Submitted:
11 February 2026
Posted:
12 February 2026
You are already at the latest version
Abstract
Keywords:
1. introduction
2. methods
2.1. patients’ selection and data collection
2.2. levosimendan administration protocol
- heart failure with reduced ejection fraction (hfref; ef <35%) refractory to guideline-directed medical therapy
- hfref patients already listed for heart transplantation with ongoing clinical instability
- hfref patients on optimized medical therapy with worsening symptoms with new york heart association (nyha) functional class>2, or recent hf hospitalization, prior to listing for transplantation, as a last attempt to achieve clinical recovery
- heart failure with preserved ejection fraction (hfpef) unresponsive to conventional therapy.
2.3. echocardiography
2.4. endpoints
- (a)
- death for any reason
- (b)
- new transplant listing/necessity of mechanical circulatory support (mcs)
- (c)
- unscheduled hospital admission for hf
- (d)
- occurrence of sustained ventricular tachycardia(vt), defined as vt>30s or non-sustained vt with symptoms, or icd appropriate therapies
- (e)
- a composite of both all the previous
2.5. statistical analysis
3. results
3.1. study population
3.2. levosimendan treatment
3.3. follow-up and secondary endpoints
3.4. outcome
4. discussion
5. conclusion
References
- Merás, P; Riesgo-Gil, F; Rybicka, J; Barradas-Pires, A; Smith, J; Kempny, A; Brookes, P; Simon, A; Gatzoulis, MA. Heart transplantation at a single tertiary adult congenital heart disease centre: Too little, too late? Int J Cardiol 2021, 322, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Meshulami, N; Shah, P; Kaushik, S; Murthy, R. Cardiac transplantation in adult congenital heart disease: a narrative review. J Thorac Dis. 2023, 15(9), 5074–5087. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cedars, A; Vanderpluym, C; Koehl, D; Cantor, R; Kutty, S; Kirklin, JK. An Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) analysis of hospitalization, functional status, and mortality after mechanical circulatory support in adults with congenital heart disease. J Heart Lung Transplant. 2018, 37(5), 619–630. [Google Scholar] [CrossRef] [PubMed]
- Masarone, D; Kittleson, MM; Pollesello, P; Marini, M; Iacoviello, M; Oliva, F; Caiazzo, A; Petraio, A; Pacileo, G. Use of Levosimendan in Patients with Advanced Heart Failure: An Update. J Clin Med. 2022, 11(21), 6408. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Masarone, D; Kittleson, MM; Martucci, ML; Valente, F; Gravino, R; Verrengia, M; Ammendola, E; Contaldi, C; Di Palma, V; Caiazzo, A; Petraio, A; Pollesello, P; Pacileo, G. Levosimendan as a "Bridge to Optimization" in Patients with Advanced Heart Failure with Reduced Ejection-A Single-Center Study. J Clin Med. 2022, 11(14), 4227. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Peled, Y; Ducharme, A; Kittleson, M; Bansal, N; Stehlik, J; Amdani, S; Saeed, D; Cheng, R; Clarke, B; Dobbels, F; Farr, M; Lindenfeld, J; Nikolaidis, L; Patel, J; Acharya, D; Albert, D; Aslam, S; Bertolotti, A; Chan, M; Chih, S; Colvin, M; Crespo-Leiro, M; D'Alessandro, D; Daly, K; Diez-Lopez, C; Dipchand, A; Ensminger, S; Everitt, M; Fardman, A; Farrero, M; Feldman, D; Gjelaj, C; Goodwin, M; Harrison, K; Hsich, E; Joyce, E; Kato, T; Kim, D; Luong, ML; Lyster, H; Masetti, M; Matos, LN; Nilsson, J; Noly, PE; Rao, V; Rolid, K; Schlendorf, K; Schweiger, M; Spinner, J; Townsend, M; Tremblay-Gravel, M; Urschel, S; Vachiery, JL; Velleca, A; Waldman, G; Walsh, J.; Hummel, J; Rücker, G; Stiller, B.; International Society for Heart and Lung Transplantation Guidelines for the Evaluation and Care of Cardiac Transplant Candidates-2024. Prophylactic levosimendan for the prevention of low cardiac output syndrome and mortality in paediatric patients undergoing surgery for congenital heart disease. J Heart Lung Transplant.;Cochrane Database Syst Rev 2024, 43(10) 8(8), 1529–1628.e54 CD011312. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wannaz, L; Boillat, L; Perez, MH; Di Bernardo, S. Prophylactic use of levosimendan in preoperative setting for surgical repair of congenital heart disease in children. Front Pediatr. 2023, 11, 1205971. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cranley, J; Hardiman, A; Freeman, LJ. Pulsed Levosimendan in advanced heart failure due to congenital heart disease: a case series. Eur Heart J Case Rep. 2020, 4(3), 1–6. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lang, RM; Badano, LP; Mor-Avi, V; et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 2015, 28, 1–39. [Google Scholar] [CrossRef]
- Menachem, JN; Schlendorf, KH; Mazurek, JA; Bichell, DP; Brinkley, DM; Frischhertz, BP; Mettler, BA; Shah, AS; Zalawadiya, S; Book, W; Lindenfeld, J. Advanced Heart Failure in Adults With Congenital Heart Disease. JACC Heart Fail 2020, 8(2), 87–99. [Google Scholar] [CrossRef] [PubMed]
- Ly, R; Karsenty, C; Amedro, P; Cohen, S; Domanski, O; Godart, F; Radojevic, J; Vaksmann, G; Naccache, N; Boubrit, A; Bataille, V; Hascoet, S; Ladouceur, M. Health-Related Quality of Life and Its Association With Outcomes in Adults With Congenital Heart Disease and Heart Failure: Insight From FRESH-ACHD Registry. J Am Heart Assoc. 2023, 12(8), e027819. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lubert, AM; Cedars, A; Almond, CS; Amdani, S; Conway, J; Friedland-Little, JM; Gajarski, RJ; Kindel, SJ; Lorts, A; Morales, DLS; O'Connor, MJ; Peng, DM; Rosenthal, DN; Smyth, L; Sutcliffe, DL; Schumacher, KR. Considerations for Advanced Heart Failure Consultation in Individuals With Fontan Circulation: Recommendations From ACTION. Circ Heart Fail 2023, 16(2), e010123. [Google Scholar] [CrossRef] [PubMed]
- Barracano, R; Merola, A; Fusco, F; Scognamiglio, G; Sarubbi, B. Protein-losing enteropathy in Fontan circulation: Pathophysiology, outcome and treatment options of a complex condition. Int J Cardiol Congenit Heart Dis 2022, 7, 100322. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Misra, A; Rodriguez-Monserrate, CP; Gauvreau, K; Dellborg, M; Fusco, F; Gupta, T; Kay, J; Kutty, S; Kauling, RM; Nicolarsen, J; Roos-Hesselink, J; John, AS; Wong, J; Burchill, LJ; Krieger, EV; Lubert, AM; Gallego, P; Kuo, M; Aboulhosn, J; Cramer, J; Antonova, P; Baker, D; Dehghani, P; Opotowsky, AR; van Dissel, A; Grewal, J; Yeung, E; Fernandes, S; Ginde, S; Khairy, P; Han, F; Muhll, IV; Wilson, WM; Kay, WA; Pylypchuk, S; Sarubbi, B; O'Donnell, C; Rodriguez, F, 3rd; Jayadeva, PS; Celermajer, DS; Shah, S; Cotts, T; DeZorzi, C; Magalski, A; Valente, AM; Broberg, CS. Propensity Score Analysis of Possible Medication Effects on Outcomes in Patients With Systemic Right Ventricles. JACC Adv. 2024, 4(1), 101443. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Fusco, F; Scognamiglio, G; Merola, A; Iannuzzi, A; Palma, M; Grimaldi, N; Sarubbi, B. Safety and Efficacy of Sacubitril/Valsartan in Patients With a Failing Systemic Right Ventricle: A Prospective Single-Center Study. Circ Heart Fail 2023, 16(2), e009848. [Google Scholar] [CrossRef] [PubMed]
- Nederend, M; Kiès, P; Regeer, MV; Vliegen, HW; Mertens, BJ; Robbers-Visser, D; Bouma, BJ; Tops, LF; Schalij, MJ; Jongbloed, MRM; Egorova, AD. Tolerability and beneficial effects of sacubitril/valsartan on systemic right ventricular failure. Heart 2023, 109(20), 1525–1532. [Google Scholar] [CrossRef] [PubMed]
- Fusco, F; Scognamiglio, G; Abbate, M; Merola, A; Grimaldi, N; Ciriello, GD; Sarubbi, B. Dapagliflozin in Patients With a Failing Systemic Right Ventricle: Results From the DAPA-SERVE Trial. JACC Heart Fail 2024, 12(4), 789–791. [Google Scholar] [CrossRef] [PubMed]
- Albertini, M; Waldmann, V; David, P; Barat, A; Legendre, A; Chaussade, AS; Iserin, L; Ladouceur, M. Safety and Efficacy of Dapagliflozin in Patients With Systemic Right Ventricular Dysfunction: DAPA-SRV Trial. J Am Heart Assoc. Epub. 2025, 14(11), e040302. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pathak, A; Lebrin, M; Vaccaro, A; Senard, JM; Despas, F. Pharmacology of levosimendan: inotropic, vasodilatory and cardioprotective effects. J Clin Pharm Ther. 2013, 38(5), 341–9. [Google Scholar] [CrossRef] [PubMed]
- Wannaz, L; Boillat, L; Perez, MH; Di Bernardo, S. Prophylactic use of levosimendan in preoperative setting for surgical repair of congenital heart disease in children. Front Pediatr. 2023, 11, 1205971. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Comín-Colet, J; Manito, N; Segovia-Cubero, J; Delgado, J; García Pinilla, JM; Almenar, L; Crespo-Leiro, MG; Sionis, A; Blasco, T; Pascual-Figal, D; Gonzalez-Vilchez, F; Lambert-Rodríguez, JL; Grau, M; Bruguera, J. LION-HEART Study Investigators. Efficacy and safety of intermittent intravenous outpatient administration of levosimendan in patients with advanced heart failure: the LION-HEART multicentre randomised trial. Eur J Heart Fail 2018, 20(7), 1128–1136. [Google Scholar] [CrossRef] [PubMed]
- García-González, MJ; Aldea Perona, A; Lara Padron, A; Morales Rull, JL; Martínez-Sellés, M; de Mora Martin, M; López Díaz, J; López Fernandez, S; Ortiz Oficialdegui, P; Jiménez Sosa, A. Efficacy and safety of intermittent repeated levosimendan infusions in advanced heart failure patients: the LAICA study. ESC Heart Fail 2021, 8(6), 4820–4831. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dobarro, D; Donoso-Trenado, V; Solé-González, E; Moliner-Abós, C; Garcia-Pinilla, JM; Lopez-Fernandez, S; Ruiz-Bustillo, S; Diez-Lopez, C; Castrodeza, J; Méndez-Fernández, AB; Vaqueriza-Cubillo, D; Cobo-Marcos, M; Tobar, J; Sagasti-Aboitiz, I; Rodriguez, M; Escolar, V; Abecia, A; Codina, P; Gómez-Otero, I; Pastor, F; Marzoa-Rivas, R; González-Babarro, E; de Juan-Baguda, J; Melendo-Viu, M; de Frutos, F; Gonzalez-Costello, J. Intermittent inotropic support with levosimendan in advanced heart failure as destination therapy: The LEVO-D registry. ESC Heart Fail 2023, 10(2), 1193–1204. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Combes, A; Saura, O; Nesseler, N; Lebbah, S; Rozec, B; Levy, B; Fellahi, JL; Beurton, A; Meslin, S; Gaudard, P; Bouglé, A; Vincentelli, A; Sonneville, R; Lebreton, G; Lévy, D; Ouattara, A; Tubach, F. LEVOECMO Trial Group and the International ECMO Network (ECMONet). Levosimendan to Facilitate Weaning From ECMO in Patients With Severe Cardiogenic Shock: The LEVOECMO Randomized Clinical Trial. JAMA 2026, 335(1), 60–69. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Burkhoff, D; Borlaug, BA; Shah, SJ; Zolty, R; Tedford, RJ; Thenappan, T; Zamanian, RT; Mazurek, JA; Rich, JD; Simon, MA; Chung, ES; Raza, F; Majure, DT; Lewis, GD; Preston, IR; Rich, S. Levosimendan Improves Hemodynamics and Exercise Tolerance in PH-HFpEF: Results of the Randomized Placebo-Controlled HELP Trial. JACC Heart Fail 2021, 9(5), 360–370. [Google Scholar] [CrossRef] [PubMed]
- Yaku, H; Burkhoff, D; Borlaug, BA; Lala, A; Butler, J; Rich, S; Shah, SJ. Oral Levosimendan for the Treatment of Pulmonary Hypertension due to Heart Failure With Preserved Ejection Fraction: Rationale and Design of the LEVEL Trial S1071-9164(25)00293-3. J Card Fail. Epub ahead of print. 2025. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y; Wei, Z; Zhang, C; Lu, C; Zeng, Z. The effect of levosimendan on right ventricular function in patients with heart dysfunction: a systematic review and meta-analysis. Sci Rep. 2021, 11(1), 24097. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
| sex (female) | 3(25%) |
| age (years) | 44.6[35-61.3] |
| weight (kg) | 71±17 |
| height (cm) | 167±9 |
bmi  (kg/m2) (kg/m2) |
25±5 |
| bsa (m2) | 2±0.3 |
| main diagnosis | 2 aortic coarctation 2 atrial septal defect 1 partial anomalous venous return 1 tetralogy of fallot 1 atrioventricular septal defect with straddling valve 1 transposition of the great arteries 4 congenitally corrected transposition of the great arteries |
| associated defects | 1 atrial septal defect 1 bicuspid aortic valve 1 right isomerism 2 ventricular septal defects 1 pulmonary stenosis 1 coarctation repair |
| surgical/percutaneous procedures | 2 coarctation repair (2 end to end anastomosis, 1 subclavian flap) 3 atrial septal defect closure (1 with a fenestrated device, 2 surgically) 1 glenn and fontan procedure 1 tetralogy of fallot repair 1 tricuspid valve annuloplasty and mitral valve edge-to-edge repair 1 correction of anomalous pulmonary veins drainage 1 percutaneous closure of left atrial appendage 1 mustard operation 2 ventricular septal defect closure 1 pulmonary stenosis relief 3 blalock taussing shunt 1 pulmonary artery banding 1 rashkind balloon atrial septostomy 1 fontan conduit stenting 1 fontan conduit replacement |
| total number of procedures01 2 5 |
2(17%) 4(33%) 5(42%) 1(8%) |
| residual defects | 2 pulmonary arterial hypertension 1 severe aortic stenosis 5 severe tricuspid regurgitation 1 severe pulmonary regurgitation 1 mitral regurgitation |
| genetic disorder | 1 turner syndrome |
| comorbidity | 1(8%) hepatocellular carcinoma 2(17%) ckd 4(33%) copd 2(17%) hypothyroidism |
| cardiovascular risk factors | 1(8%) diabetes 2 (17%) hypertension 3(25%) obesity 3 (25%) dyslipidemia 2(17%) stable coronary syndrome and previous ptca |
| pm | 1(8%) |
| icd | 4(33%) |
| nyha class (ii/iii/iv) | 2(17%)/9(75%)/1(8%) |
| previous hospitalization for hf | 12(100%) |
| hf hospitalization >1 | 6(50%) |
| history of supraventricular tachycardia | 4(33%) |
| history of ventricular tachycardia | 5(42%) |
| baseline rhythm | 8(66%) sinus 2(17%) ventricular pacing 2(17%) atrial fibrillation |
| baseline sbp (mmhg) | 108±10 |
| baseline hr (bpm) | 76±12 |
| treatment | 7(58%) arni 8 (66%) gliflozins 3(25%) acei/arb 9 (75%) beta-blockers 7(58%) mra 8(66%) diuretics 2(17%) ivabradine (25%) doac 2(17%) warfarin 2(17%) antiplateletes 2(18%) amiodaron |
| papm (mmhg) | 26±9 |
| pcwp (mmhg) | 14±5 |
| pvri (wu) | 3±1.9 |
| ci (l/min/m2) | 3±1.8 |
| baseline | follow-up | p-value | |
|---|---|---|---|
| ntprobnp (pg/ml) | 1576[705-3023] | 967[241-2263] | 0.3 |
| creatinine (mg/dl) | 0.84±0.25 | 0.88±0.17 | 0.4 |
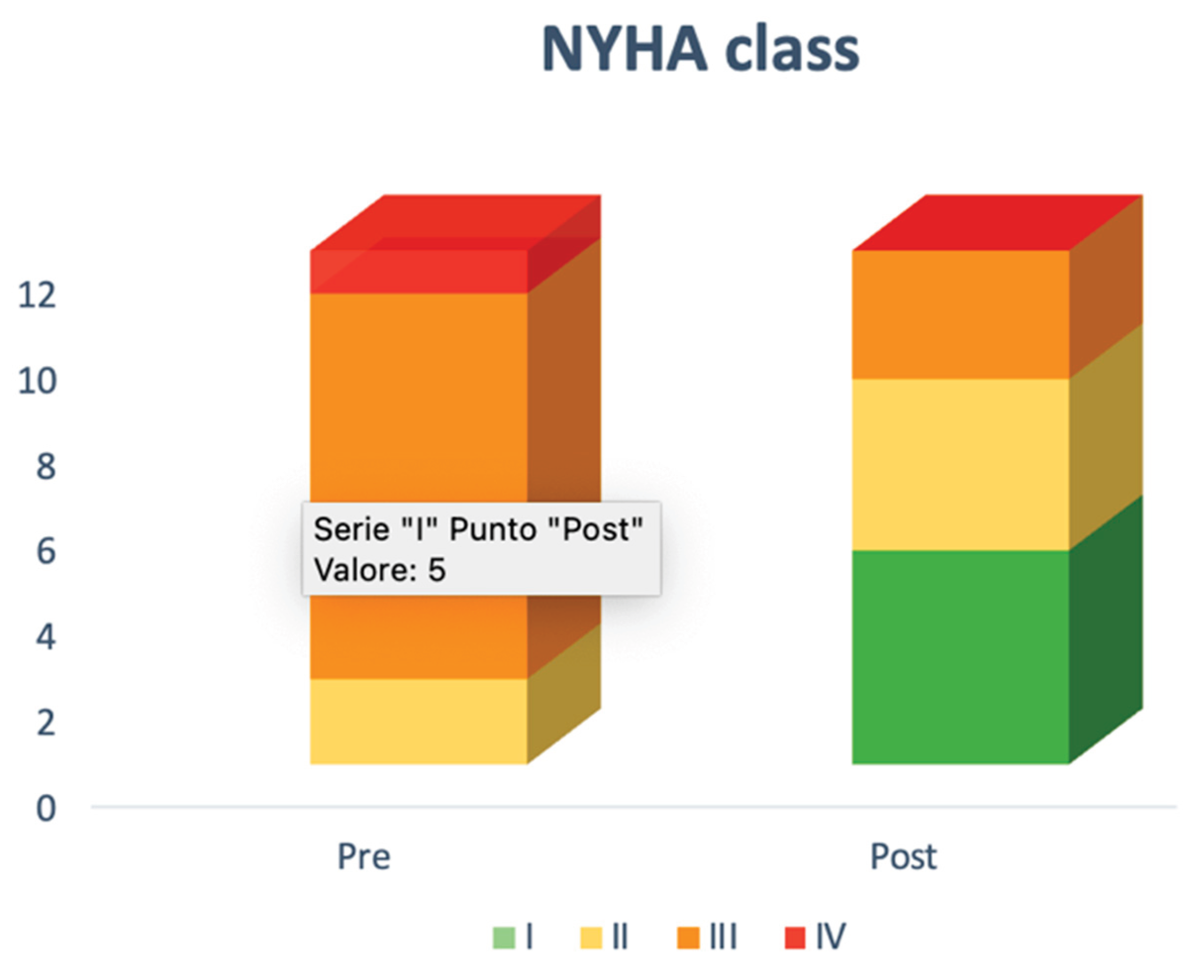
| nyha class iii-iv | 10(83%) | 3(25%) | 0.005 |
| lv ef* (%) | 37.6±8 | 43.6±5 | 0.1 |
| rv fac** (%) | 27±7.4 | 30.6±7 | 0.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




