Submitted:
11 February 2026
Posted:
15 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
Aim of the Study
2. Materials and Methods
2.1. Study Design and Population
2.2. Baseline
2.3. Enteral Nutrition and Microbiology
2.4. Outcomes
2.5. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Multivariable Logistic Regression
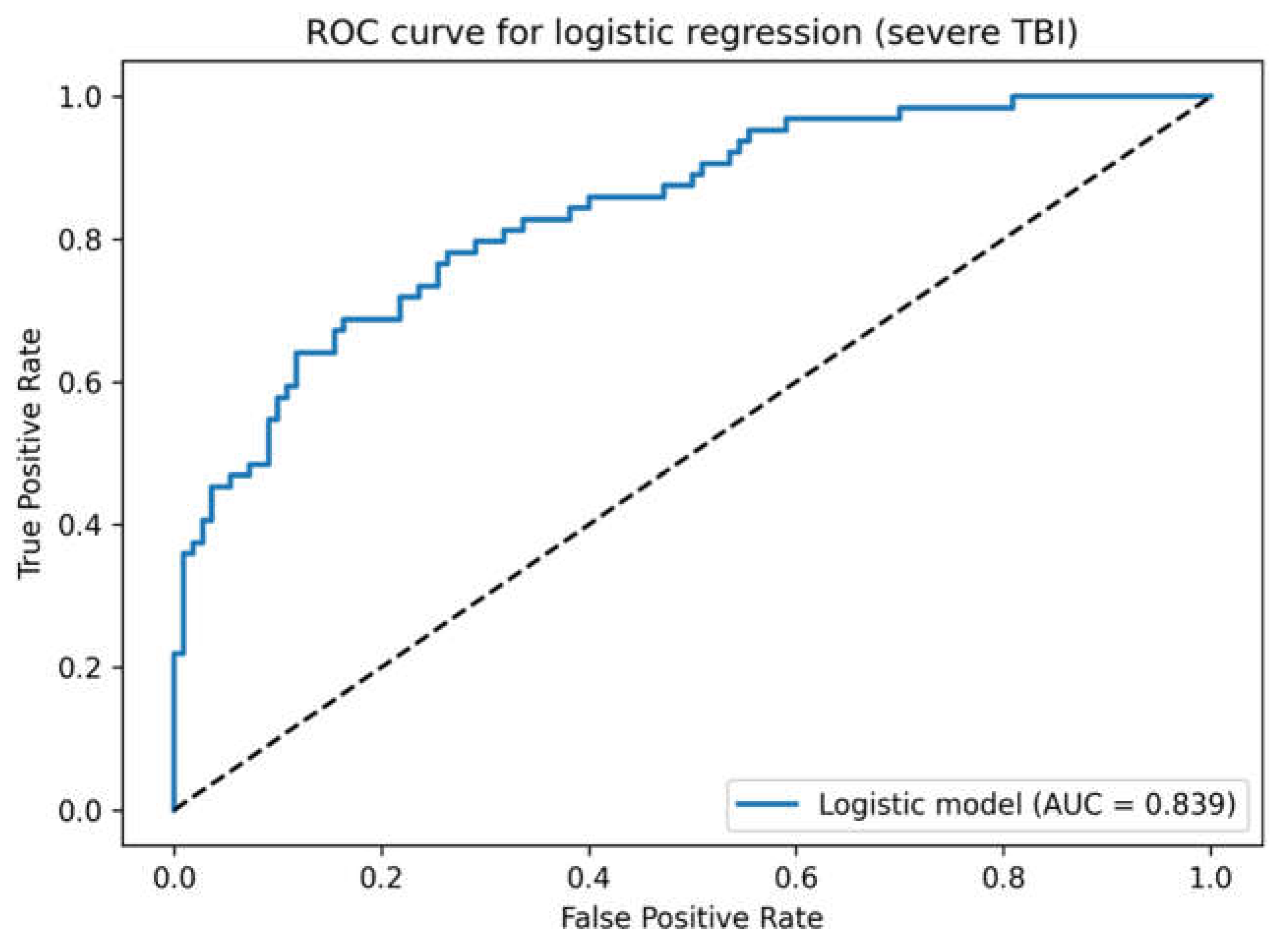
3.3. ROC Analysis
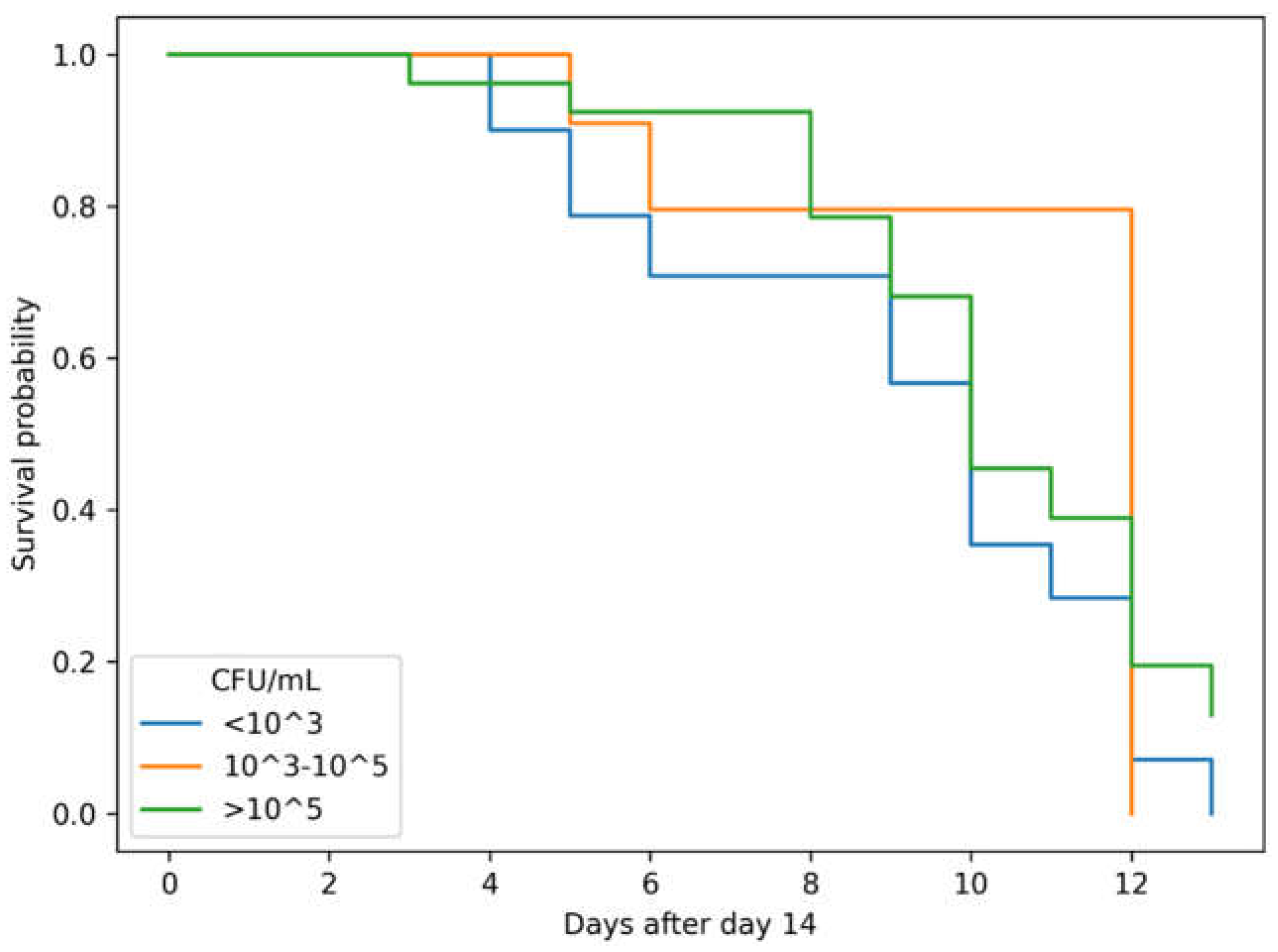
3.4. Landmark Analysis of Mortality (Day 14)
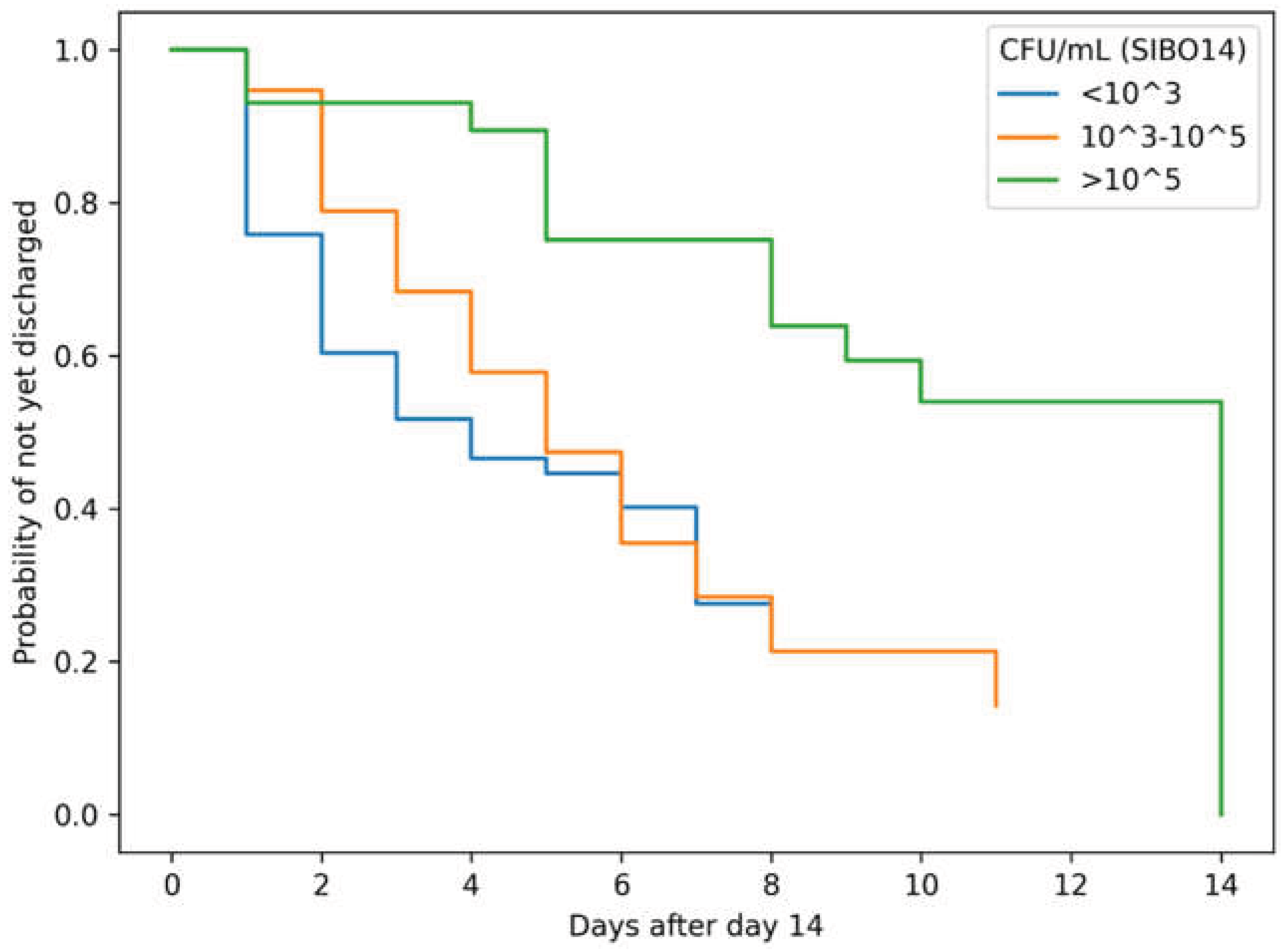
3.5. Landmark Analysis of Discharge
4. Discussion
- Gastrointestinal dysmotility in the ICU. Critically ill patients often develop impaired motility of the stomach and small intestine; key factors include mechanical ventilation, vasopressors, opioids/sedation, severe systemic inflammatory response, and metabolic disorders [21,22]. Slowing of intestinal transit and decreased propulsive peristalsis create conditions for bacterial hypercolonization and retrograde migration of flora.
- Barrier dysfunction and translocation. Critical illness is accompanied by increased intestinal permeability and the risk of bacterial/endotoxin translocation, which is discussed within the concepts of “gut-origin sepsis” and gut-induced MODS [5,23]. In this context, SIBO may be not only a local phenomenon but also part of a systemic cascade of inflammation and organ dysfunction.
- Acid suppression and antibiotics. All our patients widely received PPIs/H2-blockers (stress ulcer prophylaxis) and antibiotics, which may alter the microbial profile and facilitate excessive bacterial growth. The association between acid suppression and SIBO/increased bacterial titers has been described in clinical studies and reviews [24].
- Enteral nutrition and postpyloric tube placement. Although enteral nutrition generally supports mucosal trophism, in severe critical illness the combination of nutritional load, dysmotility, and changes in biliary-pancreatic secretion may contribute to dysbiosis. Additionally, the very fact of prolonged postpyloric tube placement (in our case beyond the ligament of Treitz) facilitates access/contamination and may change local conditions [25,26]. Collectively, these mechanisms explain well why baseline CFU1 in most patients is low, but by days 12–14, under conditions of prolonged mechanical ventilation/sedation/acid suppression/antibiotics/vasopressors and pronounced gastrointestinal dysfunction, significant bacterial growth may develop, which in our cohort is associated with worsening prognosis.
5. Conclusions
6. Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dewan, MC; Rattani, A; Gupta, S; Baticulon, RE; Hung, YC; Punchak, M.; et al. Estimating the global incidence of traumatic brain injury. J Neurosurg. 2018, 130(4), 1080–1097. [Google Scholar] [CrossRef]
- Maas, AIR; Menon, DK; Manley, GT; Abrams, M; Åkerlund, C; Andelic, N; et al. Traumatic brain injury: progress and challenges in prevention, clinical care, and research . Lancet Neurol. 2022, 21(11), 1004–1060. [Google Scholar] [CrossRef]
- Steyerberg, EW; Mushkudiani, N; Perel, P; Butcher, I; Lu, J; McHugh, GS; et al. Predicting outcome after traumatic brain injury: development and international validation of prognostic scores based on admission characteristics. PLoS Med. 2008, 5, e165. [Google Scholar] [CrossRef]
- Schuijt, TJ; Lankelma, JM; Scicluna, BP; et al. The gut microbiota plays a protective role in the host defence against pneumococcal pneumonia. Gut 2016, 65, 575–583. [Google Scholar] [CrossRef] [PubMed]
- Zaborin, A; Smith, D; Garfield, K; et al. Membership and behavior of ultra-low-diversity pathogen communities present in the gut of humans during prolonged critical illness. mBio 2014, 5(5), e01361-14. [Google Scholar] [CrossRef] [PubMed]
- Prognosis in Moderate and Severe Traumatic Brain Injury: A Systematic Review of Contemporary Models and Validation Studies . J Neurotrauma 2020, 37(1), 1–13. [CrossRef]
- Lingsma, HF; Roozenbeek, B; Steyerberg, EW; Murray, GD; Maas, AIR. Early prognosis in traumatic brain injury: from prophecies to predictions. Lancet Neurology 2010, 9, 543–554. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence. Head injury: assessment and early management (NICE guideline NG232); NICE: London, 2023. [Google Scholar]
- Carney, N; Totten, AM; O’Reilly, C; Ullman, JS; Hawryluk, GWJ; Bell, MJ; et al. Guidelines for the Management of Severe Traumatic Brain Injury, Fourth Edition. Neurosurgery 2017, 80, 6–15. [Google Scholar] [CrossRef]
- Hawryluk, GWJ; Rubiano, AM; Totten, AM; O’Reilly, C; Ullman, JS; Bratton, SL; et al. Guidelines for the Management of Severe Traumatic Brain Injury: 2020 update of the decompressive craniectomy recommendations. Neurosurgery 2020, 87(3), 427–434. [Google Scholar] [CrossRef] [PubMed]
- Acute respiratory distress syndrome: the Berlin definition. JAMA 2012, 307, 2526–2533. [CrossRef]
- Ulvik, A; Kvåle, R; Wentzel-Larsen, T; Flaatten, H. Multiple organ failure after trauma affects long-term survival and functional status. Critical Care 2007, 11, R95. [Google Scholar] [CrossRef]
- Pimentel, M; Saad, RJ; Long, MD; Rao, SSC. ACG Clinical Guideline: Small Intestinal Bacterial Overgrowth. Am J Gastroenterol. 2020, 115, 165–178. [Google Scholar] [CrossRef]
- Karakosta, A; Katsanos, K; Tsirigotis, P; Vrettou, C; Chatzimichail, A; Liaskos, C; et al. High prevalence of small intestinal bacterial overgrowth syndrome in ICU patients: an observational study. J Intensive Care Med. 2024, 39, 355–362. [Google Scholar] [CrossRef]
- Deane, AM; Chapman, MJ; Fraser, RJ; Bryant, LK; Burgstad, C; Nguyen, NQ. Mechanisms underlying feed intolerance in the critically ill: implications for treatment. World J Gastroenterol. 2007, 13(29), 3909–3917. [Google Scholar] [CrossRef] [PubMed]
- Assimakopoulos, SF; Triantos, C; Thomopoulos, K; Fligou, F; Maroulis, I; Marangos, M; et al. Gut-origin sepsis in the critically ill patient: pathophysiology and treatment. Infection 2018, 46(6), 751–760. [Google Scholar] [CrossRef] [PubMed]
- Su, T; Lai, S; Lee, A; He, X; Chen, S. Meta-analysis: proton pump inhibitors moderately increase the risk of small intestinal bacterial overgrowth. Journal of Gastroenterology 2018, 53, 27–36. [Google Scholar] [CrossRef]
- Rezaie, A; Buresi, M; Lembo, A; et al. Hydrogen and methane-based breath testing in gastrointestinal disorders: the North American Consensus. Am J Gastroenterol. 2017, 112, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Kashyap, P; Moayyedi, P; Quigley, EMM; Simren, M; Vanner, S; et al. Critical appraisal of the SIBO hypothesis and breath testing. Neurogastroenterol Motil 2024, 36, e14817. [Google Scholar] [CrossRef]
- Wischmeyer, PE. Tailoring nutrition therapy to illness and recovery. Critical Care 2017, 21 (Suppl 3), 316. [Google Scholar] [CrossRef]
- Govil, D; Pal, D. Gastrointestinal motility disorders in critically ill. Indian J Crit Care Med 2020, 24 (Suppl 4), S179–S182. [Google Scholar] [CrossRef]
- Ladopoulos, T; Giannaki, M; Alexopoulou, C; Proklou, A; Pediaditis, E; Kondili, E. Gastrointestinal dysmotility in critically ill patients. Ann Gastroenterol. 2018, 31, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Potruch, A; Schwartz, A; Ilan, Y. The role of bacterial translocation in sepsis. Ther Adv Gastroenterol. 2022, 15, 17562848221094214. [Google Scholar] [CrossRef] [PubMed]
- Dukowicz, AC; Lacy, BE; Levine, GM. Small intestinal bacterial overgrowth: a comprehensive review. Gastroenterol Hepatol (N Y) 2007, 3, 112–122. [Google Scholar] [PubMed]
- Singer, P; Blaser, AR; Berger, MM; Alhazzani, W; Calder, PC; Casaer, MP; et al. ESPEN guideline on clinical nutrition in the intensive care unit. Clinical Nutrition 2019, 38, 48–79. [Google Scholar] [CrossRef]
- Jiyong, J; Tiancha, H; Huiqin, W; Jingfen, J. Effect of gastric versus post-pyloric feeding on the incidence of pneumonia in critically ill patients: observations from traditional and Bayesian random-effects meta-analysis. Clin Nutr. 2013, 32, 8–15. [Google Scholar] [CrossRef]
| Parameter | Minimum | QI (25%) | Median | QIII (75%) | Maximum |
| Age, years | 22 | 43 | 48 | 62.75 | 74 |
| Body mass index, kg/m² | 26 | 29 | 31 | 33.0 | 36 |
| GCS at admission, points | 5 | 7 | 7 | 8.0 | 8 |
| PaO₂/FiO₂ | 305 | 345 | 356 | 378 | 411 |
| PaCO₂, mmHg | 33 | 38 | 44 | 45 | 46 |
| Blood glucose, mmol/L | 5 | 7 | 8 | 10 | 11 |
| CFU1 category | n | % |
| <10³ | 148 | 85.1 |
| 10³–10⁵ | 19 | 10.9 |
| >10⁵ | 7 | 4.0 |
| Total | 174 | 100 |
| Variable | OR | 95% CI | p-value |
| Age (per 1 year) | 1.06 | 1.03–1.10 | <0.001 |
| GCS (per 1 point) | 0.48 | 0.28–0.83 | 0.008 |
| PaO₂/FiO₂ | 0.74 | 0.68–0.91 | 0.006 |
| Glucose | 1.13 | 0.92–1.40 | 0.245 |
| CFU14 >10⁵ | 5.15 | 2.15–12.34 | <0.001 |
| CFU14 | Number of patients (n) | Deaths | Mortality risk (%) |
| <10³ | 58 | 18 | 31.0 |
| 10³–10⁵ | 19 | 4 | 21.1 |
| >10⁵ | 29 | 16 | 55.2 |
| Total | 106 | 38 | 35.8 |
| CFU14 | n | Discharged | Discharge rate (%) | Median days to discharge |
| <10³ | 58 | 40 | 69.0 | 4 |
| 10³–10⁵ | 19 | 15 | 78.9 | 5 |
| >10⁵ | 29 | 14 | 48.3 | 8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




