3. Discussion
Schwannomas are benign tumors arising from Schwann cells of the nerves. Schwannomas of the accessory nerve are rarely encountered, especially when they are located extracranially [
6].
Similarly to this clinical case, the literature on cervical accessory nerve schwannomas mostly reports an asymptomatic course—patients notice a neck mass without other complaints [
2,
7,
8]. In the aforementioned study, only one of nine patients with extracranial schwannoma localization reported pain in the mass region [
3]. Neurological symptoms are observed mainly in cases of intracranial and cervical spinal canal localization [
3].
Both the clinical presentation and the presence of a solitary node in the carotid space may pose diagnostic challenges. In clinical case reports, authors mention clarification of the diagnosis during surgery and by histological examination of the lesion [
1,
3,
9,
10]. Usually, when such a clinical picture is observed, primary hypotheses include lymphadenopathy, metastatic disease, or other more common lesions. Clinical cases have been described in the literature in which, based on objective findings (a solitary painless mass on the lateral surface of the neck) and CT results (an oval hypodense mass effect with necrosis and calcifications), the primary diagnosis was lymphadenopathy or a solitary metastasis; a significant role in verifying the correct diagnosis was played by intraoperative findings and histological examination of the material, proving accessory nerve schwannoma [
8].
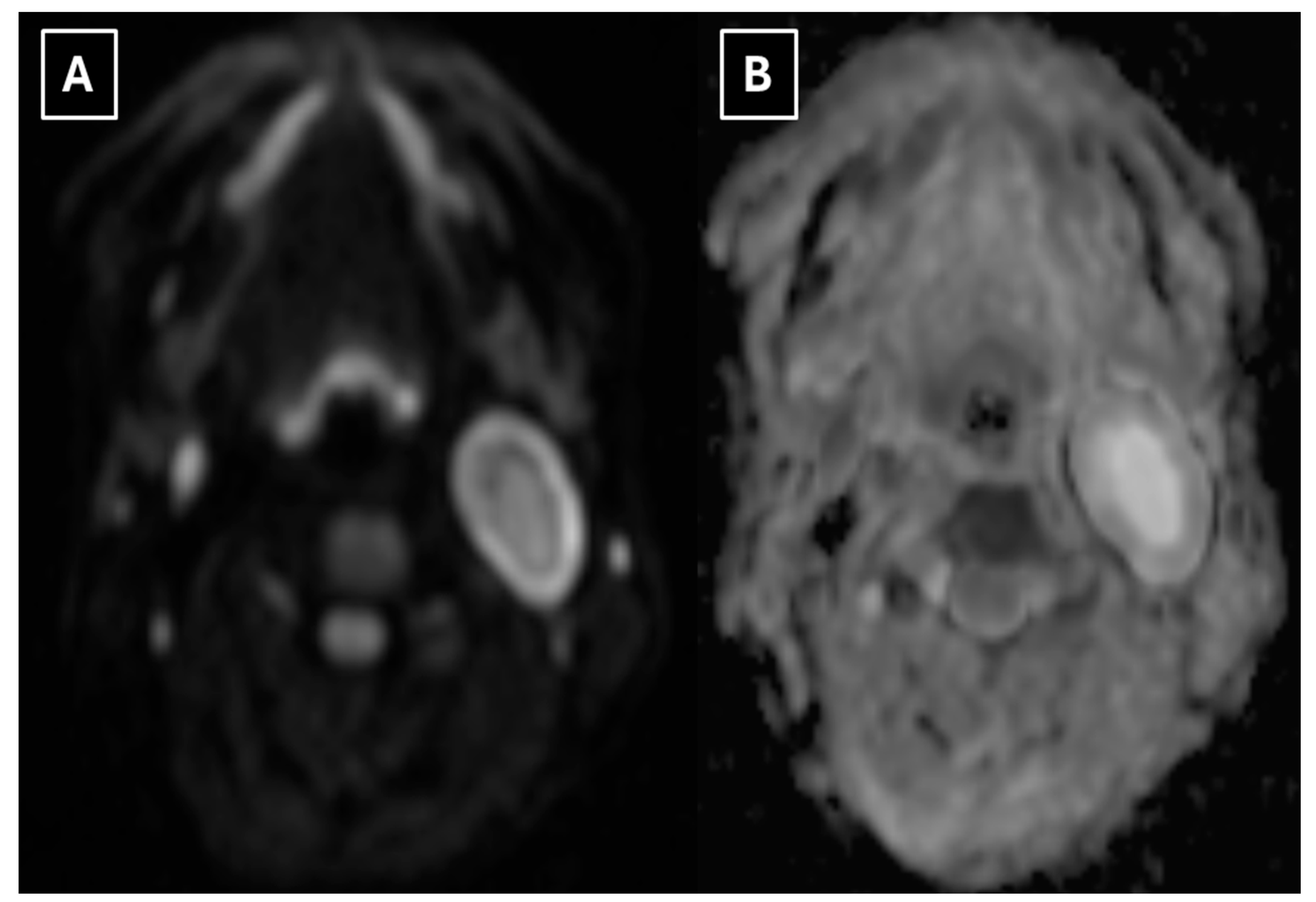
Although malignant peripheral nerve sheath tumors (MPNST) are rarely seen in the neck, given the observed diffusion restriction (
Figure 3), these tumors should be considered in the differential diagnosis. This includes neurogenic sarcomas, malignant schwannomas, and neurofibrosarcomas [
11,
12]. About half of these tumors are associated with neurofibromatosis type 1 [
12,
13]. At disease onset, the first symptom is the appearance of a mass; however, unlike schwannomas, this is followed by rapid, aggressive tumor growth and a relatively early onset of other symptoms due to compression of surrounding tissues [
12,
13]. MRI features of MPNST include a significantly greater mass effect, cystic changes within the tumor, a perifocal edema-like zone, and peripheral contrast enhancement [
14,
15]. The use of DWI sequences, applying ADC values as a biomarker, is important in the differential diagnosis of schwannomas from malignant peripheral nerve sheath tumors [
16]. The rationale for relying on these indicators is their high diagnostic accuracy in terms of both method specificity and sensitivity [
17]. A criterion for malignancy in nerve tumors is diffusion restriction and a minimum ADC value of ≤1 × 10⁻³ mm²/s. Even greater diagnostic accuracy for MPNST is demonstrated by values with a minimum ADC of 0.89 × 10⁻³ mm²/s and a mean ADC of 1.15 × 10⁻³ mm²/s [
18]. In schwannomas, high minimum ADC values > 1.1 – 1.2 (×10⁻³ mm²/s) are classically observed [
19]. Therefore, a comprehensive evaluation of all MRI findings is essential for correct identification of the pathology. In unclear cases, histopathology and immunohistochemistry play a crucial role.
In this case, the mean ADC value in the central part of the mass was 2.8 × 10⁻³ mm²/s, but in the peripheral part 1.8 – 1.9 × 10⁻³ mm²/s. Despite the diffusion restriction seen in
Figure 3, the ADC map values did not decrease significantly either centrally or peripherally, falling outside of the previously specified malignancy criteria, indicating a benign tumor.
Metastatic lesions of the carotid space also pose a diagnostic challenge, especially in cases where a primary tumor has not been diagnosed. Metastasis to cervical lymph nodes is characteristic of carcinomas of various localizations, such as breast, lung, pharynx, and thyroid [
9]. In imaging, their differentiation is aided by their effect on adjacent structures and their position relative to the carotid arteries. Metastatic lymphadenopathy is variable and does not show the characteristic effect on the carotid artery seen in schwannomas [
20]. Lymphoma often also presents as painless lymphadenopathy and is frequently localized in the neck region. CT examination with lymphoma-specific features (lymph node conglomerate) helps distinguish it from other lesions [
9,
20].
A similar appearance may be mimicked by endocrinologically inactive carotid body tumors—paragangliomas—whose only manifestation may be a mass in the neck region [
6]. Unlike schwannomas, they are well vascularized, which helps to differentiate them using CT and MR methods. Paragangliomas show intense contrast enhancement on CT (hypervascularization) and a well-defined mass effect with a high T2 signal on MR images [
9,
20]. The topography of carotid space lesions also helps in differentiation. Paragangliomas are characterized by changes in the region of the carotid bifurcation, whereas in accessory nerve schwannomas, which run along the anterior aspect of the carotid sheath, the carotid arteries are displaced anteromedially [
9,
20].
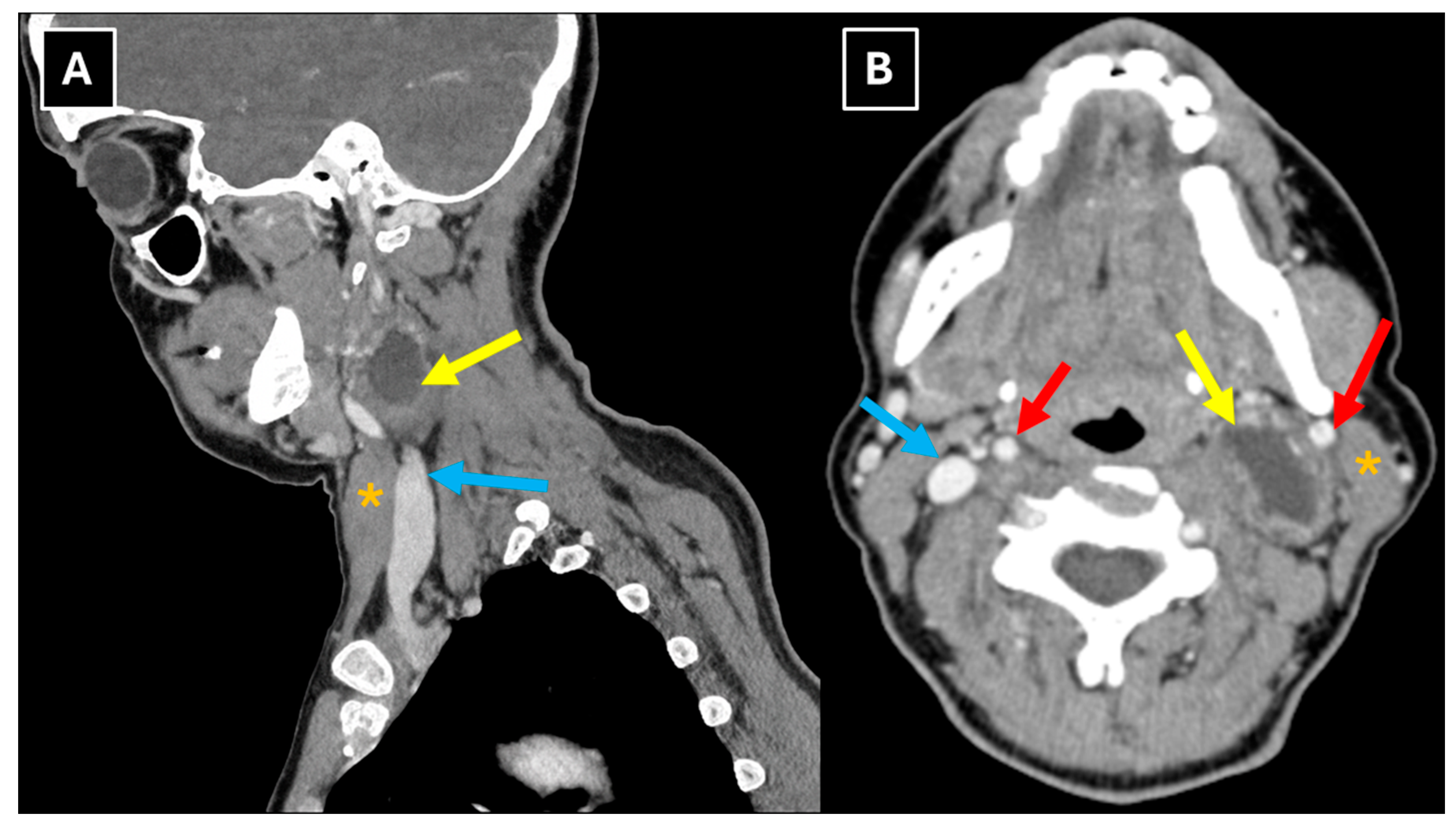
In clinical practice, the most commonly observed vascular separation patterns in the carotid space help distinguish a sympathetic chain schwannoma from an accessory nerve schwannoma. The accessory nerve passes anteriorly through the carotid sheath. In cases of schwannoma arising from this nerve, the mass is located between the carotid artery and the internal jugular vein and most often separates the vessels, displacing the artery medially and the vein laterally [
21]. The sympathetic chain is positioned posteromedial to the carotid artery, and in cases of schwannoma, it displaces the carotid artery and the jugular vein together anteriorly and laterally without separating them [
7,
21,
22]. Other atypical vascular displacement patterns are encountered less frequently; however, in clinical diagnosis, the relationship of the mass to the vessels is far more important than the displacement itself [
21]. We wish to emphasize the importance of this fact for accurate nerve identification because, in the presented case, insufficient attention was paid to the spatial relationship between the mass and the blood vessels.
Localized neurofibromas may also resemble schwannomas, but they are more closely associated with the affected nerve and characteristically grow more longitudinally. In imaging, their relationship with the affected nerve helps to differentiate them [
9].
The main diagnostic methods for accessory nerve schwannomas are CT of the neck soft tissues with intravenous contrast enhancement and magnetic resonance imaging of the neck soft tissues with contrast enhancement [
2,
10]. In some clinical case reports, ultrasound and fine-needle aspiration (FNA) biopsy were used first in primary diagnostics, followed only later by CT or MRI [
2,
10]. Many authors consider the FNA procedure to be unhelpful [
7]. A pathognomonic sign of schwannomas in CT is the presence of a high-density rim around a low-density lesion [
3]. In MR examinations, schwannomas are characterized by low T1 signal intensity and high T2 signal intensity with pronounced contrast enhancement [
9,
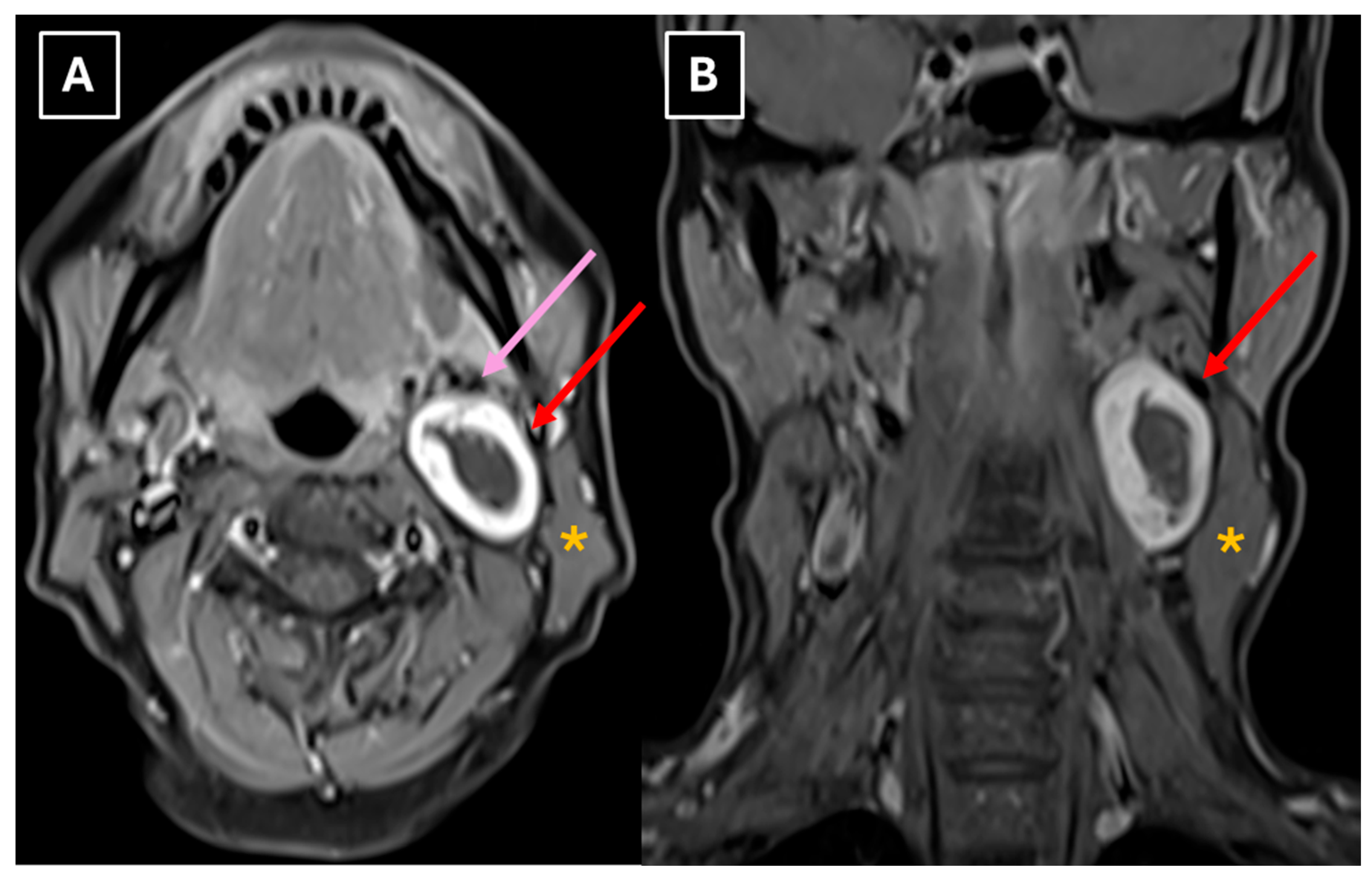
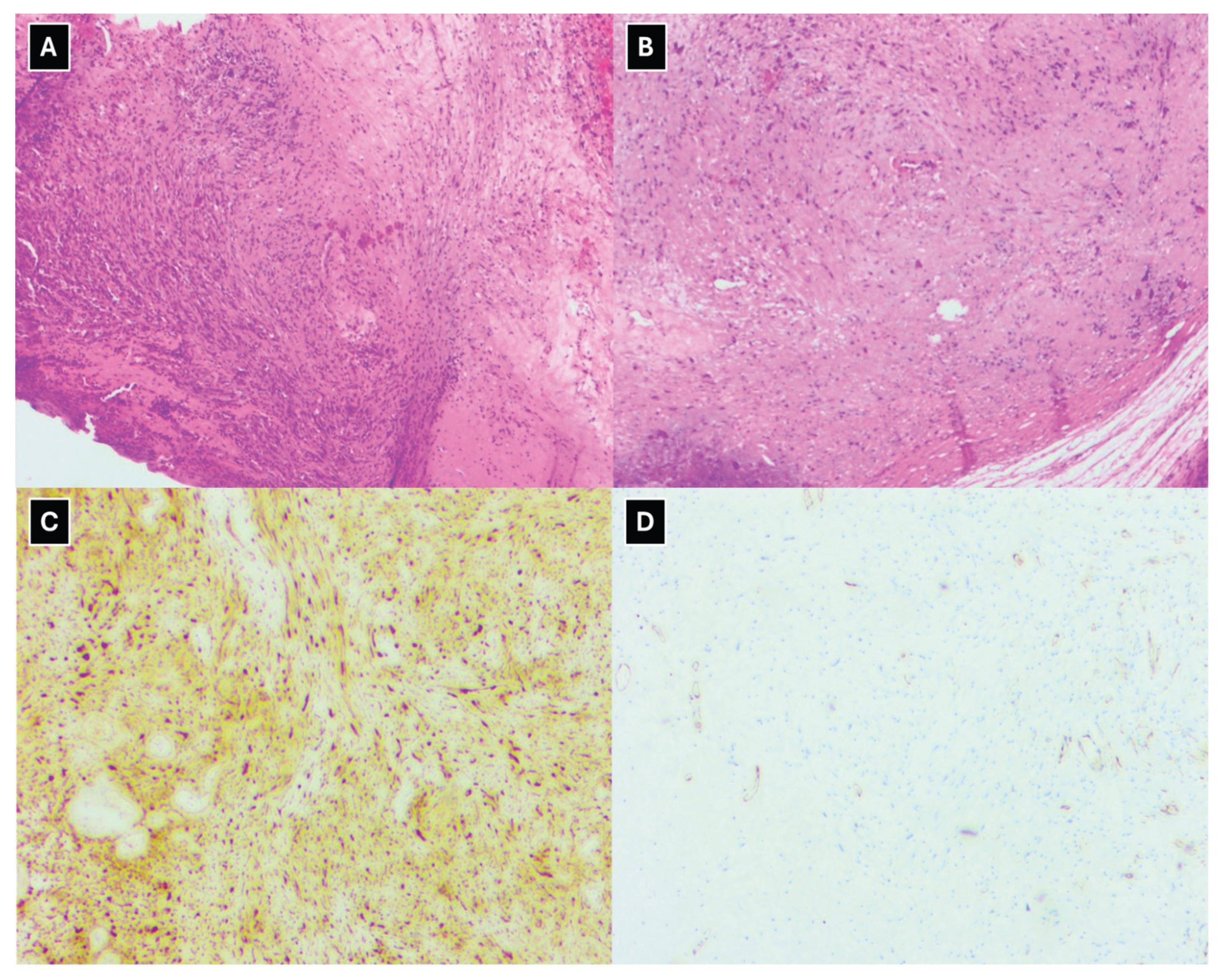
20], though the contrast enhancement is higher in Antoni type B tissue than type A. There is a known correlation between histological and MR imaging findings, which is determined by the morphological structure of the tissue. Accessory nerve schwannoma with Antoni type A tissue is characterized by increased cellularity and nuclear palisading, which on MRI appears as hyperintensity on T1W, as well as more uniform contrast enhancement [
4,
23]. Antoni type B tissue is characterized by a low number of cells, with a predominance of myxomatous stroma and cystically altered areas, which in turn are characterized by heterogeneity on MRI images [
4,
24]. This type is characterized by T1W isointensity and hyperintensity on T2W and contrast-enhanced T1W. Consequently, a heterogeneous tumor seen on MR images histologically shows a predominance of Antoni type B tissue, and this correlation is associated with cystic degeneration of the tumor and hemosiderin deposition [
4,
23,
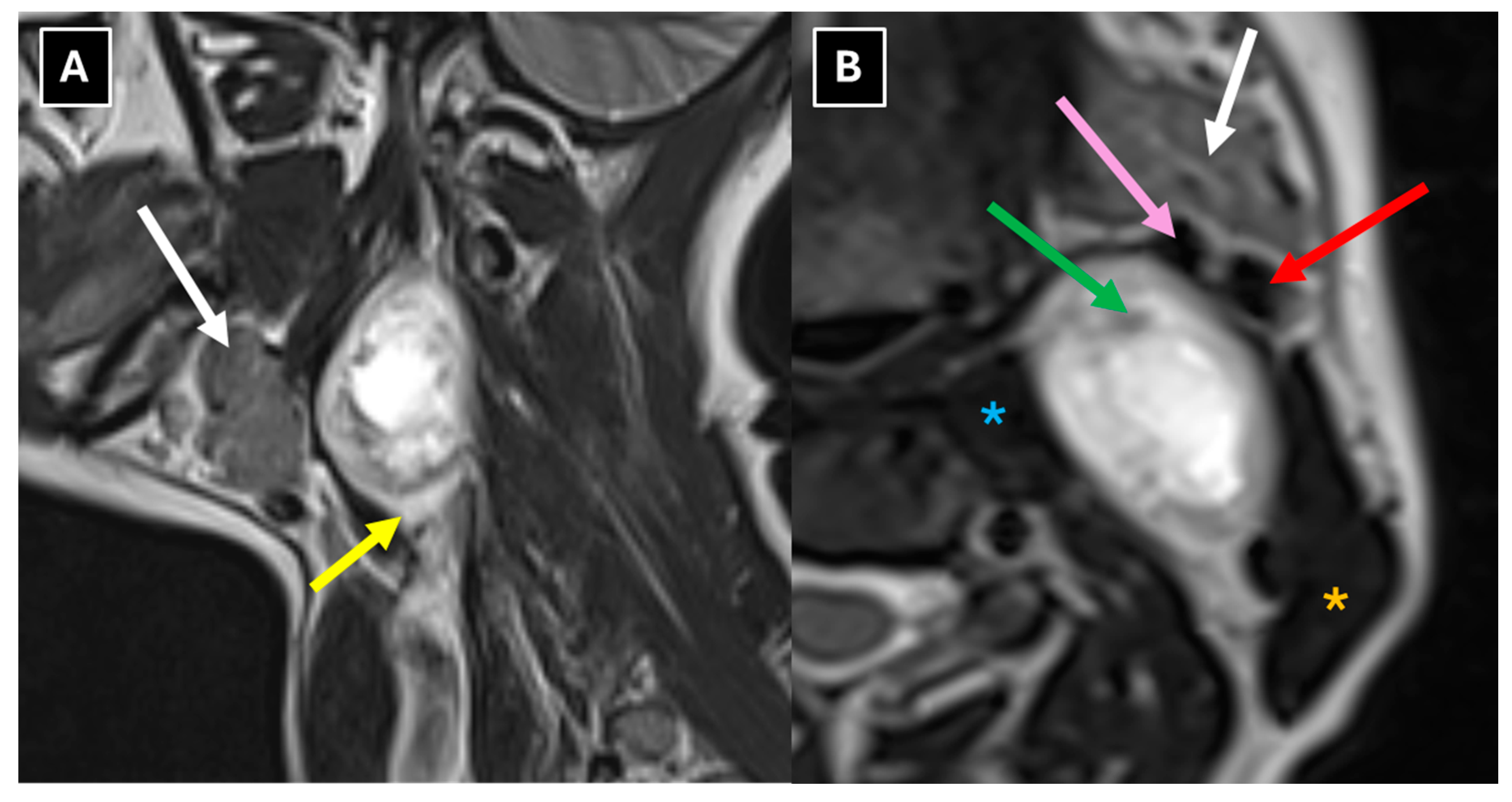
24]. In this case, Antoni type A tissue primarily made up the peripheral part of the mass, which correlates with the uniformly hyperintense contrasted rim seen in
Figure 5. Additionally, the increased cellularity of Antoni type A tissue presented as peripheral diffusion restriction seen in
Figure 3. However, Antoni type B tissue was observed in the central destructive-cystic cavity, which corresponded to the hyperintense central part of the mass seen in
Figure 2 and
Figure 4, as well as the hypointense zone in
Figure 5.
There are other characteristic MRI features that can help identify schwannomas, such as the split-fat sign, fascicular sign, and target sign. The split-fat sign has a thin perifocal fat tissue rim in non-fat-suppressed sequences and was observed in this case (
Figure 4). The fascicular sign presents with multiple small round structures with a peripheral hyperintense signal, which was also observed in this case (
Figure 4). Extracranial schwannomas exhibit the target sign more frequently than intracranial schwannomas, although it was not present in this case. The target sign is characterized by central hypointensity and peripheral hyperintensity on T2-weighted images.
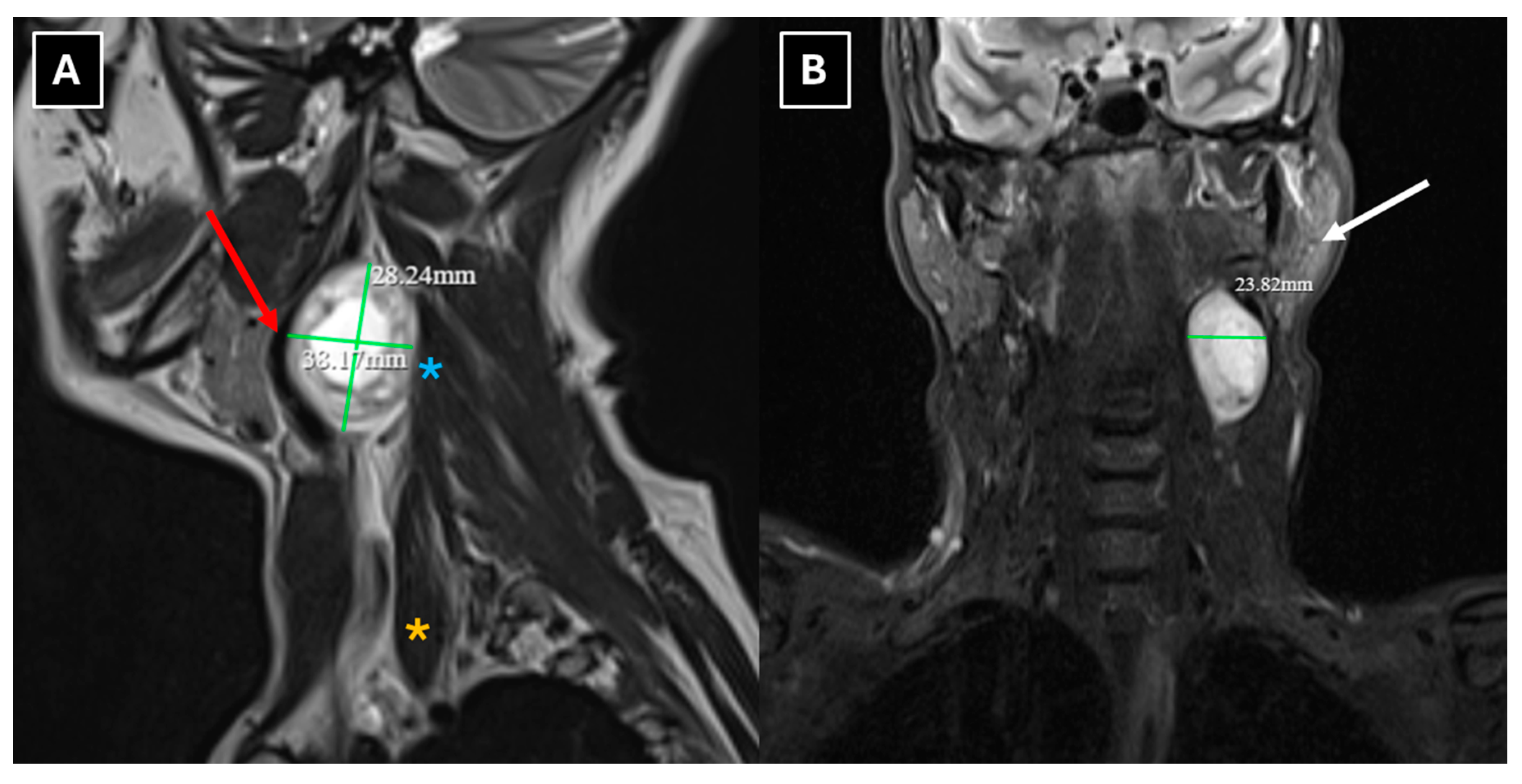
In this case, the patient first underwent a CT examination of the neck soft tissues with intravenous contrast administration, which revealed a well-defined cystic lesion on the left side of the neck, posterior to the sternocleidomastoid muscle, with a thick, contrast-enhancing capsule (
Figure 1). The radiological conclusion was a parapharyngeal neoplasm with an effect on the carotid arteries and the jugular vein. In some cases of accessory nerve schwannoma described in the literature, CT images showed a well-defined, hypodense, non–contrast-enhancing lesion [
2], but in others, an isodense lesion with hypodense areas, regions of necrosis, and calcifications [
8,
10]. It should be noted that in this clinical case, CT examination revealed an effect on the internal jugular vein, compressing it and resulting in partial venous thrombosis, which was clinically asymptomatic. Analysis of the available literature showed that only one case reported CT findings indicating involvement of the internal jugular vein [
10].
The MRI method provides relatively accurate diagnostics, and many authors consider it superior at differentiating the nerve from which the lesion originates [
8]. Both in this case and in schwannoma cases described in the literature, MRI demonstrates characteristic features such as an isointense signal on T1 (
Figure 5), a hyperintense and heterogeneous signal on T2 (
Figure 2 and
Figure 4), intense peripheral contrast enhancement, often central hypodensity, a high signal on diffusion sequences, a high ADC coefficient, and lower values peripherally [
2].
However, as demonstrated by this clinical case, the tumor type—schwannoma—was accurately determined, but the etiology was not precisely identified. Initially, it was associated with a schwannoma of the sympathetic chain rather than the accessory nerve. This can be explained by the extreme rarity of this pathology, which affects the radiologist’s ability to very precisely recognize schwannomas and the relationships between the nerve itself and the vascular structures. The literature also contains data on preoperative diagnostic difficulties in cervical schwannomas—the correct nerve pathology was identified in 83% of cases [
7].
This clinical case also demonstrates histological changes consistent with schwannoma in the surgical material, with cells of a specific nature, histochemically highlighting positive S-100 protein as a specific schwannoma biomarker, which is also noted by other authors [
3,
7,
10,
25].
In cases of schwannoma, the treatment of choice is surgery. In this case, surgery was performed with a preoperative radiological diagnosis most likely indicating a sympathetic chain schwannoma; however, intraoperatively, the lesion’s association with the accessory nerve was confirmed. Successful separation of the schwannoma from the nerve and its resection were performed without causing postoperative complications—weakness of the trapezius muscle and sternocleidomastoid muscle. The literature describes cases with complications in which weakness of the corresponding muscles persisted for several months after surgery [
2].