Submitted:
09 February 2026
Posted:
10 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Candida albicans
2.2. Mice
2.3. In Vivo VVC Model
2.4. CVL Collection
2.5. Fungal Burden (CFU/ml)
2.6. PMNs Quantification
2.7. Cytokines Quantification
2.8. Immunofluorescence for the Evaluation of mBD1 and mBD3 Expression
2.9. Statistical Analysis
3. Results
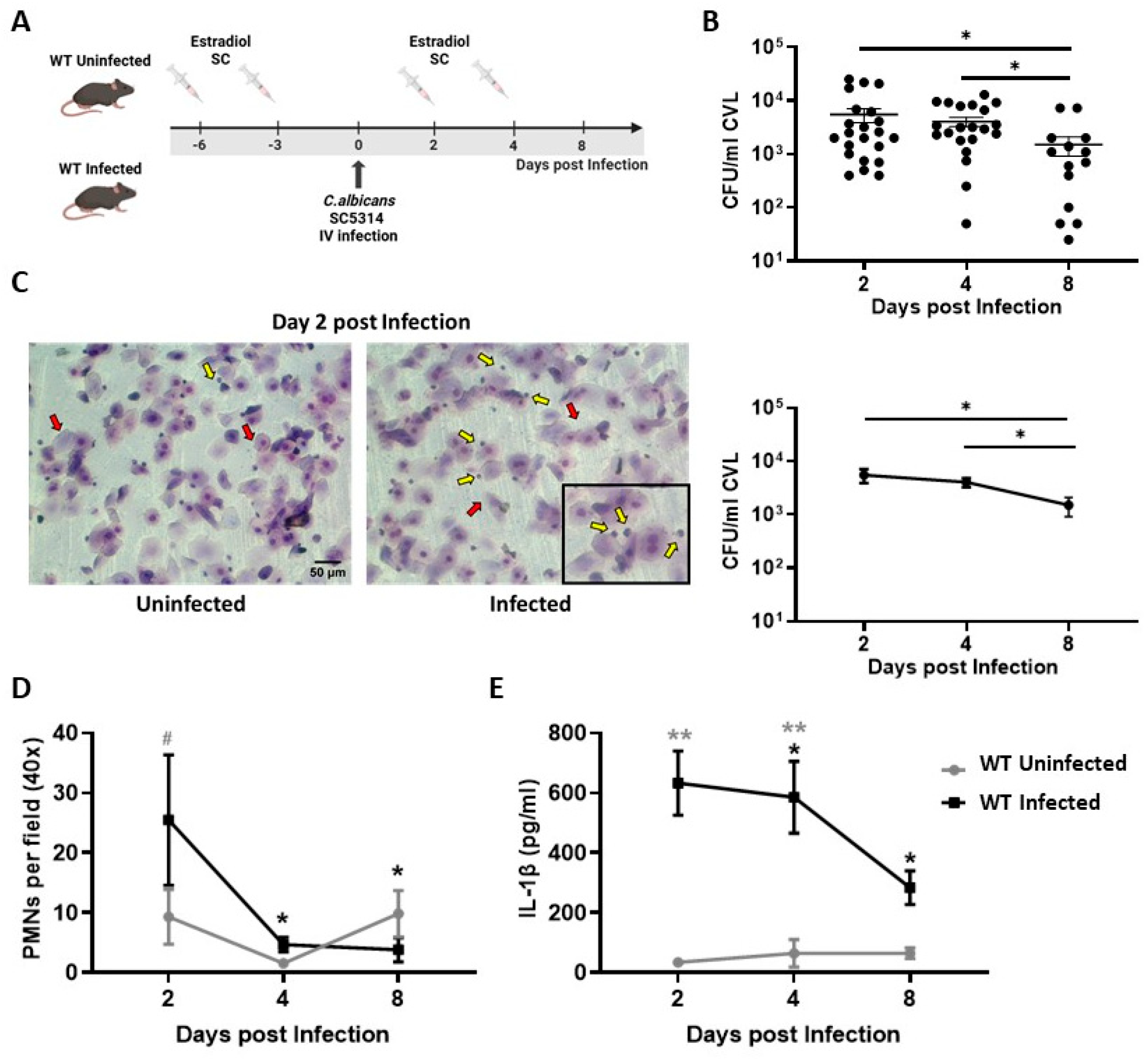
3.1. Establishment of Infection and Local Inflammatory Profile During Experimental Vulvovaginal Candidiasis in C57BL/6 (WT) Mice
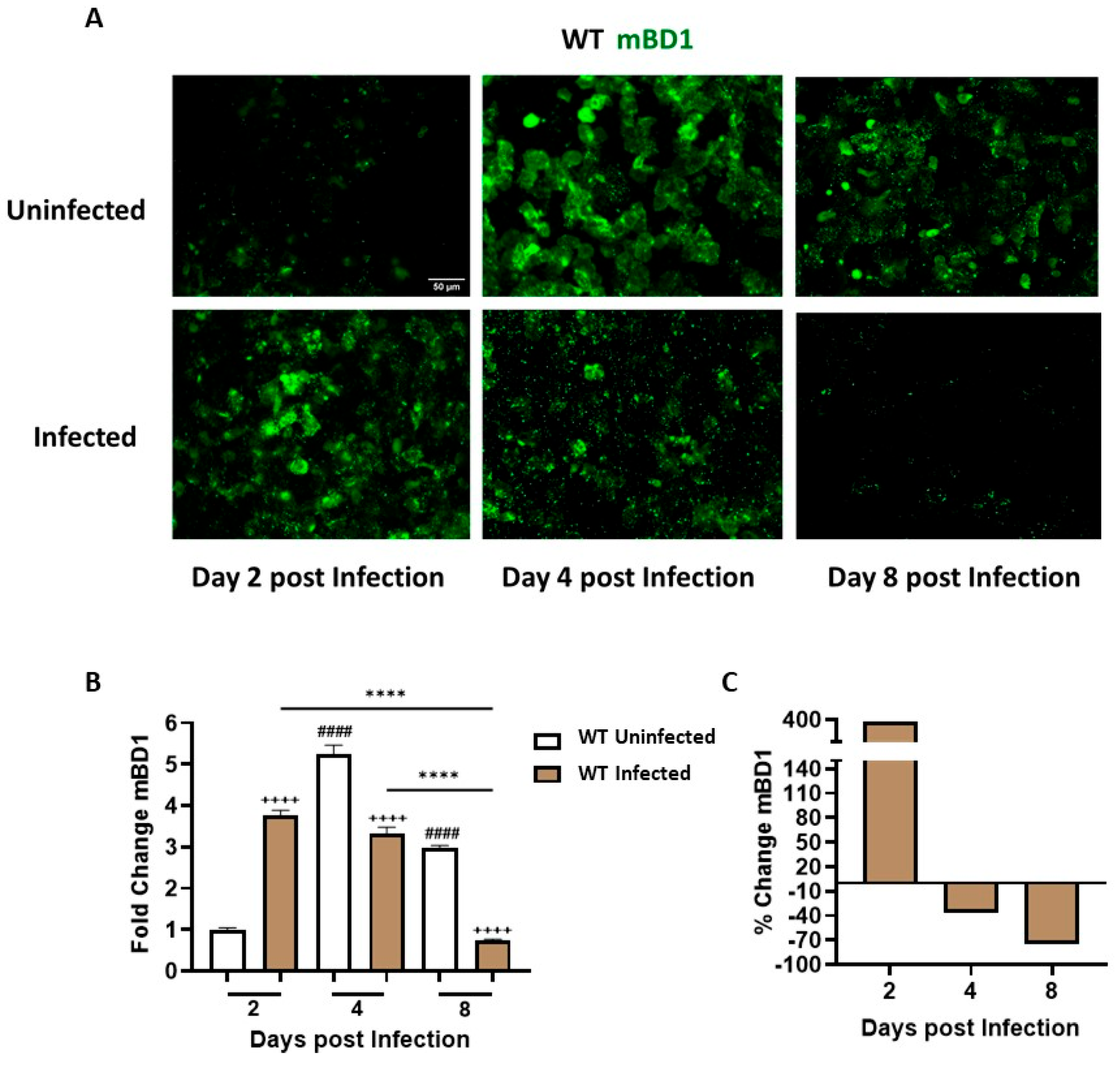
3.2. C. albicans SC5314 Infection Differentially Modulates mBD1 Expression in the Vaginal Tract
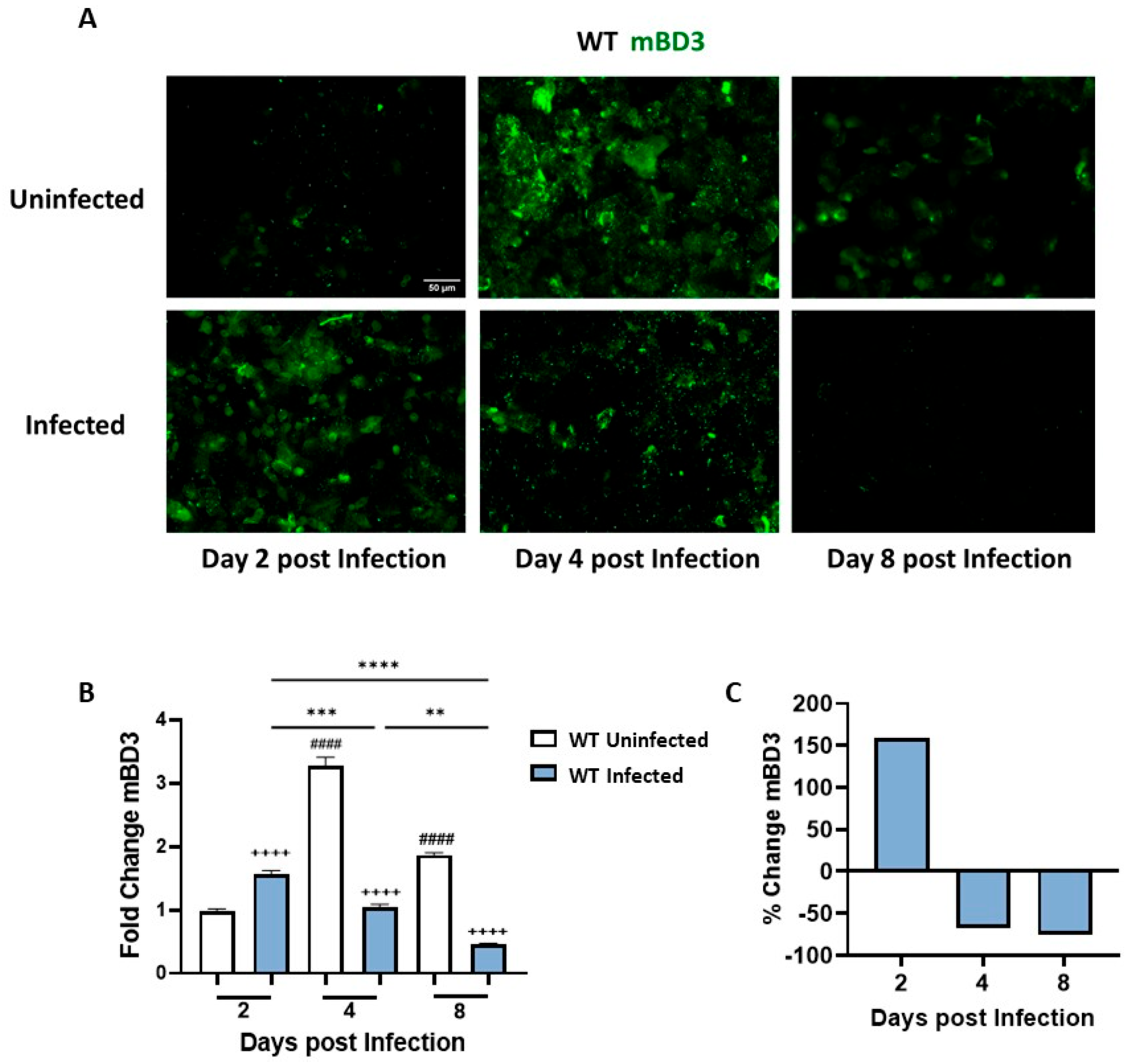
3.3. C. albicans SC5314 Infection Differentially Modulates mBD3 Expression in the Vaginal Tract
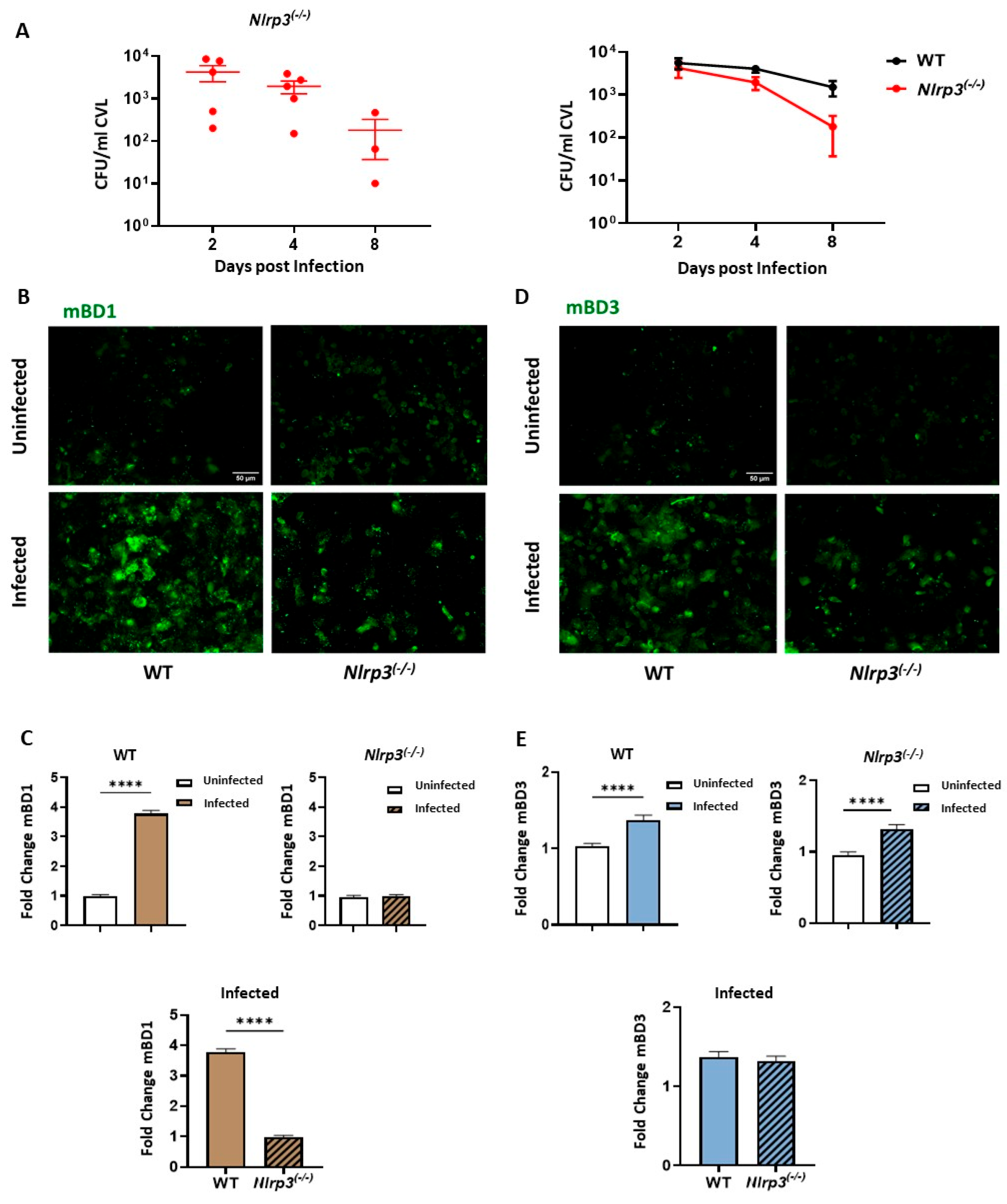
3.4. NLRP3 Inflammasome Pathway Activation Drives mBD1 but not mBD3 Induction during C. albicans Vaginal Infection
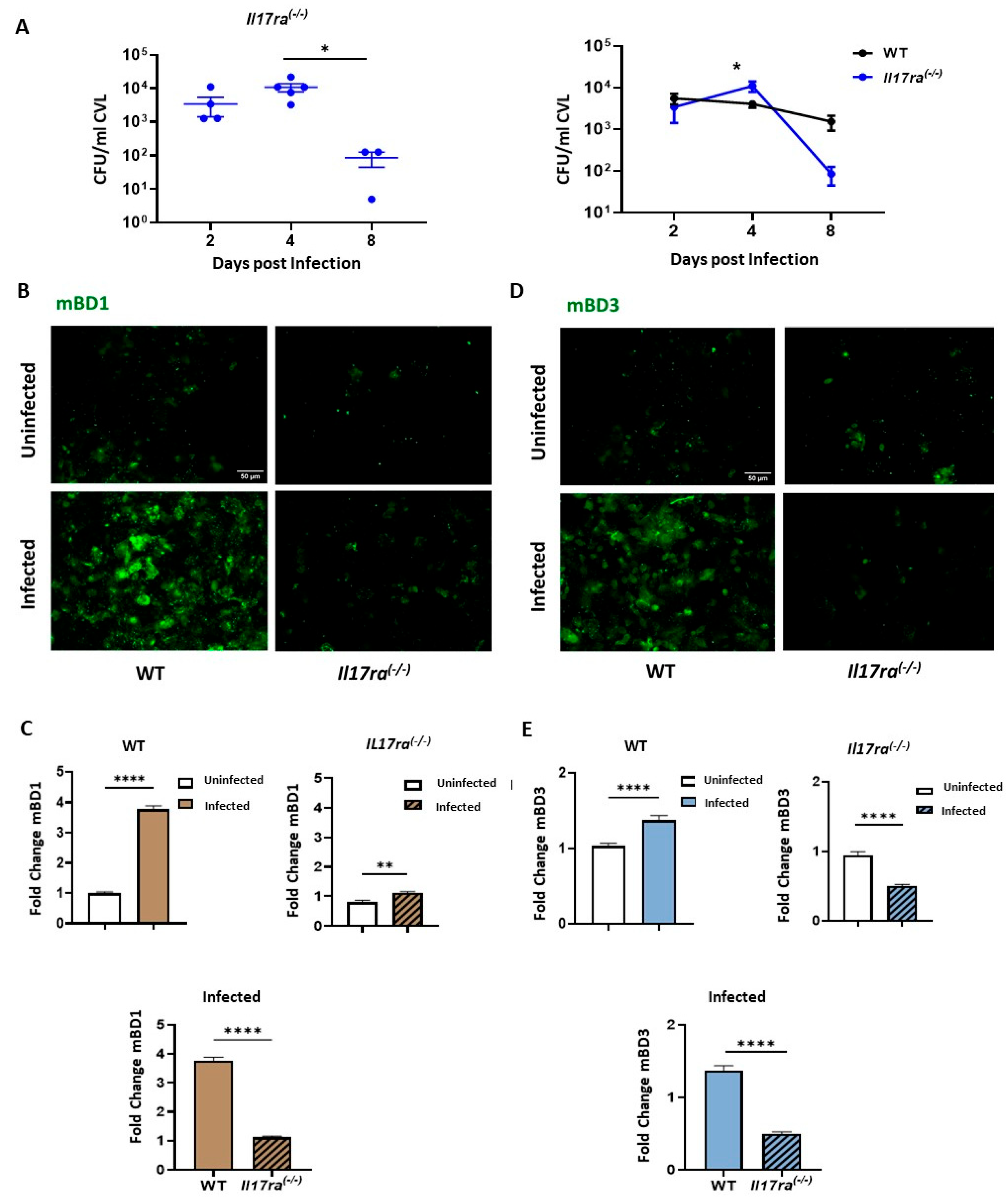
3.5. IL-17RA Signaling Differentially Regulates mBD1 and mBD3 Expression during C. albicans Vaginal Infection
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sobel JD. Vulvovaginal candidosis. The Lancet 2007;369:1961–71. [CrossRef]
- Achkar JM, Fries BC. Candida Infections of the Genitourinary Tract. Clinical Microbiology Reviews 2010;23:253–73. [CrossRef]
- Valentine M, Wilson D, Gresnigt MS, Hube B. Vaginal Candida albicans infections: host–pathogen–microbiome interactions. FEMS Microbiology Reviews 2025;49. [CrossRef]
- Denning DW, Kneale M, Sobel JD, Rautemaa-Richardson R. Global burden of recurrent vulvovaginal candidiasis: a systematic review. The Lancet Infectious Diseases 2018;18:e339–47. [CrossRef]
- MacAlpine J, Lionakis MS. Host-microbe interaction paradigms in acute and recurrent vulvovaginal candidiasis. Cell Host & Microbe 2024;32:1654–67. [CrossRef]
- Rosati D, Bruno M, Jaeger M, ten Oever J, Netea MG. Recurrent Vulvovaginal Candidiasis: An Immunological Perspective. Microorganisms 2020;8:144. [CrossRef]
- Peters BM, Palmer GE, Nash AK, Lilly EA, Fidel PL, Noverr MC. Fungal Morphogenetic Pathways Are Required for the Hallmark Inflammatory Response during Candida albicans Vaginitis. Infection and Immunity 2014;82:532–43. [CrossRef]
- Mayer FL, Wilson D, Hube B. Candida albicans pathogenicity mechanisms. Virulence 2013;4:119–28. [CrossRef]
- Roselletti E, Monari C, Sabbatini S, Perito S, Vecchiarelli A, Sobel JD, et al. A Role for Yeast/Pseudohyphal Cells of Candida albicans in the Correlated Expression of NLRP3 Inflammasome Inducers in Women With Acute Vulvovaginal Candidiasis. Frontiers in Microbiology 2019;10. [CrossRef]
- Yano J, Sobel JD, Nyirjesy P, Sobel R, Williams VL, Yu Q, et al. Current patient perspectives of vulvovaginal candidiasis: incidence, symptoms, management and post-treatment outcomes. BMC Women’s Health 2019;19:48. [CrossRef]
- Ardizzoni A, Wheeler RT, Pericolini E. It Takes Two to Tango: How a Dysregulation of the Innate Immunity, Coupled With Candida Virulence, Triggers VVC Onset. Frontiers in Microbiology 2021;12. [CrossRef]
- Cheng KO, Montaño DE, Zelante T, Dietschmann A, Gresnigt MS. Inflammatory cytokine signalling in vulvovaginal candidiasis: a hot mess driving immunopathology. Oxford Open Immunology 2024;5. [CrossRef]
- Conti HR, Shen F, Nayyar N, Stocum E, Sun JN, Lindemann MJ, et al. Th17 cells and IL-17 receptor signaling are essential for mucosal host defense against oral candidiasis. Journal of Experimental Medicine 2009;206:299–311. [CrossRef]
- Dixon BREA, Radin JN, Piazuelo MB, Contreras DC, Algood HMS. IL-17a and IL-22 Induce Expression of Antimicrobials in Gastrointestinal Epithelial Cells and May Contribute to Epithelial Cell Defense against Helicobacter pylori. PLOS ONE 2016;11:e0148514. [CrossRef]
- Pietrella D, Rachini A, Pines M, Pandey N, Mosci P, Bistoni F, et al. Th17 Cells and IL-17 in Protective Immunity to Vaginal Candidiasis. PLoS ONE 2011;6:e22770. [CrossRef]
- Peters BM, Coleman BM, Willems HME, Barker KS, Aggor FEY, Cipolla E, et al. The Interleukin (IL) 17R/IL-22R Signaling Axis Is Dispensable for Vulvovaginal Candidiasis Regardless of Estrogen Status. The Journal of Infectious Diseases 2020;221:1554–63. [CrossRef]
- Schutte BC, Mitros JP, Bartlett JA, Walters JD, Jia HP, Welsh MJ, et al. Discovery of five conserved β-defensin gene clusters using a computational search strategy. Proceedings of the National Academy of Sciences 2002;99:2129–33. [CrossRef]
- Shelley JR, Davidson DJ, Dorin JR. The Dichotomous Responses Driven by β-Defensins. Frontiers in Immunology 2020;11. [CrossRef]
- Yang D, Chertov O, Bykovskaia SN, Chen Q, Buffo MJ, Shogan J, et al. β-Defensins: Linking Innate and Adaptive Immunity Through Dendritic and T Cell CCR6. Science 1999;286:525–8. [CrossRef]
- Shimbo M, Ito N, Kadonosono K. [Investigation of beta-D-glucan values in the vitreous]. Nippon Ganka Gakkai Zasshi 2002;106:579–82.
- Funderburg N, Lederman MM, Feng Z, Drage MG, Jadlowsky J, Harding C V., et al. Human β-defensin-3 activates professional antigen-presenting cells via Toll-like receptors 1 and 2. Proceedings of the National Academy of Sciences 2007;104:18631–5. [CrossRef]
- Fesahat F, Firouzabadi AM, Zare-Zardini H, Imani M. Roles of Different β-Defensins in the Human Reproductive System: A Review Study. American Journal of Men’s Health 2023;17. [CrossRef]
- Miró MS, Caeiro JP, Rodriguez E, Vargas L, Vigezzi C, Icely PA, et al. Candida albicans Modulates Murine and Human Beta Defensin-1 during Vaginitis. Journal of Fungi 2021;8:20. [CrossRef]
- Vigezzi C, Icely PA, Dudiuk C, Rodríguez E, Miró MS, Castillo GDV, et al. Frequency, virulence factors and antifungal susceptibility of Candida parapsilosis species complex isolated from patients with candidemia in the central region of Argentina. Journal de Mycologie Médicale 2019:100907. [CrossRef]
- Pekmezovic M, Hovhannisyan H, Gresnigt MS, Iracane E, Oliveira-Pacheco J, Siscar-Lewin S, et al. Candida pathogens induce protective mitochondria-associated type I interferon signalling and a damage-driven response in vaginal epithelial cells. Nature Microbiology 2021;6:643–57. [CrossRef]
- Rodriguez E, Savid-Frontera C, Angiolini SC, Hernáez-Sánchez ML, Miró MS, Viano ME, et al. Type-I interferons in Vulvovaginal Candidiasis: Mechanism of epithelial early defense and immune regulation against Candida albicans. Mucosal Immunology 2025;18:1124–38. [CrossRef]
- Miró MS, Rodríguez E, Vigezzi C, Icely PA, Gonzaga de Freitas Araújo M, Riera FO, et al. Candidiasis vulvovaginal: una antigua enfermedad con nuevos desafíos. Revista Iberoamericana de Micología 2017;34:65–71. [CrossRef]
- Yano J, Palmer GE, Eberle KE, Peters BM, Vogl T, McKenzie AN, et al. Vaginal Epithelial Cell-Derived S100 Alarmins Induced by Candida albicans via Pattern Recognition Receptor Interactions Are Sufficient but Not Necessary for the Acute Neutrophil Response during Experimental Vaginal Candidiasis. Infection and Immunity 2014;82:783–92. [CrossRef]
- de Freitas Araújo MG, Pacífico M, Vilegas W, Dos Santos LC, Icely PA, Miró MS, et al. Evaluation of Syngonanthus nitens (Bong.) Ruhl. extract as antifungal and in treatment of vulvovaginal candidiasis. Medical Mycology 2013;51:673–82. [CrossRef]
- Renna MS, Correa SG, Porporatto C, Figueredo CM, Aoki MP, Paraje MG, et al. Hepatocellular apoptosis during Candida albicans colonization: involvement of TNF- and infiltrating Fas-L positive lymphocytes. International Immunology 2006;18:1719–28. [CrossRef]
- Yarbrough VL, Winkle S, Herbst-Kralovetz MM. Antimicrobial peptides in the female reproductive tract: a critical component of the mucosal immune barrier with physiological and clinical implications. Human Reproduction Update 2015;21:353–77. [CrossRef]
- Krishnakumari V, Rangaraj N, Nagaraj R. Antifungal Activities of Human Beta-Defensins HBD-1 to HBD-3 and Their C-Terminal Analogs Phd1 to Phd3. Antimicrobial Agents and Chemotherapy 2009;53:256–60. [CrossRef]
- Bruno VM, Shetty AC, Yano J, Fidel PL, Noverr MC, Peters BM. Transcriptomic Analysis of Vulvovaginal Candidiasis Identifies a Role for the NLRP3 Inflammasome. MBio 2015;6. [CrossRef]
- Sobel JD. Recurrent vulvovaginal candidiasis. American Journal of Obstetrics and Gynecology 2016;214:15–21. [CrossRef]
- Fidel PL, Barousse M, Espinosa T, Ficarra M, Sturtevant J, Martin DH, et al. An Intravaginal Live Candida Challenge in Humans Leads to New Hypotheses for the Immunopathogenesis of Vulvovaginal Candidiasis. Infection and Immunity 2004;72:2939–46. [CrossRef]
- Yano J, Peters BM, Noverr MC, Fidel PL. Novel Mechanism behind the Immunopathogenesis of Vulvovaginal Candidiasis: “Neutrophil Anergy.” Infection and Immunity 2018;86. [CrossRef]
- Yano J, Noverr MC, Fidel PL. Vaginal Heparan Sulfate Linked to Neutrophil Dysfunction in the Acute Inflammatory Response Associated with Experimental Vulvovaginal Candidiasis. MBio 2017;8. [CrossRef]
- Fusco A, Savio V, Donniacuo M, Perfetto B, Donnarumma G. Antimicrobial Peptides Human Beta-Defensin-2 and -3 Protect the Gut During Candida albicans Infections Enhancing the Intestinal Barrier Integrity: In Vitro Study. Frontiers in Cellular and Infection Microbiology 2021;11. [CrossRef]
- Hickey DK, Fahey J V, Wira CR. Mouse estrous cycle regulation of vaginal versus uterine cytokines, chemokines, α-/β-defensins and TLRs. Innate Immunity 2013;19:121–31. [CrossRef]
- Prado-Montes de Oca E. Human β-defensin 1: A restless warrior against allergies, infections and cancer. The International Journal of Biochemistry & Cell Biology 2010;42:800–4. [CrossRef]
- Burd RS, Furrer JL, Sullivan J, Smith AL. Murine ??-Defensin-3 Is an Inducible Peptide with Limited Tissue Expression and Broad-Spectrum Antimicrobial Activity. Shock 2002;18:461–4. [CrossRef]
- Tomalka J, Azodi E, Narra HP, Patel K, O’Neill S, Cardwell C, et al. β-Defensin 1 Plays a Role in Acute Mucosal Defense against Candida albicans. The Journal of Immunology 2015;194:1788–95. [CrossRef]
- Palrasu M, Marudamuthu A, Kakar K, Hamida H, Thada S, Gupta R, et al. AhR-Dependent Induction of β-Defensin 1 in Colonic Epithelial Cells Regulates Cross-Talk between Gut Microbiota and Immune Response Leading to Attenuation of Colitis. Advanced Science 2025;12. [CrossRef]
- Semple F, Dorin JR. β-Defensins: Multifunctional Modulators of Infection, Inflammation and More? Journal of Innate Immunity 2012;4:337–48. [CrossRef]
- Noda-Nicolau NM, Bastos LB, Bolpetti AN, Pinto GVS, Marcolino LD, Marconi C, et al. Cervicovaginal Levels of Human β-Defensin 1, 2, 3, and 4 of Reproductive-Aged Women With Chlamydia trachomatis Infection. Journal of Lower Genital Tract Disease 2017;21:189–92. [CrossRef]
- Zhai Y-J, Feng Y, Ma X, Ma F. Defensins: defenders of human reproductive health. Human Reproduction Update 2023;29:126–54. [CrossRef]
- Chakraborty K, Ghosh S, Koley H, Mukhopadhyay AK, Ramamurthy T, Saha DR, et al. Bacterial exotoxins downregulate cathelicidin (hCAP-18/LL-37) and human β-defensin 1 (HBD-1) expression in the intestinal epithelial cells. Cellular Microbiology 2008;10:2520–37. [CrossRef]
- Bauer B, Pang E, Holland C, Kessler M, Bartfeld S, Meyer TF. The Helicobacter pylori Virulence Effector CagA Abrogates Human β-Defensin 3 Expression via Inactivation of EGFR Signaling. Cell Host & Microbe 2012;11:576–86. [CrossRef]
- Patel SR, Smith K, Letley DP, Cook KW, Memon AA, Ingram RJM, et al. H elicobacter pylori downregulates expression of human β-defensin 1 in the gastric mucosa in a type IV secretion-dependent fashion. Cellular Microbiology 2013;15:2080–92. [CrossRef]
- Hise AG, Tomalka J, Ganesan S, Patel K, Hall BA, Brown GD, et al. An Essential Role for the NLRP3 Inflammasome in Host Defense against the Human Fungal Pathogen Candida albicans. Cell Host & Microbe 2009;5:487–97. [CrossRef]
- Jaeger M, Carvalho A, Cunha C, Plantinga TS, van de Veerdonk F, Puccetti M, et al. Association of a variable number tandem repeat in the NLRP3 gene in women with susceptibility to RVVC. European Journal of Clinical Microbiology & Infectious Diseases 2016;35:797–801. [CrossRef]
- Pietrella D, Pandey N, Gabrielli E, Pericolini E, Perito S, Kasper L, et al. Secreted aspartic proteases of Candida albicans activate the NLRP3 inflammasome. European Journal of Immunology 2013;43:679–92. [CrossRef]
- Pericolini E, Gabrielli E, Amacker M, Kasper L, Roselletti E, Luciano E, et al. Secretory Aspartyl Proteinases Cause Vaginitis and Can Mediate Vaginitis Caused by Candida albicans in Mice. MBio 2015;6. [CrossRef]
- Pahl R, Brunke G, Steubesand N, Schubert S, Böttner M, Wedel T, et al. IL-1β and ADAM17 are central regulators of β-defensin expression in Candida esophagitis. American Journal of Physiology-Gastrointestinal and Liver Physiology 2011;300:G547–53. [CrossRef]
- Zaga-Clavellina V, Martha RV-M, Flores-Espinosa P. In Vitro Secretion Profile of Pro-Inflammatory Cytokines IL-1β, TNF-α, IL-6, and of Human Beta-Defensins (HBD)-1, HBD-2, and HBD-3 from Human Chorioamniotic Membranes After Selective Stimulation with Gardnerella vaginalis. American Journal of Reproductive Immunology 2012;67:34–43. [CrossRef]
- Mao F-Y, Lv Y-P, Hao C-J, Teng Y-S, Liu Y-G, Cheng P, et al. Helicobacter pylori–Induced Rev-erbα Fosters Gastric Bacteria Colonization by Impairing Host Innate and Adaptive Defense. Cellular and Molecular Gastroenterology and Hepatology 2021;12:395–425. [CrossRef]
- Fidel PL, Cutright J, Steele C. Effects of Reproductive Hormones on Experimental Vaginal Candidiasis. Infection and Immunity 2000;68:651–7. [CrossRef]
- Yano J, Kolls JK, Happel KI, Wormley F, Wozniak KL, Fidel PL. The Acute Neutrophil Response Mediated by S100 Alarmins during Vaginal Candida Infections Is Independent of the Th17-Pathway. PLoS ONE 2012;7:e46311. [CrossRef]
- Zelante T, Iannitti RG, Cunha C, De Luca A, Giovannini G, Pieraccini G, et al. Tryptophan Catabolites from Microbiota Engage Aryl Hydrocarbon Receptor and Balance Mucosal Reactivity via Interleukin-22. Immunity 2013;39:372–85. [CrossRef]
- Lee AJ, Ashkar AA. The Dual Nature of Type I and Type II Interferons. Frontiers in Immunology 2018;9. [CrossRef]
- Verma A, Wuthrich M, Deepe G, Klein B. Adaptive Immunity to Fungi. Cold Spring Harbor Perspectives in Medicine 2015;5:a019612–a019612. [CrossRef]
- Rahman A, Fahlgren A, Sundstedt C, Hammarström S, Danielsson Å, Hammarström M-L. Chronic colitis induces expression of β-defensins in murine intestinal epithelial cells. Clinical and Experimental Immunology 2010;163:123–30. [CrossRef]
- Archer NK, Adappa ND, Palmer JN, Cohen NA, Harro JM, Lee SK, et al. Interleukin-17A (IL-17A) and IL-17F Are Critical for Antimicrobial Peptide Production and Clearance of Staphylococcus aureus Nasal Colonization. Infection and Immunity 2016;84:3575–83. [CrossRef]
- Kao C-Y, Chen Y, Thai P, Wachi S, Huang F, Kim C, et al. IL-17 Markedly Up-Regulates β-Defensin-2 Expression in Human Airway Epithelium via JAK and NF-κB Signaling Pathways. The Journal of Immunology 2004;173:3482–91. [CrossRef]
- Liang SC, Tan X-Y, Luxenberg DP, Karim R, Dunussi-Joannopoulos K, Collins M, et al. Interleukin (IL)-22 and IL-17 are coexpressed by Th17 cells and cooperatively enhance expression of antimicrobial peptides. The Journal of Experimental Medicine 2006;203:2271–9. [CrossRef]
- Corleis B, Lisanti AC, Körner C, Schiff AE, Rosenberg ES, Allen TM, et al. Early type I Interferon response induces upregulation of human β-defensin 1 during acute HIV-1 infection. PLOS ONE 2017;12:e0173161. [CrossRef]
- Smeekens SP, Ng A, Kumar V, Johnson MD, Plantinga TS, van Diemen C, et al. Functional genomics identifies type I interferon pathway as central for host defense against Candida albicans. Nature Communications 2013;4:1342. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



