Submitted:
10 February 2026
Posted:
12 February 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
Study Design
End Points and Sample Size Estimation
Quantitation and Characterization of MNPs, and Immunohistochemistry
Statistical Analysis
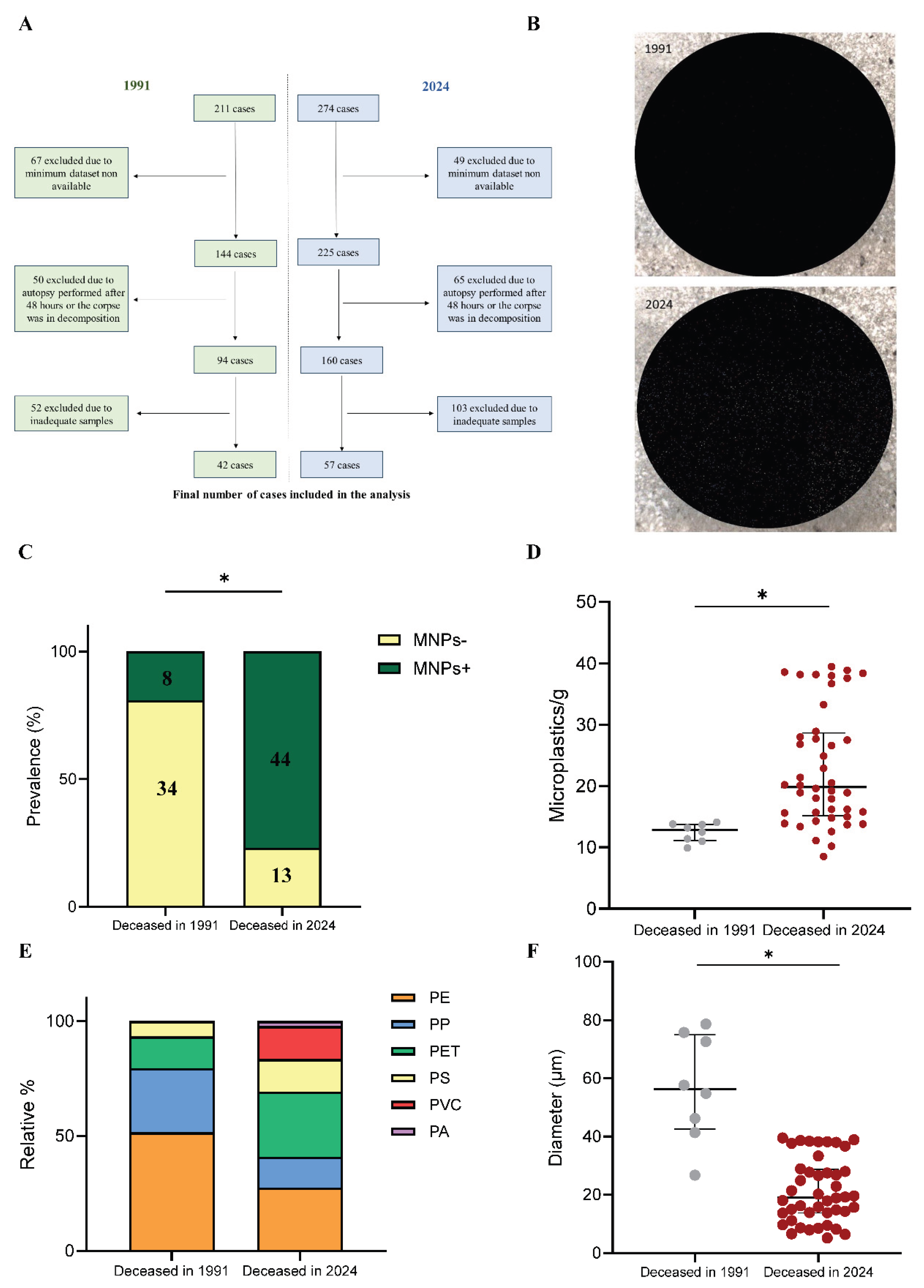
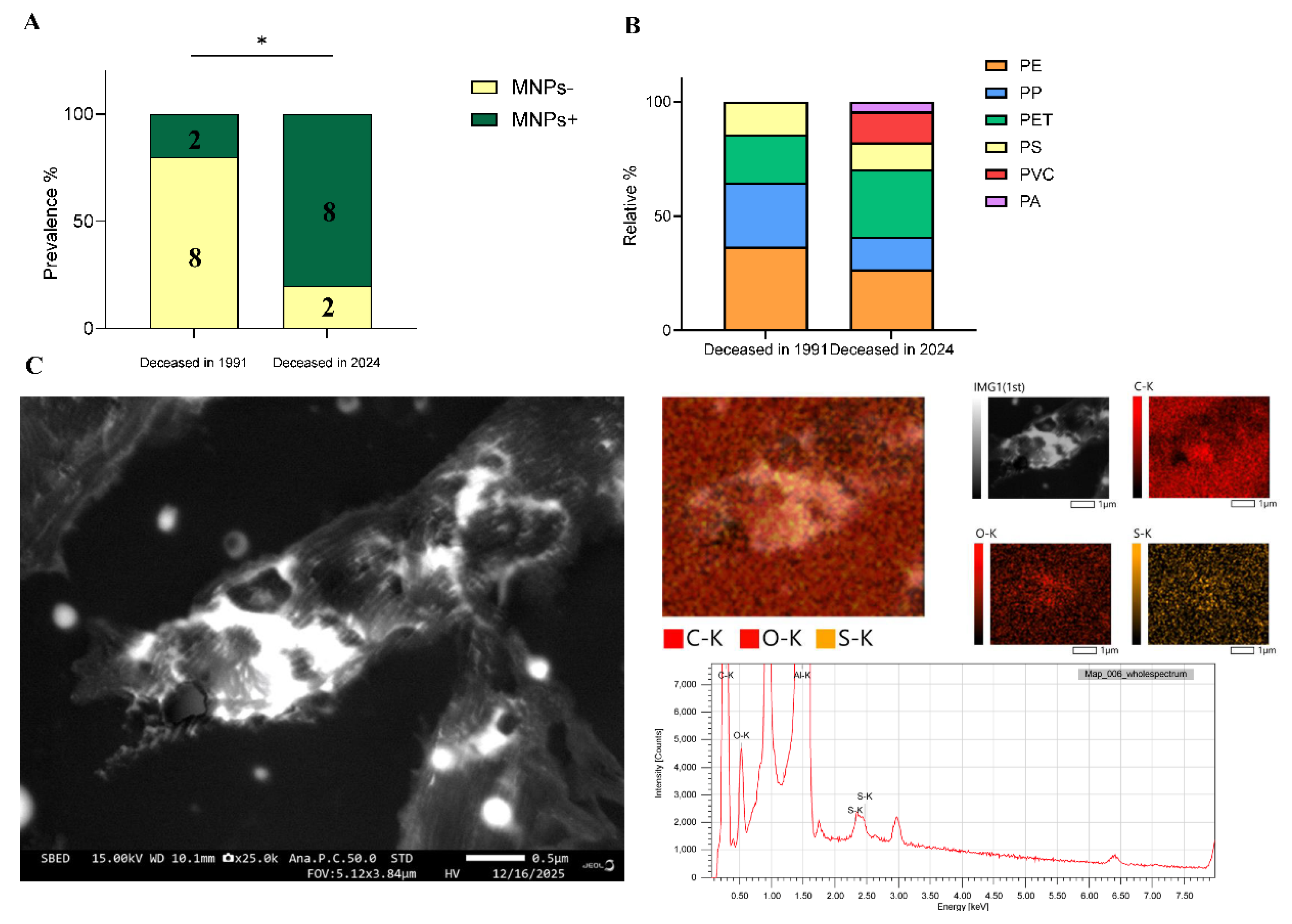
Results
Discussion
Supplementary Materials
Author Contributions
Funding
Conflict of Interest
References
- Landrigan, P. J.; et al. The Lancet Countdown on health and plastics. Lancet 2025, 406, 1044–1062. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; et al. Potential Health Impact of Microplastics: A Review of Environmental Distribution, Human Exposure, and Toxic Effects. Environment & health 2023, 1, 249–257. [Google Scholar] [CrossRef]
- Marfella, R.; et al. Microplastics and Nanoplastics in Atheromas and Cardiovascular Events. The New England journal of medicine 2024, 390, 900–910. [Google Scholar] [CrossRef] [PubMed]
- Roslan, N. S.; et al. Detection of microplastics in human tissues and organs: A scoping review. Journal of global health 2024, 14, 04179. [Google Scholar] [CrossRef] [PubMed]
- Nihart, A. J.; et al. Bioaccumulation of microplastics in decedent human brains. Nature medicine 2025, 31, 1114–1119. [Google Scholar] [CrossRef] [PubMed]
- Prattichizzo, F.; et al. Micro-nanoplastics and cardiovascular diseases: evidence and perspectives. European heart journal 2024, 45, 4099–4110. [Google Scholar] [CrossRef] [PubMed]
- Geyer, R.; Jambeck, J. R.; Law, K. L. Production, use, and fate of all plastics ever made. Science advances 2017, 3, e1700782. [Google Scholar] [CrossRef] [PubMed]
- Miyazono, K.; et al. Long-Term Changes in the Abundance, Size, and Morphotype of Marine Plastics in the North Pacific. Environmental science & technology 2025, 59, 4608–4617. [Google Scholar] [CrossRef]
- Eberhard, T.; Casillas, G.; Zarus, G. M.; Barr, D. B. Systematic review of microplastics and nanoplastics in indoor and outdoor air: identifying a framework and data needs for quantifying human inhalation exposures. Journal of exposure science & environmental epidemiology 2024, 34, 185–196. [Google Scholar] [CrossRef]
- Laumbach, R. J.; Kipen, H. M. Respiratory health effects of air pollution: update on biomass smoke and traffic pollution. The Journal of allergy and clinical immunology 2012, 129, 3-11; quiz 12-13. [Google Scholar] [CrossRef] [PubMed]
- Thomas, K.V.; et al. Communicating Confidence in the Reliability of Micro- and Nanoplastic Identification in Human Health Studies. Environment & Health Article ASAP. [CrossRef]
- Amato-Lourenco, L. F.; et al. Presence of airborne microplastics in human lung tissue. Journal of hazardous materials 2021, 416, 126124. [Google Scholar] [CrossRef] [PubMed]
- Jenner, L. C.; et al. Detection of microplastics in human lung tissue using muFTIR spectroscopy. The Science of the total environment 2022, 831, 154907. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; et al. Microplastics in the Lung Tissues Associated with Blood Test Index. Toxics 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Vasse, G. F.; Melgert, B. N. Microplastic and plastic pollution: impact on respiratory disease and health. European respiratory review: an official journal of the European Respiratory Society 2024, 33. [Google Scholar] [CrossRef] [PubMed]
| Variable | Deceased in 1991 (n=42) |
Deceased in 2024 (n=57) |
p-value |
|---|---|---|---|
| Age (years) | 63,5 (54.3-67.8) | 65 (59-71) | 0.111 |
| Males, n (%) | 29 (69.1) | 34 (59.6) | 0.401 |
| Nationality | |||
| Italian, n (%) | 39 (92.9) | 50 (87.7) | 0.518 |
| Other, n (%) | 3 (7.1) | 7 (12.3) | 0.518 |
| Manner of death | |||
| Natural cause, n (%) | 29 (69.1) | 37 (64.9) | 0.830 |
| Non-natural cause, n (%) | 13 (30.9) | 20 (35.1) | 0.830 |
| Mechanism of death | |||
| Cardiac cause, n (%) | 15 (51.7) | 19 (51.4) | >0.999 |
| Cerebral cause, n (%) | 6 (20.7) | 7 (18.9) | >0.999 |
| Others, n (%) | 8 (27.6) | 11 (29.7) | >0.999 |
| Place of death | |||
| Home, n (%) | 29 (69) | 38 (66.7) | 0.831 |
| Hospital, n (%) | 2 (4.8) | 5 (8.8) | 0.695 |
| Others, n (%) | 11 (26.2) | 14 (24.5) | >0.999 |
| Season of death | |||
| Spring, n (%) | 9 (21.4) | 14 (24.6) | 0.812 |
| Summer, n (%) | 10 (23.8) | 15 (26.3) | 0.819 |
| Autumn, n (%) | 12 (28.6) | 13 (22.8) | 0.641 |
| Winter, n (%) | 11 (26.2) | 15 (26.3) | >0.999 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




