Submitted:
13 February 2026
Posted:
14 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
3.1. Dementia or Cognitive Impairment
3.2. Frailty
3.3. Depression
3.4. Falls
3.5. Low Muscle Strength or Sarcopenia
3.6. Multimorbidity
3.7. Functional Decline
3.8. Other Diagnoses or Several Geriatric Syndromes
4. Discussion
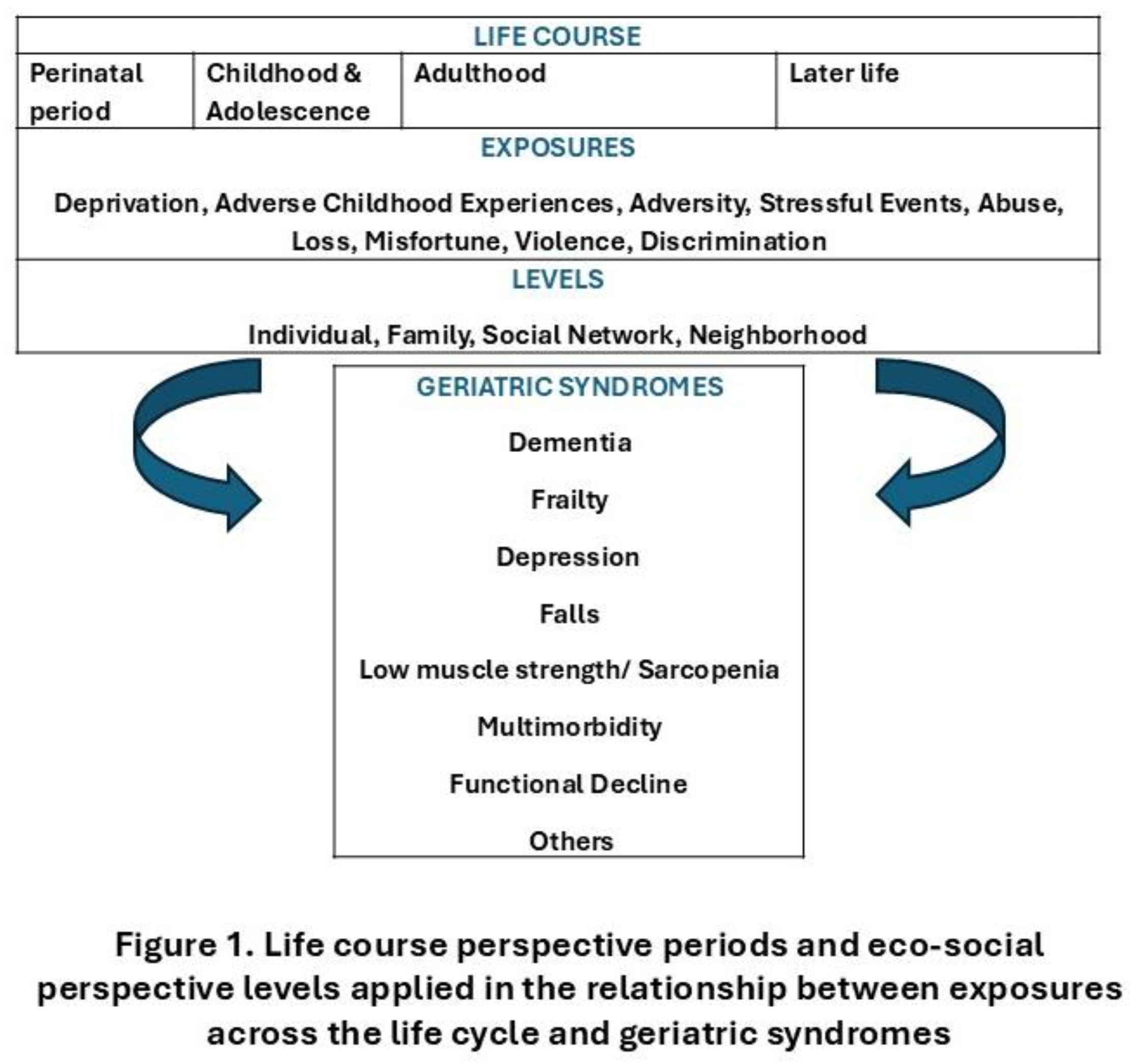
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tinetti ME, Inouye SK, Gill TM, Doucette JT. Shared risk factors for falls, incontinence, and functional dependence. Unifying the approach to geriatric syndromes. JAMA. 1995;273(17):1348-1353.
- Wang LY, Hu ZY, Chen HX, Tang ML, Hu XY. Multiple geriatric syndromes in community-dwelling older adults in China. Sci Rep. 2024;14(1):3504. Published 2024 Feb 12. [CrossRef]
- Inouye SK, Studenski S, Tinetti ME, Kuchel GA. Geriatric syndromes: clinical, research, and policy implications of a core geriatric concept. J Am Geriatr Soc. 2007;55(5):780-791. [CrossRef]
- Wu D, Yu X, Li F, Qiao W, Chen X. Geriatric syndrome awareness and its determinants in China: a cross-sectional study. BMC Geriatr. 2024;24(1):712. Published 2024 Aug 26. [CrossRef]
- Morley JE. A brief history of geriatrics. J Gerontol A Biol Sci Med Sci. 2004;59(11):1132-1152. [CrossRef]
- Crome P, Lally F. Frailty: joining the giants. CMAJ. 2011;183(8):889-890. [CrossRef]
- Rockwood K, Song X, Mitnitski A. Changes in relative fitness and frailty across the adult lifespan: evidence from the Canadian National Population Health Survey. CMAJ. 2011;183(8):E487-E494. [CrossRef]
- Sanford AM, Morley JE, Berg-Weger M, et al. High prevalence of geriatric syndromes in older adults. PLoS One. 2020;15(6):e0233857. Published 2020 Jun 5. [CrossRef]
- Kujawska A, Kujawski S, Zupkauskienė J, et al. Prevalence, co-existence, and factors related to a change in geriatric giant syndromes over 2 years: results of the Second Wave of Cognition of Older People, Education, Recreational Activities, Nutrition, Comorbidities, Functional Capacity Studies (COPERNICUS). Psychogeriatrics. 2025;25(3):e70018. [CrossRef]
- Shultz JM, Sullivan LM, Galea S. Public Health: An Introduction to the Science and Practice of Population Health. New York: Springer Publishing Company, 2019, pages 30-55.
- Hughes K, Bellis MA, Hardcastle KA, Sethi D, Butchart A, Mikton C, et al. The effect of multiple adverse childhood experiences on health: a systematic review and meta-analysis. Lancet Public Health 2017;2(8):e356–66.
- Morris G, Berk M, Maes M, Carvalho AF, Puri BK. Socioeconomic Deprivation, Adverse Childhood Experiences and Medical Disorders in Adulthood: Mechanisms and Associations. Mol Neurobiol. 2019;56(8):5866-5890. [CrossRef]
- Petruccelli K, Davis J, Berman T. Adverse childhood experiences and associated health outcomes: A systematic review and meta-analysis. Child Abuse Negl. 2019;97:104127.
- Rothman EF, Lynch AK. The State of the Science on Adverse Childhood Experiences. OTJR (Thorofare N J). 2023;43(1):6-13. [CrossRef]
- Moore CR, Polick CS, Stoddard SA. Effects of adverse childhood experiences and childhood socioeconomic disadvantage on youth depression: A scoping review. J Child Adolesc Ment Health. Published online September 29, 2025. [CrossRef]
- Tao Y, Wang H, Luo J, et al. The Association between Adverse Childhood Experiences and Frailty: A Systematic Review. J Am Med Dir Assoc. 2024;25(7):105042.
- Abouelmagd ME, AbdelMeseh M, Elrosasy A, Eldeeb HA, Nabil Y. Adverse childhood experiences and risk of late-life dementia: a systematic review and meta-analysis. Soc Psychiatry Psychiatr Epidemiol. 2025;60(5):1087-1098. [CrossRef]
- Donley GAR, Lönnroos E, Tuomainen TP, Kauhanen J. Association of childhood stress with late-life dementia and Alzheimer’s disease: the KIHD study. Eur J Public Health. 2018;28(6):1069-1073. [CrossRef]
- Conde-Sala JL, Garre-Olmo J. Early parental death and psychosocial risk factors for dementia: A case-control study in Europe. Int J Geriatr Psychiatry. 2020;35(9):1051-1059. [CrossRef]
- Tani Y, Fujiwara T, Kondo K. Association Between Adverse Childhood Experiences and Dementia in Older Japanese Adults. JAMA Netw Open. 2020;3(2):e1920740.
- Grainger SA, Crawford JD, Kochan NA, et al. An investigation into early-life stress and cognitive function in older age. Int Psychogeriatr. 2020;32(11):1325-1329. [CrossRef]
- Yang L, Wang Z. Early-Life Conditions and Cognitive Function in Middle-and Old-Aged Chinese Adults: A Longitudinal Study. Int J Environ Res Public Health. 2020;17(10):3451. Published 2020 May 15. [CrossRef]
- Nilaweera D, Freak-Poli R, Ritchie K, Chaudieu I, Ancelin ML, Ryan J. The long-term consequences of trauma and posttraumatic stress disorder symptoms on later life cognitive function and dementia risk. Psychiatry Res. 2020 Dec;294:113506.
- Macpherson JM, Gray SR, Ip P, et al. Child maltreatment and incident mental disorders in middle and older ages: a retrospective UK Biobank cohort study. Lancet Reg Health Eur. 2021;11:100224. Published 2021 Sep 27. [CrossRef]
- Gold AL, Meza E, Ackley SF, et al. Are adverse childhood experiences associated with late-life cognitive performance across racial/ethnic groups: results from the Kaiser Healthy Aging and Diverse Life Experiences study baseline. BMJ Open. 2021;11(2):e042125. Published 2021 Feb 5. [CrossRef]
- Lin L, Cao B, Chen W, Li J, Zhang Y, Guo VY. Association of Adverse Childhood Experiences and Social Isolation With Later-Life Cognitive Function Among Adults in China. JAMA Netw Open. 2022;5(11):e2241714. Published 2022 Nov 1. [CrossRef]
- Nilaweera D, Freak-Poli R, Gurvich C, et al. The association between adverse childhood events and later-life cognitive function and dementia risk. J Affect Disord. 2022;304:128-132. [CrossRef]
- Stebbins RC, Maselko J, Yang YC, Plassman BL, Edwards JK, Aiello AE. Lifecourse Traumatic Events and Cognitive Aging in the Health and Retirement Study. Am J Prev Med. 2022;63(5):818-826. [CrossRef]
- Voyer H, Aytur S, Tanda N. Linking Adverse Childhood Experiences and Other Risk Factors to Subjective Cognitive Decline in an Aging Population. Prev Chronic Dis. 2023;20:E115. Published 2023 Dec 21. [CrossRef]
- Nilaweera D, Gurvich C, Freak-Poli R, et al. Adverse events in older adults and the risk of dementia and cognitive decline. J Affect Disord Rep. 2023;13:100592. [CrossRef]
- Zuelsdorff M, Sonnega A, Barnes LL, et al. Childhood and Adulthood Trauma Associate With Cognitive Aging Among Black and White Older Adults. Am J Geriatr Psychiatry. 2024;32(3):373-385. [CrossRef]
- Hu W, Zhang WB, Liu BP, Jia CX. Associations and Mediating Pathways Between Childhood Adversity and Risk of Dementia: A Cohort Study in the UK Biobank. J Gerontol A Biol Sci Med Sci. 2024;79(8):glae121. [CrossRef]
- Wang Y, Yu J, Yang Y, Chen S. Childhood Adversity and Cognitive Function Across Physical-Mental-Cognitive Health Trajectories: A 10-Year Longitudinal Study of Chinese Older Adults. Int J Geriatr Psychiatry. 2025;40(10):e70162. [CrossRef]
- Assuras S, Courtney K, Maxfield M, et al. Childhood maltreatment confers long-term risk for cognitive impairment: A prospective investigation. J Prev Alzheimers Dis. 2025;12(9):100303. [CrossRef]
- Choi EY, Cho G, Chang VW. Association of Early Life Risk Factors and APOE ε4 With Incident Dementia: Evidence From 14 Years of U.S. Data. J Am Geriatr Soc. 2025;73(11):3406-3415. [CrossRef]
- Li J, Ortí-Casañ N, Bayraktaroglu I, et al. Psychosocial stressors and cognitive function: An analysis using data from the English longitudinal study of ageing. J Prev Alzheimers Dis. 2025;12(8):100232. [CrossRef]
- Lian J, Crowe M, Anstey KJ, Kiely KM, Luisa Dávila A, Andel R. Impact of Childhood Adversity on Late-Life Cognition in Older Puerto Rican Adults. J Gerontol B Psychol Sci Soc Sci. 2025;80(4):gbae199. [CrossRef]
- Liu G, Bao T, Gao C, et al. The mediating role of life course cognitive reserve-enhancing factors in the relationship between adverse childhood experiences and dementia among older adults: evidence from a prospective cohort study in the United States. Soc Psychiatry Psychiatr Epidemiol. 2025;60(5):1063-1075. [CrossRef]
- Haapanen, M.J.; Perälä, M.M.; Salonen, M.K.; Kajantie, E.; Simonen, M.; Pohjolainen, P.; Pesonen, A.K.; Räikkönen, K.; Eriksson, J.G.; von Bonsdorff, M.B. Early life stress and frailty in old age: The Helsinki birth cohort study. BMC Geriatr. 2018, 18, 179. [CrossRef]
- Dos Santos Gomes C, Pirkle CM, Zunzunegui MV, et al. Frailty and life course violence: The international mobility in aging study. Arch Gerontol Geriatr. 2018;76:26-33. [CrossRef]
- Van Der Linden BWA, Sieber S, Cheval B, et al. Life-Course Circumstances and Frailty in Old Age Within Different European Welfare Regimes: A Longitudinal Study With SHARE. J Gerontol B Psychol Sci Soc Sci. 2020;75(6):1326-1335. [CrossRef]
- Li Y, Xue QL, Odden MC, Chen X, Wu C. Linking early life risk factors to frailty in old age: evidence from the China Health and Retirement Longitudinal Study. Age Ageing. 2020;49(2):208-217. [CrossRef]
- Ye C, Aihemaitijiang S, Wang R, Halimulati M, Zhang Z. Associations between Early-Life Food Deprivation and Risk of Frailty of Middle-Age and Elderly People: Evidence from the China Health and Retirement Longitudinal Study. Nutrients. 2021;13(9):3066. Published 2021 Aug 31. [CrossRef]
- Baranyi G, Welstead M, Corley J, et al. Association of Life-Course Neighborhood Deprivation With Frailty and Frailty Progression From Ages 70 to 82 Years in the Lothian Birth Cohort 1936. Am J Epidemiol. 2022;191(11):1856-1866. [CrossRef]
- Yan Y, Cai L, Lu N. Childhood experiences and frailty trajectory among middle-aged and older adults in China. Eur J Ageing. 2022;19(4):1601-1615. Published 2022 Nov 24. [CrossRef]
- Haapanen MJ, Jylhävä J, Kortelainen L, et al. Early-Life Factors as Predictors of Age-Associated Deficit Accumulation Across 17 Years From Midlife Into Old Age. J Gerontol A Biol Sci Med Sci. 2022;77(11):2281-2287. [CrossRef]
- Mian O, Anderson LN, Belsky DW, et al. Associations of Adverse Childhood Experiences with Frailty in Older Adults: A Cross-Sectional Analysis of Data from the Canadian Longitudinal Study on Aging. Gerontology. 2022;68(10):1091-100.
- Gao T, Han S, Mo G, Sun Q, Zhang M, Liu H. A positive association between hunger in childhood and frailty in old age: Findings from the Chinese longitudinal healthy longevity survey. Front Med (Lausanne). 2022;9:955834. Published 2022 Oct 24. [CrossRef]
- Wang Q. Association of Adverse Childhood Experiences With Frailty Index Level and Trajectory in China. JAMA Netw Open. 2022;5(8):e2225315. Published 2022 Aug 1. [CrossRef]
- Dimitriadis MM, Jeuring HW, Marijnissen RM, Wieringa TH, Hoogendijk EO, Oude Voshaar RC. Adverse Childhood Experiences and frailty in later life: a prospective population-based cohort study. Age Ageing. 2023;52(2):afad010. [CrossRef]
- Wang Q. Social contexts and cross-national differences in association between adverse childhood experiences and frailty index. SSM Popul Health. 2023;22:101408. Published 2023 Apr 17. [CrossRef]
- Yang G, Cao X, Yu J, et al. Association of Childhood Adversity With Frailty and the Mediating Role of Unhealthy Lifestyle: A Lifespan Analysis. Am J Geriatr Psychiatry. 2024;32(1):71-82. [CrossRef]
- Xian X, Ji X, Chen X, Cheng Y, Shen K. Exposure to the Chinese famine during early life increases the risk of frailty in adulthood: Evidence from the China Health and Retirement Longitudinal Study. Prev Med Rep. 2025;56:103145. Published 2025 Jun 18. [CrossRef]
- Ege MA, Messias E, Thapa PB, Krain LP. Adverse childhood experiences and geriatric depression: results from the 2010 BRFSS. Am J Geriatr Psychiatry. 2015;23(1):110-114. [CrossRef]
- Cheong EV, Sinnott C, Dahly D, Kearney PM. Adverse childhood experiences (ACEs) and later-life depression: perceived social support as a potential protective factor. BMJ Open. 2017;7(9):e013228. Published 2017 Sep 1. [CrossRef]
- Flores RJ, Campo-Arias A, Stimpson JP, Chalela CM, Reyes-Ortiz CA. The Association Between Past Sexual Abuse and Depression in Older Adults From Colombia. J Geriatr Psychiatry Neurol. 2018;31(1):13-18. [CrossRef]
- Iob E, Lacey R, Steptoe A. Adverse childhood experiences and depressive symptoms in later life: Longitudinal mediation effects of inflammation. Brain Behav Immun. 2020;90:97-107. [CrossRef]
- Yazawa A, Shiba K, Inoue Y, et al. Early childhood adversity and late-life depressive symptoms: unpacking mediation and interaction by adult socioeconomic status. Soc Psychiatry Psychiatr Epidemiol. 2022;57(6):1147-1156. [CrossRef]
- Guo X, Lin L, Qin K, Li J, Chen W, Guo VY. Adverse childhood experiences and depressive symptoms among middle-aged or older adults in China and the mediating role of short sleep duration. J Affect Disord. 2023;340:711-718. [CrossRef]
- Li S, Yin Y, Cui G, Zhang C, Zhu H, Yao Y. The mediating and moderating effects of resilience between childhood trauma and geriatric depressive symptoms among Chinese community-dwelling older adults. Front Public Health. 2023;11:1137600. Published 2023 Apr 14. [CrossRef]
- Dai Q, Li M, Wang Z, Xu Q, Zhang X, Tao L. The Mediating Effects of Chronic Diseases in the Relationship Between Adverse Childhood Experiences and Trajectories of Depressive Symptoms in Later Life: A Nationwide Longitudinal Study. Healthcare (Basel). 2024;12(24):2539. Published 2024 Dec 16. [CrossRef]
- Babatunde OA, Ramkumar SP, Nguyen SA, et al. Association between number of Adverse Childhood Experiences and depression among older adults is moderated by race. Prev Med. 2024;181:107921. [CrossRef]
- Jiang F, Guan X, Zhu Z, Liu N, Gu H, Li X. The relationship between self-reported adverse experiences and depressive symptoms among middle-aged and elderly individuals: A longitudinal study based on the CHARLS database. Brain Behav Immun Health. 2025;46:101001. Published 2025 Apr 22. [CrossRef]
- Rhee MK, Park J, Park H, Lee I, Jang Y. Adverse Childhood Experiences and Depressive Symptoms Among Older Korean Americans in Affordable Senior Housing: The Moderating Role of Social Networks and Family Solidarity. J Appl Gerontol. Published online December 29, 2025. [CrossRef]
- Li Q, Chen L, Wang R. Adverse childhood experiences and late-life depression in older Chinese adults: the mediating roles of sleep disorders and chronic diseases. BMC Geriatr. 2025;25(1):1076. Published 2025 Dec 30. [CrossRef]
- Dai S, Dai C, Wang D, Ma S. Adverse childhood friendship and depression in later life: findings from a national survey in China. BMC Psychol. 2025;13(1):856. Published 2025 Aug 1. [CrossRef]
- Chen W, Sheng Y, Wang W, Wang X. Associations between exposure to adverse childhood experiences and depressive symptom trajectories among Chinese middle-aged and older adults: A nationally representative cohort study. J Affect Disord. 2026;394(Pt B):120654. [CrossRef]
- Reyes-Ortiz CA, Ocampo-Chaparro JM, Campo-Arias A, Holmes H, Halphen J. Association Between History of Abuse and Falling in Older Adults. J Am Geriatr Soc. 2018;66(8):1603-7. [CrossRef]
- Reyes-Ortiz CA, Perez-Zepeda MU, Ocampo-Chaparro JM, et al. Polyvictimization and Recurrent Falling in Older Ecuadorian Adults: The Mediation Role of Depressive Symptoms. J Aging Health. 2021;33(1-2):27-38. [CrossRef]
- Reyes-Ortiz CA, Campo-Arias A, Ocampo-Chaparro JM, Moncayo-Hernández BA, Lee T, Luque JS. The Association Between Discrimination and Falling. A National Sample of Older Adults. J Aging Health. 2022;34(4-5):614-25. [CrossRef]
- Reyes-Ortiz CA, Robinson CC, Williams DR, et al. Perceived Ageism is Associated With Recurrent Falling Among Older Colombian Adults. J Appl Gerontol. 2024;43(9):1343-1354. [CrossRef]
- Huang R, Li S, Hu J, et al. Adverse childhood experiences and falls in older adults: The mediating role of depression. J Affect Disord. 2024;365:87-94.
- Tan H, Liu M, Ren H, Zhou J, Guo Y, Jiang X. Associations of Adverse Childhood Experiences with Falls and Fall Risk Factors Among Middle-Aged and Older Adults in China. Am J Prev Med. Published online February 11, 2025. [CrossRef]
- Reyes-Ortiz CA, Payan-Villamizar CM, Campo-Arias A, et al. Adverse childhood experiences are associated with falling among middle-aged and older adults, US 2023 BRFSS data. J Gerontol A Biol Sci Med Sci. 2026;81(3):glaf292. [CrossRef]
- Cheval B, Boisgontier MP, Orsholits D, et al. Association of early- and adult-life socioeconomic circumstances with muscle strength in older age. Age Ageing. 2018;47(3):398-407. [CrossRef]
- Cheval B, Chabert C, Sieber S, et al. Association between Adverse Childhood Experiences and Muscle Strength in Older Age. Gerontology. 2019;65(5):474-484. [CrossRef]
- Smith NR, Ferraro KF, Kemp BR, Morton PM, Mustillo SA, Angel JL. Childhood Misfortune and Handgrip Strength Among Black, White, and Hispanic Americans. J Gerontol B Psychol Sci Soc Sci. 2019;74(3):526-535. [CrossRef]
- Lin L, Sun W, Lu C, Chen W, Guo VY. Adverse childhood experiences and handgrip strength among middle-aged and older adults: a cross-sectional study in China. BMC Geriatr. 2022;22(1):118. Published 2022 Feb 12. [CrossRef]
- Huang R, Li Y, Ma C, et al. Adverse childhood experiences, sarcopenia, and social participation in older adults: a cohort study. BMC Public Health. 2024;24(1):711. Published 2024 Mar 5. [CrossRef]
- Dimitriadis MM, Kokkeler KJE, Hoogendijk EO, et al. Adverse Childhood Experiences and Sarcopenia in Later Life: Baseline Data from the Canadian Longitudinal Study on Aging. Geriatrics (Basel). 2025;10(4):111. Published 2025 Aug 15. [CrossRef]
- Vásquez E, Quiñones A, Ramirez S, Udo T. Association Between Adverse Childhood Events and Multimorbidity in a Racial and Ethnic Diverse Sample of Middle-Aged and Older Adults. Innov Aging. 2019;3(2):igz016. Published 2019 Jun 29. [CrossRef]
- Lin L, Wang HH, Lu C, Chen W, Guo VY. Adverse Childhood Experiences and Subsequent Chronic Diseases Among Middle-aged or Older Adults in China and Associations With Demographic and Socioeconomic Characteristics. JAMA Netw Open. 2021;4(10):e2130143. Published 2021 Oct 1. [CrossRef]
- Reyes-Ortiz CA, Lee T, Campo-Arias A, Ocampo-Chaparro JM, Luque JS. Racial Discrimination and Multimorbidity Among Older Adults in Colombia: A National Data Analysis. Prev Chronic Dis. 2023;20:E34. Published 2023 May 4. [CrossRef]
- Chandrasekar R, Lacey RE, Chaturvedi N, Hughes AD, Patalay P, Khanolkar AR. Adverse childhood experiences and the development of multimorbidity across adulthood-a national 70-year cohort study. Age Ageing. 2023;52(4), afad062. [CrossRef]
- Liu Y, Dai W, Yang Y, et al. Adverse childhood experiences and multimorbidity among middle-aged and older adults: Evidence from China. Child Abuse Negl. 2024;158:107100. [CrossRef]
- Guedes DT, Vafaei A, Alvarado BE, et al. Experiences of violence across life course and its effects on mobility among participants in the International Mobility in Aging Study. BMJ Open. 2016;6(10):e012339. Published 2016 Oct 13. [CrossRef]
- Amemiya A, Fujiwara T, Murayama H, Tani Y, Kondo K. Adverse Childhood Experiences and Higher-Level Functional Limitations Among Older Japanese People: Results From the JAGES Study. J Gerontol A Biol Sci Med Sci. 2018;73(2):261-266. [CrossRef]
- Li J, Lin S, Yan X, Pei L, Wang Z. Adverse Childhood Experiences and Trajectories of ADL Disability among Middle-Aged and Older Adults in China: Findings from the CHARLS Cohort Study. J Nutr Health Aging. 2022;26(12):1034-1041. [CrossRef]
- Lee VM, Hargrave AS, Lisha NE, Huang AJ. Adverse Childhood Experiences and Aging-Associated Functional Impairment in a National Sample of Older Community-Dwelling Adults. J Gen Intern Med. 2023;38(15):3362-3371. [CrossRef]
- Wang B, Dong X. Life Course Violence: Child Maltreatment, IPV, and Elder Abuse Phenotypes in a US Chinese Population. J Am Geriatr Soc. 2019;67(S3):S486-S492. [CrossRef]
- Payne CF, Mall S, Kobayashi L, Kahn K, Berkman L. Life-Course Trauma and Later Life Mental, Physical, and Cognitive Health in a Postapartheid South African Population: Findings From the HAALSI study. J Aging Health. 2020;32(9):1244-1257. [CrossRef]
- Lv S, Shen Z, Zhang H, et al. Association between exposure to the Chinese famine during early life and the risk of chronic kidney disease in adulthood. Environ Res. 2020;184:109312. [CrossRef]
- Dorji N, Dunne M, Deb S. Adverse childhood experiences: association with physical and mental health conditions among older adults in Bhutan. Public Health. 2020;182:173-178. [CrossRef]
- Sheffler JL, Burchard V, Pickett S. Adverse Childhood Experiences and Poor Sleep Quality in Older Adults: The Influence of Emotion Regulation. J Gerontol A Biol Sci Med Sci. 2023;78(10):1919-1924. [CrossRef]
- Ren Z, Du Y, Lian X, Luo Y, Zheng X, Liu J. Bidirectional longitudinal associations between depressive symptoms and somatic conditions after adverse childhood experiences in middle-aged and older Chinese: A causal mediation analysis. Soc Sci Med. Published online October 27, 2023. [CrossRef]
- Li J, Sun Q, Zhang H, et al. Depressive symptoms mediate associations of adverse childhood experiences and chronic lung diseases: A mediation effect analysis. J Affect Disord. 2024;345:342-348. [CrossRef]
- He SY, Zhang WS, Jiang CQ, et al. Association of adverse childhood experiences with anemia in older Chinese: Guangzhou Biobank Cohort Study. Sci Rep. 2024;14(1):4729. Published 2024 Feb 27. [CrossRef]
- Cheng C, Wang X, Huang M, He J, Li Y, Cui Y. The association between adverse childhood experiences and chronic respiratory diseases in the Chinese middle-aged and elderly population. BMC Public Health. 2025;25(1):2942. Published 2025 Aug 27. [CrossRef]
- Jin Y, Lin H. Relationship between adverse childhood experiences and musculoskeletal pain: the mediating effect of depressive symptoms. BMC Psychol. 2025;13(1):854. Published 2025 Aug 1. [CrossRef]
- Chen B, Xue E, Li Y, et al. Life-Course Psychosocial Stress and Risk of Dementia and Stroke in Middle-Aged and Older Adults. JAMA Netw Open. 2026;9(1):e2556012. Published 2026 Jan 2. [CrossRef]
- Karhu E, Gurland B, Craven MR, Lamothe D, Neshatian L. Psychological Symptoms and Adverse Childhood Experiences Negatively Impact Coexisting Obstructed Defecation and Fecal Incontinence Symptoms. Neurogastroenterol Motil. 2026;38(1):e70243. [CrossRef]
- Krieger N. Theories for social epidemiology in the 21st century: an ecosocial perspective. Int J Epidemiol. 2001;30(4):668-677. [CrossRef]
- Suominen H, Heikkinen E. A life course approach to physical activity, health, and aging. Eur Rev Aging Phys Act 2011;8(1): 5–6. [CrossRef]
- Wagner C, Carmeli C, Jackisch J, et al. Life course epidemiology and public health. Lancet Public Health. 2024;9(4):e261-e269. [CrossRef]
- Champagne FA, Courtney K, Bellatin A, Miller M, Widom CS. Association between childhood maltreatment, stressful life events and hair cortisol concentration in late midlife: A prospective investigation. Psychoneuroendocrinology. 2025;180:107561. [CrossRef]
- Fleck L, Buss C, Bauer M, et al. Early-Life Adversity Predicts Markers of Aging-Related Neuroinflammation, Neurodegeneration, and Cognitive Impairment in Women. Ann Neurol. 2025;97(4):642-656. [CrossRef]
- Herzog JI, Schmahl C. Adverse Childhood Experiences and the Consequences on Neurobiological, Psychosocial, and Somatic Conditions Across the Lifespan. Front Psychiatry. 2018;9:420. Published 2018 Sep 4. [CrossRef]
- Godoy LC, Frankfurter C, Cooper M, Lay C, Maunder R, Farkouh ME. Association of Adverse Childhood Experiences With Cardiovascular Disease Later in Life: A Review. JAMA Cardiol. 2021;6(2):228-35. [CrossRef]
- Brownell P. Trauma Theory and Abuse, Neglect and Violence Across the Life Course. J Interpers Violence. 2024;39(19-20):4041-4064. [CrossRef]
| Authors | Design, population | Exposures | Geriatric Syndromes or other Outcomes | Main outcome’s findings | Additional comments |
|---|---|---|---|---|---|
| Donley, 2018, Finland | Longitudinal cohort, 2,682 men aged ≥42; KIHD | Childhood stress (interviews 1984 & 1989) included living in custody or an orphanage, experience of crisis in childhood, having problems with teachers and emigrating because of war | Incident cases of dementia, including AD, were obtained through 2014 via national health register linkages | Childhood stress was associated with increased risk of dementia (HR=1.93 95% CI 1.14–3.25). |
|
| Conde-Sala, 2020, Spain | Case–control study 65, 997 participants aged ≥50; SHARE | Parental death (mother or father or both) at the age of ≤16 years (childhood & adolescence) | Diagnosis of dementia by a doctor | Early parental death associated with increased odds of dementia (OR=1.50 95% CI 1.31-1.72). | |
| Tani, 2020, Japan | Longitudinal (3-yr follow-up), 17,412 participants aged ≥65; JAGES | ACEs 7 questions, before the age of 18 interpersonal loss (parental loss or parental divorce), family psychopathology (parental mental illness or family violence), and abuse and neglect (physical abuse, psychological neglect, or psychological abuse). |
Dementia was defined as level II or higher on the dementia scale (it corresponds to a 16-point rating on the Mini-Mental State Examination) | Participants who experienced ≥3 ACEs (vs. 0) had a greater risk of developing dementia (HR= 1.78 95% CI 1.15-2.75; p=.009) | |
| Grainger, 2020, Australia | Cohort study, 484 older adults (mean age 83.4 SD=4.3); Sydney MAS | Early-life stress (ELS), using the short form of the Childhood Trauma Questionnaire, including 28 items and assesses 5 traumatic experiences: physical abuse, physical neglect, sexual abuse, emotional abuse, emotional neglect. | Global cognitive function measured by 10 neuropsychological tests with domains: attention/processing speed, memory, language, visuospatial reasoning, and executive functioning. (standardized z-scores) |
Global cognition was poorer in those who had experienced physical neglect (mean − .48 SD=1.04) relative to those who had not (mean − .14 SD=1.08; p<.05). | |
| Yang, 2020, China | Longitudinal, 16,258 participants aged ≥45; CHARLS | Early-life conditions (ELCs): early parental death, childhood SES, food deprivation, and childhood health | Cognitive function was assessed using episodic memory (immediate & delayed recall) & mental intactness (serial subtractions, date, day of week, redraw a picture) |
Early maternal death was associated with lower cognitive function among middle- and old-aged Chinese adults (β range between -0.44 & -0.35, p<0.05). Less healthy participants during childhood had lower cognitive performance than those who had enjoyed good health (β range between -0.36 and -0.14, p < 0.05). | |
| Nilaweera, 2020, France | Longitudinal (14 years), 1,700 participants aged ≥65, ESPRIT study cohort | Lifetime and current post-traumatic stress disorder (PTSD) diagnoses were assessed using the Watson’s PTSD Inventory | Cognitive tests assessed global cognition, visual memory, verbal fluency, psychomotor speed and executive function. Incident dementia (Dx DSM-IV criteria) |
Men with lifetime PTSD or without re-experiencing symptoms had lower odds for low global cognition (OR=0.61 95% CI 0.42-0.88; and OR=0.57 95% CI 0.38-0.84, respectively). Women with re-experiencing symptoms had higher odds for low global cognition (OR=1.46 95% CI 1.03-2.09) | Women without re-experiencing symptoms had lower risk for dementia (HR=0.49 95% CI 0.29-0.80) |
| Macpherson, 2021, UK | Retrospective cohort study 56,082 people age 55.5 (±7.7); UK Biobank | Childhood maltreatment (CM) included physical abuse, physical neglect, emotional abuse, emotional neglect, and sexual abuse | Incident dementia diagnosis – AD-8: The Washington University Dementia Screening Test |
CM was associated with dementia (HR = 1.32, 95% CI 1.02–1.71) | |
| Gold, 2021, USA | Cohort 1,661 participants aged ≥65; KHANDLE | ACEs (score 0-9) before age 16: parents divorced; parents remarried. Domestic violence; family member substance abuse or serious illness, parent loss of a job; parent was at jail, death of mother or father. | Spanish and English Neuropsychological Assessment Scales (SENAS) including Verbal episodic memory, semantic memory and executive function |
ACEs associated with worse cognition: parent remarried (β= -0.11 95% CI -0.20 to -0.03); death of mother (β= -0.18 95% CI -0.30 to -0.07); and death of a father (β= -0.11 95% CI -0.20 to -0.01) | ACEs total score was not associated with cognition |
| Lin, 2022, China | Prospective cohort, 6,466 participants mean age 57.2 (SD=8.3); CHARLS | ACEs 10 types, 5 threat-related adversities (i.e., physical abuse, household substance abuse, domestic violence, unsafe neighborhood, bullying) and 5 deprivation-related adversities (i.e., emotional neglect, household mental illness or someone incarcerated, parental divorce or death) | Episodic memory (immediate & delayed recall (score 0-10). Executive function (orientation, calculation & visuospatial ability (score 0-11). Global cognition (total score 0-21) | Participants with ≥2 childhood deprivations had faster cognitive decline in all cognitive tests (β=−0.035 [95% CI −0.050 to−0.019] SD/y for global cognition; β=−0.047 [95% CI −0.068 to −0.025] SD/y for episodic memory; β=−0.019 [95% CI −0.034 to−0.004] SD/y for executive function) | |
| Nilaweera, 2022, France | Longitudinal (14 years), 1,562 participants aged ≥65, ESPRIT study cohort | 25-item questionnaire to assess ACEs, using a modified version of the Childhood Trauma Questionnaire | Cognitive tests assessed global cognition, visual memory, verbal fluency, psychomotor speed (PS) and executive function. Incident dementia (Dx DSM-IV criteria) | At baseline, women having ≥5 ACEs (vs. 0-2) had poor psychomotor speed (OR= 1.52 95% CI 1.07–2.17). Also, participants with 3-4 ACEs (vs. 0-2) had worse verbal fluency (OR= 1.34 95% CI 1.00–1.78). Early abuse/maltreatment & poverty/financial difficulties were associated with worse PS. | No associations with incident dementia. |
| Stebbins, 2022, USA | Longitudinal cohort, 7,785 participants aged ≥65; HRS | Life course (ever have or been) traumatic events (TEs), e.g., child ever died? ever been in a major fire, flood, earthquake, or other natural disaster? ever have a life-threatening illness or accident? Before age 18: did your parents drink or use drugs? ever physically abused by parents? | Telephone Interview for Cognitive Status (HRS-TICS) | ≥1 TEs over the life course was associated with accelerated cognitive decline (β=−0.05 95% CI: −0.07, −0.02) HRS-TICS units/year; 1 vs. 0 events) compared to experiencing no events. | Experiencing TEs was associated with better cognitive function cross-sectionally. No associations with incident dementia. |
| Voyer, 2023, USA | Cross-sectional, 17,042 participants aged ≥45; 2020 BRFSS | Eleven questions for ACEs were converted to a summed ACEs score. As binary variable was used ≥2 ACEs vs. 0-1. | Subjective cognitive decline (SCD): had confusion or memory loss more often or is getting worse in the past 12 months | Having ≥2 ACEs (vs. 0-1) increased the odds of SCD (OR= 1.69 95% CI 1.36–2.10). | |
| Nilaweera, 2023, Australia | Longitudinal 12,789 participants aged ≥ 70; ASPREE & ALSOP studies |
Ten adverse life events related to interpersonal relationships (e.g. death of a spouse), finances (e.g. major money problems) and external factors (e.g. major accident or disaster). |
Cognitive tests assessed global cognition, episodic memory, delayed recall, executive function, and psychomotor speed. Incident cognitive decline: those whose cognitive score dropped by >1.5 SD. Diagnosis of dementia - DSM-IV criteria. | Experiencing death of a spouse/partner (HR=1.72 95% CI 1.17-2.52) and major financial problems (HR=1.53 95% CI 1.05-2.23) were associated with increased risk of dementia. Experiencing financial problems was associated with increased risk cognitive decline in men (HR=1.43 95% CI 1.10-1.86). | Women with some events (e.g. close family or friends lost their job/retired) had low risk of dementia (HR=0.62 95% CI 0.40-0.95). |
| Zuelsdorff, 2024, USA | Longitudinal cohort, 13,952 participants aged ≥55; HRS | Childhood traumatic events (CT): 4 items about childhood (e.g., before you were 18 years old, were you ever physically abused by parents?). 7 events at any age (e.g., Were you the victim of a serious physical attack or assault in your life?) and, if yes, the year that occurred. CT index (0-11). | 27-item adapted Telephone Interview on Cognitive Status (TICS) | In White participants only (n=11,607), greater childhood trauma exposure predicted worse baseline cognition (global cognition, immediate recall & delay recall) but slower change over time (global cognition & immediate recall). | |
| Hu, 2024, UK | Longitudinal, cohort, mean age 55.9 (7.7) at baseline; UK Biobank (n=150,152) | Childhood adversity (CA) (abuse or neglect; childhood trauma screen, 0-5) | Dementia (ICD-10 diagnosis) | Individuals with any CA had a greater risk for dementia (OR=1.30, 95% CI 1.13-1.50) compared with those who did not experience CA | Depression, smoking, low grip strength & biomarkers were mediators. |
| Wang, 2025, China | Longitudinal, 6,178 participants aged ≥60; CHARLS | 13 childhood adversities (before age 17), e.g., parental death, anxiety, depression, mental illness, disability, drug abuse, alcohol abuse, or involvement in criminal activities; a bedridden parent, lack of affection, neglect, physical abuse, domestic violence | Cognitive function evaluation comprises of an episodic memory test (immediate & delayed recall) and an attention test (total score 0-25) | Multiple childhood adversities were associated with lower cognitive function (β = -0.36, 95% CI [-0.58, -0.14]). Participants having rapid cognitive decline with moderate physical-mental deterioration, experiencing 2 childhood adversities predicted lower cognitive function (β = -0.88, 95% CI [-1.62, -0.14]). | |
| Assuras, 2025, USA | Prospective cohort, n=908 (matched control group, n=667); New York | Documented physical, sexual abuse or neglect; 5 interviews from mean age 29.2 to 59.4 | Comprehensive neuropsychological assessment battery (12 tests). Cognitive impairment with no dementia (CIND) or dementia | Maltreated individuals in both age groups were at increased risk for CIND [older (> age 59, OR=2.08 95% CI 1.52–2.86) and younger (< age 59, OR=1.63 95% CI 1.11–2.40)], compared to controls | Any maltreatment was associated with amnestic or non-amnestic CIND |
| Choi, 2025, USA | Cohort of 8,678 dementia-free participants aged ≥60 and <90 at baseline with data on APOE genotype; HRS |
Retrospective early life conditions (deficits in financial, social, and human capital, as well as poor childhood health) | Dementia incidence (Langa–Weir Classification of Cognitive Function) | Low social capital (cause-specific HR=1.24 95% CI 1.06–1.45), low human capital (HR=1.46, 95% CI 1.29–1.66), and any health conditions (HR=1.12 95% CI 1.01–1.25) were also associated with greater risk of dementia. | |
| Li, 2025, UK | Cohort of 10,893 participants aged ≥50 at baseline; ELSA | Psychosocial stressors (financial strain, caregiving, disability & limiting long-term illness) | Cognitive function: an overall global cognition score and domains scores (memory, executive function & orientation) | Multiple stressors were associated with steeper declines in global cognition (B= -0.34, 95% CI [-0.45, -0.23], p<.001), memory (B= -0.12, 95% CI [-0.16, -0.07], p<.001), and executive function (B= -0.20, 95% CI [-0.29, -0.11], p<.001). | |
| Lian, 2025, Puerto Rico | Longitudinal, cohort of, 3,713 participants aged ≥60; PREHCO | Childhood adversity, 13 items (e.g., household SES, indicators of health status during early childhood and adoles cence, neighborhood characteristics) | Mini-mental Cabán (MMC) (score 0-20) for global cognition. Cognitive impairment was defined as MMC scores 1.5 SD or more below the expected score at baseline | At baseline, parental illiteracy (β= −0.35, p<.001), neigh borhood disadvantage (β=−0.27, p< .001), economic hardship (β=−0.10, p= .003) and childhood illness (β =−0.21, p < .001) were associated with MMC scores. Neighborhood disadvantage was associated with incident cognitive impairment (OR=1.19 95% CI 1.06-1.34, p= .003). | Depression and self-rated health were mediators |
| Liu, 2025, USA | Longitudinal, cohort of, 51,327 participants aged ≥50; HRS | ACEs include 6 questions from two dimensions: financial adversity and traumatic events (total score 0-6) | Dementia was evaluated using the modified Telephone Interview for Cognitive Status (TICS-m) | ACEs were associated with increased risk of dementia (HR= 1.08 95% CI 1.02-1.16) | The association of ACEs and dementia was fully mediated by early-life stage cognitive reserve enhancing factor and partially mediated by adulthood enhancing factor |
| Authors | Design, population | Exposures | Geriatric Syndromes or other Outcomes | Main outcome’s findings | Additional comments |
|---|---|---|---|---|---|
| Haapanen, 2018, Finland | Cases (n=117; separated age 72.8 SD=2.6)- controls (n=855 non-separated 70.5 SD=2.5); HBCS | Early life stress (Wartime parental separation- World War II) | Frailty at a mean age of 71 years (Fried’s criteria: weight loss, low p. activity, exhaustion, weakness and slowness between 2011 and 2013) | Compared to the non-separated men, men who had been separated had an increased frailty (RRR= 5.18 95% CI 1.16- 23.17 p=0.031). | No associations were observed among women |
| dos Santos Gomes, 2018, Albania, Brazil, Canada, Colombia | Cross-sectional 2,002 participants aged 65-74; IMIAS | Physical abuse during the first 15 years of life. Adulthood domestic physical and psychological violence by family and intimate partners was assessed by the Hurt, Insult, Threaten and Scream (HITS) scale. | Frailty (Fried’s phenotype) | Childhood physical abuse (OR=1.68; 95% CI 1.01-2.78) and psychological violence by their intimate partner (OR= 2.07 95% CI 1.37-3.12) were associated with frailty. | |
| Van Der Linden, 2020; Switzerland | Longitudinal cohort 23,358 participants aged ≥50; SHARE | ACEs score 0-7: e.g., child in foster care, parental (death, mental illness, or drinking abuse), period of hunger, and property taken away (≥1 vs. 0). Adverse childhood health experiences (ACHE) e.g., serious health conditions, long hospitalization, (≥1 vs. 0) | Frailty (Fried’s phenotype): weakness, shrinking, exhaustion, slowness, or low activity. | Participants having at least one ACE (OR=1.30 95% CI 1.14–1.48 p< .001) or one ACHE (OR=1.40 95% CI 1.24–1.57 p< .001) had increased odds for frailty. | |
| Li, 2020; China | Longitudinal cohort 6,806 participants aged ≥60; CHARLS | Early life risk factors (childhood or adolescence): starvation, domestic violence, neighborhood quality [0 (lowest) to 4 (highest)], childhood health status (e.g. healthier vs. same as others) | Frailty: slowness, weakness, exhaustion, inactivity and weight loss (robust, pre-frail, frail) | Better childhood neighborhood quality (e.g., 4 vs. 0, OR= 0.28 95% CI 0.15-0.52) and better childhood health status (OR=0.74 95% CI 0.57-0.96) had lower risk of being frail. Childhood severe starvation was associated with higher risk of prefrailty (OR=1.30 95% CI 1.04-1.62) | |
| Ye, 2021; China | Longitudinal cohort 11,615 individuals aged ≥45; CHARLS | Childhood food deprivation (CFD): not enough food to eat before age 12, or born & brought in famine affected areas and in famine periods in China. | FRAIL scale as a sum of fatigue, resistance, ambulation, illness, and loss of weight (robust, pre-frail, frail) | CFD was associated with frailty at old age (OR=1.30 95% CI 1.26–1.36). Those with extreme CFD (vs. mild CFD) had increased risks of frailty (OR=1.34 95% CI 1.26–1.43). | |
| Baranyi, 2022, UK | Longitudinal cohort, 363 participants, mean age 69.3 (SD=0.74); LBC1936 | Neighborhood social deprivation (NSD): population density, overcrowding, infant mortality, households renting; male unemployment, overcrowding, car ownership, and social class. During childhood, young adulthood, late adulthood | Frailty Index (FI) 30 deficits covering physical, psychological, and cognitive systems | Among males, greater accumulated NSD was associated with higher FI at baseline (B= 0.017, 95% CI 0.005, 0.029). Among females, in late adulthood, higher NSD was associated with widening frailty trajectories (B= 0.005, 95% CI 0.0004, 0.009). | |
| Yan, 2022, China | Longitudinal cohort 10,963 participants aged ≥45; CHARLS | ACEs (before age 17) with 17 indicators criteria; including abuse and neglect, death, illness/disability, living environment outside the home -score 0 to 1, with greater values indicating severe ACEs), childhood SES, and childhood health and health care | Frailty Index (32 deficits in six domains were chosen to construct FI, score 0 to 1) | ACEs, childhood SES, and indicators of childhood health and health care were associated with baseline & change rate of FI. ACEs (B= 0.018 [SE=0.004], p< 0.001), self-rated childhood SES (−0.001 [0.000], p < 0.01), and objective health & health care (−0.002 [0.001], p< 0.05) were significantly associated with the slope (latent growth curve, FI trajectory). | |
| Haapanen, 2022, Finland | Longitudinal cohort 2,000 participants aged 57-84 years (3 waves over 17 years); HBCS | Early life stress (Wartime parental separation- World War II) | Frailty Index (FI) assessed at a mean age of 57 years (Fried’s criteria). | Women separated from their parents during War had steeper increase in FI levels percentage point differences of change per year from midlife to old age (B=0.211 95% CI 0.009, 0.414; p=0.041) | No associations were observed among men |
| Mian, 2022, Canada | Cross-sectional, 23,354 people aged 45–85 years; CLSA | ACEs (8-item) (3 abuse types; neglect; parental divorce, mental illness or death; intimate partner violence) | Frailty Index (76-item FI) (based on Rockwood & Mitnitski) | ACEs was associated with FI (≥3 ACEs vs. 0, B=0.04 [95% CI 0.037, 0.044] | |
| Gao, 2022, China | Cross-sectional, 7,342 participants aged ≥65; CLHLS | Childhood hunger (CH) “Did you often go to bed hungry as a child?” “yes” or “no.” |
Frailty Index (FI) using 44 health deficits, including daily life events, chronic illness, and psychological functioning | Childhood hunger was associated with frailty among participants 65–79 years (OR=1.21 95% CI 1.03–1.43), | CH at aged ≥80 years had lower odds of frailty (OR= 0.80 95% CI 0.65–0.98). |
| Wang, 2022, China | Longitudinal, 11,568 participants aged ≥45, CHARLS | ACEs covering childhood intrafamilial aggression, family dynamics, SE deprivation, loss or threat of loss within the family, and neighborhood quality (score 0-18) | Frailty Index (FI) using 41 measures. Robust, prefrail, and frail, with its trajectories classified as stable at robust and prefrail and rapidly rising to frail. | An increased number of ACEs was associated with a frail status (OR=1.20 95% CI 1.16-1.23) and being in the rapidly rising trajectory (OR=1.19 95% CI 1.16-1.23) . | |
| Dimitriadis, 2023, the Netherlands | Baseline (n=2,176) aged 58-89, longitudinal 17-yr follow-up (n=1,427); LASA | ACEs: War experiences, a parent death, excessive alcohol use of a relative, sexual abuse, severe problems at home, parents’ poverty, physical illness of respondent | LASA-FI (score 0.0 to 0.7; Frailty is defined as an FI ≥0.25) | ACE and frailty were associated at baseline (OR=1.88 95% CI 1.46–2.42; p<.001) and at follow-up (HR=1.28 95% CI 1.01–1.64; p=0.044) | |
| Wang, 2023, China | Longitudinal 43,928 participants from Europe (aged ≥50) and China (aged ≥45); SHARE and CHARLS | ACEs covering childhood intrafamilial aggression, family dynamics, SE deprivation, loss or threat of loss within the family, and neighborhood quality (score 0-18) | Frailty Index (FI) using 35 attributes (score 0 to 1). Robust (FI ≤0.10), prefrail (FI >0.10 & <0.25), and frail (FI ≥0.25). Trajectories: stable at robust and prefrail, rapidly increasing to frail. | ACEs>3 (vs. ≤1) was associated with a frail status for men (OR=1.69 95% CI 1.36-2.09) and women (OR=2.38 95% CI 1.93-2.94). ACEs>3 (vs. ≤1) was associated with FI trajectory of rapidly rising to frail for men (OR=1.70 95% CI 1.24-2.34) and women (OR=2.12 95% CI 1.70-2.63). | |
| Yang, 2024, UK | Cross-sectional, 152,914 adults aged 40–69; from the UK Biobank | Childhood adversity 5-item: physical neglect, emotional neglect, sexual abuse, physical abuse, and emotional abuse | Frailty index (FI) using 49 items covering sensory, cranial, mental well-being, infirmity, cardiometabolic, musculoskeletal, immunological, cancer, pain, and gastrointestinal. A frailty score (0 to 1) | Childhood adversity was associated with frailty (OR=1.38 95% CI 1.36-1.40; p<.001) | Unhealthy lifestyle score (0–5; (calculated based on BMI, smoking, alcohol, activity, and diet) was a mediator |
| Xian, 2025, China | Cross-sectional (2017-2018), 4,473 participants with mean age 60.0 ± 5.4; CHARLS | Famine fetal exposed (1959–1962), preschool exposed (1954–1957), and school-aged (1950–1953), compared to the non-exposed group (1964–1967). | Frailty index (FI) based on 32 items for disability, comorbidity, physical functioning, cognitive impairment, and depressive symptoms. Robustness (FI ≤0.10), prefrailty (FI >0.10 & <0.25), and frailty (FI ≥0.25) | There was a significant increase in the probability of frailty compared to the non-exposed group in fetal (OR=2.84 95% CI 1.73-4.65) and school-age (OR=2.81 95% CI 1.73-4.57) exposed groups. |
| Authors | Design, population | Exposures | Geriatric Syndromes or other Outcomes | Main outcome’s findings | Additional comments |
|---|---|---|---|---|---|
| Ege, 2015, USA | Cross-sectional, 8,051 participants aged ≥60; 2010 BRFSS data | ACEs: parents being physically abusive to each other, being physically harmed by a parent, being sworn at by the parent, being touched sexually by an adult, being forced to sexually touch an adult, and being forced into a sexual encounter | Depressive symptoms 8-item the Patient Health Questionnaire (PHQ). Depression a score of ≥10. |
A single occurrence of being forced sexual intercourse was associated with late-life depression (OR=2.92 95% CI 1.06-8.02). Repeated physical abuse and repeated forced sexual intercourse were also associated with depression (OR=2.94 95% CI 1.68-5.13; OR=3.66 95% CI 1.01-13.2, respectively). | |
| Cheong, 2017; Ireland | Cross-sectional, 2047 participants aged 50–69; Livinghealth Clinic Mitchelstown (Ireland) 2010–2011 cohort | ACEs (0-10), three domains: abuse, neglect, household dysfunction. Perceived social support (Oslo Social Support Scale), scores 3-8 (poor, 9-11 (moderate) and 12-14 (strong) |
CES-D scale (0-60) ≥16 was defined as having depressive symptoms |
Among individuals reporting poor perceived social support (PSS), having any ACE (vs 0) was associated with depressive symptoms (OR=2.85 95% CI 1.64- 4.95). Among those reporting moderate PSS, the odds were OR=2.21 95% CI 1.52-3.22 | |
| Flores, 2018, USA | Cross-sectional, 2,000 participants aged ≥60; SABE Bogota | History of sexual abuse (ever being a victim) | Using the GDS (score 0-15), depression was defined as score ≥6 | Higher odds of depression for past sexual abuse (OR= 3.91 95% CI 2.13-7.16) | Being displaced by violence was also associated with depression |
| Iob 2020; England | Longitudinal, 4,382 participants aged ≥50; ELSA | ACEs before age 16, with 4 dimensions: threat (e.g., abuse), household dysfunction (e.g., parent mental illness or substance abuse), low parental bonding (maternal and paternal), and loss experiences (e.g., parent death, or foster care). |
Depressive symptoms were assessed using the 8-item Centre for Epidemiological Studies Depression scale (CESD-8) (score 0-8) |
Greater ACEs cumulative exposure was associated with higher depressive symptoms at baseline (β=0.149 95% CI 0.115–0.183) and with their increase over time (β=0.355 95% CI 0.184–0.526). |
Higher baseline CRP levels mediated ACEs cumulative exposure on the baseline value and change in depressive symptoms. |
| Yazawa, 2022; Japan | Cohort, 7,271 participants aged ≥65; JAGES | ACEs, having ≥ 2 experiences: parental loss, parental divorce, parental mental illness, domestic violence, physical abuse, psychological neglect or abuse, & economic disadvantage | Depressive symptoms were assessed using the GDS-15. Score 0-15; or ≥5 mild-to-severe depressive symptoms |
ACEs was associated with greater depressive symptoms after adjusting for low adult SES (B= 0.41 95% CI 0.25–0.57). | Adult SES was a mediator |
| Guo, 2023; China | Cross-sectional, 11,452 participants aged ≥45, CHARLS | ACEs 12 items that occurred before the age of 17 years (score 0-12) | Depressive symptoms were measured by the CESD-10 (Epidemiologic Studies Depression Scale) (score 0-30). Cut-off point of 10 for clinically significant levels | Having ≥4 ACEs (vs. 0) was associated with depressive symptoms OR=3.38 95% CI 2.92–3.90) | Short sleep duration was a mediator |
| Li, 2023; China | Cross-sectional, 1,091 participants aged ≥60, in Jinan, China. | Childhood traumatic events as the number of traumatic events (0-15) before age 18. Their impact on their lives (0=not at all to 4=very hard; total score 0-60 points for childhood trauma severity [CTS]). | Depressive symptoms were assessed using the GDS-15 (Score 0-15) | Childhood traumatic events (β=0.131 (SE=0.041) 95% CI= 0.048-0.209, p=0.003), childhood trauma severity (β=0.109 (SE=0.033) 95% CI= 0.043-0.171, p=0.002). | Resilience mediated the relationship between CTS and depressive symptoms |
| Dai, 2024, China | Longitudinal; 6,921 participants aged ≥45; CHARLS | ACEs (10- item) 5 threats (e.g., physical abuse) and 5 deprivations (e.g., emotional neglect). |
Depressive symptoms (CES-D-10 scale) trajectory | Compared to individuals without ACEs, those with ≥4 ACEs had a significantly higher odds of following the continuing-high trajectory (OR=20.22 95% CI 12.11–33.74), rather than the continuing-low trajectory | Mediators were arthritis, digestive, and respiratory diseases |
| Babatunde, 2024; USA | Cross-sectional, 60,122 participants aged ≥60; 2020 BRFSS | ACE score (0, 1, 2-3, & ≥4) included exposure to eight types of ACEs before age 18 | Self-reported history of depression (health professional diagnosis) | Experiencing ≥4 ACEs (vs. 0) had higher odds for depression among Whites (OR=3.83 95% CI 3.07-4.79), Blacks (OR=3.39 95% CI 1.71-6.71), and Hispanics (OR=12.61 95% CI 4.75-33.43). | |
| Jiang, 2025; China | Longitudinal, 3,941 participants aged ≥45; CHARLS |
ACEs 10 items before the age of 18 (e.g., parental divorce, childhood hunger). Five items for Adverse Adult Experiences (AAEs) (e.g., death of a child, lifetime discrimination) | Depressive symptoms (DS) were assessed using the 10-item Center for Epidemiologic Studies Depression Scale (CES-D 10) (score 0-30). A cutoff score >10 indicated probable depression | Six ACEs were associated with DS [e.g., childhood hunger (OR=1.23 95% CI 1.03–1.47), dangerous growth environments (OR= 1.34 95% CI 1.09–1.65)] & 2 AAEs like prolonged bed rest (OR=1.39 95% CI 1.08–1.79), & lifetime discrimination (OR=1.37 95% CI 1.12–1.66) | Arthritis or rheumatism was a partial mediator |
| Rhee, 2025; USA | Cross-sectional, 315 Korean Americans residing in affordable senior housing. | ACEs 10 items before the age of 18, such as maltreatment (e.g., neglect, physical, verbal, or sexual abuse) and household dysfunction (e.g., parental separation/incarceration, substance use, or mental health issues). | Depressive symptoms (DS) using the Patient Health Questionnaire-9 (PHQ-9) | ACEs were associated with DS in multivariate analysis (B=0.84 SE= 0.18, p<.001); adding potential protective factors (B=0.68 SE=0.17, p<.001) and interactions (B=0.46 SE=0.19, p<.05) | |
| Li, 2025; China | Cross-sectional, 6,585 participants aged ≥60; CHARLS |
ACEs (range 0–12): 7 conventional (physical abuse, emotional neglect, household substance abuse, mental illness, domestic violence, incarcerated household mem ber, parental separation/divorce); 2 expanded (unsafe neighborhood, bullying); and 3 novel indicators (parental death, sibling death, parental disability) | Depression via the CES-D-10 (cutoff ≥ 12). | ACEs was associated to depression (OR=1.17 95% CI 1.11–1.24). | Mediators were sleep duration and chronic diseases |
| Dai, 2025, China | Longitudinal; 6,395 participants aged ≥60; CHARLS | Poor childhood friendship experiences (CFE) (self-report) | Depressive symptoms (CES-D-10 scale, score 0-30); ≥10 significant depressive symptoms | Individuals with poor CFE had a greater risk for depressive symptoms (OR=1.64, 95% CI 1.22-2.21) compared with those with better CFE | |
| Chen, 2026; China | Longitudinal, 3,734 participants aged ≥45; CHARLS | ACEs score 0-10; (e.g., adverse peer relationships, emotional abuse, domestic violence, household substance abuse or incarceration, parental mental illness, family discord, neglect, family member death, family burden, and adverse outside-family environment. | Depressive symptoms (DS)- Center for Epidemiologic Studies Depression Scale (CES-D 10) score 0-30, ≥10 significant depressive symptomatology | Having ≥4 ACEs was associated with DS trajectories, stable increasing vs. low-stable type (OR=2.43 95% CI 1.78-3.32, p<.001). For each one-point increase in ACE scores, there is an increase of 0.501 (95% CI 0.430-0.570, < .001) CES-D score. |
| Authors | Design, population | Exposures | Geriatric Syndromes or other Outcomes | Main outcome’s findings | Additional comments |
|---|---|---|---|---|---|
| Reyes-Ortiz, 2018; USA | Cross-sectional, 2,000 participants aged ≥60; SABE Bogota | History of abuse (ever being a victim): emotional, physical, or sexual. Polyvictimization (≥2 types of abuse vs. 0-1) | Falls during past 12 months. Any fall (≥1 vs. 0) or recurrent falls (≥2 vs. 0-1) | Abuse types: emotional (OR=1.43 95% CI 1.07-1.91), physical (OR=2.05 95% CI 1.51-2.77), or sexual (OR=2.11 95% CI 1.12-3.99), and polyvictimization (OR=2.39 95% CI 1.72- 3.33) were associated with recurrent falls. | |
| Reyes-Ortiz, 2021; USA | Cross-sectional, 5,227 participants aged ≥60; SABE Ecuador | History of abuse (ever being a victim): physical, emotional, sexual, financial, or caregiver neglect. Polyvictimization (≥2 types of abuse vs. 0-1) | Falls during past 12 months. Recurrent falls (≥2 vs. 0-1) | Polyvictimization was associated with higher odds of recurrent falling (OR=1.45 95% CI 1.20-1.76) | Depressive symptoms were a mediator |
| Reyes-Ortiz, 2022; USA | Cross-sectional, 19,004 participants aged ≥60; SABE Colombia | Everyday discrimination (ever had discrimination e.g., racial, SES, gender, age, religion, sexual orientation, or disability), childhood discrimination score (0-3), and discrimination last 5 years due to skin color (score 0-4; at group activities, public places, family, or health centers) |
Falls during past 12 months. Recurrent falls (≥2 vs. 0-1) | Everyday discrimination (OR=1.27 95% CI 1.21–1.33), childhood discrimination score (OR=1.23 95% CI 1.13–1.33), and number of situations for discrimination (OR=1.12 95% CI 1.08–1.17) were associated with recurrent falling. | |
| Reyes-Ortiz, 2024; USA | Cross-sectional, 18,875 participants aged ≥60; SABE Colombia | History of age discrimination or ageism by 1) the neighborhood 2) family 3) health services 4) public services | Falls during past 12 months. Recurrent falls (≥2 vs. 0-1). | Any ageism (OR=1.81 95% CI 1.61–2.02, p <.0001) was associated with recurrent falling. |
Mediators were depressive symptoms, low IADL & multimorbidity |
| Huang, 2024, China | Longitudinal; 12,061 participants aged ≥45, CHARLS | ACEs (12- item, self-report) (physical abuse, childhood environment & adversity [e.g., health status, death of parents, neighborhood safety, hunger, bullying] | Falls since last survey | Those with ≥ 5 ACEs vs. ≤2 ACEs (OR=1.85 95% CI 1.65-2.08) were more likely to experience falls; higher number of ACEs (OR=1.16 95% CI 1.13-1.19) was associated with higher risk of fall. | Depressive symptoms were a mediator |
| Tan, 2025, China | Cross-sectional; 14,582 middle-aged, older adults, CHARLS | ACEs (12- item, self-report) (e.g., physical abuse, emotional neglect, household dysfunction, parental divorce or death, bullying) | Falls since last survey | ACEs were associated with falls (≥4 ACEs vs. 0; OR=2.56 95% CI 2.12-3.09, p<.001) | Pain and depressive symptoms were mediators |
| Reyes-Ortiz, 2025, USA | Cross-sectional; 38,437 participants age 45 to 80, BRFSS | ACEs (11- item, self-report) abuse domain (5 questions) and household dysfunction domain (6 questions) | Falls during past 12 month (Any falls: ≥1 vs. 0) among middle-aged (45-64 years) and older adults (≥65) | Those with ≥2 ACEs (vs. 0-1) have increased odds of falling among middle-aged (OR=1.34 95% CI 1.18-1.52) and older adults (OR=1.28 95% CI 1.15-1.43) | Mediators: depression, functional difficulties, multimorbidity & difficulty remembering |
| Authors | Design, population | Exposures | Geriatric Syndromes or other Outcomes | Main outcome’s findings | Additional comments |
|---|---|---|---|---|---|
| Cheval, 2018, Switzerland | Cohort, 24,179 aged 50-96, SHARE | Early-life SEC (socioeconomic circumstances) at age 10; based on 4 SEC indicators, 5-level variable from most disadvantaged to most advantaged. | Handgrip strength, defined low muscle strength (LMS) (cut-off obtained after being stratified by gender and BMI quartiles) | Among women early-life SEC disadvantaged (OR=0.79 95% CI 0.64–0.99), middle (OR=0.69 95% CI 0.54–0.86), and advantaged (OR=0.58 95% CI 0.44–0.77) had lower risk for low muscle strength compared to those most disadvantaged. | In men those disadvantaged (OR=0.67 95% CI 0.51–0.88), & middle (OR=0.74 95% CI 0.56–0.98) had lower risk for low muscle strength compared to those most disadvantaged. |
| Cheval, 2019, Switzerland | Cohort, 24,179 aged 50-96, SHARE | ACEs (child in care, parental death, parental mental illness, parental drinking, period of hunger, or property taken away) | Handgrip strength, defined low muscle strength (LMS) (cut-off obtained after being stratified by gender and BMI quartiles) | For women, there was a gradual increase in the risk of LMS with the number of experienced ACEs (OR=1.22 for 1 ACE, OR=1.74 for ≥2 ACEs compared to no ACE) | No significant association among men. |
| Smith, 2019, USA | Cohort, 4,459 aged ≥50, HRS | Childhood misfortune (conditions experienced before age 18) 5 domains: infectious disease, chronic disease, impairment, SES, and risky parental behavior) | Handgrip strength (HGS) in Kg | For men, at follow-up (Time 2), there was a steeper decline in HGS (B= -0.60, p<.05) when having one childhood impairment | At follow-up, no significant association among women. |
| Lin, 2022, China | Cross-sectional, 7,209 participants aged ≥45, CHARLS | ACEs before age 17: physical abuse, emotional neglect, household substance abuse, household mental illness, domestic violence, incarcerated household member, parental separation or divorce, unsafe neighborhood, bullying, parental death, sibling death, and parental disability |
Handgrip strength (HGS) in kilograms (continuous); low muscle strength (LMS; defined based in sex and BMI) | Having ≥ 3 ACEs (vs. 0) was negatively associated with lower HGS in Kg (β = -0.93, 95% CI -1.37, -0.49) and positively associated with having LMS (OR=1.34 95% CI 1.12-1.61). | |
| Huang, 2024, China | Longitudinal, 6,859 participants aged ≥45; CHARLS | ACEs, two adversities domains, 5 threat-related (physical abuse, household substance abuse, domestic violence, unsafe neighborhood, or bullying), and 5 deprivation-related (emotional neglect, household mental illness or incarceration, parental or death). | Sarcopenia (Asian Working Group for Sarcopenia 2019 algorithm), was diagnosed when low muscle mass, and low muscle strength or poor physical performance were detected | ACEs were associated with increased risk for sarcopenia: ≥ 3 ACEs (vs. 0) (HR=1.31 95% CI 1.10–1.56); participants with ≥ 2 threat-related ACEs (vs. 0) (HR=1.22 95% CI 1.04–1.43) or with ≥ 2 deprivation-related ACEs (vs. 0) (HR=1.22 95% CI 1.02–1.46) | Active social participation modified the association between ACEs, (especially threat-related), and sarcopenia |
| Dimitriadis, 2025, Canada | Cross-sectional, 25,327 participants aged 45–85 years; CLSA | ACEs: War experiences, a parent death, excessive alcohol use of a relative, sexual abuse, severe problems at home, parents’ poverty, physical illness of respondent | Sarcopenia was defined using the revised EWGSOP2 guidelines | Significant protective association for ACEs (Two ACEs vs. No ACE) on sarcopenia emerged in the oldest group (75–85 years; OR=0.72 95% CI 0.54–0.95 p=0.021) | Findings might be explained by bias due to survival of the fittest |
| Authors | Design, population | Exposures | Geriatric Syndromes or other Outcomes | Main outcome’s findings | Additional comments |
|---|---|---|---|---|---|
| Vásquez, 2019, USA | Cross-sectional, 10,727 participants aged ≥55, 2012-13 NESARC-III | Before age 18 a) maltreatment (#5): physical, emotional/sexual abuse; physical/emotional neglect; b) others (#11): i.e., witnessing domestic violence; parents divorced, parents died; c) past year stressful life events (#9), i.e., unemployed, bankruptcy. ACEs defined any maltreatment or other adverse events (1 = Yes, 0 = No) | Multimorbidity based on # of somatic conditions (i.e., heart disease, hypertension, stroke, diabetes, arthritis, cancer, osteoporosis & chronic lung problems) & DSM-5 psychiatric disorders (i.e., depression) during past 12 months, adjusted means ± SE |
History of ACEs, compared with no history, had greater numbers of somatic & psychiatric multimorbidity (1.37 ± 0.04 vs. 1.13 ± 0.04) among racial and ethnic middle-aged adults (55-64) | No association among older adults (≥65) |
| Lin, 2021, China | Cross-sectional, 11,972 participants aged ≥45, CHARLS | ACEs (0-12), 7 conventional (physical abuse, emotional neglect, household substance abuse, household mental illness, domestic violence, incarcerated household member, & parental separation or divorce), 2 expanded (unsafe neighborhood & bullying), & 3 others (parental death, sibling death, and parental disability) | Multimorbidity, defined as ≥2 of 14 chronic diseases. | Having ≥ 4 ACEs (vs. 0) was associated with higher odds for multimorbidity (OR=2.03 95% CI 1.70-2.41). In addition, using only the 7 conventional ACEs, having ≥ 4 ACEs (vs. 0) was also associated with multimorbidity (OR=2.52 95% CI 1.81-3.50). | |
| Reyes-Ortiz, 2023; USA | Cross-sectional, 18,873 participants aged ≥60; SABE Colombia | Three racial discrimination measures: 1) everyday discrimination (ever had), 2) childhood discrimination score (0-3), and 3) situations of discrimination in the last 5 years (score 0-4 [group activities, public places, inside the family, health centers]). | Multimorbidity, defined as having 2 or more chronic conditions | Multimorbidity was associated with experiencing everyday discrimination (OR=2.21 95% CI 1.62–3.02), childhood discrimination (OR=1.27 95% CI 1.10–1.47), and the number of situations of discrimination (OR=1.56 95% CI 1.22–2.00). | |
| Chandrasekar, 2023, UK | Longitudinal, 3,264 cohort members, ages 36-, 43-, 53-, 63- & 69- years. NSHD | Nine ACEs in 3 groups: a) psychosocial, b) parental health, and c) childhood health. | Multimorbidity, based on 18 health disorders, its score was defined as the unweighted sum of disorders accumulated by an individual at each assessment | Combined childhood health ACEs were associated with higher and progressively increasing multimorbidity scores across adulthood (from 0.18 at age 36 to 0.45 at age 69). | Psychosocial ACE accumulation was associated with steeper multimorbidity trajectories at follow-up |
| Liu, 2024, China | Longitudinal cohort, 6,428 participants aged ≥45, CHARLS | ACEs: physical abuse, emotional neglect, household substance abuse, household mental illness, domestic violence, incarcerated household member, parental separation or divorce, unsafe neighborhood, bullying, parental death, sibling death & parental disability | Multimorbidity was the number of physician diagnoses of 14 chronic diseases, the Chinese multimorbidity-weighted index (CMWI) | ACEs were associated with an increased number of chronic diseases at the baseline (intercept= 0.28, 95% CI 0.20-0.36) and a more rapid increase in the number of chronic diseases over 7 years (intercept= 0.03, 95% CI 0.01-0.05). |
| Authors | Design, population | Exposures | Geriatric Syndromes or other Outcomes | Main outcome’s findings | Additional comments |
|---|---|---|---|---|---|
| Guedes, 2016, Albania, Brazil, Canada, Colombia | Cross-sectional, 1,995 participants aged 65-74, IMIAS | Childhood physical abuse (CPA) (first 15 years of life). Adulthood domestic violence as intimate partner/family member violence (physical or psychological) (PA=physical abuse) | Lower extremity functioning measured by the SPPB= Short Physical Performance Battery including standing balance, 4-m walk, & chair stand test. Mobility disability was defined as SPPB<8 or self-reported limitation in walking/climbing stairs. | CPA (OR=1.69 95% CI 1.14-2.51), partner PA (OR=1.78 95% CI 1.15-2.28) and family PA (OR=1.62 95% CI 1.16-2.27) were associated with SPPB<8. CPA (OR=1.43 95% CI 1.03-1.99) and family PA (OR=1.39 95% CI 1.06-1.83) were associated with mobility disability. | Chronic conditions & depression were mediators between physical violence and both mobility measures. |
| Amemiya, 2018, Japan | Cross-sectional 19,220 participants aged ≥65; JAGES |
ACEs before age of 18 including seven adversities: parental death, parental divorce, parental mental illness, family violence, physical abuse, psychological neglect, and psychological abuse | Higher-level functional limitations (HLFL), using the Lawton IADLs scale, 13 items with instrumental self-maintenance (5 items), intellectual activities (4 items), & social role (4 items). | ACEs (1 vs. 0) (PR=1.06 95% CI = 1.02–1.10) and ACEs (≥2 vs. 0) (PR=1.19 95% CI = 1.12–1.27). were associated with HLFL |
Socio-demographics, adult health behaviors, and health status were mediators |
| Li, 2022, China | Longitudinal, 10,651 participants aged ≥45, CHARLS | ACEs: 13 items including physical abuse, emotional neglect, domestic violence, household substance abuse, household mental illness, incarcerated household member, parental separation or divorce, parental death, sibling death, parental disability, bullying, peer rejection, & unsafe neighborhood. | ADLs disability, having difficulty or could not perform the activity. A 6-item summary ADLs, score 0-6. | Participants who had ≥4 ACEs had increased risk for being on low-mild trajectory (OR=1.32 95% CI 1.11-1.57) and mild-increasing trajectory (OR=1.41 95% CI 1.06-1.89), compared with those with 0 ACEs. | Chronic diseases were mediators: arthritis, digestive system disease, respiratory disease, and cardio-metabolic disease. |
| Lee, 2023, USA | Cross-sectional, 3,387 participants aged ≥50, NSHAP | ACEs= childhood experience of violence/abuse, witnessing violence, financial insecurity, parental separation, or serious illness (exposures between ages 6 to 16) | Lower extremity functioning (tandem balance, 3-m walk & chair stand tests); physical mobility impairment was defined if those tests fell below clinical standards. Also, cognitive testing (MoCA) and reported functional disability (difficulty with ADLs or IADLs). | Childhood experience of violence was associated with greater physical mobility impairment (OR=1.38, 95% CI 1.11–1.71) and functional disability (OR=1.86, 95% CI 1.49–2.33). |
| Authors | Design, population | Exposures | Geriatric Syndromes or other Outcomes | Main outcome’s findings | Additional comments |
|---|---|---|---|---|---|
| Wang, 2019, USA | Cross-sectional, 3,157 participants aged ≥60, PINE | Child maltreatment (CM) and intimate partner violence (IPV) cases (been physically hurt, insulted, threatened with harm, screamed and cursed at, and a private part touched when it was unwanted during each age-related period. |
Elder abuse (EA) phenotype (psychological, physical & sexual abuse; financial exploitation & caregiver neglect) (total 56 items | CM physical/sexual was associated with increased risks for IPV physical/sexual (OR=1.86 95% CI 1.02-3.38), EA psychological (OR=1.70 95% CI 1.20-2.42), and EA financial exploitation (OR=2.38 95% CI 1.72-3.30). |
|
| Payne, 2020, South Africa | Cross-sectional, 2,473 participants aged ≥40, HAALSI | 16 Traumatic Experiences (TEs) during their lifetime- five categories: childhood environment, community violence, illness/accident/disaster, social/family environment, and war violence | Depressive symptoms (7-item CES-D) score ≥3. PTSD symptoms (Short Screening Scale for PTSD battery; score ≥4). Katz ADLs scale. Cognitive impairment (<1.5 SD below the mean in the HAALSI cognitive battery | TEs were associated with depressive symptoms (OR=1.08 95% CI 1.03-1.14), PTSD symptoms (OR=1.17 95% CI 1.07-1.29), ADL limitations OR=1.11 95% CI 1.03, 1.18). | No association between TEs and later life cognitive impairment |
| Dorji, 2020, Buthan | Cross-sectional, 337 participants aged ≥60, | WHO ACEs International Questionnaire (13-item): physical or emotional abuse, neglect, someone chronically depressed in a household, incarcerated household member, parental separation or divorce, alcohol or drug abuser or member treated violently in the household, bullying, witnessing of community violence, forced to physical labor, ≥2 weeks had difficulty with basic needs of life | Heart disease, high blood pressure (HBP), diabetes, depression, mobility impairment, lung disease, visual impairment (VI), insomnia, memory decline (MD) | Having ≥7 ACEs [vs. 0-2] was associated with higher odds for lung disease (OR=2.15 95% CI 1.03-4.49), VI (OR=2.38 95% CI 1.16-4.85), insomnia (OR=2.35 95% CI 1.11-4.98), MD (OR=2.30 95% CI 1.10-4.78), HBP (OR=3.21 95% CI 1.39-7.38), diabetes (OR=5.12 95% CI 1.06-24.72). | |
| Lv, 2020, China | Cross-sectional, 6,267 participants aged ≥45, CHARLS | Famine fetal exposed, preschool exposed, and school-aged, exposed groups when compared to the non-exposed group | Chronic kidney disease (CKD) was defined as eGFR less than 60 mL/min per 1.73 m2. | Fetal exposure to the severe famine was associated with the elevated risk of CKD among male participants (OR=4.44 95% CI 1.10–17.92) | |
| Sheffler, 2023, USA | Cross-sectional, 348 participants aged ≥55; North Florida |
ACEs, 10-item questionnaire including childhood abuse, parental psychopathology and divorce, and violence in the home occurring before the age of 18. | Sleep quality: Pittsburgh Sleep Quality Index (PSQI). Rating their “sleep quality overall” during the past month from 1 (Very Good) to 4 (Very Bad) | ACEs were associated with significantly worse sleep quality (B= 0.156, p= .008); higher PSQI score indicates lower sleep quality | Adaptive emotion regulation skills had a moderation effect |
| Ren, 2023, China | Longitudinal; 10,695 participants aged ≥45, CHARLS | ACEs (14- item, self-report) (physical abuse, emotional neglect, family or community violence, parental [divorce, disability, substance abuse, incarceration or death], economic adversity, loneliness, bullying | New-onset pain, falls, chronic diseases, multimorbidity, ADL limitations, and IADL limitations | ≥4 ACEs vs. 0 were associated with new-onset pain (HR=1.57 95% CI 1.37–1.79), falls (HR=1.84 95% CI 1.55–2.18), chronic diseases (HR=1.43 95% CI 1.19–1.72), multimorbidity (HR=1.55 95% CI 1.32–1.83), ADL limitations (HR=1.85 95% CI 1.54–2.23), and IADL limitations, (HR=1.46 95% CI 1.24–1.71) | For all outcomes, depressive symptoms was a mediator |
| Li, 2024, China | Cross-sectional, 12,277 participants aged ≥45, CHARLS | ACEs (12- item) physical abuse, parental mental health, guardians’ bad habits, hunger, feel alone, peer bullied, self-reported health status, the health limitations, death of parents, death of siblings, childhood neighborhood quality, and childhood neighborhood safety | Chronic lung diseases (CLDs) such as chronic bronchitis, emphysema | ACEs that were associated with CLDs include physical abuse (OR=1.28), parents mental health (OR=1.50), hunger (OR=1.20), feel alone (OR=1.33), peer bullied (OR=1.56), poor health status (OR=1.86), health limitation (OR=1.97), parents’ death (OR=1.27), siblings’ death (OR=1.42), unsafe neighborhood (OR=1.23), & poor neighborhood quality (OR=1.11) | Depressive symptoms were a partial mediator on the association between CLDs and 7 ACEs |
| He, 2024, China | Cross-sectional, 24,116 participants aged ≥50, GBCS | ACEs: 5 items reflected separation, traumatic experience, emotional abuse, domestic violence and parental death | Hemoglobin (Hb)(g/dL). Anemia defined as Hb <12.0 g/dL for men and <11.0 g/dL for women | Participants with ≥2 ACEs, (vs. 0 ACEs), had lower Hb (β= - 0.08, 95% CI - 0.12 to - 0.03) & higher odds of anemia (OR=1.26 95% CI 1.01-1.59). | |
| Cheng, 2025, China | Cross-sectional; 14,582 participants middle-aged, older adults, CHARLS | ACEs (11- item, before age 17)- bullying, corpo ral punishment, domestic violence, parental substance abuse, poor parental mental health, parental criminal ity, parental divorce, parental disability, parental death, emotional neglect, and insecurity. | Chronic respiratory diseases [CRD] (COPD & asthma) | Individuals with ≥4 ACEs had OR=1.48 (95% CI 1.10-1.99) for CRD compared to those with no ACEs | Long sleep duration combined with ACEs increased the risk of CRD |
| Jin, 2025, China | Cross-sectional; 11,905 participants middle-aged, older adults, CHARLS | ACEs (12- item)- physical abuse, emotional neglect, domestic violence, peer bullying, unsafe neigh borhood, parental death, parental disability, sibling death, household mental illness, substance abuse, parental separation or divorce, and incarcerated household mem bers. | Musculoskeletal pain (MSP) | Individuals with ≥4 ACEs had OR=2.46 (95% CI 2.13-2.83) for MSP compared to those with 0-1 ACEs; depressive symptoms were a partial mediator | Depressive symptoms were a mediator |
| Chen, 2025, China | Longitudinal; 11,601 participants aged ≥45, CHARLS | ACEs 10 items before the age of 18 (e.g., parental divorce, childhood hunger). Five items for Adverse Adult Experiences (AAEs) (e.g., death of a child, lifetime discrimination) | Dementia diagnosis based on cognitive battery and ADL scale. Stroke (self-reported physician diagnosis) |
ACEs (HR=1.11 95% CI 1.05-1.18) and AAEs (HR=1.23 95% CI 1.14-1.33) were associated with dementia. AAEs (HR=1.19 95% CI 1.12-1.26) were associated with stroke. | Depressive symptoms were a mediator |
| Karhu, 2025, USA | Cross-sectional; 128 patients aged 55 (±16), Cleveland Clinic |
ACEs are a 10-item screening tool that assesses exposure to various forms of abuse, neglect, and house hold dysfunction during the first 18 years of life. | Obstructed Defecation Syndrome (ODS): ex cessive straining, incomplete rectal evacuation, enemas and laxatives’ use, vaginal-anal-perineal maneuvers to attempt def ecation, and abdominal discomfort/pain caused by obstructed defecation. CCFI measures severity of fecal incontinence. | Patients with ACEs had more severe ODS (not CCFI) scores which correlated with higher HADS anxiety and depression scores. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




