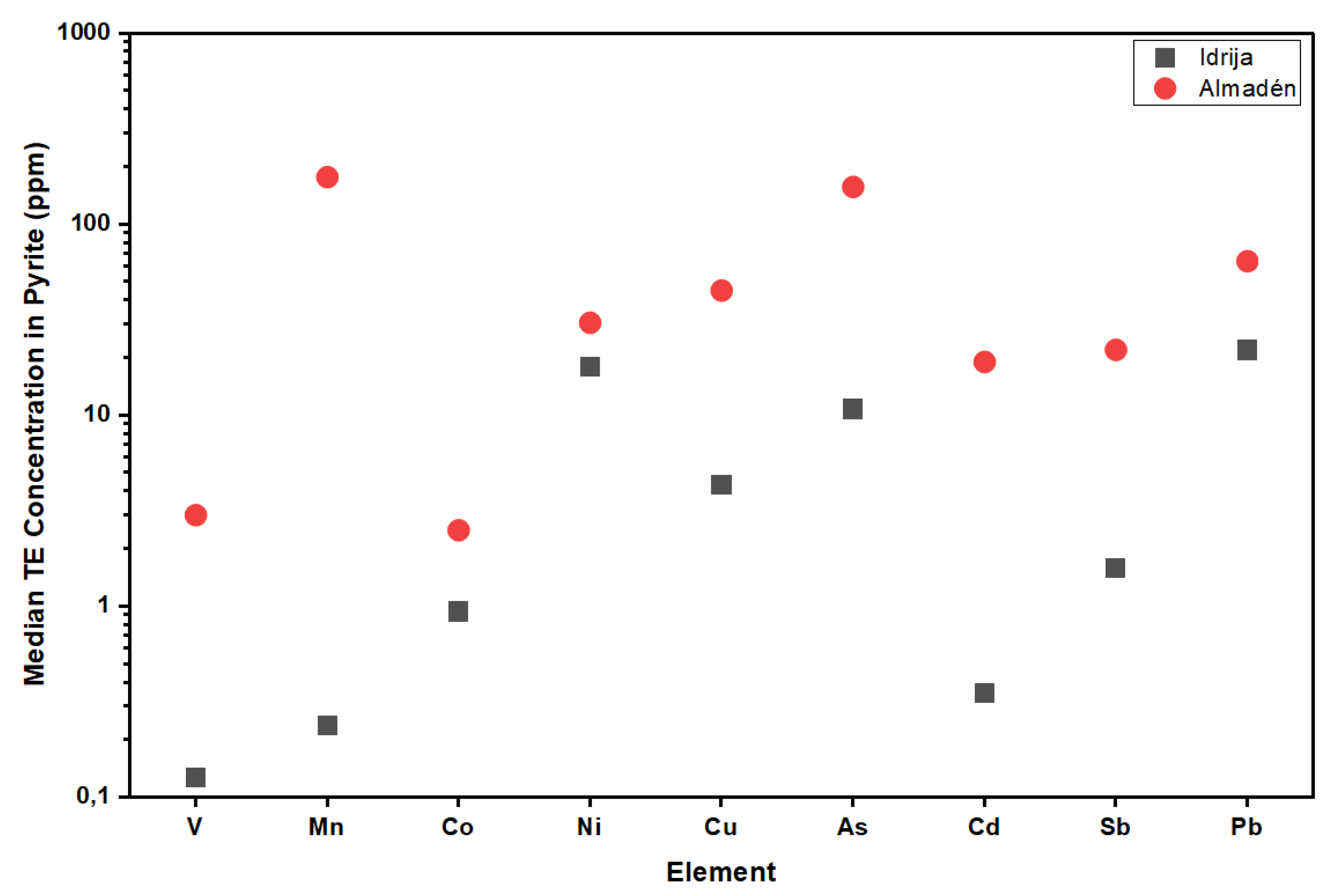
6.1. Trace Element Composition
6.2. Sulfur and Mercury Isotopes
6.3. Implications for Ore Genesis
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Čar, J.; Placer, L. The Middle Triassic structure of the Idrija region. Geologija 1977, 20, 141–166. [Google Scholar]
- Higueras, P.; Oyarzun, R.; Lillo, J.; Morata, D. Intraplate mafic magmatism, degasification, and deposition of mercury: The giant Almadén mercury deposit (Spain) revisited. Ore Geology Reviews 2013, 51, 93–102. [Google Scholar] [CrossRef]
- Rimondi, V.; Chiarantini, L.; Lattanzi, P.; Benvenuti, M.; Beutel, M.; Colica, A.; Costagliola, P.; Di Benedetto, F.; Gabbani, G.; Gray, J.E.; et al. Metallogeny, exploitation and environmental impact of the Mt. Amiata mercury ore district (Southern Tuscany, Italy). Italian Journal of Geosciences 2015, 134, 323–336. [Google Scholar] [CrossRef]
- Mlakar, I.; Drovenik, M. Strukturne in genetske posebnosti idrijskega rudišča. Geologija 1971, 14, 67–126. [Google Scholar]
- Drovenik, M.; Pleničar, M.; Drovenik, F. The origin of Slovenian ore deposits. Geologija 1980, 23, 1–157. [Google Scholar]
- Čadež, F. Gypsum and anhydrite occurrences in Idria region. Geologija 1977, 20, 289–301. [Google Scholar]
- Čadež, F. Najmlajše diskordantne sedimentne kamenine na karbonskih plasteh v Idriji. Geologija 1980, 23, 163–172. [Google Scholar]
- Mlakar, I. Relations between the lower and the upper structure of the Idrija ore deposit. Geologija 1967, 10, 87–126. [Google Scholar]
- Placer, L.; Čar, J. Rekonstrukcija srednjetriadnih razmer na idrijskem prostoru. Geologija 1975, 18, 197–209. [Google Scholar]
- Placer, L. Structural history of the Idrija mercury deposit. Geologija 1982, 25, 7–94. [Google Scholar]
- Čar, J. Angular tectonic-erosional unconformity in the deposit’s part of the Idrija Middle Triassic tectonic structure. Geologija 1988, 31, 267–284. [Google Scholar]
- Lavrič, J.V.; Spangenberg, J.E. Stable isotope (C, O, S) systematics of the mercury mineralization at Idrija, Slovenia: constraints on fluid source and alteration processes. Mineralium Deposita 2003, 38, 886–899. [Google Scholar] [CrossRef]
- Božič, D.; Živković, I.; Dizdarević, T.; Peljhan, M.; Štrok, M.; Horvat, M. Insights into the heterogeneity of the mercury isotopic fingerprint of the Idrija Mine (Slovenia). Minerals 2023, 13, 1227. [Google Scholar] [CrossRef]
- Agangi, A.; Hofmann, A.; Wohlgemuth-Ueberwasser, C.C. Pyrite zoning as a record of mineralization in the Ventersdorp Contact Reef, Witwatersrand Basin, South Africa. Economic Geology 2013, 108, 1243–1272. [Google Scholar] [CrossRef]
- Zhang, J.; Deng, J.; Chen, H.y.; Yang, L.q.; Cooke, D.; Danyushevsky, L.; Gong, Q.j. LA-ICP-MS trace element analysis of pyrite from the Chang’an gold deposit, Sanjiang region, China: Implication for ore-forming process. Gondwana Research 2014, 26, 557–575. [Google Scholar] [CrossRef]
- Thomas, H.V.; Large, R.R.; Bull, S.W.; Maslennikov, V.; Berry, R.F.; Fraser, R.; Froud, S.; Moye, R. Pyrite and pyrrhotite textures and composition in sediments, laminated quartz veins, and reefs at Bendigo gold mine, Australia: Insights for ore genesis. Economic Geology 2011, 106, 1–31. [Google Scholar] [CrossRef]
- Large, R.R.; Danyushevsky, L.; Hollit, C.; Maslennikov, V.; Meffre, S.; Gilbert, S.; Bull, S.; Scott, R.; Emsbo, P.; Thomas, H.; et al. Gold and trace element zonation in pyrite using a laser imaging technique: Implications for the timing of gold in orogenic and Carlin-style sediment-hosted deposits. Economic Geology 2009, 104, 635–668. [Google Scholar] [CrossRef]
- Large, R.R.; Halpin, J.A.; Danyushevsky, L.V.; Maslennikov, V.V.; Bull, S.W.; Long, J.A.; Gregory, D.D.; Lounejeva, E.; Lyons, T.W.; Sack, P.J.; et al. Trace element content of sedimentary pyrite as a new proxy for deep-time ocean–atmosphere evolution. Earth and Planetary Science Letters 2014, 389, 209–220. [Google Scholar] [CrossRef]
- Li, W.; Cook, N.J.; Xie, G.Q.; Mao, J.W.; Ciobanu, C.L.; Li, J.W.; Zhang, Z.Y. Textures and trace element signatures of pyrite and arsenopyrite from the Gutaishan Au–Sb deposit, South China. Mineralium Deposita 2019, 54, 591–610. [Google Scholar] [CrossRef]
- Deditius, A.P.; Utsunomiya, S.; Reich, M.; Kesler, S.E.; Ewing, R.C.; Hough, R.; Walshe, J. Trace metal nanoparticles in pyrite. Ore Geology Reviews 2011, 42, 32–46. [Google Scholar] [CrossRef]
- Gao, F.; Du, Y.; Pang, Z.; Du, Y.; Xin, F.; Xie, J. LA-ICP-MS trace-element analysis of pyrite from the Huanxiangwa gold deposit, Xiong’ershan district, China: Implications for ore genesis. Minerals 2019, 9, 157. [Google Scholar] [CrossRef]
- Gregory, D.D.; Large, R.R.; Halpin, J.A.; Baturina, E.L.; Lyons, T.W.; Wu, S.; Danyushevsky, L.; Sack, P.J.; Chappaz, A.; Maslennikov, V.V.; et al. Trace element content of sedimentary pyrite in black shales. Economic Geology 2015, 110, 1389–1410. [Google Scholar] [CrossRef]
- Seal, R.R. Sulfur isotope geochemistry of sulfide minerals. Reviews in mineralogy and geochemistry 2006, 61, 633–677. [Google Scholar] [CrossRef]
- Hoefs, J. Stable isotope geochemistry; Springer: Berlin, Germany, 2009. [Google Scholar]
- Bergquist, B.A.; Blum, J.D. Mass-dependent and-independent fractionation of Hg isotopes by photoreduction in aquatic systems. Science 2007, 318, 417–420. [Google Scholar] [CrossRef] [PubMed]
- Blum, J.D.; Sherman, L.S.; Johnson, M.W. Mercury isotopes in earth and environmental sciences. Annual Review of Earth and Planetary Sciences 2014, 42, 249–269. [Google Scholar] [CrossRef]
- Foucher, D.; Ogrinc; Hintelmann, H. Tracing mercury contamination from the Idrija mining region (Slovenia) to the Gulf of Trieste using Hg isotope ratio measurements. Environmental Science & Technology 2009, 43, 33–39. [Google Scholar]
- Grasby, S.E.; Them, T.R., II; Chen, Z.; Yin, R.; Ardakani, O.H. Mercury as a proxy for volcanic emissions in the geologic record. Earth-Science Reviews 2019, 196, 102880. [Google Scholar] [CrossRef]
- Moynier, F.; Jackson, M.G.; Zhang, K.; Cai, H.; Halldórsson, S.A.; Pik, R.; Day, J.M.; Chen, J. The mercury isotopic composition of Earth’s mantle and the use of mass independently fractionated Hg to test for recycled crust. Geophysical Research Letters 2021, 48, e2021GL094301. [Google Scholar] [CrossRef]
- Buser, S. Stratigrafske vrzeli v paleozojskih in mezozojskih plasteh v Sloveniji. In Proceedings of the Regional geology and palaeontology symposium, 1980; pp. 335–345. [Google Scholar]
- Buser, S. Development of the Dinaric and the Julian carbonate platforms and of the intermediate Slovenian Basin (NW Yugoslavia). Mem. Soc. Geol. Ital 1989, 40, 313–320. [Google Scholar]
- Placer, L. Principles of the tectonic subdivision of Slovenia. Geologija 2008, 51, 205–217. [Google Scholar] [CrossRef]
- Gosar, A. Monitoring of micro-deformations along Idrija and Raša faults in W Slovenia. Geologija 2007, 50, 45–54. [Google Scholar] [CrossRef]
- Placer, L. Rekonstrukcija krovne zgradbe idrijsko žirovskega ozemlja. Geologija 1973, 16, 317–334. [Google Scholar]
- Berce, B. Geologija živosrebrnega rudišča Idrija. Geologija 1958, 4, 5–62. [Google Scholar]
- Čar, J. Ladinian skonca beds of the Idrija Ore Deposit (W Slovenia). Geologija 2013, 56, 151–174. [Google Scholar] [CrossRef]
- Čar, J. Razvoj srednjetriasnih sedimentov v idrijskem tektonskem jarku. Ph.d. thesis, University of Ljubljana, Univerza Edvarda Kardelja v Ljubljani, Fakulteta za naravoslovje in tehnologijo, Oddelek za geologijo, 1985. [Google Scholar]
- Rečnik, A.; Čar, J.; Mirtič, B. Minerals of the mercury ore deposit Idria; Springer: Berlin, Germany, 2013. [Google Scholar]
- Castroviejo, R. Fundamentos de petrografía: Rocas ígneas, sedimentarias y metamórficas; Red ALEMYMA-Programa ALFA (UE/DGI), Escuela Técnica Superior de Ingenieros de Minas, Universidad Politécnica de Madrid: Madrid, Spain, 1998; 84, Unpublished teaching material from the Dpto. Ingeniería Geológica. [Google Scholar]
- Šoster, A.; Erlandsson, V.B.; Ajuaba, S.; Šmuc, N.R. Selective trace element uptake in iron sulfides during burial metamorphism of coal: Insights from the coal-bearing Socka Beds, Northeastern Slovenia. International journal of coal geology 2025, 104805. [Google Scholar] [CrossRef]
- Morse, J.; Luther Iii, G. Chemical influences on trace metal-sulfide interactions in anoxic sediments. Geochimica et Cosmochimica Acta 1999, 63, 3373–3378. [Google Scholar] [CrossRef]
- Koglin, N.; Frimmel, H.E.; Lawrie Minter, W.; Brätz, H. Trace-element characteristics of different pyrite types in Mesoarchaean to Palaeoproterozoic placer deposits. Mineralium Deposita 2010, 45, 259–280. [Google Scholar] [CrossRef]
- Reich, M.; Deditius, A.; Chryssoulis, S.; Li, J.W.; Ma, C.Q.; Parada, M.A.; Barra, F.; Mittermayr, F. Pyrite as a record of hydrothermal fluid evolution in a porphyry copper system: A SIMS/EMPA trace element study. Geochimica et Cosmochimica Acta 2013, 104, 42–62. [Google Scholar] [CrossRef]
- Large, R.R.; Maslennikov, V.V. Invisible gold paragenesis and geochemistry in pyrite from orogenic and sediment-hosted gold deposits. Minerals 2020, 10, 339. [Google Scholar] [CrossRef]
- Large, R.R.; Maslennikov, V.V.; Robert, F.; Danyushevsky, L.V.; Chang, Z. Multistage sedimentary and metamorphic origin of pyrite and gold in the giant Sukhoi Log deposit, Lena gold province, Russia. Economic Geology 2007, 102, 1233–1267. [Google Scholar] [CrossRef]
- Ai Zamruddin, N.N.S.; Zainal Abidin, N.S.; Endut, Z.; Makoundi, C.; Lok, L.K.; Ismail, M.S. Trace Element Analysis of Pyrite and Arsenopyrite Using the LA-ICPMS Technique in Pulai, Central Belt of Peninsular Malaysia. Minerals 2023, 13, 1026. [Google Scholar] [CrossRef]
- Bajwah, Z.; Seccombe, P.; Offler, R. Trace element distribution, Co: Ni ratios and genesis of the Big Cadia iron-copper deposit, New South Wales, Australia. Mineralium Deposita 1987, 22, 292–300. [Google Scholar] [CrossRef]
- Reich, M.; Kesler, S.E.; Utsunomiya, S.; Palenik, C.S.; Chryssoulis, S.L.; Ewing, R.C. Solubility of gold in arsenian pyrite. Geochimica et Cosmochimica Acta 2005, 69, 2781–2796. [Google Scholar] [CrossRef]
- Fleet, M.E.; Mumin, A.H. Gold-bearing arsenian pyrite and marcasite and arsenopyrite from Carlin Trend gold deposits and laboratory synthesis. American Mineralogist 1997, 82, 182–193. [Google Scholar] [CrossRef]
- Qian, G.; Brugger, J.; Testemale, D.; Skinner, W.; Pring, A. Formation of As (II)-pyrite during experimental replacement of magnetite under hydrothermal conditions. Geochimica et Cosmochimica Acta 2013, 100, 1–10. [Google Scholar] [CrossRef]
- Herazo, A.; Reich, M.; Barra, F.; Morata, D.; Real, I.d. Trace element geochemistry of pyrite from bitumen-bearing stratabound Cu–(Ag) deposits, Northern Chile. ACS Earth and Space Chemistry 2021, 5, 566–579. [Google Scholar] [CrossRef]
- Keith, M.; Häckel, F.; Haase, K.M.; Schwarz-Schampera, U.; Klemd, R. Trace element systematics of pyrite from submarine hydrothermal vents. Ore Geology Reviews 2016, 72, 728–745. [Google Scholar] [CrossRef]
- Nyström, E.; Thomas, H.; Wanhainen, C.; Alakangas, L. Occurrence and release of trace elements in pyrite-rich waste rock. Minerals 2021, 11, 495. [Google Scholar] [CrossRef]
- Wang, Y.Y.; Long, T.; Fougerouse, D.; Liu, X.M.; Li, Q.; Yu, X.F.; Ma, M.Z.; Liu, D.Y. Heterogeneous incorporation of trace elements at the microscale and nanoscale during episodic epitaxial growth of pyrite. Geological Society of America Bulletin 2025, 137, 899–910. [Google Scholar] [CrossRef]
- Yu, Q.; Wang, Z.; Sun, Q.; Wang, K. In Situ Trace Element and Sulfur Isotope Composition of Pyrite from the Beiwagou Pb-Zn Deposit, Liaodong Peninsula, Northeast China: Implications for Ore Genesis. Minerals 2023, 13, 1176. [Google Scholar] [CrossRef]
- Zhang, Z.; Xie, G.; Olin, P. Texture, in-situ geochemical, and S isotopic analyses of pyrite and arsenopyrite from the Longshan Sb-Au deposit, southern China: Implications for the genesis of intrusion-related Sb-Au deposit. Ore Geology Reviews 2022, 143, 104781. [Google Scholar] [CrossRef]
- Maslennikov, V.; Maslennikova, S.; Large, R.; Danyushevsky, L. Study of trace element zonation in vent chimneys from the Silurian Yaman-Kasy volcanic-hosted massive sulfide deposit (Southern Urals, Russia) using laser ablation-inductively coupled plasma mass spectrometry (LA-ICPMS). Economic Geology 2009, 104, 1111–1141. [Google Scholar] [CrossRef]
- Abraitis, P.; Pattrick, R.; Vaughan, D. Variations in the compositional, textural and electrical properties of natural pyrite: a review. International Journal of Mineral Processing 2004, 74, 41–59. [Google Scholar] [CrossRef]
- Qin, Z.W.; Wang, L.; Qin, K.Z.; Li, G.M.; Xue, S.C.; Song, G.X. Geochemistry of trace elements of pyrite and its implications in intermediate sulfidation epithermal systems: Case study based on the Zhengguang Au–Zn deposit, North-East China. Journal of Geochemical Exploration 2025, 107810. [Google Scholar] [CrossRef]
- Revan, M.K.; Genc, Y.; Maslennikov, V.V.; Maslennikova, S.P.; Large, R.R.; Danyushevsky, L.V. Mineralogy and trace-element geochemistry of sulfide minerals in hydrothermal chimneys from the Upper-Cretaceous VMS deposits of the eastern Pontide orogenic belt (NE Turkey). Ore Geology Reviews 2014, 63, 129–149. [Google Scholar] [CrossRef]
- Martín, J.A.; Lespagnard, J.M.; Maroto, A.G.; Galiano, J.G. Distribución geoquímica de elementos traza en los sulfuros pirita y cinabrio y en las intercalaciones pizarrosas de la capa de San Nicolás. Yacimiento de Almaden (Ciudad Real). Estudios Geológicos 1988, 44, 181–187. [Google Scholar] [CrossRef]
- Deng, C.; Lehmann, B.; Xiao, T.; Tan, Q.; Chen, D.; Tian, Z.; Wang, X.; Sun, G.; Yin, R. Intracontinental and arc-related hydrothermal systems display distinct δ202Hg and Δ199Hg features: Implication for large-scale mercury recycling and isotopic fractionation in different tectonic settings. Earth and Planetary Science Letters 2022, 593, 117646. [Google Scholar] [CrossRef]
- Wang, Q.; Morse, J.W. Pyrite formation under conditions approximating those in anoxic sediments I. Pathway and morphology. Marine Chemistry 1996, 52, 99–121. [Google Scholar] [CrossRef]
- Wang, X.; Shi, M.; Zhang, J.; Pang, Y.; Zhao, Y. Significance of trace elements in marine shale pyrite for reconstructing the sedimentary environment: a case study of Niutitang and Hongshuizhuang Formations. ACS Earth and Space Chemistry 2021, 5, 3210–3225. [Google Scholar] [CrossRef]
- Jones, D.S.; Fike, D.A. Dynamic sulfur and carbon cycling through the end-Ordovician extinction revealed by paired sulfate–pyrite δ34S. Earth and Planetary Science Letters 2013, 363, 144–155. [Google Scholar] [CrossRef]
- Audétat, A. The metal content of magmatic-hydrothermal fluids and its relationship to mineralization potential. Economic Geology 2019, 114, 1033–1056. [Google Scholar] [CrossRef]

| SAMPLE | PYRITE (%) | CINNABAR (%) | GANGUE (%) |
|---|---|---|---|
| S-K-a | 30 | 5 | 65 |
| S-K-b | 10 | 30 | 60 |
| S-K-c | 60 | 25 | 15 |
| S-K-d | 35 | 20 | 45 |
| S-K-e | 55 | 5 | 40 |
| No. | V | Cr | Mn | Co | Ni | Cu | Ga | As | Ag | Cd | Sb | Au | Tl | Pb |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PY1 | 0.24 | 0.74 | 0.27 | 0.94 | 17.95 | 4.26 | - | 9.17 | 0.019 | - | 2.02 | 0.025 | 0.62 | 27.57 |
| PY2 | 0.11 | 0.39 | 0.24 | 1.76 | 36.18 | 4.22 | - | 12.93 | 0.035 | - | 2.73 | - | 0.55 | 39.38 |
| PY3 | 0.12 | 0.60 | 0.22 | 2.21 | 46.66 | 4.74 | - | 15.30 | - | - | 6.18 | - | 0.30 | 65.15 |
| PY4 | 0.26 | - | 0.54 | 1.41 | 29.31 | 8.35 | 0.067 | 14.26 | - | - | 2.00 | - | 0.82 | 33.79 |
| PY5 | 0.13 | - | 0.24 | 0.87 | 17.88 | 8.91 | - | 9.16 | 0.048 | - | 2.81 | - | 1.01 | 39.18 |
| PY6 | 0.42 | 0.70 | 0.24 | 0.78 | 15.28 | 5.22 | 0.060 | 8.03 | - | - | 1.59 | - | 0.56 | 23.46 |
| PY7 | 0.099 | - | 0.26 | 1.75 | 33.63 | 4.48 | - | 14.09 | 0.027 | - | 0.89 | - | 0.24 | 21.81 |
| PY8 | 0.14 | - | 0.38 | 2.22 | 44.54 | 8.35 | - | 18.93 | - | - | 2.13 | - | 1.68 | 49.83 |
| PY9 | 0.14 | - | 0.23 | 0.53 | 10.63 | 3.63 | - | 9.89 | 0.014 | - | 0.68 | - | 0.64 | 12.87 |
| PY10 | 0.13 | - | 0.22 | 0.63 | 12.86 | 1.67 | - | 9.82 | 0.016 | - | 1.36 | - | 0.32 | 16.74 |
| PY11 | 0.11 | 0.60 | 0.18 | 0.85 | 17.14 | 1.62 | - | 9.73 | - | - | 0.78 | - | 0.05 | 11.60 |
| PY12 | 0.12 | - | 0.25 | 0.61 | 13.64 | 1.58 | - | 10.37 | - | - | 2.79 | - | 0.45 | 28.63 |
| PY13 | 0.16 | - | 0.38 | 0.72 | 14.64 | 2.08 | - | 11.19 | 0.028 | - | 1.66 | - | 0.30 | 20.92 |
| PY14 | 0.23 | 0.58 | 0.33 | 0.40 | 7.41 | 4.32 | - | 6.48 | 0.014 | 0.35 | 1.19 | - | 2.21 | 11.20 |
| PY15 | 0.15 | 0.54 | 0.23 | - | 4.30 | - | 0.027 | 4.71 | 0.020 | - | 1.01 | - | 2.20 | 8.18 |
| PY16 | 0.085 | - | 0.18 | 3.72 | 46.98 | 4.53 | - | 13.68 | 0.032 | - | 1.59 | - | 0.27 | 20.25 |
| PY17 | 0.051 | - | 0.18 | 3.52 | 52.72 | - | - | 21.73 | 0.023 | - | 0.85 | - | 0.25 | 13.78 |
| PY18 | 0.040 | 0.38 | 0.18 | 4.38 | 76.69 | - | - | 23.41 | 0.026 | - | 1.12 | - | 0.16 | 22.02 |
| ELEMENT | V | Cr | Mn | Co | Ni | Cu | As | Ag | Sb | Tl |
|---|---|---|---|---|---|---|---|---|---|---|
| Cr | 0.72 | |||||||||
| Mn | 0.44 | 0.34 | ||||||||
| Co | ||||||||||
| Ni | 0.96 | |||||||||
| Cu | 0.23 | 0.13 | 0.48 | 0.31 | 0.40 | |||||
| As | 0.88 | 0.93 | 0.40 | |||||||
| Ag | 0.23 | 0.29 | 0.70 | 0.18 | ||||||
| Sb | 0.06 | 0.06 | 0.01 | 0.18 | 0.21 | 0.08 | 0.73 | |||
| Tl | 0.28 | 0.05 | 0.36 | 0.51 | ||||||
| Pb | 0.24 | 0.13 | 0.36 | 0.53 | 0.32 | 0.80 | 0.88 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




