Submitted:
11 February 2026
Posted:
12 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
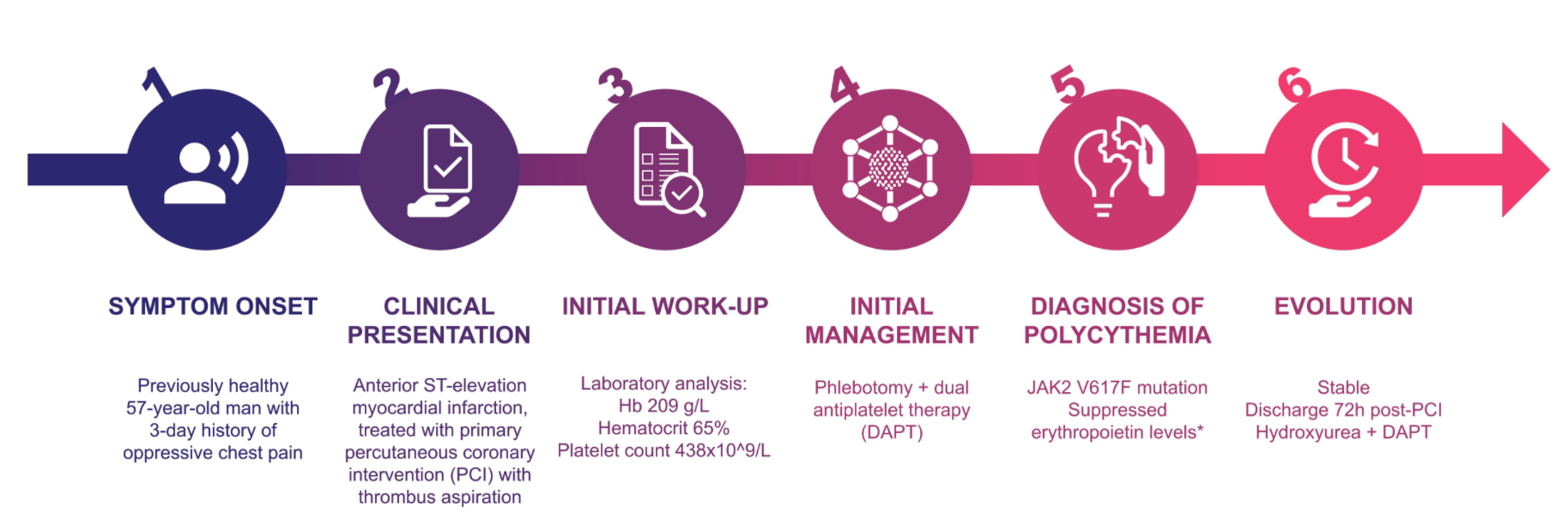
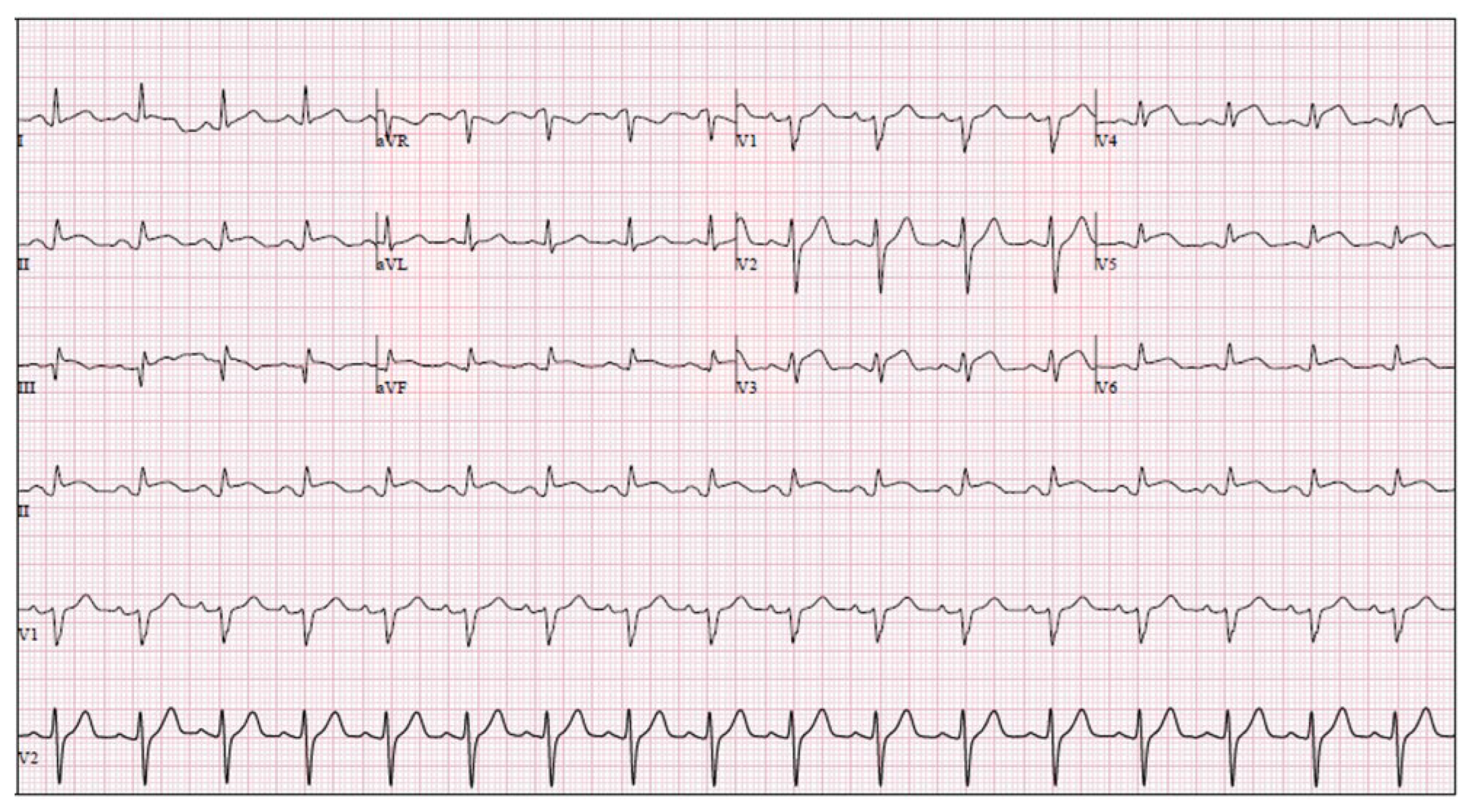
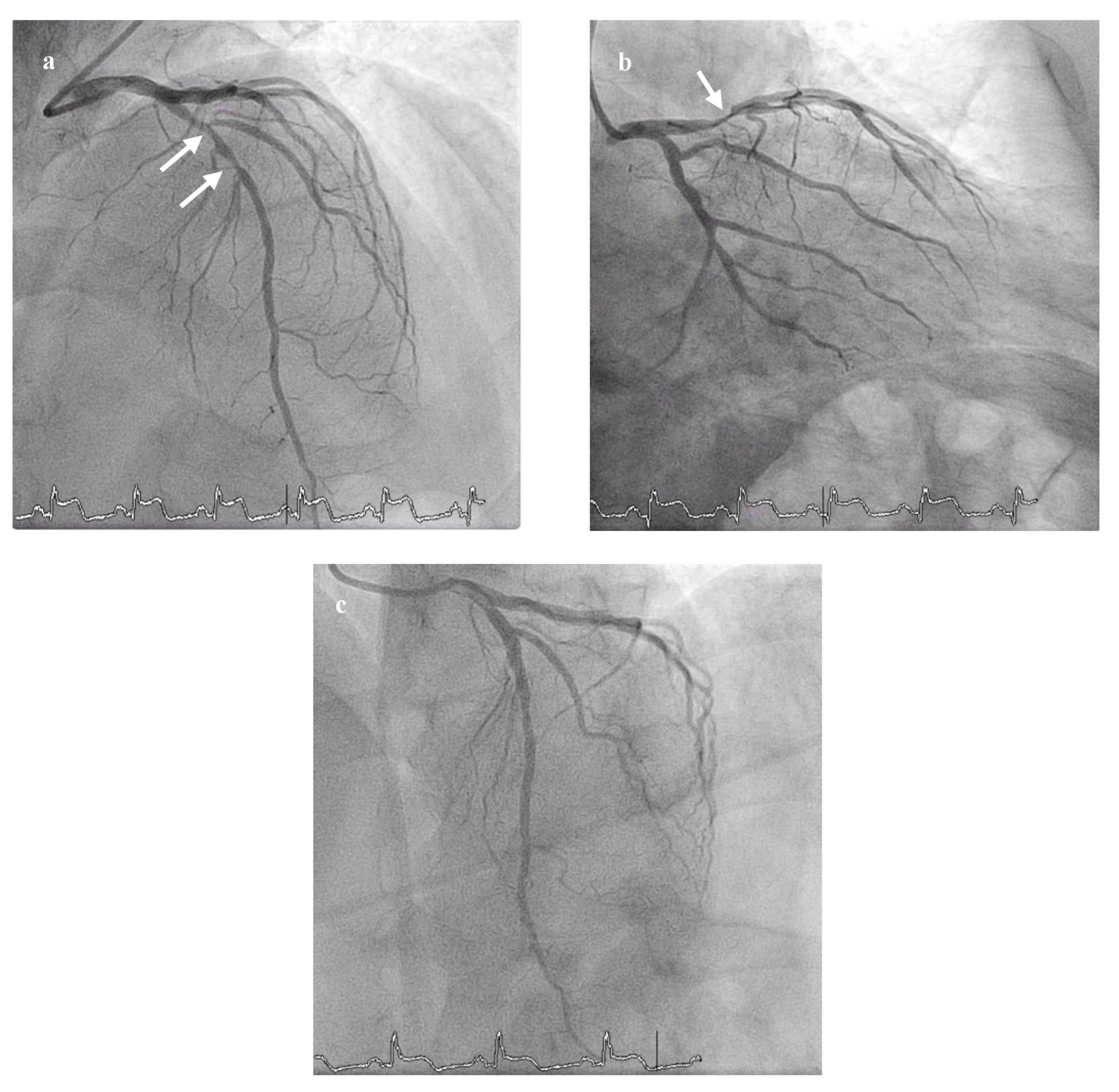
2. Case Description
4. Discussion
5. Conclusions
Supplementary Materials
Funding
Informed Consent Statement
Conflicts of Interest
Abbreviations
| ACS MDPI |
Acute Coronary Syndrome Multidisciplinary Digital Publishing Institute |
| DOAJ LAD PCI PV STEMI |
Directory of open access journals Left Anterior Descending Artery Percutaneous Coronary Intervention Polycythemia vera ST-Elevation Myocardial Infarction |
References
- Waggoner, M. Polycythemia vera: thinking beyond the hematocrit. J Adv Pract Oncol. 2023, 14(5), 405. [Google Scholar] [CrossRef] [PubMed]
- Fox, S.; Griffin, L.; Harris, D.R. Polycythemia vera: rapid evidence review. Am Fam Physician 2021, 103(11), 680–687. [Google Scholar] [CrossRef] [PubMed]
- Mesa, R.A. New guidelines from the NCCN for polycythemia vera. Clin Adv Hematol Oncol. 2017, 15(11), 848. [Google Scholar] [PubMed]
- Tefferi, A.; Barbui, T. Polycythemia vera: 2024 update on diagnosis, risk-stratification, and management. Am J Hematol. 2023, 98(9), 1465–1487. [Google Scholar] [CrossRef] [PubMed]
- Marchioli, R.; Finazzi, G.; Specchia, G.; Cacciola, R.; Cavazzina, R.; Cilloni, D.; De Stefano, V.; Elli, E.; Iurlo, A.; Latagliata, R.; Lunghi, F. Cardiovascular events and intensity of treatment in polycythemia vera. N Engl J Med. 2013, 368(1), 22–33. [Google Scholar] [CrossRef] [PubMed]
- Cerquozzi, S.; Barraco, D.; Lasho, T.; Finke, C.; Hanson, C.A.; Ketterling, R.P.; Pardanani, A.; Gangat, N.; Tefferi, A. Risk factors for arterial versus venous thrombosis in polycythemia vera: a single center experience in 587 patients. Blood Cancer J. 2017, 7(12), 662. [Google Scholar] [CrossRef] [PubMed]
- Kroll, M.H.; Michaelis, L.C; Verstovsek, S. Mechanisms of thrombogenesis in polycythemia vera. Blood Rev. 2015, 29(4), 215–221. [Google Scholar] [CrossRef] [PubMed]
- Wolach, O.; Sellar, R.S.; Martinod, K.; Cherpokova, D.; McConkey, M.; Chappell, R.J.; Silver, A.J.; Adams, D.; Castellano, C.A.; Schneider, R.K.; Padera, R..F. Increased neutrophil extracellular trap formation promotes thrombosis in myeloproliferative neoplasms. Sci Transl Med. 2018, 10(436), eaan8292. [Google Scholar] [CrossRef] [PubMed]
- Landolfi, R.; Di Gennaro, L.; Barbui, T.; De Stefano, V.; Finazzi, G.; Marfisi, R.; Tognoni, G.; Marchioli, R. Leukocytosis as a major thrombotic risk factor in patients with polycythemia vera. Blood 2007, 109(6), 2446–2452. [Google Scholar] [CrossRef] [PubMed]
- Manan, M.R.; Kipkorir, V.; Nawaz, I.; Waithaka, M.W.; Srichawla, B.S.; Gãman, A.M.; Diaconu, C.C.; Gãman, M.A. Acute myocardial infarction in myeloproliferative neoplasms. World J Cardiol. 2023, 15(11), 571. [Google Scholar] [CrossRef] [PubMed]
- Silveira, C.F.D.S.M.P.D.; Vitali, L.B.S.L.; Faustino, F.G.; Maurício, A.D.C.V.; Teixeira, R.; Bazan, S.G.Z. Acute myocardial infarction as first onset of polycythemia vera. Arq Bras Cardiol. 2020, 114, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Cengiz, B.; Aytekin, V.; Bildirici, U.; Sahin, S.T.; Yurdakul, S.; Aytekin, S.; Kucukkaya, R. A rare cause of acute coronary syndromes in young adults - myeloproliferative neoplasms: A case series. Rev Port Cardiol. 2019, 38(9), 613–617. [Google Scholar] [CrossRef] [PubMed]
- Okabe, H.; Sonoda, S.; Abe, K.; Doi, H.; Matsumura, T.; Otsuji, Y. Acute myocardial infarction following sequential multi-vessel occlusion in a case of polycythemia vera. J Cardiol Cases 2019, 20(4), 111–114. [Google Scholar] [CrossRef] [PubMed]
- Barbui, T.; Thiele, J.; Gisslinger, H.; Kvasnicka, H.M.; Vannucchi, A.M.; Guglielmelli, P.; Orazi, A.; Tefferi, A. The 2016 WHO classification and diagnostic criteria for myeloproliferative neoplasms: document summary and in-depth discussion. Blood Cancer J. 2018, 8(2), 15. [Google Scholar] [CrossRef] [PubMed]
| Blood test | Admission | Discharge | 1st Cardiology Appointment |
|---|---|---|---|
| Hemoglobin | 209 g/L | 179 g/L | |
| Haematocrit | 64.9% | 55.2% | |
| Erythrocytes | 7.91 x 109/L | 6.77 x 109/L | |
| Leucocytes | 9.0 x 109/L | 6.5 x 109/L | |
| Platelets | 438 x 109/L | 448 x 109/L | |
| LDL cholesterol | 3.47 mmol/L | ||
| HDL cholesterol | 1.06 mmol/L | ||
| Triglycerides | 0.97 mmol/L | ||
| hs-Troponin I | 6796 pg/mL | 2752 pg/mL | |
| Erythropoietin | < 0,6 mU/mL | ||
| JAK2 kinase mutation | JAK-2 V617F mutation: positive |
| Major criteria |
|---|
| Hemoglobin > 16.5 g/dL (men) / > 16.0 g/dL (women) or Haematocrit > 49% (men) / > 48% (women) or increased red cell mass (RCM) |
| Bone Marrow biopsy showing hypercellularity for age with trilineage growth (panmyelosis) including prominent erythroid, granulocytic and megakaryocytic proliferation with pleomorphic, mature megakaryocytes (differences in size) |
| Presence of JAK2 or JAK2 exon 12 mutation |
| Minor criteria |
| Subnormal serum erythropoietin level |
| A diagnosis of PV can be established by satisfying the complete triad of major criteria, or by meeting the first two major criteria plus any one minor criteria |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




