Submitted:
11 February 2026
Posted:
12 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Sample Selection
2.3. Data Acquisition and Evaluation
2.4. Statistical Analyses
3. Results
3.1. Signalment
3.2. Bacterial Isolates, Severity of Cystitis, and Timing of Radiographic Examinations
3.3. Urinary Bladder Size, Urolithiasis, and Mineral Density
3.4. Urinary Bladder Height and the VBS
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| VBS | Vertebral bladder score |
| LVU | Lumbar vertebral units |
References
- Azevedo, S.; O’Malley, B.; Greene, C.; Moran, H.; Magalhães, T.R.; Queiroga, F.L. Lower urinary tract diseases in guinea pigs: a 14-year retrospective study (2004–2018). Animals (Basel) 2022, 13, 112. [Google Scholar] [CrossRef]
- Reavill, D.R.; Lennox, A.M. Disease overview of the urinary tract in exotic companion mammals and tips on clinical management. Vet. Clin. North Am. Exot. Anim. Pract. 2020, 23, 169–193. [Google Scholar] [CrossRef]
- Peng, X.; Griffith, J.W.; Lang, C.M. Cystitis, urolithiasis and cystic calculi in ageing guinea pigs. Lab. Anim. 1990, 24, 159–163. [Google Scholar] [CrossRef]
- Turner, P.V.; Brash, M.L.; Smith, D.A. Chapter 3. Guinea pigs. In Pathology of Small Mammal Pets, 1st ed.; Turner, P.V., Brash, M.L., Smith, D.A., Eds.; John Wiley & Sons: Hoboken, NJ, 2017; pp. 147–191. [Google Scholar]
- Barak Pressler, J.W.; Westropp, J.L. Urinary tract infections. In Textbook of Veterinary Internal Medicine: Diseases of the Dog and Cat, 7th ed.; Ettinger, S.J., Feldmann, E.C., Eds.; Elsevier Saunders: Philadelphia, PA, 2012; pp. 2036–2047. [Google Scholar]
- Grauer, G.F. Leitsymptome von Erkrankungen der Harnwege. In Innere Medizin der Kleintiere, 2nd ed.; Nelson, R.W., Couto, C.G., Eds.; Elsevier: Munich, Germany, 2010; pp. 631–645. [Google Scholar]
- Rooney, T.A.; Eshar, D.; Wong, A.D.; Gardhouse, S.; Beaufrère, H. The association between bloodwork, signalment, and urolithiasis in guinea pigs (Cavia porcellus). J. Exot. Pet. Med. 2021, 38, 26–31. [Google Scholar] [CrossRef]
- Edell, A.S.; Vella, D.G.; Sheen, J.C.; Carotenuto, S.E.; McKee, T.; Bergman, P.J. Retrospective analysis of risk factors, clinical features, and prognostic indicators for urolithiasis in guinea pigs: 158 cases (2009–2019). J. Am. Vet. Med. Assoc. 2022, 260, 95–100. [Google Scholar] [CrossRef]
- Hawkins, M.G.; Ruby, A.L.; Drazenovich, T.L.; Westropp, J.L. Composition and characteristics of urinary calculi from guinea pigs. J. Am. Vet. Med. Assoc. 2009, 234, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Coutant, T.; Dunn, M.; Langlois, I.; Maccolini, E. Cystoscopic-guided lithotripsy for the removal of a urethral stone in a guinea pig. J. Exot. Pet. Med. 2019, 28, 111–114. [Google Scholar] [CrossRef]
- Fehr, M.; Rappold, S. Urine matter formation in 20 guinea pigs (Cavia porcellus). Vet. Pract. 1997, 5, 553–557. [Google Scholar]
- Martin, L. Retrospective Study on the Species Distribution and Common Diseases in Exotic Pets Presented to the Clinic for Zoo Animals, Exotic Pets and Wildlife, University of Zurich from 2005 to 2014; 2018; Volume 90. [Google Scholar]
- Sykes, J.E.; Westropp, J.L. Bacterial infections of the genitourinary tract. In Greene’s Infectious Diseases of the Dog and Cat, 5th ed.; Sykes, J.E., Ed.; Elsevier Saunders: St Louis, MO, 2021; pp. 1669–1687. [Google Scholar]
- Barsanti, J.A.; Brown, S.A. Urinary tract infections. In Textbook of Veterinary Internal Medicine, 8th ed.; Ettinger, S.J.F., Côté, E.C.E., Eds.; Elsevier: St. Louis, MO, 2017; pp. 2004–2014. [Google Scholar]
- Weese, J.S.; Blondeau, J.; Boothe, D.; Guardabassi, L.G.; Gumley, N.; Papich, M.; Jessen, L.R.; Lappin, M.; Rankin, S.; Westropp, J.L.; et al. International Society for Companion Animal Infectious Diseases (ISCAID) guidelines for the diagnosis and management of bacterial urinary tract infections in dogs and cats. Vet. J. 2019, 247, 8–25. [Google Scholar] [CrossRef] [PubMed]
- Roberts-Steel, S.; Oxley, J.A.; Carroll, A.; Wills, A.P. Frequency of owner-reported bacterial infections in pet guinea pigs. Animals (Basel) 2019, 9, 649. [Google Scholar] [CrossRef]
- Minarikova, A.; Hauptman, K.; Jeklova, E.; Knotek, Z.; Jekl, V. Diseases in pet guinea pigs: a retrospective study in 1000 animals. Vet. Rec. 2015, 177, 200–200. [Google Scholar] [CrossRef]
- DeCubellis, J. Common emergencies in rabbits, guinea pigs, and chinchillas. Vet. Clin. North Am. Exot. Anim. Pract. 2016, 19, 411–429. [Google Scholar] [CrossRef]
- Vella, D. Emergency presentations of exotic mammal herbivores. J. Exot. Pet. Med. 2012, 21, 293–299. [Google Scholar] [CrossRef]
- Petite, A.; Busoni, V.; Heinen, M.P.; Billen, F.; Snaps, F. Radiographic and ultrasonographic findings of emphysematous cystitis in four nondiabetic female dogs. Vet. Radiol. Ultrasound 2006, 47, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Maggi, C.A.; Meli, A.; Santicioli, P. Neuroeffector mechanisms in the voiding cycle of the guinea pig urinary bladder. J. Auton. Pharmacol. 1987, 7, 295–308. [Google Scholar] [CrossRef]
- Martinez-Ruzafa, I.; Kruger, J.M.; Miller, R.; Swenson, C.L.; Bolin, C.A.; Kaneene, J.B. Clinical features and risk factors for development of urinary tract infections in cats. J. Feline Med. Surg. 2012, 14, 729–740. [Google Scholar] [CrossRef] [PubMed]
- Wolf, P.; Schröder, A.; Wenger, A.; Kamphues, J. The nutrition of the chinchilla as a companion animal – basic data, influences and dependences. J. Anim. Physiol. Anim. Nutr. (Berl). 2003, 87, 129–133. [Google Scholar] [CrossRef]
- Böttcher, A.; Müller, K. Radiological and laboratory prognostic parameters for gastric dilation in rabbits (Oryctolagus cuniculus). Vet. Rec. 2024, 194, e3827. [Google Scholar] [CrossRef]
- Dadah, H.J.; Hawkins, M.G.; Munk, A.W.; Westropp, J.L.; Kass, P.H.; Beaufrère, H. Risk and protective factors associated with clinical urolithiasis in pet guinea pigs (Cavia porcellus). J. Exot. Pet. Med. 2025, 54, 28–34. [Google Scholar] [CrossRef]
- Alves, D.A. Pathology in practice: urolithiasis. J. Am. Vet. Med. Assoc. 2012, 241, 185–187. [Google Scholar] [CrossRef]
- Mische, A.; Landmann, K.; Mundhenk, L.; Lübke-Becker, A.; Müller, K. Sepsis secondary to cystitis in a guinea pig (Cavia porcellus). Vet. Rec. Case Rep. 2026, 14, e70271. [Google Scholar] [CrossRef]
- Smith, J.S.; Krull, A.C.; Schleining, J.A.; Derscheid, R.J.; Kreuder, A.J. Clinical presentations and antimicrobial susceptibilities of Corynebacterium cystitidis associated with renal disease in four beef cattle. J. Vet. Intern. Med. 2020, 34, 2169–2174. [Google Scholar] [CrossRef]
- Bichler, K.H.; Eipper, E.; Naber, K.; Braun, V.; Zimmermann, R.; Lahme, S. Urinary infection stones. Int. J. Antimicrob. Agents. 2002, 19, 488–498. [Google Scholar] [CrossRef]
- Osanai, T.; Miyoshi, I.; Hiramune, T.; Kasai, N. Spontaneous urinary calculus in young LEW rats caused by Corynebacterium renale. J. Urol. 1994, 152, 1002–1004. [Google Scholar] [CrossRef] [PubMed]
- De Silva, M.; Bo, P.; Dora Genocchi, E.; Tagliavia, C.; Roccaro, M.; Grandis, A.; Baron Toaldo, M. Radiographic and anatomical morphometric assessments of heart size in presumed healthy pet guinea pigs. Vet. Radiol. Ultrasound 2022, 63, 8–17. [Google Scholar] [CrossRef] [PubMed]
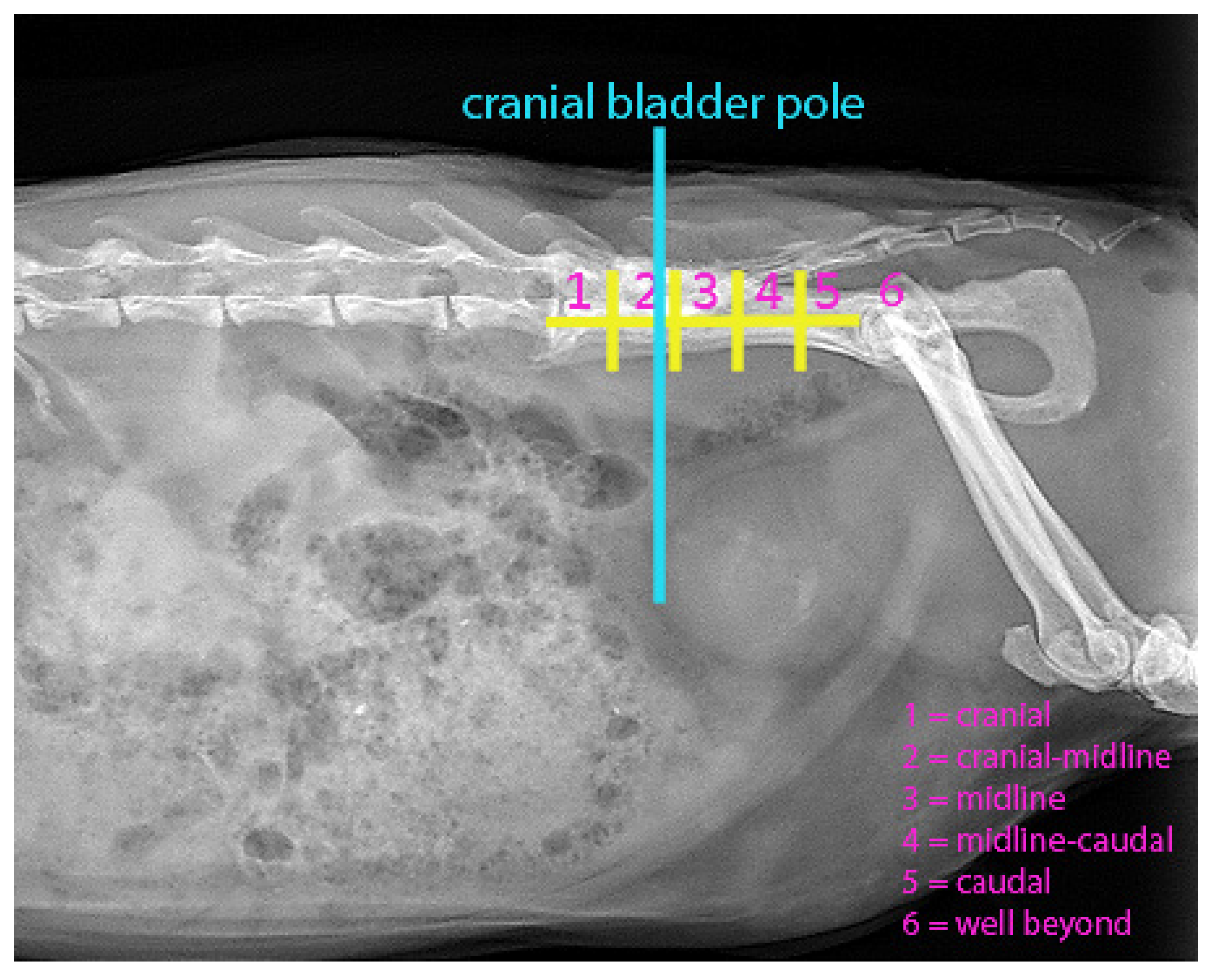
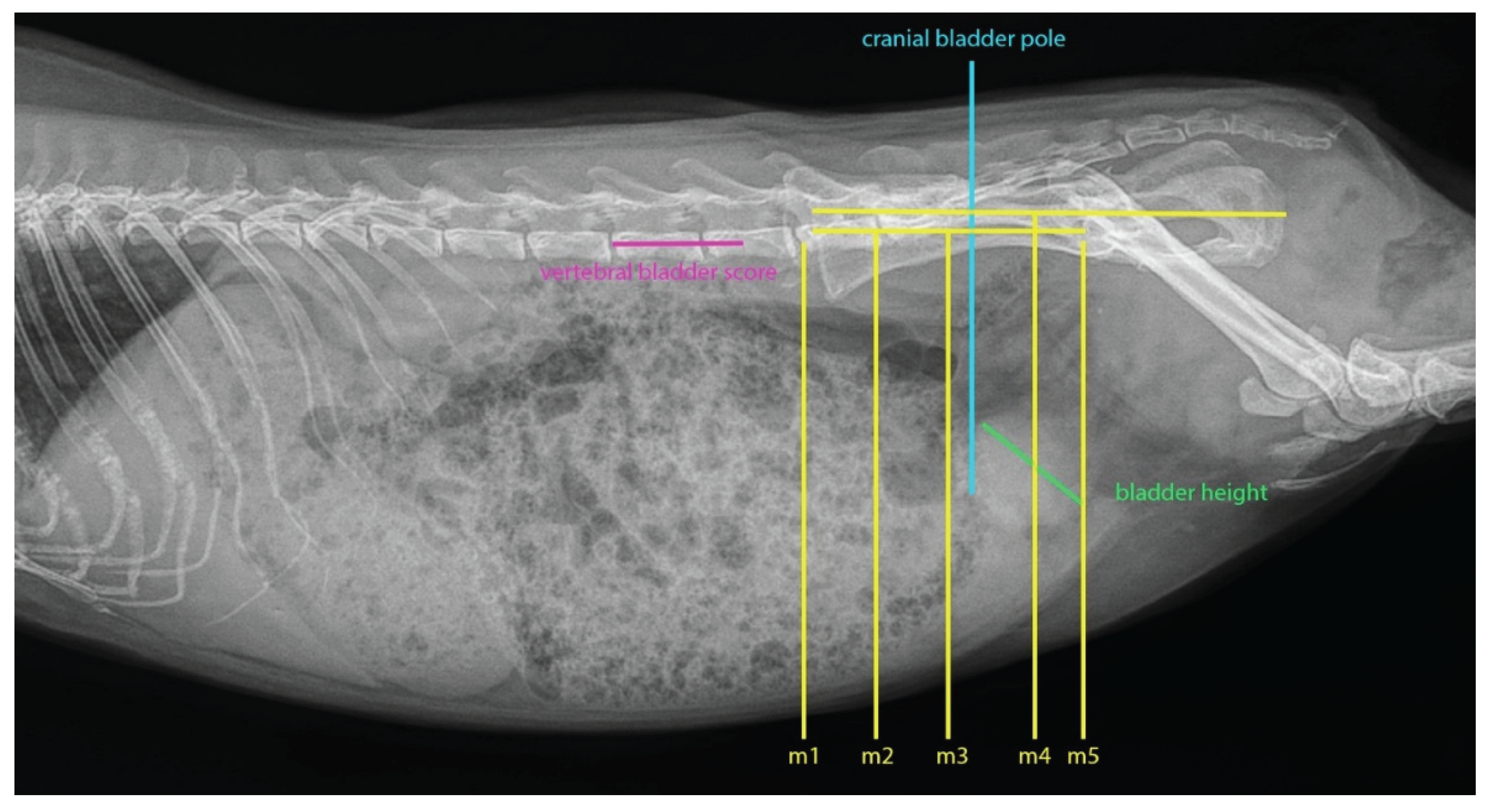
| Position |
Control n% |
Cystitis group n% |
|||||
| 1 | Cranial | 2 | 8.3 | 3 | 12.5 | ||
| 2 | Cranial-midline | 5 | 20.8 | 10 | 41.7 | ||
| 3 | Midline | 11 | 45.8 | 11 | 45.8 | ||
| 4 | Midline-caudal | 3 | 12.5 | 0 | 0 | ||
| 5 | Caudal | 2 | 8.3 | 0 | 0 | ||
| 6 | Well beyond | 1 | 4.2 | 0 | 0 | ||
| Total | 24 | 100 | 24 | 100 | |||
|
Control n% |
Cystitis group n% |
||||
| Mineral-dense shading | Present | 0 | 0 | 11 | 45.8 |
| Not present | 24 | 100 | 13 | 54.2 | |
| Total | 24 | 100 | 24 | 100 | |
| Uroliths | Present | 0 | 0 | 6 | 25 |
| Not present | 24 | 100 | 18 | 75 | |
| Total | 24 | 100 | 24 | 100 | |
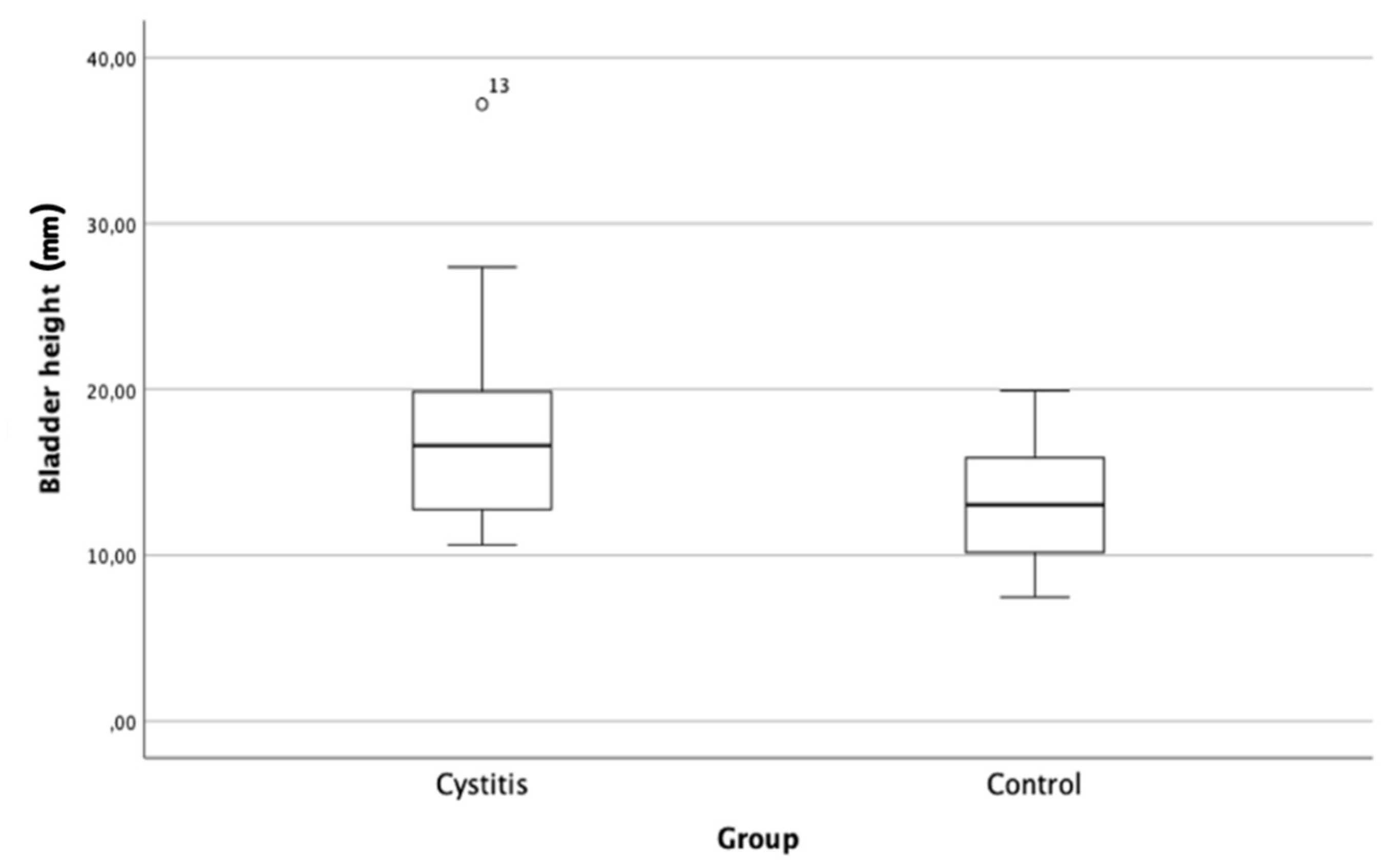
| Parameter | Control | Cystitis group |
| Median (mm) | 13.0 | 16.6 |
| Minimum (mm) | 7.5 | 10.6 |
| Maximum (mm) | 19.9 | 37.2 |
| Mean (mm) | 13.1 | 17.7 |
| Standard deviation (mm) | 3.6 | 6.3 |
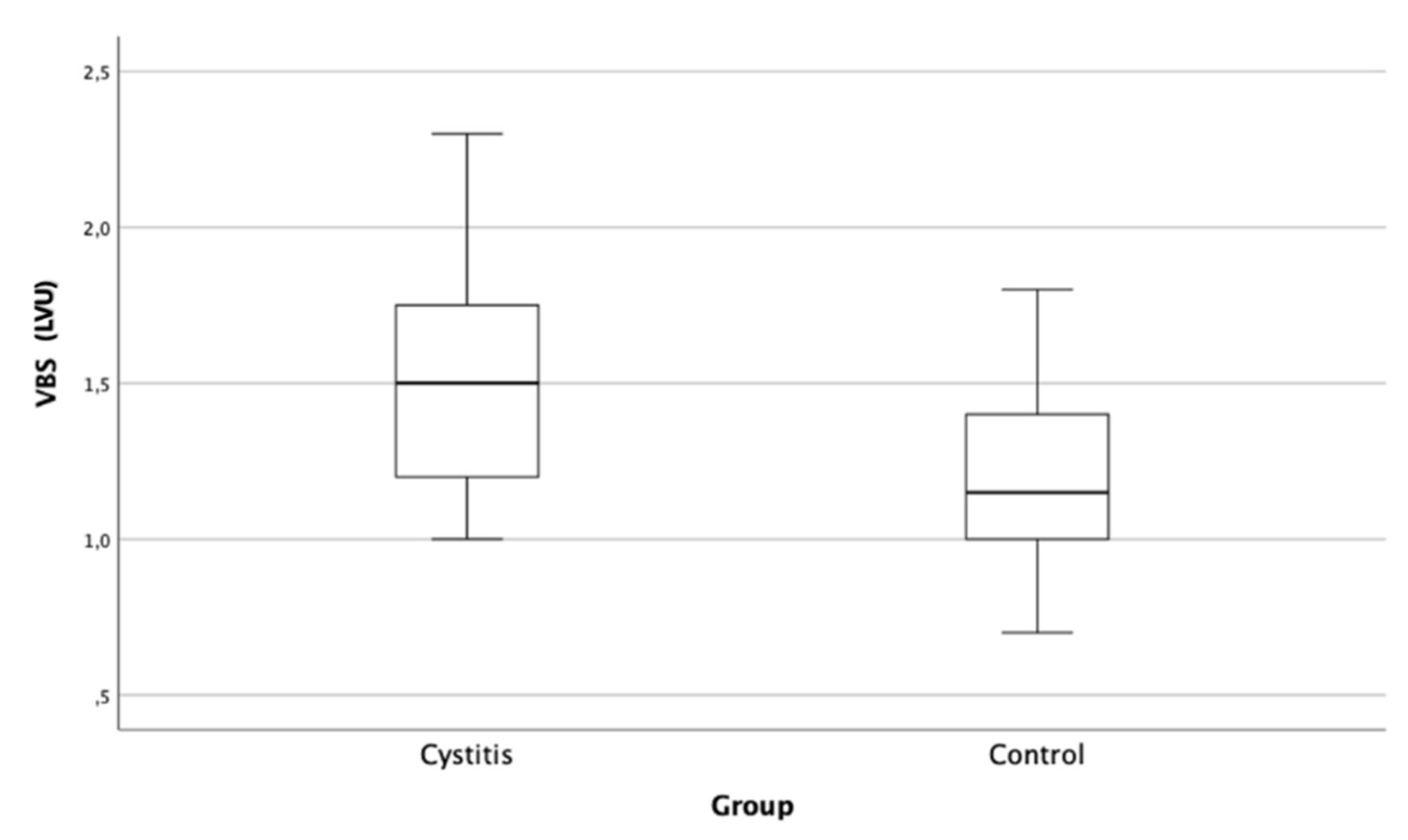
| Parameter | Control | Cystitis group |
| Median (LVU) | 1.2 | 1.5 |
| Minimum (LVU) | 0.7 | 1.0 |
| Maximum (LVU) | 1.8 | 2.3 |
| Mean (LVU) | 1.2 | 1.5 |
| Standard deviation (LVU) | 0.3 | 0.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




