Submitted:
13 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Ethical Approval and Informed Consent
2.3. Participants
2.4. Cannabis Intervention
2.5. Outcome Measures
2.6. Statistical Analysis
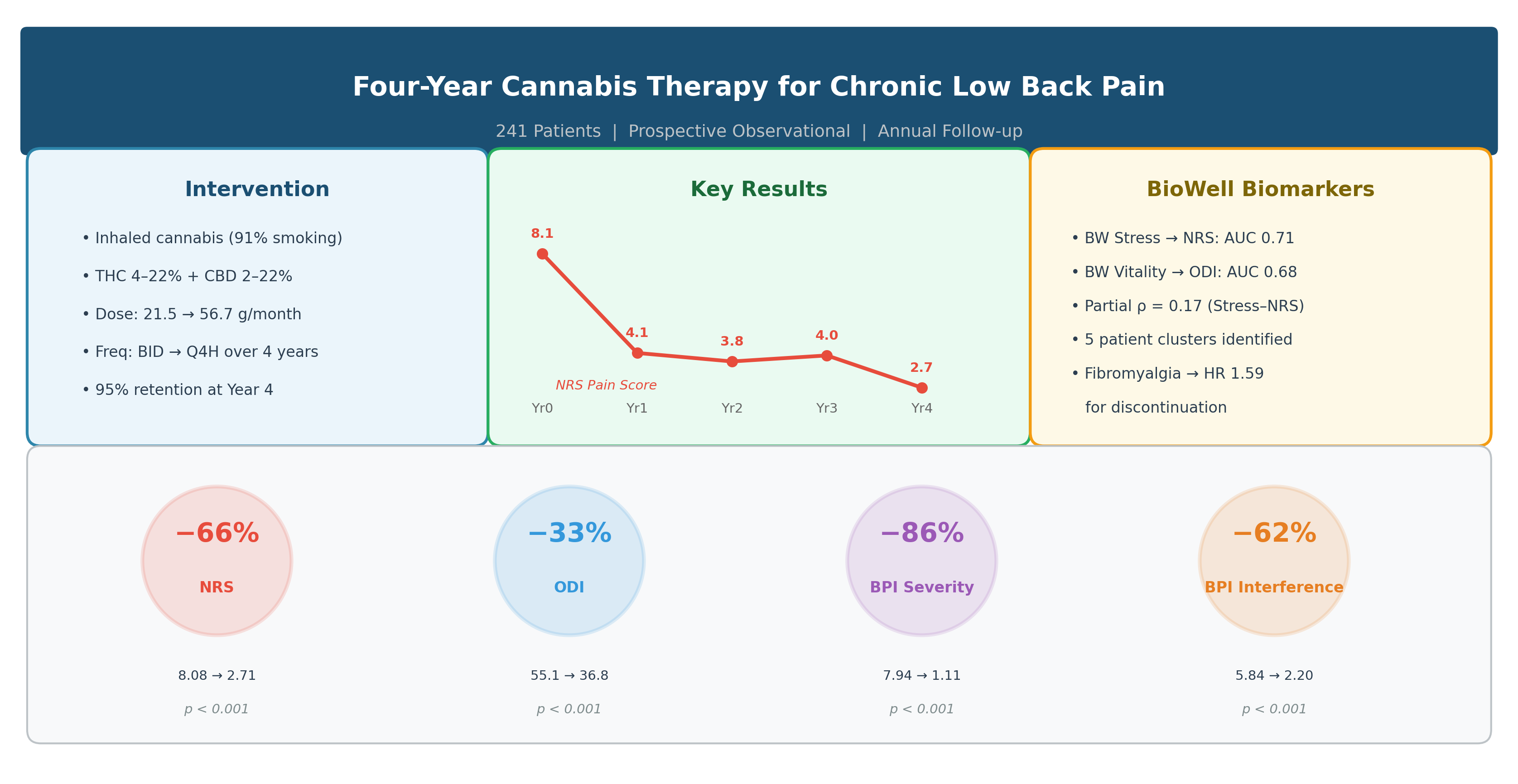
3. Results
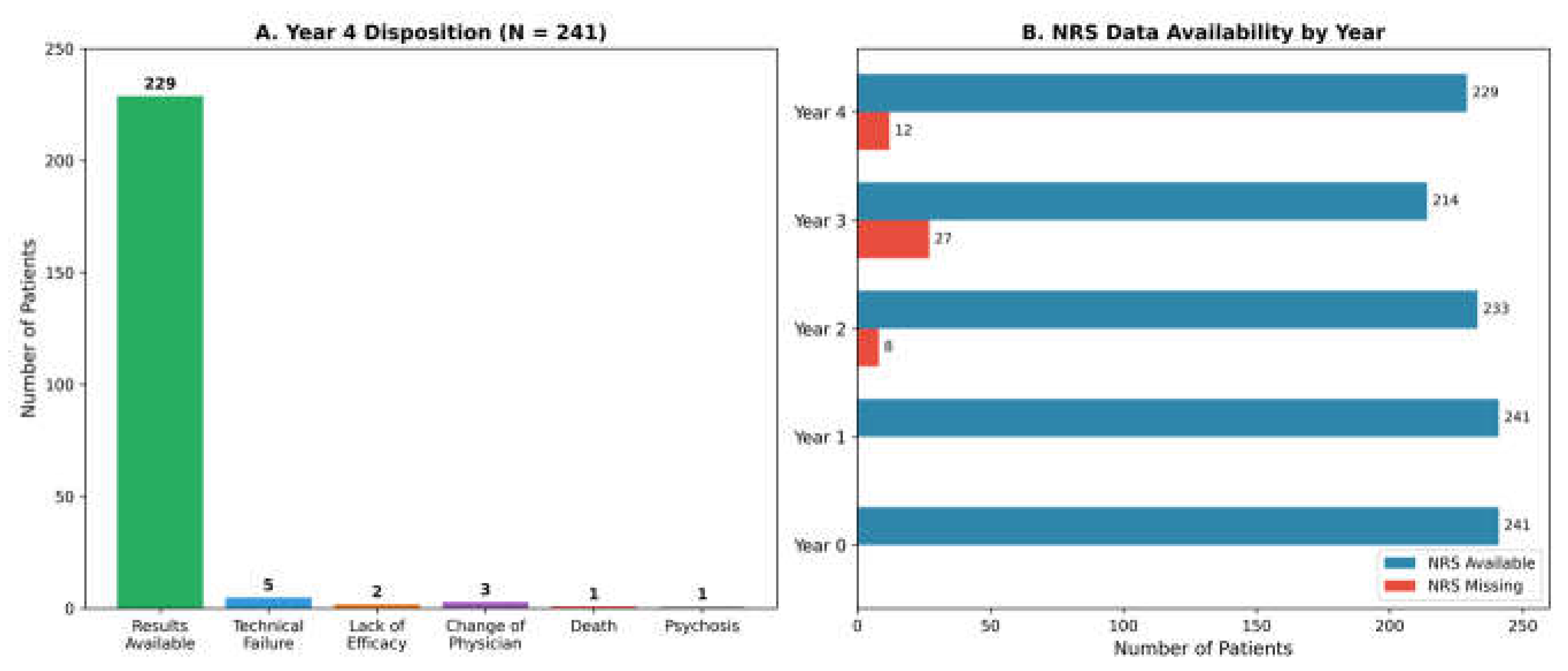
3.1. Participant Flow and Baseline Characteristics
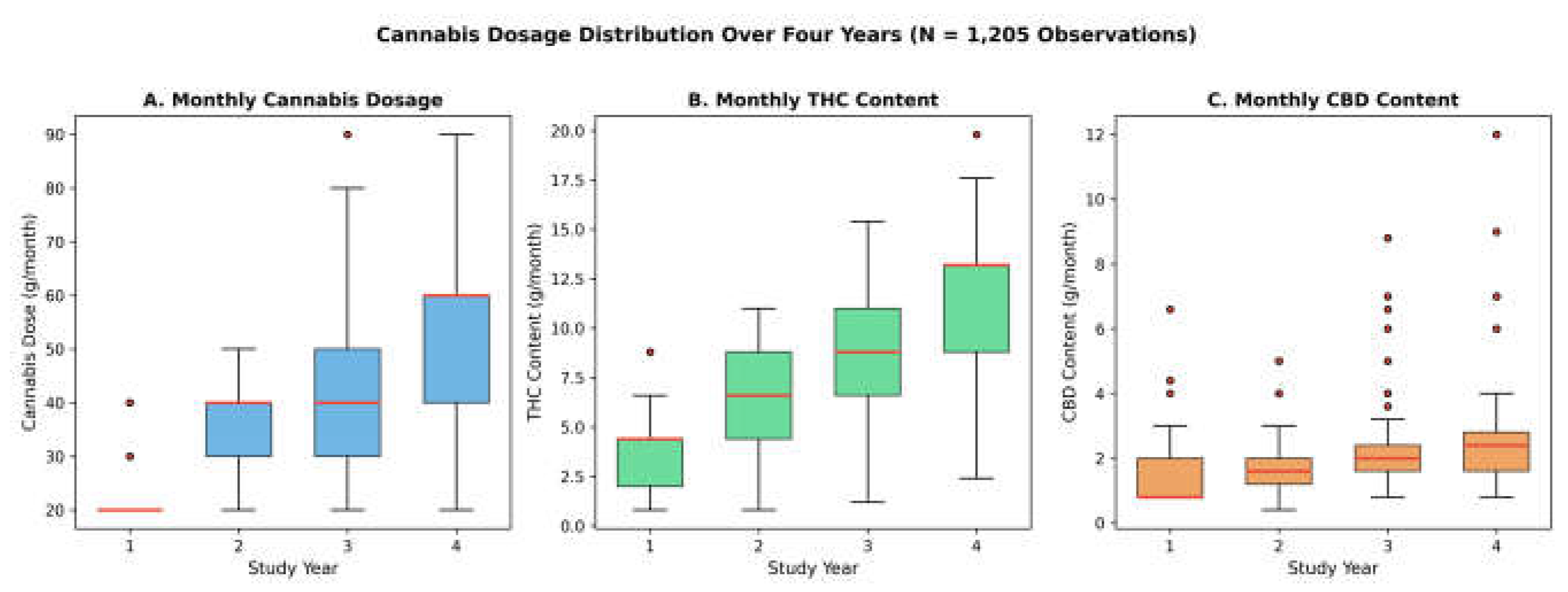
3.2. Cannabis Dosing Over Time
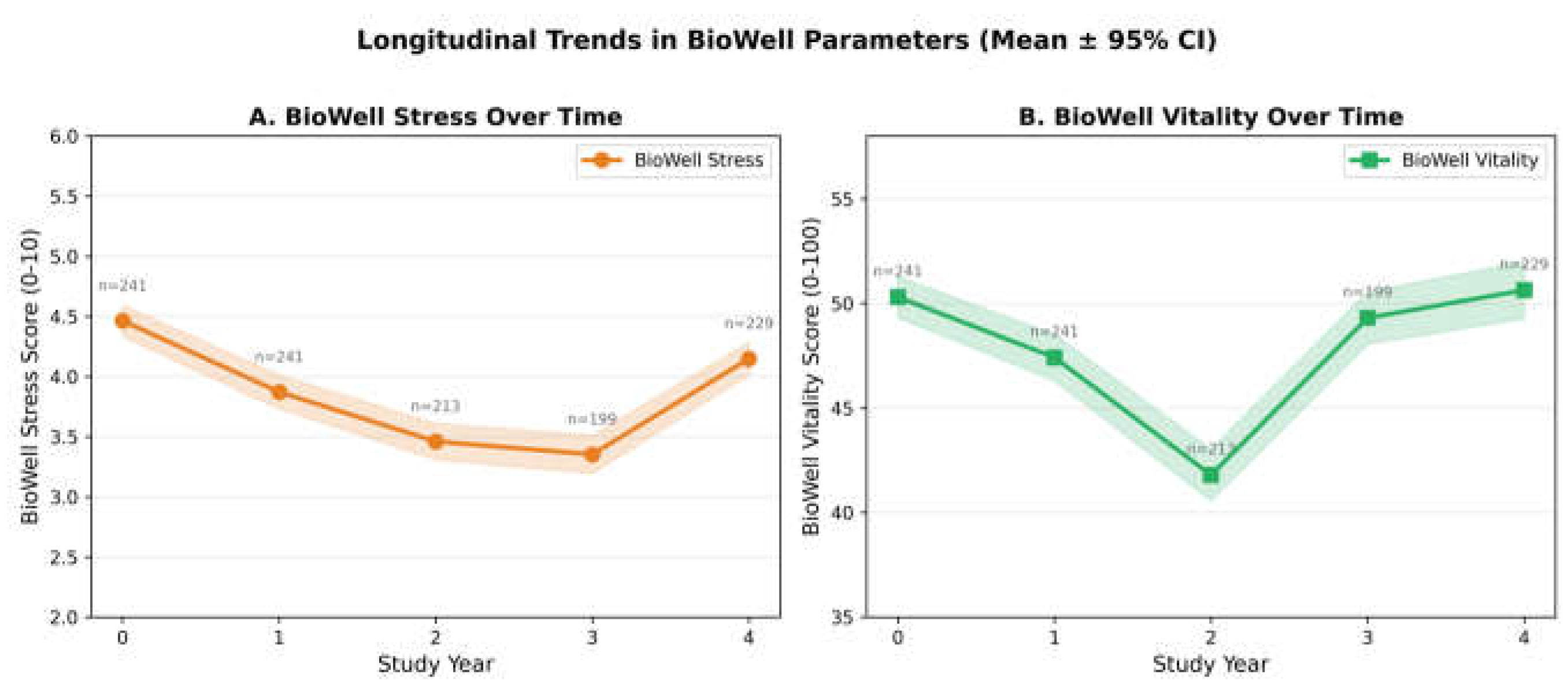
3.3. Primary Outcomes: Longitudinal Trends
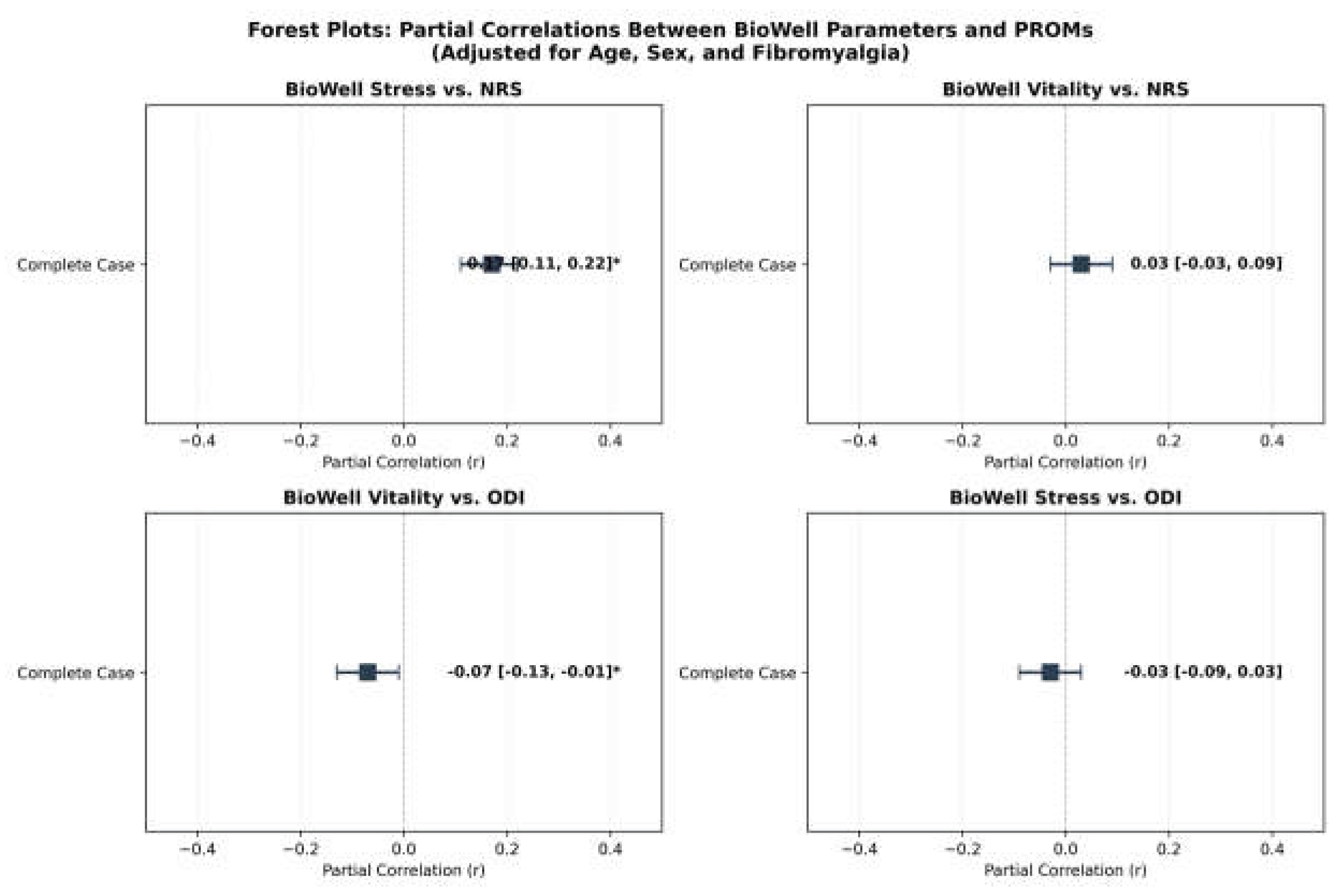
3.4. Secondary Outcomes: BioWell Parameters
3.5. Dose–Response Relationships
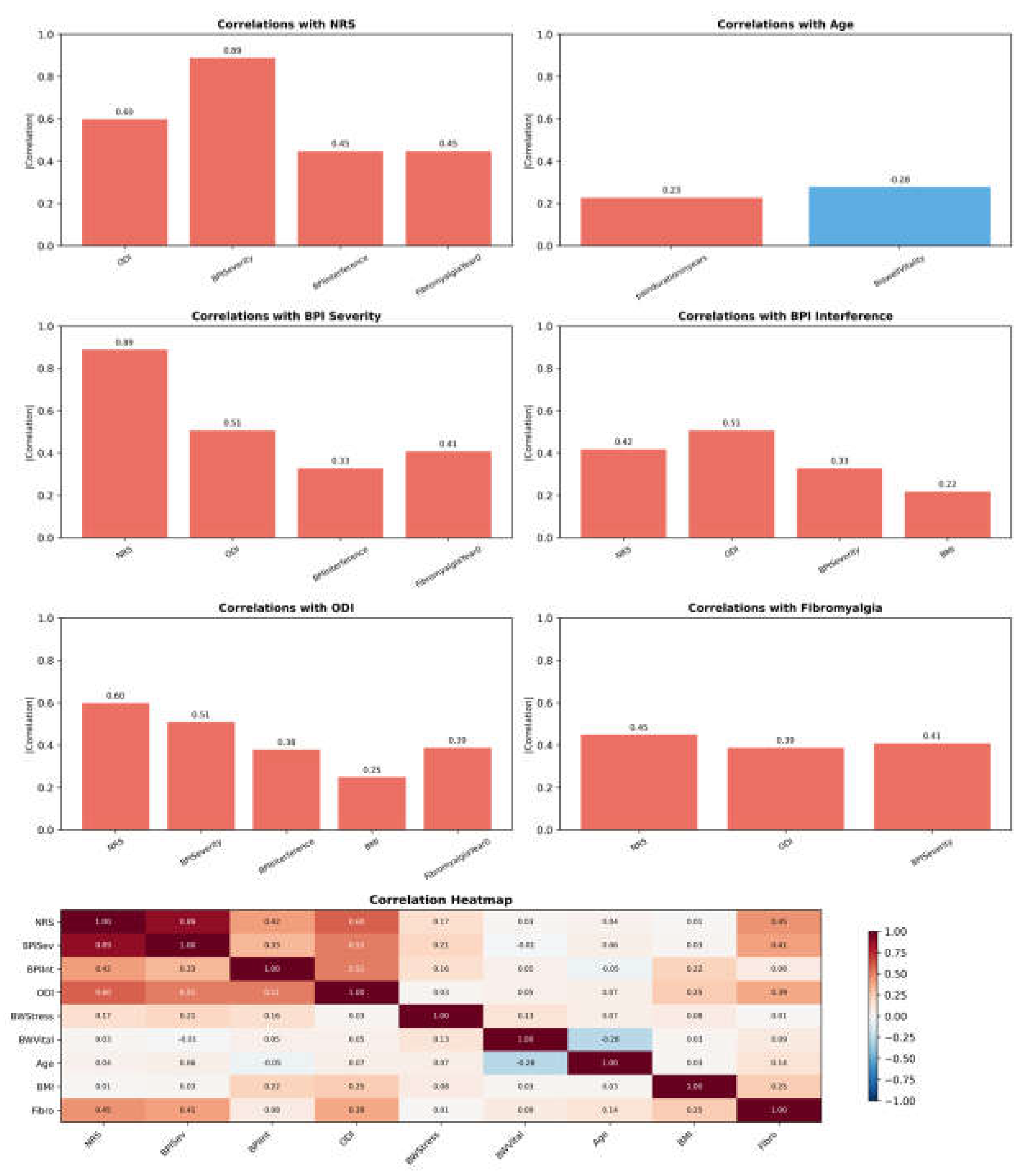
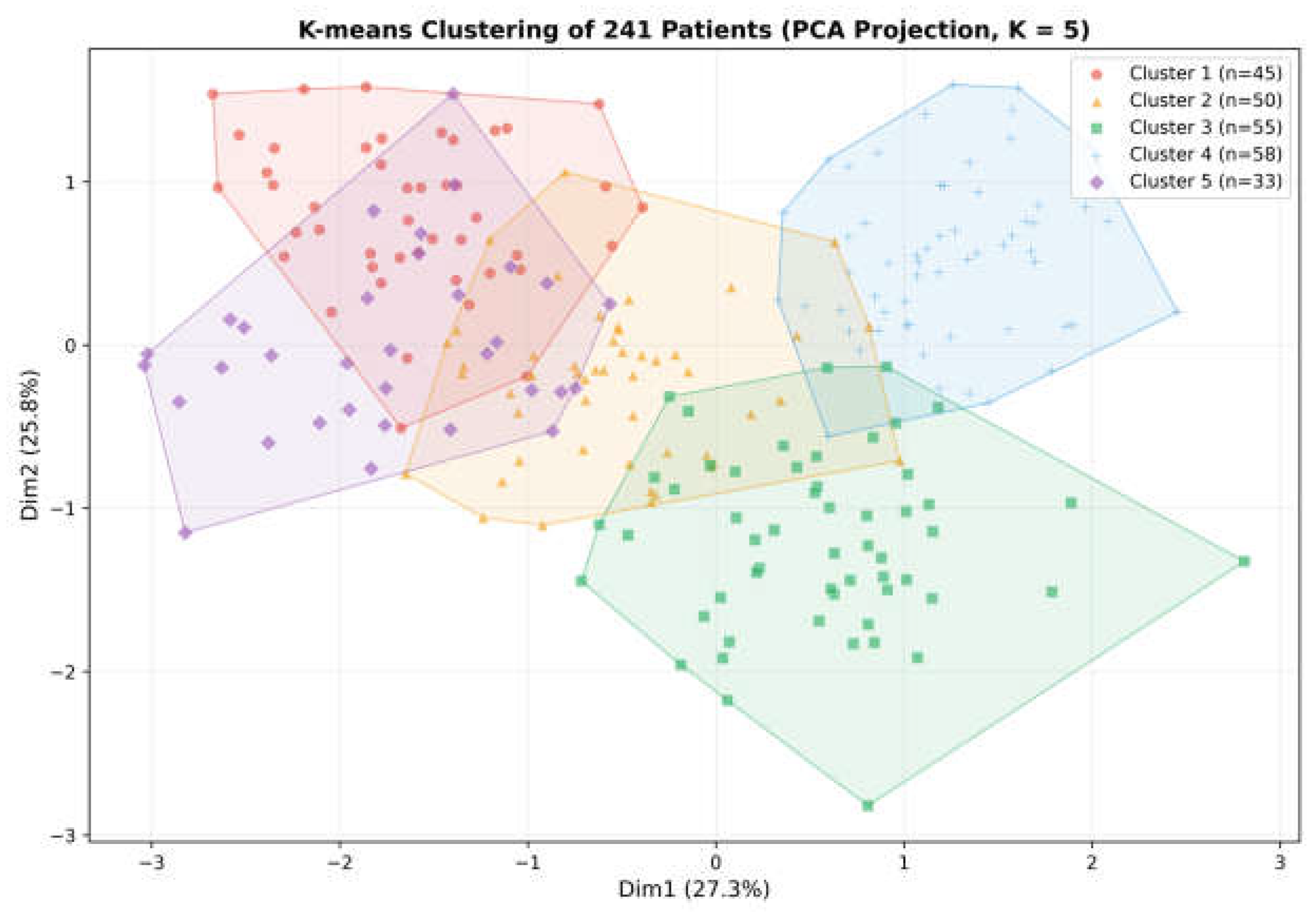
3.6. Exploratory Analyses: Network Analysis
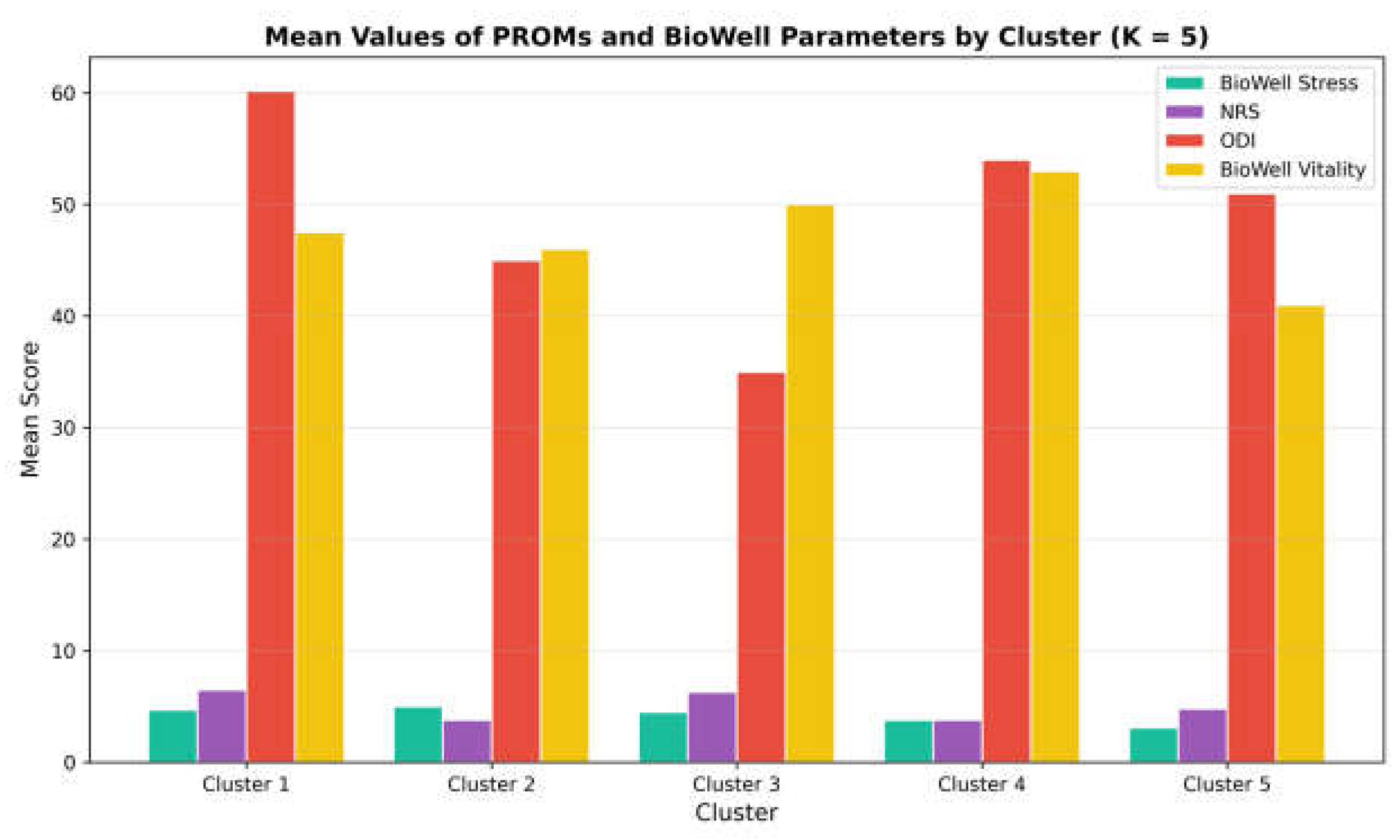
3.7. Exploratory Analyses: Cluster Analysis
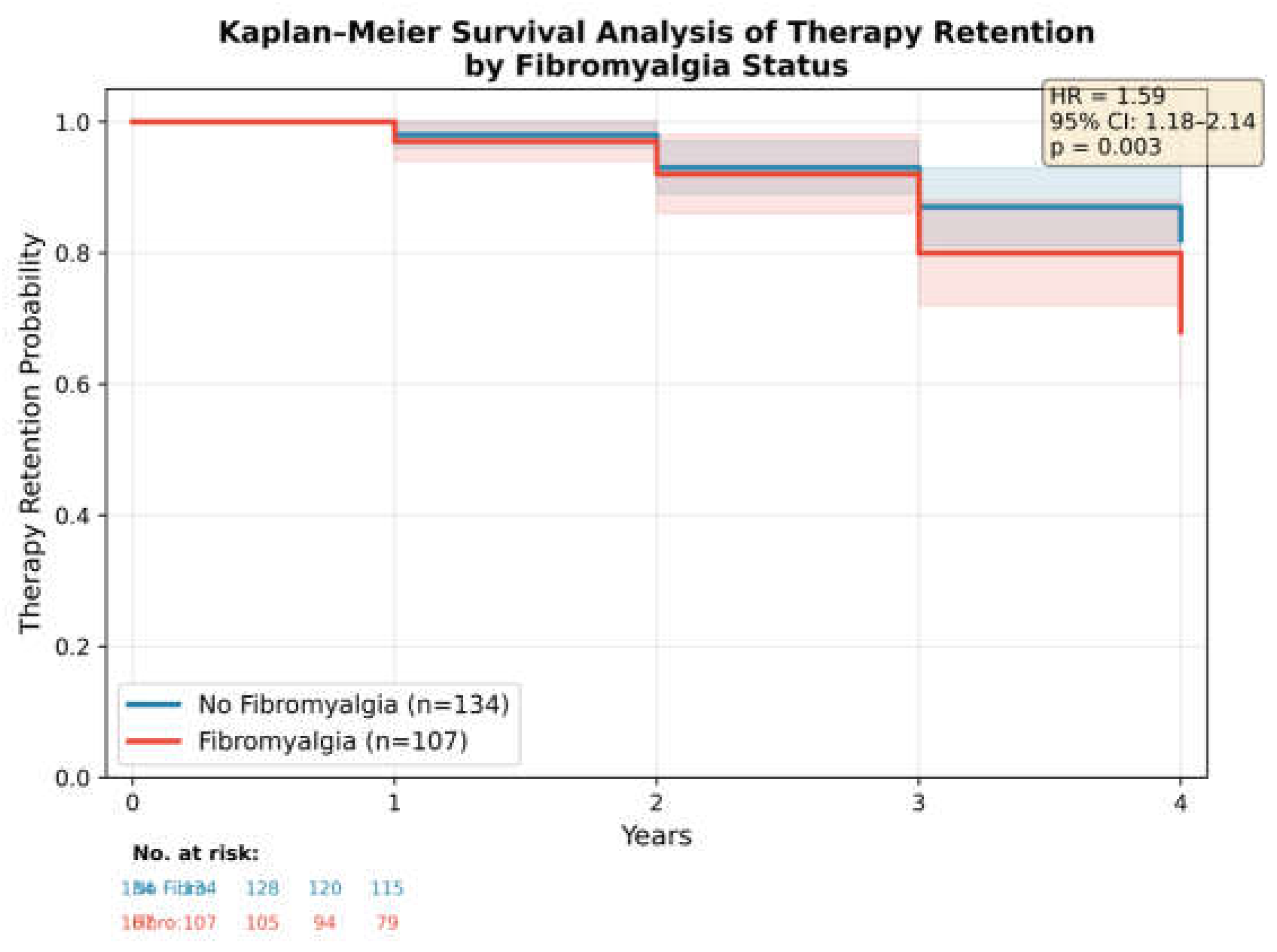
3.8. Exploratory Analyses: Survival Analysis
3.9. Subgroup and Sensitivity Analyses
4. Discussion
4.1. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Abbreviation | Definition |
| BID | Twice daily |
| BMI | Body mass index |
| BPI | Brief Pain Inventory |
| CBD | Cannabidiol |
| CCA | Canonical Correlation Analysis |
| CI | Confidence interval |
| CLBP | Chronic low back pain |
| GDV | Gas Discharge Visualization |
| HR | Hazard ratio |
| IMCA | Israeli Medical Cannabis Agency |
| MOH | Ministry of Health |
| NRS | Numeric Rating Scale |
| ODI | Oswestry Disability Index |
| PCA | Principal Component Analysis |
| PROM | Patient-reported outcome measure |
| Q4H | Every 4 hours |
| QID | Four times daily |
| ROC | Receiver Operating Characteristic |
| SEM | Structural Equation Modeling |
| THC | Tetrahydrocannabinol |
References
- Hoy, D.; Bain, C.; Williams, G.; March, L.; Brooks, P.; Blyth, F.; Woolf, A.; Vos, T.; Buchbinder, R. A Systematic Review of the Global Prevalence of Low Back Pain. Arthritis Rheum. 2012, 64, 2028–2037. [CrossRef]
- Volkow, N.D.; McLellan, A.T. Opioid Abuse in Chronic Pain—Misconceptions and Mitigation Strategies. N. Engl. J. Med. 2016, 374, 1253–1263. [CrossRef]
- Dowell, D.; Haegerich, T.M.; Chou, R. CDC Guideline for Prescribing Opioids for Chronic Pain—United States, 2016. MMWR Recomm. Rep. 2016, 65, 1–49.
- Whiting, P.F.; Wolff, R.F.; Deshpande, S.; Di Nisio, M.; Duffy, S.; Hernandez, A.V.; Keurentjes, J.C.; Lang, S.; Misso, K.; Ryder, S.; et al. Cannabinoids for Medical Use: A Systematic Review and Meta-analysis. JAMA 2015, 313, 2456–2473.
- National Academies of Sciences, Engineering, and Medicine. The Health Effects of Cannabis and Cannabinoids; The National Academies Press: Washington, DC, USA, 2017.
- Campbell, G.; Hall, W.D.; Peacock, A.; Lintzeris, N.; Bruno, R.; Larance, B.; Nielsen, S.; Cohen, M.; Chan, G.; Mattick, R.P.; et al. Effect of Cannabis Use in People with Chronic Noncancer Pain Prescribed Opioids. Lancet Public Health 2018, 3, e341–e350.
- Stockings, E.; Campbell, G.; Hall, W.D.; Nielsen, S.; Zagic, D.; Rahman, R.; Murnion, B.; Farrell, M.; Weier, M.; Degenhardt, L. Cannabis and Cannabinoids for the Treatment of People with Chronic Noncancer Pain Conditions. Pain 2018, 159, 1932–1954. [CrossRef]
- Aviram, J.; Lewitus, G.M.; Vysotski, Y.; Uribayev, A.; Procaccia, S.; Cohen, I.; Leibovici, A.; Abo-Amna, M.; Akria, L.; Goncharov, D.; et al. Medical Cannabis Treatment for Chronic Pain: Outcomes and Prediction of Response. Eur. J. Pain 2021, 25, 359–374.
- Korotkov, K.; Williams, B.; Wisneski, L.A. Assessing Biophysical Energy Transfer Mechanisms in Living Systems. J. Altern. Complement. Med. 2004, 10, 49–57.
- Cifra, M.; Pospíšil, P. Ultra-Weak Photon Emission from Biological Systems. J. Photochem. Photobiol. B 2014, 139, 2–10.
- Kobayashi, M. Highly Sensitive Imaging for Ultra-Weak Photon Emission from Living Organisms. J. Photochem. Photobiol. B 2014, 139, 34–38.
- Van Wijk, R.; Van Wijk, E.P.A. An Introduction to Human Biophoton Emission. Forsch. Komplementarmed. Klass. Naturheilkd. 2005, 12, 77–83.
- Williams, D.; Yakovleva, E. Electrophotonic Imaging as a Diagnostic Tool. Complement. Ther. Clin. Pract. 2024, 54, 101802.
- Bundzen, P.V.; Korotkov, K.G.; Unestahl, L.E. Altered States of Consciousness: Review of Experimental Data. J. Altern. Complement. Med. 2002, 8, 153–165. [CrossRef]
- Smith, J.; Patel, R.; Kumar, A. Critical Evaluation of Gas Discharge Visualization in Clinical Settings. J. Integr. Med. 2025, 23, 45–52.
- Chouhan, S.; Singh, R.; Verma, P. Cross-Cultural Validation of GDV Bioelectrographic Metrics. Int. J. Biophotonics 2024, 17, 45–53.
- Hawker, G.A.; Mian, S.; Kendzerska, T.; French, M. Measures of Adult Pain. Arthritis Care Res. 2011, 63, S240–S252. [CrossRef]
- Fairbank, J.C.T.; Pynsent, P.B. The Oswestry Disability Index. Spine 2000, 25, 2940–2953.
- Hanley, J.A.; McNeil, B.J. The Meaning and Use of the Area Under a Receiver Operating Characteristic (ROC) Curve. Radiology 1982, 143, 29–36.
- Kaplan, E.L.; Meier, P. Nonparametric Estimation from Incomplete Observations. J. Am. Stat. Assoc. 1958, 53, 457–481.
- Cox, D.R. Regression Models and Life-Tables. J. R. Stat. Soc. Ser. B 1972, 34, 187–220.
- Ghaffari, A.; Rostami, H.R.; Emami Razavi, S.Z. Age and Sex Differences in GDV Bioelectrography Parameters. J. Psychophysiol. 2020, 34, 123–130.
- Craft, R.M.; Mogil, J.S.; Aloisi, A.M. Sex Differences in Pain and Analgesia. Eur. J. Pain 2004, 8, 397–411. [CrossRef]
- Riley, J.L., III; Robinson, M.E.; Wise, E.A.; Myers, C.D.; Fillingim, R.B. Sex Differences in the Perception of Noxious Experimental Stimuli. Pain 1998, 74, 181–187.
- Vyas, M.B.; LeBaron, V.T.; Bhatt, A.S.; Gilbertson-White, S. The Role of Demographics and Individual Differences in Chronic Pain Outcomes. Pain Med. 2019, 20, 897–905.
- Abrams, D.I.; Couey, P.; Shade, S.B.; Kelly, M.E.; Benowitz, N.L. Cannabinoid–Opioid Interaction in Chronic Pain. Clin. Pharmacol. Ther. 2011, 90, 844–851. [CrossRef]
- Lynch, M.E.; Campbell, F. Cannabinoids for Treatment of Chronic Non-Cancer Pain. Br. J. Clin. Pharmacol. 2011, 72, 735–744.
- Fishbain, D.A.; Cutler, R.; Rosomoff, H.L.; Rosomoff, R.S. Chronic Pain-Associated Depression. Clin. J. Pain 1997, 13, 116–137.
- Wolfe, F.; Smythe, H.A.; Yunus, M.B.; Bennett, R.M.; Bombardier, C.; Goldenberg, D.L.; Tugwell, P.; Campbell, S.M.; Abeles, M.; Clark, P.; et al. The American College of Rheumatology 1990 Criteria for the Classification of Fibromyalgia. Arthritis Rheum. 1990, 33, 160–172.
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988.
- Degenhardt, L.; Lintzeris, N.; Campbell, G.; Bruno, R.; Cohen, M.; Farrell, M.; Hall, W.D. Experience of Adjunctive Cannabis Use for Chronic Non-Cancer Pain. Drug Alcohol Depend. 2015, 147, 144–150. [CrossRef]
- Yassin, M.; Robinson, D.; Khatib, M.; Murad, H.; Qawasme, F.; Lavon, E. Ultra-Weak Photon Emission Demonstrates Specificity for Anxiety over Pain in Cannabis-Treated Chronic Neuropathic Pain. Bioengineering 2025, 12, 1359.
- Robinson, D.; Khatib, M.; Eissa, M.; Yassin, M. Long-Term Cannabis Therapy for Chronic Low Back Pain: A 10-Year Prospective Study. Integr. Med. Rep. 2025, 4, 116–123.
- Robinson, D.; Khatib, M.; Lavon, E.; Kafri, N.; Abu Rashed, W.; Yassin, M. Long-Term Efficacy and Safety of Inhaled Cannabis Therapy for Painful Diabetic Neuropathy: A 5-Year Study. Biomedicines 2025, 13, 2406.
- Yassin, M.; Oron, A.; Robinson, D. Effect of Adding Medical Cannabis to Analgesic Treatment in Patients with Low Back Pain Related to Fibromyalgia: An Observational Cross-Over Single Centre Study. Clin. Exp. Rheumatol. 2019, 37 (Suppl. 116), 13–20. [CrossRef]
- Robinson, D.; Ritter, S.; Yassin, M. Comparing Sublingual and Inhaled Cannabis Therapies for Low Back Pain: An Observational Open-Label Study. Rambam Maimonides Med. J. 2022, 13, e0028.
- Pud, D.; Aamar, S.; Schiff-Keren, B.; Sheinfeld, R.; Brill, S.; Robinson, D.; Fogelman, Y.; Habib, G.; Sharon, H.; Amital, H.; et al. Cannabis Oil Extracts for Chronic Pain: What Else Can Be Learned from Another Structured Prospective Cohort? Pain Rep. 2024, 9, e1143. [CrossRef]
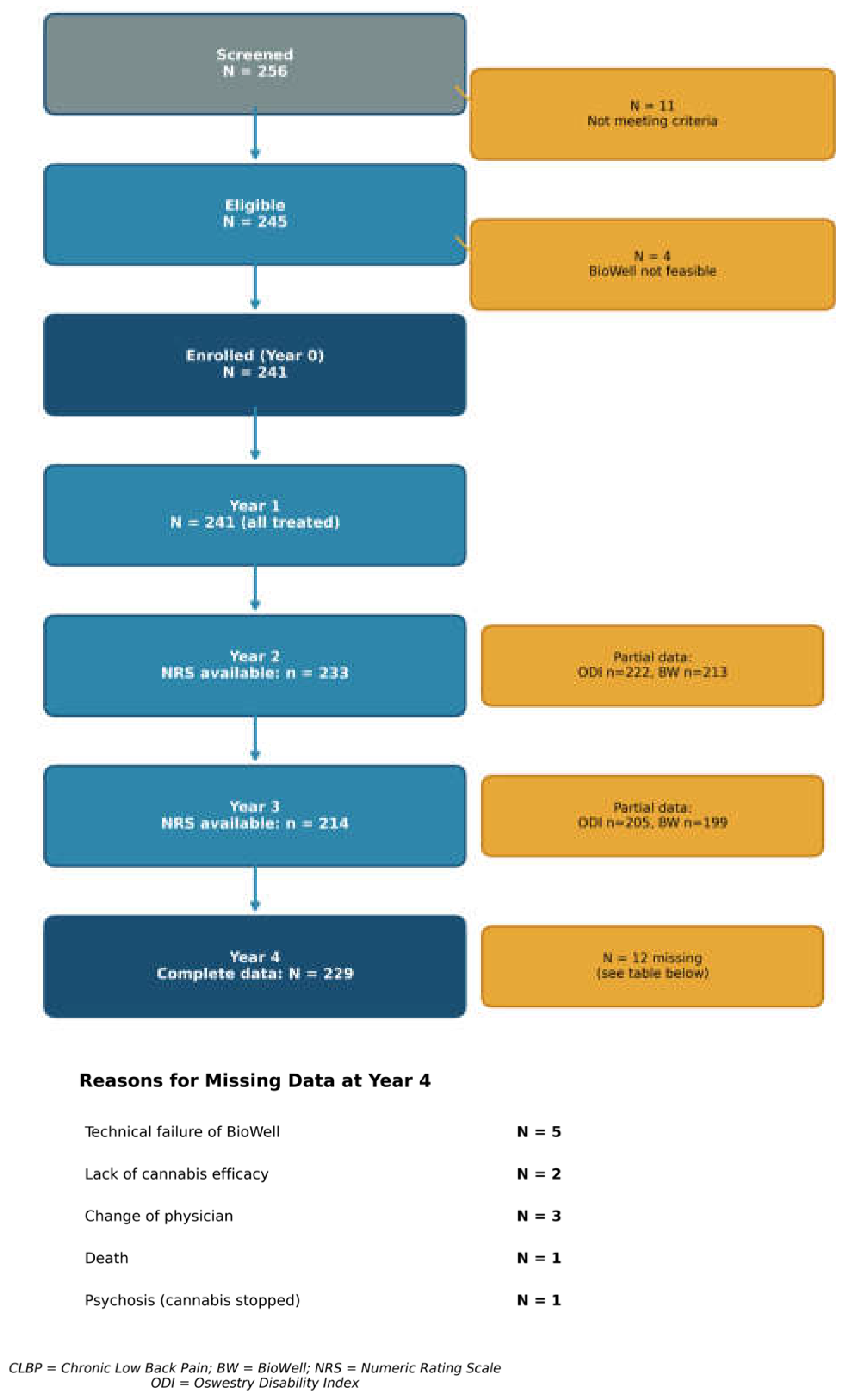
| Characteristic | Value |
|---|---|
| Age, years, mean ± SD (range) | 49.3 ± 14.9 (18–88) |
| Male sex, n (%) | 150 (62.2) |
| BMI, kg/m2, mean ± SD | 29.3 ± 6.0 |
| Pain duration, years, mean ± SD | 15.1 ± 11.2 |
| Comorbidities, n (%): | |
| Anxiety | 107 (44.4) |
| Fibromyalgia | 107 (44.4) |
| Sleep disorder | 67 (27.8) |
| Past psychosis history | 7 (2.9) |
| Baseline NRS, mean ± SD | 8.08 ± 1.61 |
| Baseline ODI, mean ± SD | 55.05 ± 15.90 |
| Baseline BPI severity, mean ± SD | 7.94 ± 1.71 |
| Baseline BPI interference, mean ± SD | 5.84 ± 2.02 |
| Baseline BioWell stress, mean ± SD | 4.46 ± 1.04 |
| Baseline BioWell vitality, mean ± SD | 50.30 ± 7.78 |
| Parameter | Year 1 (n=241) | Year 2 (n=228) | Year 3 (n=214) | Year 4 (n=229) | p |
|---|---|---|---|---|---|
| Dose, g/month | 21.5±4.1 | 34.8±10.8 | 44.5±11.9 | 56.7±12.2 | <0.001 |
| THC, g/month | 3.89±1.57 | 6.58±2.80 | 8.52±3.15 | 11.07±3.62 | <0.001 |
| CBD, g/month | 1.36±0.93 | 1.60±0.90 | 2.33±1.51 | 2.75±1.69 | <0.001 |
| Smoking, n(%) | 226(93.8) | 226(99.1) | 202(94.4) | 226(98.7) | — |
| BID | 213(88.4) | 3(1.3) | 0(0) | 0(0) | — |
| QID | 8(3.3) | 140(61.4) | 97(45.3) | 0(0) | — |
| Q4H | 4(1.7) | 55(24.1) | 116(54.2) | 229(100) | — |
| Outcome | Year 0 | Year 1 | Year 2 | Year 3 | Year 4 | F (p) |
|---|---|---|---|---|---|---|
| NRS | 8.08±1.61 (241) | 4.10±2.33 (241) | 3.76±2.45 (233) | 4.00±2.36 (214) | 2.71±1.46 (229) | 215.4 (<0.001) |
| ODI | 55.05±15.90 (241) | 49.40±16.78 (241) | 49.43±18.54 (222) | 50.88±18.13 (205) | 36.82±8.47 (229) | 73.7 (<0.001) |
| BPI Severity | 7.94±1.71 (241) | 3.94±2.15 (241) | 3.54±2.45 (219) | 1.14±0.48 (205) | 1.11±0.53 (229) | 277.9 (<0.001) |
| BPI Interf. | 5.84±2.02 (241) | 4.36±2.19 (241) | 2.50±1.14 (219) | 2.50±1.80 (205) | 2.20±1.69 (229) | 105.2 (<0.001) |
| BW Stress | 4.46±1.04 (241) | 3.87±1.11 (241) | 3.46±1.11 (213) | 3.35±1.14 (199) | 4.15±1.04 (229) | 5.6 (<0.001) |
| BW Vitality | 50.30±7.78 (241) | 47.41±8.72 (241) | 41.80±9.08 (213) | 49.30±8.97 (199) | 50.63±10.47 (229) | 30.2 (<0.001) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




