Submitted:
13 February 2026
Posted:
14 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Areas
2.2. Sampling Strategy and Sample Preservation
2.3. Microplastics Extraction
2.4. Microplastics Identification
2.5. Quality Assurance/Quality Control
3. Results and Discussion
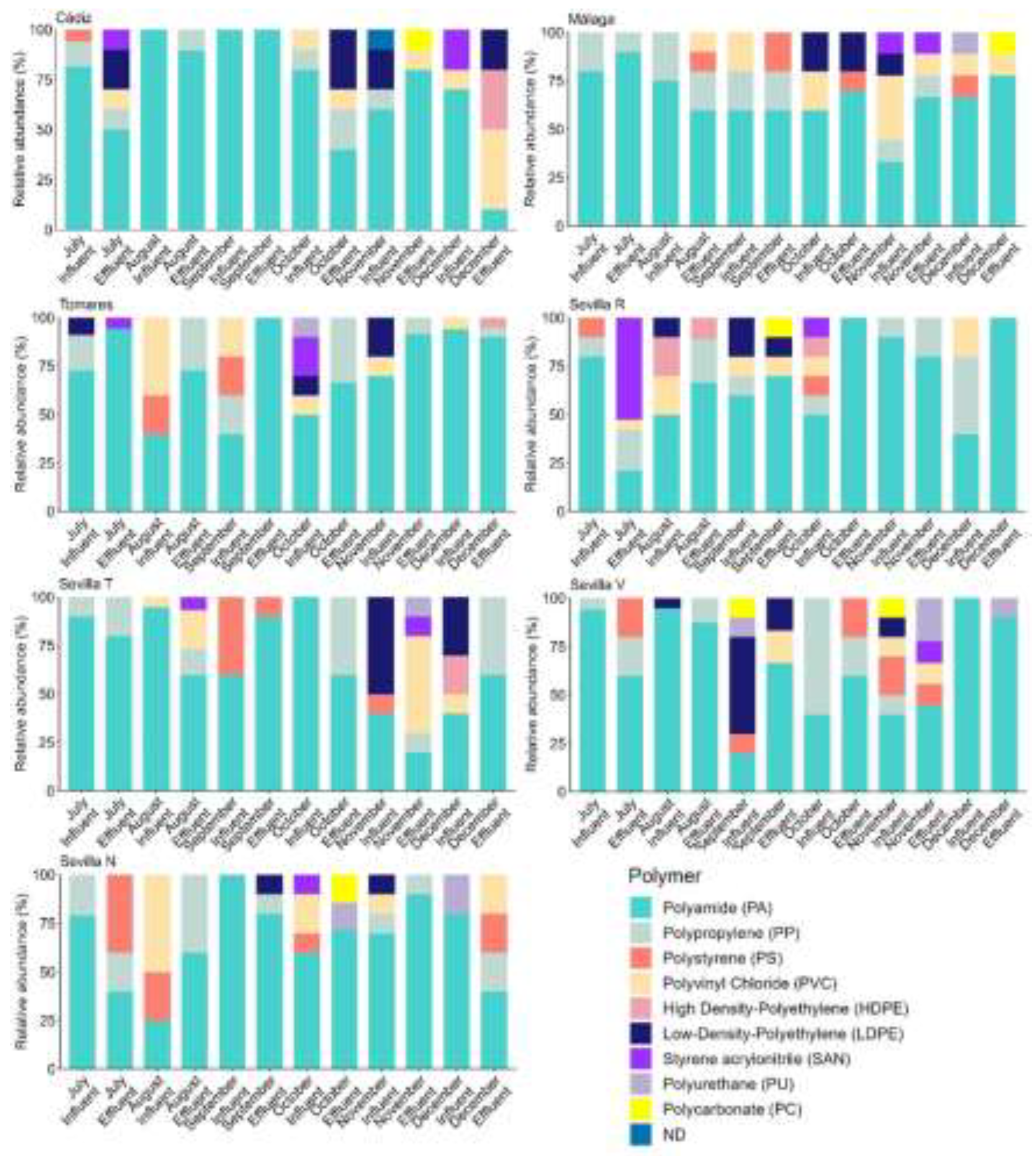
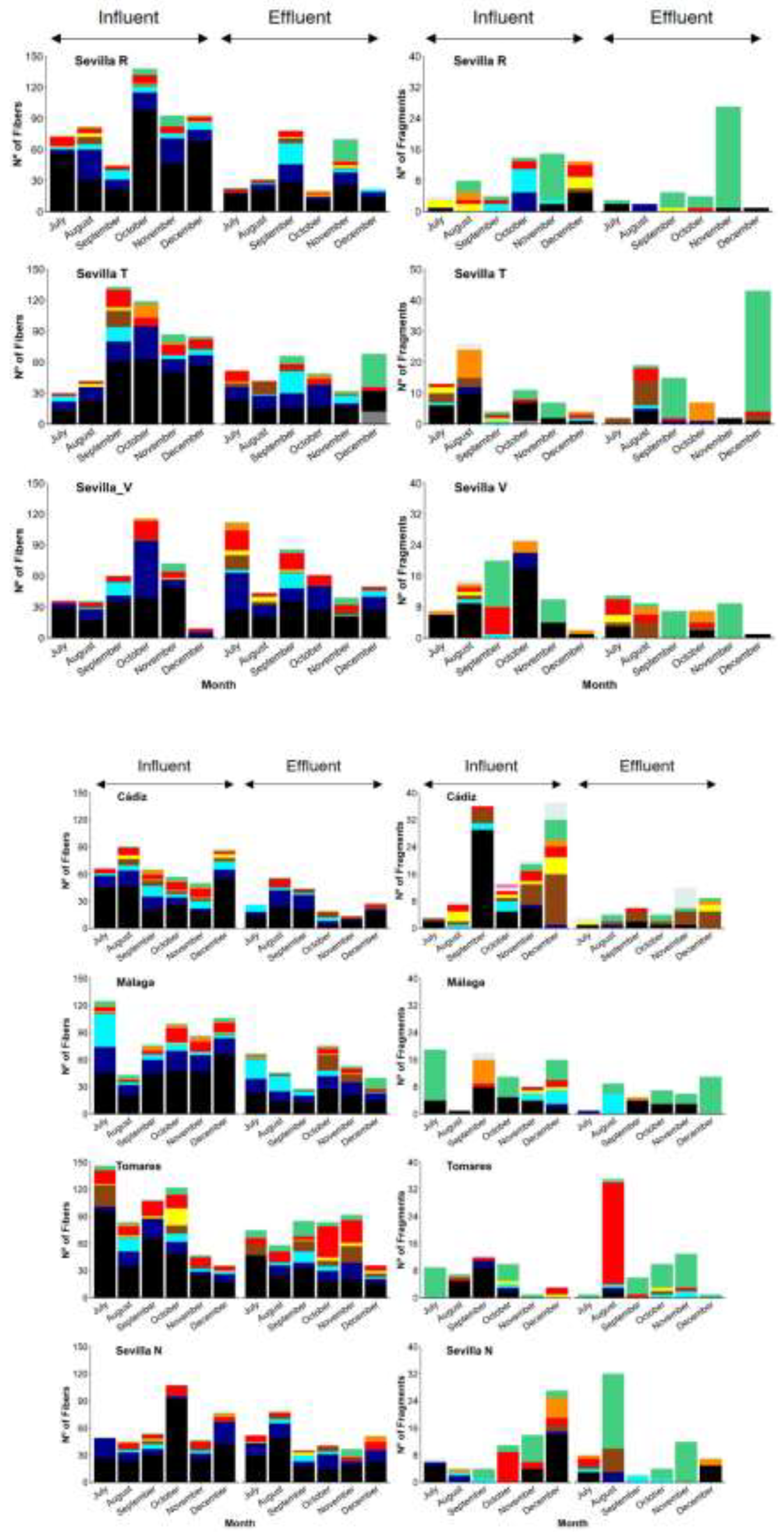
3.1. MP Polymer Composition
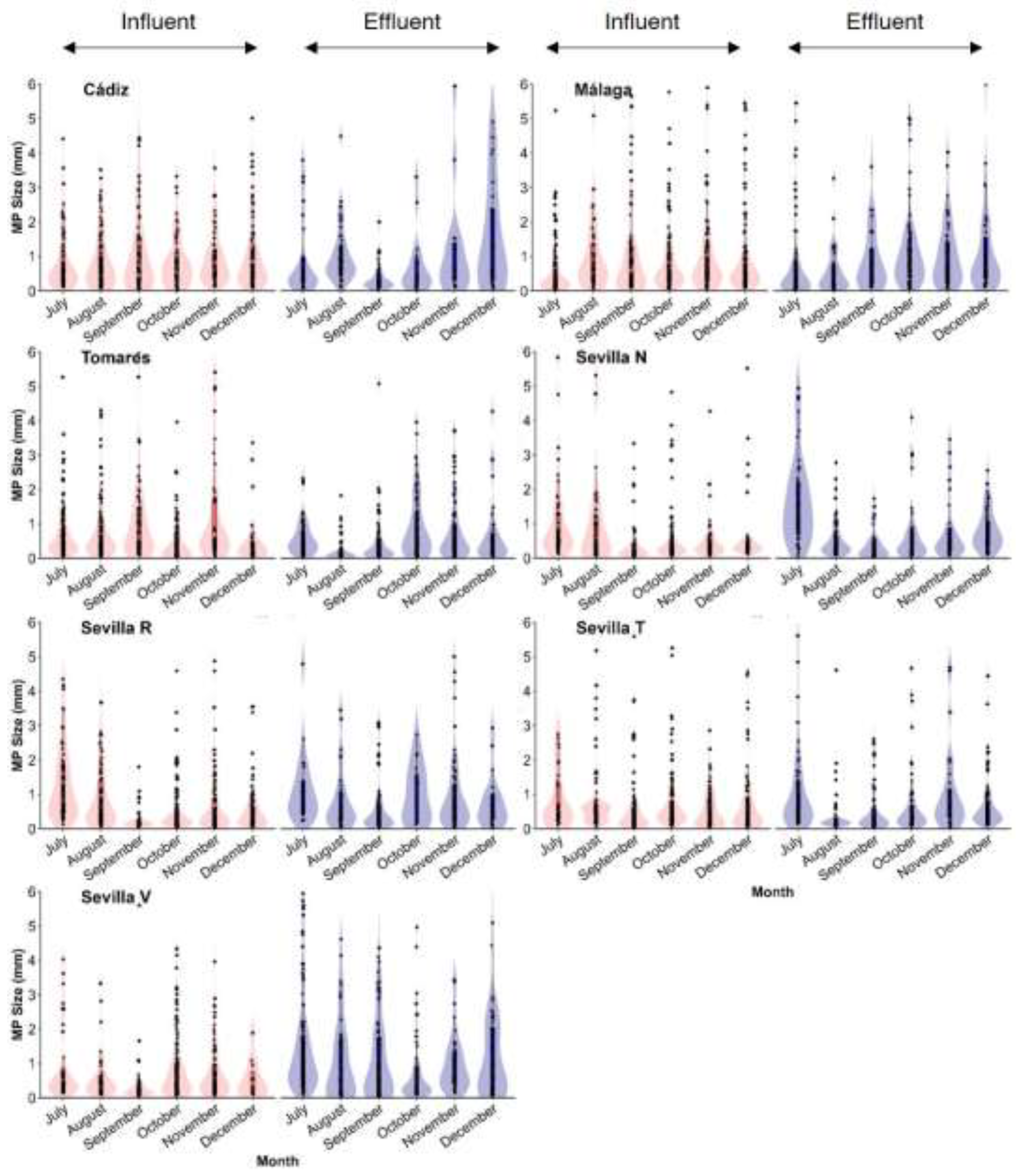
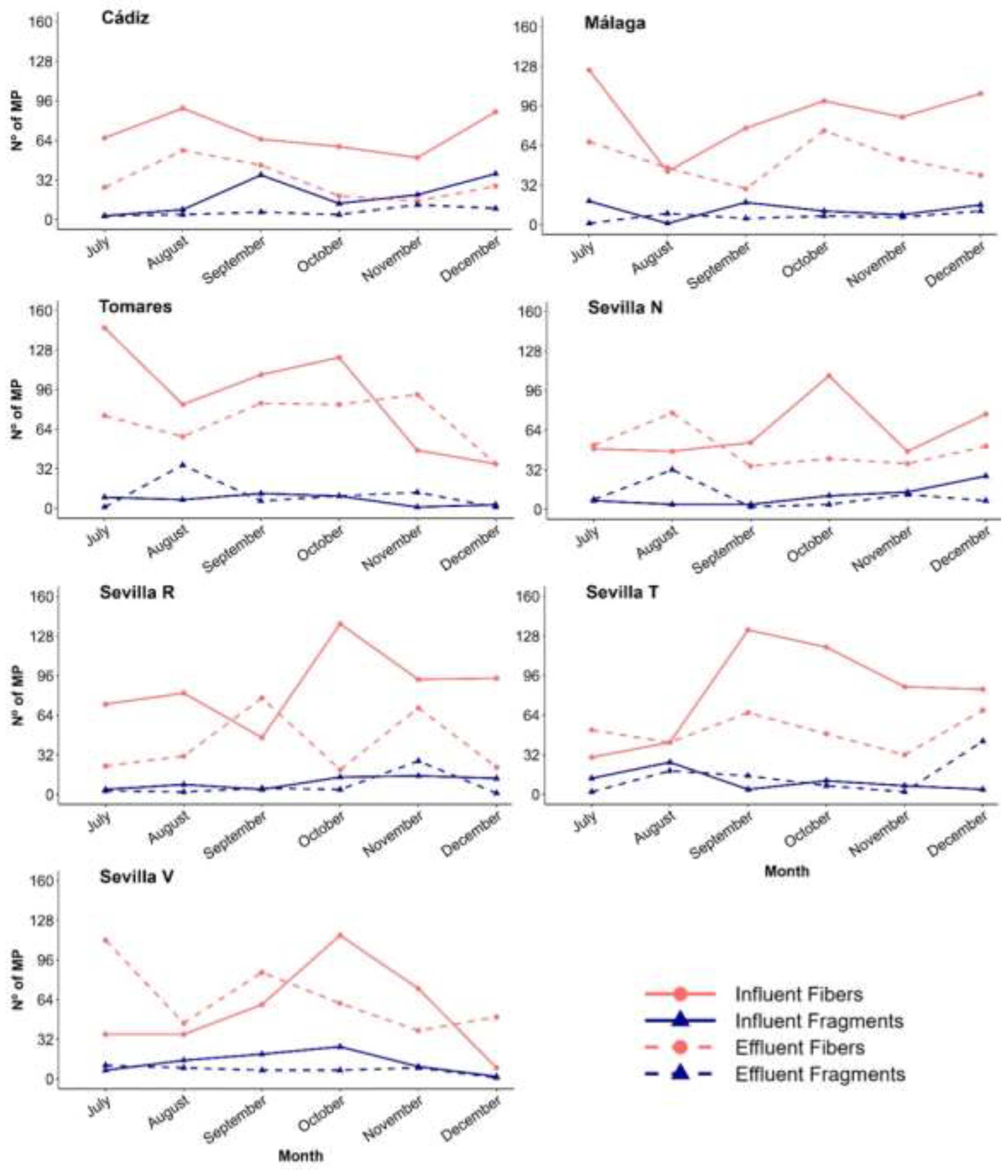
3.2. MP Characteristics (Size, Shape and Colour)
3.3. MP Removal Efficiencies (RE%)
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| MPs | Microplastics |
| NPs | Nanoplastics |
| WWTPs | Wastewater Treatment Plants |
| PP | Polypropylene |
| PE | Polyethylene |
| LDPE | Low-density polyethylene |
| HDPE | High-density polyethylene |
| PVC | Polyvinyl Chloride |
| SAN | Styrene Acrylonitrile |
| PU | Polyurethane |
| PET | Polyethylene Terephthalate |
| PC | Polycarbonate |
| PS | Polystyrene |
| PA | Polyamide |
| HRT | Hydraulic Retention Time |
| RE | Removal Efficiency |
| QA | Quality Assurance |
| QC | Quality Control |
References
- PlasticEurope_TheFacts_2024_digital-1pager.
- OECD Global Plastics Outlook: Policy Scenarios to 2060; OECD, 2022; ISBN 978-92-64-97364-0.
- Hildebrandt, J.; Thünemann, A.F. Aqueous Dispersions of Polypropylene: Toward Reference Materials for Characterizing Nanoplastics. Macromol. Rapid Commun. 2023, 44, 2200874. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, S.; Alves, L.; Medronho, B.; Romano, A.; Rasteiro, M.D.G. Microplastics in Ecosystems: From Current Trends to Bio-Based Removal Strategies. Molecules 2020, 25, 3954. [Google Scholar] [CrossRef] [PubMed]
- Gigault, J.; Halle, A.T.; Baudrimont, M.; Pascal, P.-Y.; Gauffre, F.; Phi, T.-L.; El Hadri, H.; Grassl, B.; Reynaud, S. Current Opinion: What Is a Nanoplastic? Environmental Pollution 2018, 235, 1030–1034. [Google Scholar] [CrossRef] [PubMed]
- Duis, K.; Coors, A. Microplastics in the Aquatic and Terrestrial Environment: Sources (with a Specific Focus on Personal Care Products), Fate and Effects. Environ Sci Eur 2016, 28, 2. [Google Scholar] [CrossRef]
- Peng, J.; Wang, J.; Cai, L. Current Understanding of Microplastics in the Environment: Occurrence, Fate, Risks, and What We Should Do. Integrated Environmental Assessment and Management 2017, 13, 476–482. [Google Scholar] [CrossRef]
- Luo, G.; Liang, B.; Cui, H.; Kang, Y.; Zhou, X.; Tao, Y.; Lu, L.; Fan, L.; Guo, J.; Wang, A.; et al. Determining the Contribution of Micro/Nanoplastics to Antimicrobial Resistance: Challenges and Perspectives. Environ. Sci. Technol. 2023, 57, 12137–12152. [Google Scholar] [CrossRef]
- Jeong, E.; Lee, J.-Y.; Redwan, M. Animal Exposure to Microplastics and Health Effects: A Review. Emerging Contaminants 2024, 10, 100369. [Google Scholar] [CrossRef]
- European Parliament Microplastics: Sources, Effects and Solutions; 2018.
- Bodzek, M.; Pohl, A.; Rosik-Dulewska, C. Microplastics in Wastewater Treatment Plants: Characteristics, Occurrence and Removal Technologies. Water 2024, 16, 3574. [Google Scholar] [CrossRef]
- Freeman, S.; Booth, A.M.; Sabbah, I.; Tiller, R.; Dierking, J.; Klun, K.; Rotter, A.; Ben-David, E.; Javidpour, J.; Angel, D.L. Between Source and Sea: The Role of Wastewater Treatment in Reducing Marine Microplastics. Journal of Environmental Management 2020, 266, 110642. [Google Scholar] [CrossRef]
- Tadsuwan, K.; Babel, S. Unraveling Microplastics Removal in Wastewater Treatment Plant: A Comparative Study of Two Wastewater Treatment Plants in Thailand. Chemosphere 2022, 307, 135733. [Google Scholar] [CrossRef]
- Hu, Z.; Sun, Y.; Zhou, J.; Sun, W.; Shah, K.J. Microplastics in Wastewater Plants: A Review of Sources, Characteristics, Distribution and Removal Technologies. Journal of Contaminant Hydrology 2024, 267, 104448. [Google Scholar] [CrossRef]
- Ormaniec, P.; Mikosz, J. Circulation of Microplastics in a Municipal Wastewater Treatment Plant with Multiphase Activated Sludge. Desalination and Water Treatment 2024, 317, 100265. [Google Scholar] [CrossRef]
- Kurt, Z.; Özdemir, I.; James R., A.M. Effectiveness of Microplastics Removal in Wastewater Treatment Plants: A Critical Analysis of Wastewater Treatment Processes. Journal of Environmental Chemical Engineering 2022, 10, 107831. [Google Scholar] [CrossRef]
- Franco, A.A.; Martín-García, A.P.; Egea-Corbacho, A.; Arellano, J.M.; Albendín, G.; Rodríguez-Barroso, R.; Quiroga, J.M.; Coello, M.D. Assessment and Accumulation of Microplastics in Sewage Sludge at Wastewater Treatment Plants Located in Cádiz, Spain. Environmental Pollution 2023, 317, 120689. [Google Scholar] [CrossRef] [PubMed]
- Edo, C.; González-Pleiter, M.; Leganés, F.; Fernández-Piñas, F.; Rosal, R. Fate of Microplastics in Wastewater Treatment Plants and Their Environmental Dispersion with Effluent and Sludge. Environmental Pollution 2020, 259, 113837. [Google Scholar] [CrossRef] [PubMed]
- Conley, K.; Clum, A.; Deepe, J.; Lane, H.; Beckingham, B. Wastewater Treatment Plants as a Source of Microplastics to an Urban Estuary: Removal Efficiencies and Loading per Capita over One Year. Water Research X 2019, 3, 100030. [Google Scholar] [CrossRef]
- Kazour, M.; Terki, S.; Rabhi, K.; Jemaa, S.; Khalaf, G.; Amara, R. Sources of Microplastics Pollution in the Marine Environment: Importance of Wastewater Treatment Plant and Coastal Landfill. Marine Pollution Bulletin 2019, 146, 608–618. [Google Scholar] [CrossRef]
- Sun, J.; Dai, X.; Wang, Q.; Van Loosdrecht, M.C.M.; Ni, B.-J. Microplastics in Wastewater Treatment Plants: Detection, Occurrence and Removal. Water Research 2019, 152, 21–37. [Google Scholar] [CrossRef]
- Murphy, F.; Ewins, C.; Carbonnier, F.; Quinn, B. Wastewater Treatment Works (WwTW) as a Source of Microplastics in the Aquatic Environment. Environ. Sci. Technol. 2016, 50, 5800–5808. [Google Scholar] [CrossRef]
- Franco, A.A.; Iglesias-Arroyo, D.; Egea-Corbacho, Á.; Martín-García, A.P.; Quiroga, J.M.; Coello, M.D. Influence of Tourism on Microplastic Contamination at Wastewater Treatment Plants in the Coastal Municipality of Chiclana de La Frontera. Science of The Total Environment 2023, 900, 165573. [Google Scholar] [CrossRef]
- Magni, S.; Binelli, A.; Pittura, L.; Avio, C.G.; Della Torre, C.; Parenti, C.C.; Gorbi, S.; Regoli, F. The Fate of Microplastics in an Italian Wastewater Treatment Plant. Science of The Total Environment 2019, 652, 602–610. [Google Scholar] [CrossRef]
- Prasetya, K.D.; Hassan, F.; Yen, Y.-T.; Chen, P.-Y.; Jiang, J.-J.; Wang, Y.-F.; You, S.-J. Assessing the Fate of Microplastics in Multi-Stage Treatment Units through Distribution Patterns and Settling Dynamics Models. Journal of Contaminant Hydrology 2026, 276, 104734. [Google Scholar] [CrossRef] [PubMed]
- Ridall, A.; Farrar, E.; Dansby, M.; Ingels, J. Influence of Wastewater Treatment Plants and Water Input Sources on Size, Shape, and Polymer Distributions of Microplastics in St. Andrew Bay, Florida, USA. Marine Pollution Bulletin 2023, 187, 114552. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liu, J.; Xie, Y.; Zhong, S.; Gao, P. Occurrence and Removal of Microplastics from Wastewater Treatment Plants in a Typical Tourist City in China. Journal of Cleaner Production 2021, 291, 125968. [Google Scholar] [CrossRef]
- Yaseen, A.; Assad, I.; Sofi, M.S.; Hashmi, M.Z.; Bhat, S.U. A Global Review of Microplastics in Wastewater Treatment Plants: Understanding Their Occurrence, Fate and Impact. Environmental Research 2022, 212, 113258. [Google Scholar] [CrossRef]
- AF-Plastics-the-Facts-2021_250122.
- Nematollahi, M.J.; Mobasheri, M.; Esmaeili, Z.; Mahmoudi, M.; Yousefi, N.; Busquets, R. Distribution and Abundance of Microplastics in Urban and Industrial Wastewater Treatment Plants in Tabriz Metropolis. Sci Rep 2025, 15, 24577. [Google Scholar] [CrossRef]
- Andrady, A.L. Microplastics in the Marine Environment. Marine Pollution Bulletin 2011, 62, 1596–1605. [Google Scholar] [CrossRef]
- Akarsu, C.; Kumbur, H.; Gökdağ, K.; Kıdeyş, A.E.; Sanchez-Vidal, A. Microplastics Composition and Load from Three Wastewater Treatment Plants Discharging into Mersin Bay, North Eastern Mediterranean Sea. Marine Pollution Bulletin 2020, 150, 110776. [Google Scholar] [CrossRef]
- Ziajahromi, S.; Neale, P.A.; Rintoul, L.; Leusch, F.D.L. Wastewater Treatment Plants as a Pathway for Microplastics: Development of a New Approach to Sample Wastewater-Based Microplastics. Water Research 2017, 112, 93–99. [Google Scholar] [CrossRef]
- Hajji, S.; Ben-Haddad, M.; Abelouah, M.R.; De-la-Torre, G.E.; Alla, A.A. Occurrence, Characteristics, and Removal of Microplastics in Wastewater Treatment Plants Located on the Moroccan Atlantic: The Case of Agadir Metropolis. Science of The Total Environment 2023, 862, 160815. [Google Scholar] [CrossRef]
- Luo, Y.; Xie, H.; Xu, H.; Zhou, C.; Wang, P.; Liu, Z.; Yang, Y.; Huang, J.; Wang, C.; Zhao, X. Wastewater Treatment Plant Serves as a Potentially Controllable Source of Microplastic: Association of Microplastic Removal and Operational Parameters and Water Quality Data. Journal of Hazardous Materials 2023, 441, 129974. [Google Scholar] [CrossRef]
- Üstün, G.E.; Bozdaş, K.; Can, T. Abundance and Characteristics of Microplastics in an Urban Wastewater Treatment Plant in Turkey. Environmental Pollution 2022, 310, 119890. [Google Scholar] [CrossRef] [PubMed]
- Napper, I.E.; Parker-Jurd, F.N.F.; Wright, S.L.; Thompson, R.C. Examining the Release of Synthetic Microfibres to the Environment via Two Major Pathways: Atmospheric Deposition and Treated Wastewater Effluent. Science of The Total Environment 2023, 857, 159317. [Google Scholar] [CrossRef]
- Gao, Z.; Cizdziel, J.V.; Wontor, K.; Vianello, A. Spatiotemporal Characteristics of Microplastics in a University Wastewater Treatment Plant: Influence of Sudden on-Campus Population Swings. Journal of Environmental Chemical Engineering 2022, 10, 108834. [Google Scholar] [CrossRef]
- Okoffo, E.D.; Rauert, C.; Thomas, K.V. Mass Quantification of Microplastic at Wastewater Treatment Plants by Pyrolysis-Gas Chromatography–Mass Spectrometry. Science of The Total Environment 2023, 856, 159251. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Li, D.; Zhang, Z.; Wen, L.; Liu, S.; Hu, E.; Li, M.; Gao, L. Characteristics and the Potential Impact Factors of Microplastics in Wastewater Originated from Different Human Activity. Process Safety and Environmental Protection 2022, 166, 78–85. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, W.; Gao, Q.; Huang, F.; Kang, Y.; Pan, Y.; Luo, G.; Zhou, X.; Fan, L.; Wang, A.; et al. Microplastics Extraction from Wastewater Treatment Plants: Two-Step Digestion Pre-Treatment and Application. Water Research 2023, 230, 119569. [Google Scholar] [CrossRef]
- Martín-García, A.P.; Egea-Corbacho, Á.; Franco, A.A.; Rodríguez-Barroso, R.; Coello, M.D.; Quiroga, J.M. Grab and Composite Samples: Variations in the Analysis of Microplastics in a Real Wastewater Treatment Plant in the South of Spain. Journal of Environmental Chemical Engineering 2023, 11, 109486. [Google Scholar] [CrossRef]
- Bayo, J.; Olmos, S.; López-Castellanos, J. Microplastics in an Urban Wastewater Treatment Plant: The Influence of Physicochemical Parameters and Environmental Factors. Chemosphere 2020, 238, 124593. [Google Scholar] [CrossRef]
- Blair, R.M.; Waldron, S.; Gauchotte-Lindsay, C. Average Daily Flow of Microplastics through a Tertiary Wastewater Treatment Plant over a Ten-Month Period. Water Research 2019, 163, 114909. [Google Scholar] [CrossRef]
- Browne, M.A.; Crump, P.; Niven, S.J.; Teuten, E.; Tonkin, A.; Galloway, T.; Thompson, R. Accumulation of Microplastic on Shorelines Woldwide: Sources and Sinks. Environmental Science and Technology 2011, 45, 9175–9179. [Google Scholar] [CrossRef]
- De Falco, F.; Gullo, M.P.; Gentile, G.; Di Pace, E.; Cocca, M.; Gelabert, L.; Brouta-Agnésa, M.; Rovira, A.; Escudero, R.; Villalba, R.; et al. Evaluation of Microplastic Release Caused by Textile Washing Processes of Synthetic Fabrics. Environmental Pollution 2018, 236, 916–925. [Google Scholar] [CrossRef]
- Schell, T.; Hurley, R.; Nizzetto, L.; Rico, A.; Vighi, M. Spatio-Temporal Distribution of Microplastics in a Mediterranean River Catchment: The Importance of Wastewater as an Environmental Pathway. Journal of Hazardous Materials 2021, 420, 126481. [Google Scholar] [CrossRef]
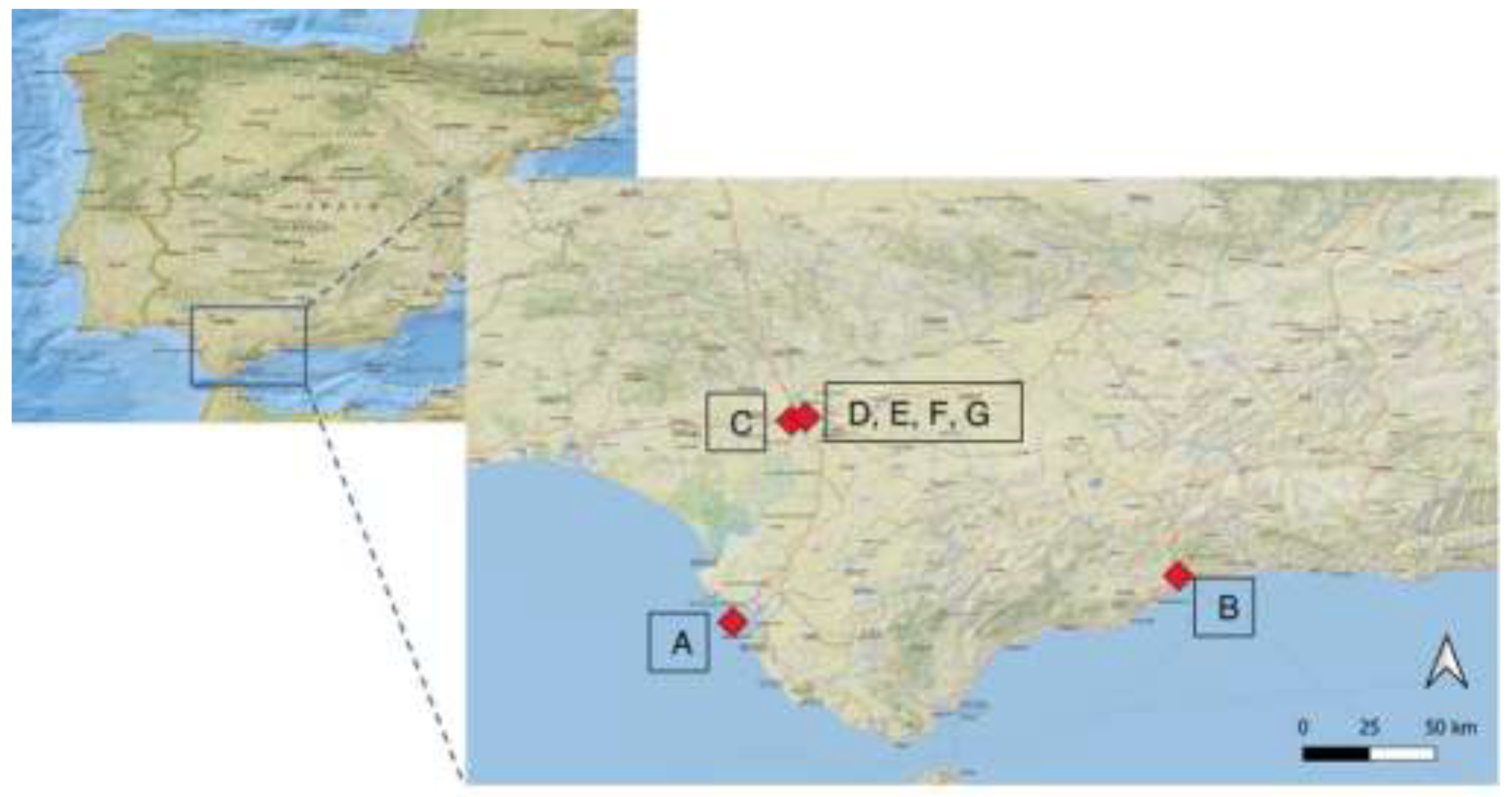
| WWTP | Location | Treatment capacity (m3/d) |
Population equivalent | Influx composition | Type of process | Water treatment processs |
|---|---|---|---|---|---|---|
| A Cádiz | Coast | 75,000 | 375,000 | Urban | Activated sludge, anaerobic digestion | Primary, secondary |
| B Málaga | Coast | 177,000 | 1,300,000 | Urban | Activated sludge, anaerobic digestion, ultrafiltration | Primary, secondary |
| C Tomares | Inland area | 112,000 | 350,000 | Urban | Activated sludge, anaerobic digestion | Primary, secondary |
| D Sevilla N | Inland area | 90,000 | 350,000 | Urban | Activated sludge, anaerobic digestion, | Primary, secondary |
| E Sevilla R | Inland area | 90,000 | 350,000 | Urban | Activated sludge, anaerobic digestion, N and P removal, tertiary treatment | Primary, secondary, tertiary |
| F Sevilla T | Inland area | 50,000 | 200,000 | Urban | Activated sludge, anaerobic digestion | Primary, secondary |
| G Sevilla V | Inland area | 9,220 | 41,000 | Urban | Carrousel system, N and P removal, sludge dewatering | Primary, secondary |
| Cádiz | Málaga | Tomares | Sevilla N | Sevilla R | Sevilla T | Sevilla V | |||
|---|---|---|---|---|---|---|---|---|---|
| July | Influent | 35 | 72 | 78 | 28 | 39 | 22 | 22 | |
| Effluent | 13 | 34 | 38 | 30 | 13 | 28 | 62 | ||
| August | Influent | 49 | 22 | 46 | 26 | 45 | 34 | 26 | |
| Effluent | 30 | 28 | 47 | 55 | 17 | 31 | 27 | ||
| September | Influent | 51 | 48 | 60 | 29 | 25 | 69 | 40 | |
| Effluent | 25 | 17 | 46 | 19 | 42 | 41 | 47 | ||
| October | Influent | 36 | 56 | 66 | 60 | 76 | 65 | 71 | |
| Effluent | 12 | 42 | 47 | 23 | 12 | 28 | 34 | ||
| November | Influent | 35 | 48 | 24 | 31 | 54 | 47 | 42 | |
| Effluent | 14 | 30 | 53 | 25 | 49 | 17 | 24 | ||
| December | Influent | 62 | 61 | 20 | 52 | 54 | 45 | 6 | |
| Effluent | 18 | 26 | 19 | 29 | 12 | 65 | 26 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




