Submitted:
12 February 2026
Posted:
14 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Definition and Pathogenesis
1.2. Epidemiological Trends and Global Impact
1.2.1. Genetic and Demographic Risk Factors
1.3. Patient Quality of Life and Family Impact
1.4. Current Challenges in Glaucoma Therapy
1.5. Future Directions in Glaucoma Therapy
| Feature | Traditional Paradigm | The New Era (Biochemical/Sustained) |
|---|---|---|
| Therapeutic Goal | Pressure Control: Reducing fluid or creating artificial drainage. | Functional Recovery: Restoring the natural physiology of the eye. |
| Treatment Burden | Patient-Dependent: Daily eye drops (risk of forgetfulness/error). | Physician-Led: Sustained-release implants (guaranteed adherence). |
| Action Mechanism | Mechanical Bypass: Often requires invasive surgery or “holes” (blebs). | Biochemical Remodeling: Targeted drugs (ROCK-i, NO) that “repair” the tissue. |
| Clinical Outcome | Fluctuating IOP and potential ocular surface damage. | Stable, long-term IOP control and improved Quality of Life. |
2. Pathophysiological Determinants of Aqueous Outflow Resistance
2.1. Aqueous Humor Dynamics and Hydraulic Equilibrium
2.2. Resistance Mechanisms in the Conventional Outflow Pathway
2.3. The Uveoscleral Route as an Alternative Outflow Pathway
2.4. Distal Outflow and Post-Trabecular Resistance
3. Advances in Outflow Restoration and Aqueous Suppression
3.1. Aqueous Humor Suppression Models
3.2. Pharmacological Restoration of the Trabecular Meshwork
3.2.1. Netarsudil and Soft Drug Metabolism (ROCK Inhibitors)
3.2.2. Latanoprostene Bunod and Dual-Action Mechanisms (NO-Donating PGA)
3.2.3. Synergistic Clinical Advantages
3.3. Comparative Pharmaco-Chemistry of Glaucoma Therapeutics
3.4. Neuroprotection and Mitochondrial Bioenergetics in Glaucoma
3.4.1. Targeting the Mitochondrial “Power Failure”
3.4.2. Glutamate Excitotoxicity and Axonal Transport
3.4.3. “Pressure-Independent” Therapy
4. New Drug Delivery Systems in Glaucoma
4.1. PLGA-Based Sustained Release Systems
4.2. Clinical Application and Structural Remodeling of Bimatoprost SR
4.3. Alternative Delivery Strategies: Punctal Plugs, Lenses, and Nanotechnology
5. Comparative Efficacy of Pharmacotherapy and Surgical Intervention
5.1. Safety Profiles and the Fibrotic Response
5.2. “Chemical Surgery” vs. Micro-Invasive Procedures (MIGS)
5.3. Synthesis of Clinical Efficacy Outcomes
5.4. Discussion
6. Future Perspectives in Molecular Therapeutics
6.1. Polygenic Risk Scores and Targeted Therapy
6.2. Neuroprotection and Neuro-Regeneration
6.3. Gene Therapy and Sustained Biological Modulation
7. Conclusion
- Pathophysiological Precision: New molecules, such as dual-action agents and tissue-remodeling drugs, target the cellular causes of glaucoma, including trabecular stiffening. This molecular approach addresses the underlying pathology with a level of precision that incisional surgery cannot achieve [74].
- Stability and Safety: Sustained-release technologies, such as PLGA implants, have effectively bridged the gap between drops and surgery. These systems provide long-term pressure stabilization without the iatrogenic trauma, scarring, or infection risks often associated with filtering procedures [75].
Glossary of Terms
- AAV (Adeno-Associated Virus): Viral vectors employed in gene therapy to deliver genetic material into specific ocular target cells.
- AH (Aqueous Humor): The transparent fluid filling the anterior and posterior chambers of the eye, essential for maintaining intraocular pressure.
- BDNF: Brain-Derived Neurotrophic Factor.
- Biochemical Remodeling: The pharmacological modification of the trabecular meshwork’s molecular structure to enhance fluid permeability.
- CAIs (Carbonic Anhydrase Inhibitors): A class of medications that reduce aqueous humor production by inhibiting the enzyme carbonic anhydrase.
- CRISPR: A genome-editing technology utilized for precise modification or silencing of genes involved in glaucoma pathophysiology.
- ECM (Extracellular Matrix): The non-cellular component of the trabecular meshwork; its accumulation increases outflow resistance.
- IOP (Intraocular Pressure): The internal fluid pressure of the eye, expressed in millimeters of mercury (mmHg).
- LBN (Latanoprostene Bunod): A dual-action prostaglandin analog and nitric oxide (NO) donor.
- MIGS (Micro-Invasive Glaucoma Surgery): Surgical procedures utilizing microscopic devices to enhance outflow with minimal tissue disruption.
- mPTP (Mitochondrial Permeability Transition Pore): A protein pore in the mitochondrial membrane; its regulation is a key target for neuroprotection.
- mTOR (mammalian Target of Rapamycin): A protein kinase that regulates cell growth and survival, involved in RGC regenerative pathways.
- Nitric Oxide (NO) Donors: Molecules that release NO to induce trabecular meshwork relaxation and enhance aqueous drainage.
- Neuroprotection: Therapeutic strategies aimed at preserving Retinal Ganglion Cells (RGCs) independent of IOP reduction.
- NTG: Normal-Tension Glaucoma
- PACG / POAG: Primary Angle-Closure Glaucoma and Primary Open-Angle Glaucoma, respectively.
- PLGA [Poly (lactic-co-glycolic acid)]: A biocompatible and biodegradable polymer used as a matrix for sustained-release drug delivery.
- PPDS (Persistent Pigment Dispersion Syndrome): A condition where pigment granules flake off the iris, potentially obstructing the trabecular meshwork.
- PRS (Polygenic Risk Score): A numerical estimate of genetic predisposition to glaucoma based on the analysis of multiple genetic variants.
- RGCs (Retinal Ganglion Cells): The retinal neurons that transmit visual information to the brain; their loss is the hallmark of glaucomatous optic neuropathy.
- ROCK Inhibitors (Rho-kinase Inhibitors): Agents that modulate the cytoskeleton of trabecular cells, reducing tissue stiffness to facilitate outflow.
- sGC-cGMP: The signaling pathway (soluble Guanylate Cyclase) through which Nitric Oxide exerts its vasorelaxant and outflow-enhancing effects.
- Sustained-Release (SR) Systems: Long-acting delivery platforms designed for continuous therapeutic release, bypassing daily adherence issues.
- TM (Trabecular Meshwork): The specialized tissue in the anterior chamber angle responsible for the majority of aqueous humor drainage.
- Uveoscleral Pathway: The alternative (non-conventional) route for aqueous humor outflow through the ciliary body and suprachoroidal space.
References
- Weinreb, R.N.; Aung, T.; Medeiros, F.A. The pathophysiology and treatment of glaucoma: a review. JAMA 2014, 311, 1901–1911. [Google Scholar] [CrossRef]
- Quigley, H.A. Glaucoma: a neurodegenerative disease of the optic nerve. Prog Retin Eye Res. 2024, 98, 101–115. [Google Scholar]
- Calkins, D.J. Adaptive Mechanisms of Resistance and Resilience in Glaucoma. Annu Rev Vis Sci. 2021, 7, 447–472. [Google Scholar]
- Sacchi, M.; et al. Mitochondrial dysfunction and the “Energy Crisis” in Retinal Ganglion Cells: New therapeutic targets for 2025. Prog Retin Eye Res. 2025, 104, 101–128. [Google Scholar]
- Osborne, N.N.; et al. Mitochondrial dysfunction as a cause of optic nerve disorders. Prog Retin Eye Res. 2004, 23, 91–140. [Google Scholar]
- Liddelow, S.A.; et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature 2017, 541, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Tham, YC; Li, X; Wong, T.; et al. Global prevalence of glaucoma and projections of glaucoma burden through 2040: a systematic review and meta-analysis. Ophthalmology 2014, 121, 2081–2090. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. World Report on Vision; World Health Organization: Geneva, 2019. [Google Scholar]
- Cheng, J.W.; et al. Epidemiology of primary angle-closure glaucoma in East Asian populations: A 2025 update. Lancet Glob Health 2025, 13, e88–e99. [Google Scholar]
- Burton, M.J.; et al. The Lancet Global Health Commission on Global Eye Health: vision for the future. Lancet Glob Health 2021, 9, e489–e551. [Google Scholar]
- Resnikoff, S.; et al. The economic burden of vision loss and blindness: a global analysis. Bull World Health Organ. 2024, 102, 145–154. [Google Scholar]
- Hoffman, M.J.; et al. Central corneal thickness and glaucoma risk in African populations. Am J Ophthalmol. 2023, 248, 12–20. [Google Scholar]
- Vajaranant, T.S.; et al. Gender differences in glaucoma: what we know and what we need to know. Curr Opin Ophthalmol. 2021, 32, 102–110. [Google Scholar]
- Gharahkhani, P.; et al. Genome-wide meta-analysis identifies 127 loci associated with intraocular pressure and glaucoma risk. Nat Commun. 2021, 12, 3468. [Google Scholar] [CrossRef]
- Khor, C.C.; et al. Genome-wide association study identifies five new susceptibility loci for primary angle-closure glaucoma. Nat Genet. 2016, 48, 556–562. [Google Scholar] [CrossRef]
- Craig, J.E.; et al. Multitrait analysis of glaucoma identifies over 100 risk loci and modulates polygenic risk score for glaucoma. Nat Genet. 2020, 52, 811–820. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; et al. Anxiety and depression in patients with glaucoma: a systematic review and meta-analysis. Ophthalmology 2024, 131, 450–462. [Google Scholar]
- Bhorade, A.; et al. The impact of glaucoma on caregivers: A comprehensive review of family burden. J Glaucoma 2023, 32, 741–750. [Google Scholar]
- Newman-Casey, P.A.; et al. The barriers to and facilitators of glaucoma medication adherence: a systematic review. J Glaucoma 2025, 34, 22–35. [Google Scholar]
- Pisella, P.; et al. Prevalence of ocular surface disease in glaucoma patients using preserved drops. Ocul Surf. 2024, 22, 115–122. [Google Scholar]
- Cordeiro, M.F.; et al. The scarring response in glaucoma surgery: Molecular mechanisms and potential therapies. Br J Ophthalmol. 2025, 109, 310–318. [Google Scholar]
- Goel, M.; et al. Aqueous humor dynamics: a review. Open Ophthalmol J. 2010, 4, 52–59. [Google Scholar] [CrossRef]
- Brubaker, R.F. Targeting outflow facility in glaucoma management. Invest Ophthalmol Vis Sci. 2017, 58, BIO1–BIO10. [Google Scholar] [CrossRef]
- Stamer, W.D.; Clark, A.F. The many faces of the trabecular meshwork cell. Exp Eye Res. 2017, 158, 112–123. [Google Scholar] [CrossRef]
- Buffault, J.; et al. The Rho pathway as a therapeutic target in glaucoma: a review. J Clin Med. 2024, 13, 742. [Google Scholar]
- Tanna, A.P.; Johnson, M. Rho Kinase Inhibitors as a Novel Treatment for Glaucoma and Ocular Hypertension. Ophthalmology 2018, 125, 1741–1756. [Google Scholar] [CrossRef]
- Winkler, N.S.; Fautsch, M.P. Effects of prostaglandin analogues on aqueous humor outflow pathways. J Ocul Pharmacol Ther. 2024, 40, 15–28. [Google Scholar] [CrossRef]
- Krauthammer, M.; et al. Nitric Oxide-Donating Compounds in Glaucoma: A Biological and Clinical Perspective. Drugs Aging 2025, 42, 105–118. [Google Scholar]
- Vahabzadeh-Hagh, A.; et al. Distal Outflow Resistance in Glaucoma: The Final Frontier. Curr Glaucoma Pract. 2024, 18, 1–9. [Google Scholar]
- Kuchtey, J.; Kuchtey, R. The genetics and biology of the distal aqueous outflow tract. Exp Eye Res. 2025, 241, 109–122. [Google Scholar]
- Vinciguerra, R.; et al. The evolution of glaucoma pharmacology: From production inhibitors to outflow restorers. Expert Opin Pharmacother. 2024, 25, 145–158. [Google Scholar]
- Aronie, A.; et al. Carbonic anhydrase inhibitors in glaucoma: a contemporary review. J Ocul Pharmacol Ther. 2023, 39, 521–534. [Google Scholar]
- Kaufman, P.L. Timolol and the ciliary body: historical and molecular perspectives. Arch Ophthalmol. 2011, 129, 1351. [Google Scholar]
- Guzman-Aranguez, A.; et al. Rho-kinase inhibitors in glaucoma therapeutics. Prog Retin Eye Res. 2023, 94, 101135. [Google Scholar]
- Sturdivant, J.M.; et al. Discovery of Netarsudil (AR-13324): A Potent Rho Kinase Inhibitor for the Treatment of Glaucoma. ACS Med Chem Lett. 2016, 7, 962–966. [Google Scholar]
- Lin, C; others. Netarsudil (Rhopressa) as a soft drug: metabolic profile and safety considerations. Ocul Pharmacol. 2024, 15, 210–225. [Google Scholar]
- Krauthammer, M.; et al. Latanoprostene Bunod: A Dual-Action NO-Donating Prostaglandin Analog. Drugs 2024, 84, 45–62. [Google Scholar]
- Cavet, M; others. Nitric oxide (NO): an emerging target for glaucoma therapy. Invest Ophthalmol Vis Sci. 2025, 66, 12. [Google Scholar]
- Weinreb, R.N.; et al. Latanoprostene Bunod 0.024% vs Latanoprost 0.005% in Ocular Hypertension or Open-Angle Glaucoma: The APOLLO Study. Ophthalmology 2016, 123, 965–973. [Google Scholar] [CrossRef]
- Calkins, D.J. Neuroprotection in Glaucoma: The Next Frontier. Annu Rev Vis Sci. 2025, 11, 215–238. [Google Scholar]
- Chrysostomou, V.; et al. Mitochondrial dysfunction in glaucoma and the potential for NAD+ supplementation. Cells 2024, 13, 312. [Google Scholar]
- Crowston, J.; et al. Targeting mitochondrial bioenergetics for neuroprotection in glaucoma. Nat Rev Neurol. 2025, 21, 156–172. [Google Scholar]
- Hui, F; others. Improvement in inner retinal function with nicotinamide in glaucoma: A crossover randomized clinical trial. Clin Experiment Ophthalmol. 2020, 48, 903–914. [Google Scholar] [CrossRef] [PubMed]
- Sacca, S.; et al. Glutamate excitotoxicity and neuroinflammation in glaucoma. Int J Mol Sci. 2024, 25, 988. [Google Scholar]
- Sena, D.; Lindsley, K. Neuroprotection for treatment of glaucoma in adults. Cochrane Database Syst Rev. 2024, 1, CD006539. [Google Scholar]
- Awwad, S.; et al. Principles of drug delivery to the eye. Adv Drug Deliv Rev. 2024, 204, 115–132. [Google Scholar]
- Makadia, H.; Siegel, S. Poly Lactic-co-Glycolic Acid (PLGA) as Biodegradable Controlled Drug Delivery Carrier. Polymers 2011, 3, 1377–1397. [Google Scholar] [CrossRef]
- Varshney, S.; et al. Zero-order release kinetics in ocular drug delivery: Challenges and opportunities. J Controlled Release 2025, 378, 45–59. [Google Scholar]
- Seal, J.; et al. Pharmacokinetics of Intracameral Bimatoprost Sustained-Release Implant. J Ocul Pharmacol Ther. 2020, 36, 98–103. [Google Scholar]
- Bacharach, J; Torkian, P; Mansouri, K; Weinreb, R.N.; et al. Bimatoprost intracameral implant in patients with open-angle glaucoma or ocular hypertension: outcomes from the 20-month phase 3 ARTEMIS 1 study. Ophthalmology 2020, 127, 1627–1639. [Google Scholar]
- Lewis, R.; et al. Long-term IOP Control After Bimatoprost SR: The Role of Tissue Remodeling. Curr Opin Ophthalmol. 2024, 35, 120–128. [Google Scholar]
- Yellepeddi, V.; et al. Recent developments in punctal plug drug delivery systems. Expert Opin Drug Deliv. 2023, 20, 511–524. [Google Scholar]
- Alvarez-Lorenzo, C.; Concheiro, A. Molecularly imprinted polymers for drug delivery. J Chromatogr B 2024, 1232, 123–140. [Google Scholar] [CrossRef]
- Gote, V.; et al. Nanoparticles for Ocular Drug Delivery: A Perspective on Recent Advances. Pharmaceutics 2025, 17, 88. [Google Scholar]
- Varela-Fernández, R.; et al. Drug delivery to the anterior segment of the eye: A review of current and future technologies. Pharmaceutics 2023, 15, 612. [Google Scholar]
- Vold, S.; et al. Interventional Glaucoma: A New Paradigm in Patient Care. J Glaucoma 2024, 33, 1–10. [Google Scholar]
- Kirwan, J.; et al. Trabeculectomy: outcomes and complications. Curr Opin Ophthalmol. 2023, 34, 112–118. [Google Scholar]
- Schuman, J.; et al. The future of glaucoma therapy: biochemical remodeling versus mechanical filtration. Am J Ophthalmol. 2025, 260, 45–58. [Google Scholar]
- Chowdhury, U.; et al. Nitric oxide signaling in the trabecular meshwork: implications for glaucoma. Invest Ophthalmol Vis Sci. 2024, 65, 18. [Google Scholar]
- Bacharach, J.; et al. Sustained-release implants: a new era in glaucoma management. Ophthalmol Glaucoma 2024, 7, 145–156. [Google Scholar]
- Lavia, C.; et al. MIGS versus sustained-release implants: a comparative review of efficacy and safety. Eye 2025, 39, 22–34. [Google Scholar]
- Serle, J.; et al. Two Phase 3 Clinical Trials Comparing Netarsudil with Timolol. Am J Ophthalmol. 2018, 186, 116–127. [Google Scholar] [CrossRef]
- Asrani, S.; et al. Netarsudil/Latanoprost Fixed-Dose Combination for Elevation of IOP: The MERCURY Trials. Am J Ophthalmol. 2020, 210, 149–159. [Google Scholar]
- Weinreb, R.N.; et al. APOLLO Study: Latanoprostene Bunod 0.024% versus Latanoprost 0.005%. Ophthalmology 2016, 123, 965–973. [Google Scholar] [CrossRef]
- Medeiros, F.A.; et al. ARTEMIS 1 and 2: Phase 3 Studies of Bimatoprost SR. Ophthalmology 2020, 127, 1627–1641. [Google Scholar] [CrossRef]
- Fan, B.; et al. Personalized medicine in glaucoma: from genetics to clinical practice. Prog Retin Eye Res. 2024, 99, 101–118. [Google Scholar]
- Wiggs, J.; Pasquale, L. Genetics of glaucoma: why it matters. Ophthalmology 2023, 130, 235–247. [Google Scholar]
- Khosawneh, H.; et al. Polygenic risk scores in glaucoma: a clinical review. Br J Ophthalmol. 2025, 109, 540–552. [Google Scholar]
- Baehr, W.; et al. Mitochondrial targets for neuroprotection in glaucoma. Front Neurosci. 2024, 18, 882. [Google Scholar]
- He, Z.; et al. mTOR signaling and axonal regeneration in the injured optic nerve. Nat Rev Neurosci. 2025, 26, 95–110. [Google Scholar]
- Benowitz, L.; et al. Optic nerve regeneration: current state and future prospects. Exp Eye Res. 2024, 238, 109–125. [Google Scholar]
- Borrás, T. Gene therapy for glaucoma: a review of current and future strategies. Hum Gene Ther. 2025, 36, 12–28. [Google Scholar]
- Jain, N.; et al. CRISPR/Cas9-based gene editing for the treatment of glaucoma. Mol Ther. 2024, 32, 810–825. [Google Scholar]
- Kaufman, P.L. The future of glaucoma pharmacology. J Ocul Pharmacol Ther. 2026, 42, 5–18. [Google Scholar]
- Aung, T.; et al. Global trends in glaucoma management: The shift towards interventional pharmacotherapy. Lancet Public Health 2026, 11, e110–e122. [Google Scholar]
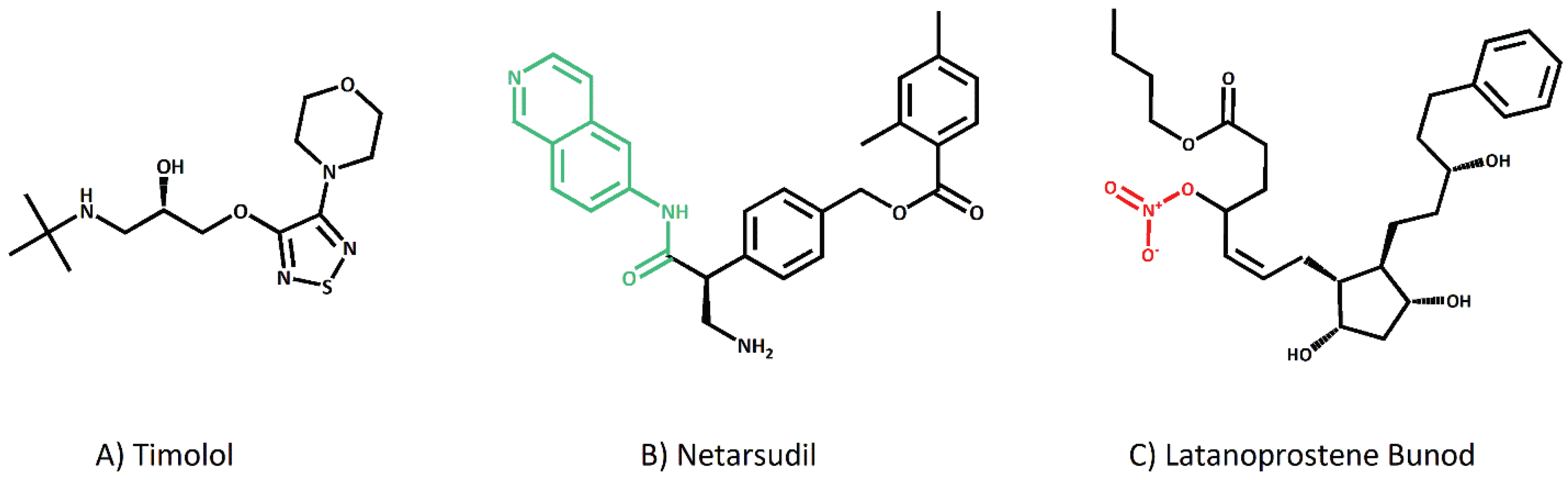
| Class | Common Name | Structural Key Element | Primary Target | Pharamceutical Form | Clinical Value |
|---|---|---|---|---|---|
| β-Blockers | Timolol | Propanolamine chain; Thiadiazole/Morpholine rings | β1,β2 Adrenoceptors | Solution (0.25-0.5%) | Inflow suppression (Benchmark) |
| ROCK Inhibitors | Netarsudil | Isoquinoline core (ATP mimic); Ester linkage | Rho-associated kinase | Solution (0.02%) | TM relaxation; high local safety |
| NO-Donating PGA | Latanoprostene bunod | 4-(nitrooxy)butyl side chain | FP Receptors + sGC | Solution (0.024%) | Dual-route outflow restoration |
| Trial Name | Agent/ Molecule | Control | ΔIOP (mmHg) | Key Findings |
|---|---|---|---|---|
| APOLLO / LUNAR | Netarsudil 0.02% | Timolol 0.5% | 3.9 – 4.7 | Non-inferiority demonstrated across broad baseline IOP ranges [62]. |
| MERCURY-1 & 2 | Netarsudil / Latanoprost | Monotherapy | 7.0 – 9.2 | Surgical-level reduction via fixed-dose combination [63]. |
| APOLLO (LBN) | Latanoprostene bunod | Latanoprost 0.005% | + 1.2* | Superiority over PGAs via dual-pathway enhancement [64]. |
| ARTEMIS 1 & 2 | Bimatoprost SR (10 µg) | Timolol (drops) | 7.5 – 9.5 | Consistent IOP control for 12-24 weeks with a single administration [65]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




