Submitted:
09 February 2026
Posted:
10 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
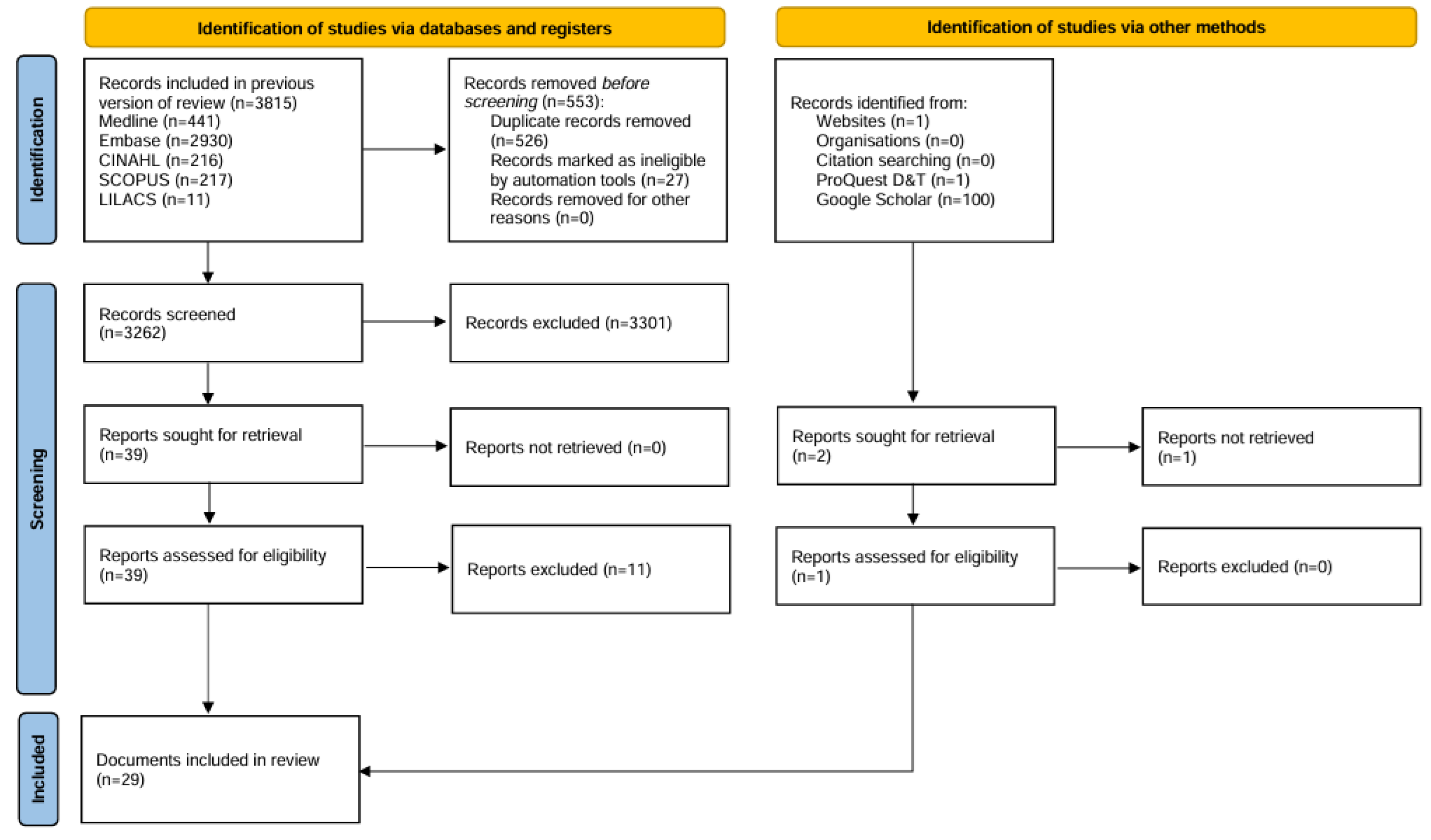
2. Materials and Methods
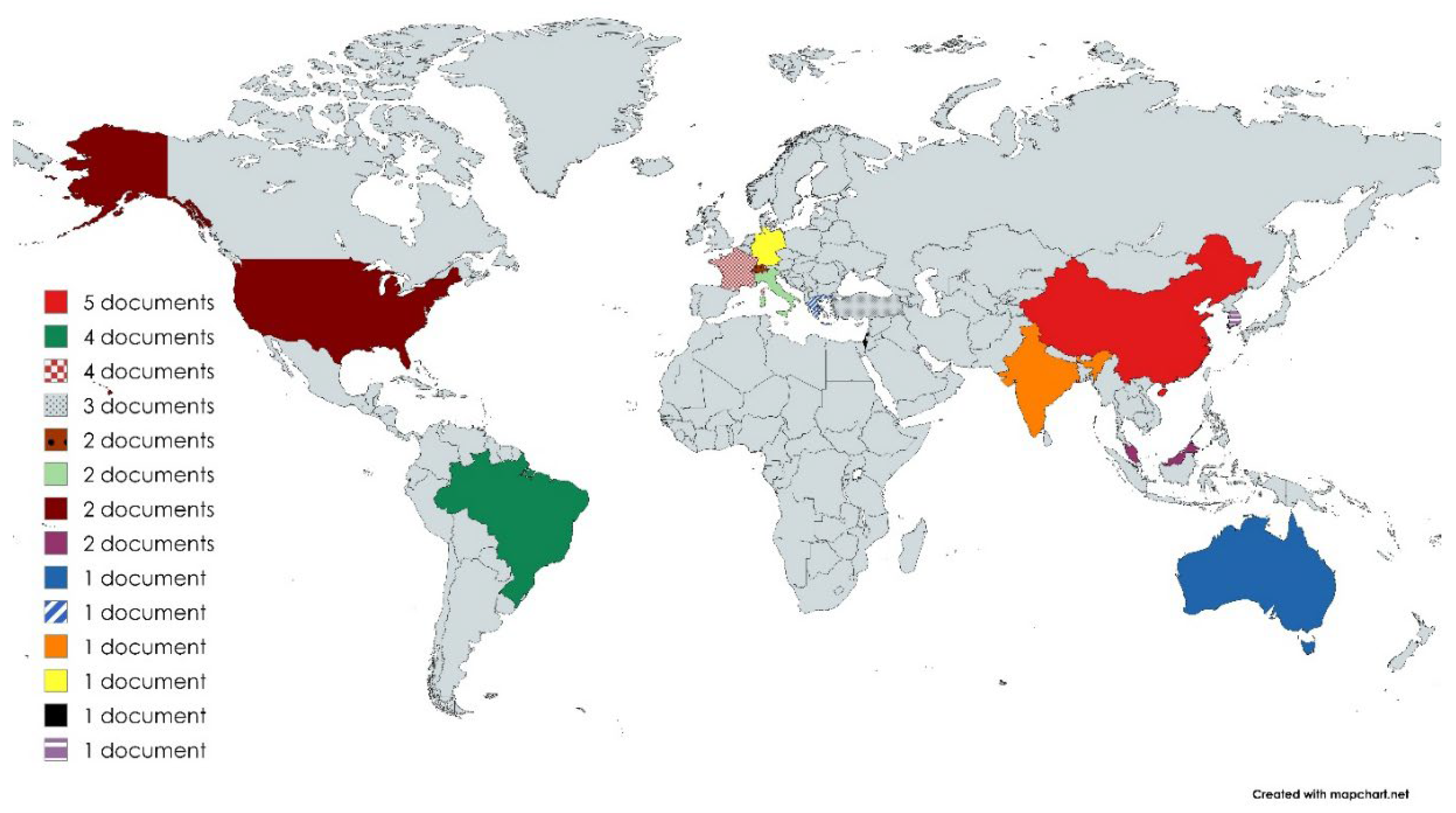
3. Results
3.1. Clinical Settings
3.2. Indication (I)
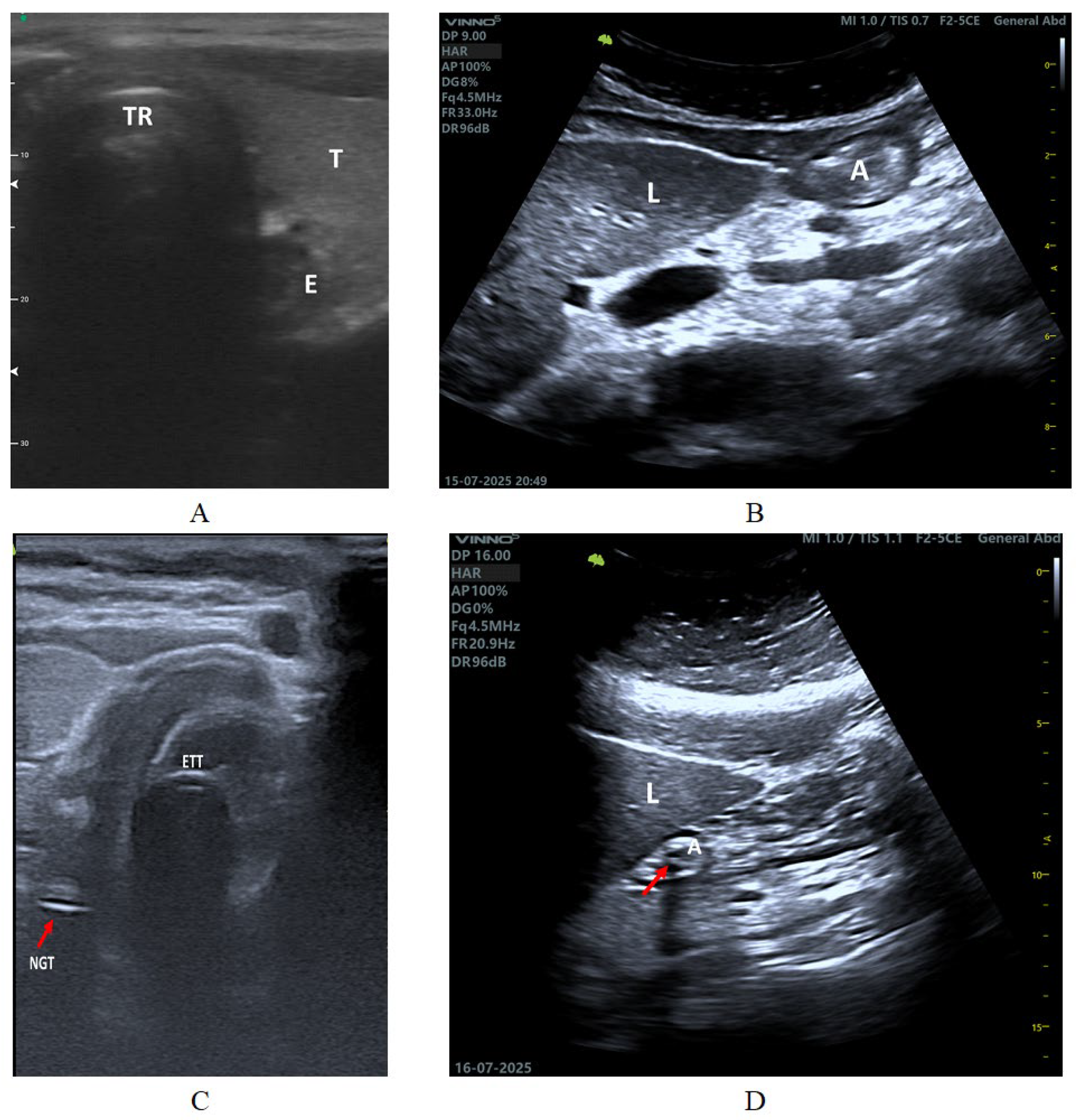
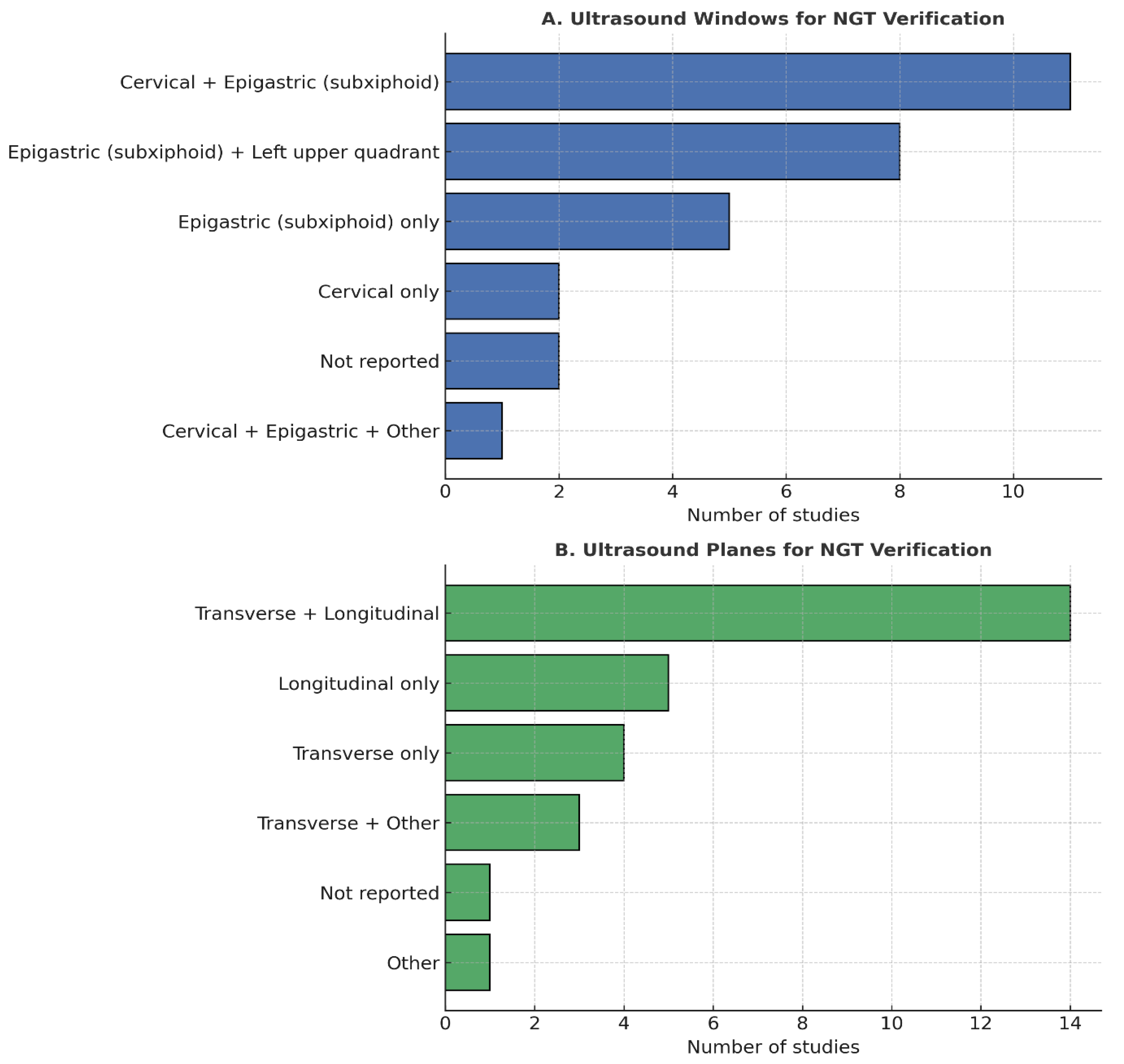
3.3. Acquisition (A)
3.4. Interpretation (I)
3.5. Decision-Making
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BVS | Biblioteca Virtual de Saúde |
| CINAHL | Cumulative Index to Nursing and Allied Health Literature |
| DeCS | Descritores em Saúde (Health Sciences Descriptors) |
| GRV | Gastric Residual Volume |
| I-AIM | Indication, Acquisition, Interpretation, and decision-Making |
| ICU | Intensive Care Unit |
| LILACS | Literatura Latino-Americana e do Caribe em Ciências da Saúde |
| MeSH | Medical Subject Headings |
| NGT | Nasogastric Tube |
| PoCUS | Point-of-Care Ultrasonography |
| PRISMA | Preferred Reporting Items for Systematic reviews and Meta-Analyses |
References
- World Health Organization. Global Patient Safety Action Plan 2021–2030: Towards Eliminating Avoidable Harm in Health Care; WHO: Geneva, Switzerland, 2021. [Google Scholar]
- NHS England. Patient Safety Alert: Nasogastric Tube Misplacement—Continuing Risk of Death and Severe Harm; NHS England: London, UK, 2016. [Google Scholar]
- Zafran, M.; Crook, R.; Tuck, A.; Banerjee, A. Nasogastric tube placement perforating the nasopharynx causing mediastinal passage and feeding into the pleural space. BMJ Case Rep. 2024, 17, e254771. [Google Scholar] [CrossRef]
- Gok, F.; Kilicaslan, A.; Yosunkaya, A. Ultrasound-guided nasogastric feeding tube placement in critical care patients. Nutr. Clin. Pract. 2015, 30, 257–260. [Google Scholar] [CrossRef]
- NHS England. Never Events List 2018; NHS England: London, UK, 2021. [Google Scholar]
- Metheny, N.A.; Krieger, M.M.; Healey, F.; Meert, K.L. A review of guidelines to distinguish between gastric and pulmonary placement of nasogastric tubes. Heart Lung 2019, 48, 226–235. [Google Scholar] [CrossRef]
- Bourgault, A.M.; Heath, J.; Hooper, V.; Sole, M.L.; Nesmith, E.G. Methods used by critical care nurses to verify feeding tube placement in clinical practice. Crit. Care Nurse 2015, 35, e1–e7. [Google Scholar] [CrossRef]
- Fan, X.-N.; et al. Application of nurse-led enteral nutritional feeding strategies in neurological intensive care unit patients. Chin. J. Gen. Pract. 2022, 20, 2151–2155. [Google Scholar] [CrossRef]
- Taylor, S.; Manara, A.R. X-ray checks of NG tube position: A case for guided tube placement. Br. J. Radiol. 2021, 94, 20210432. [Google Scholar] [CrossRef]
- Boeykens, K.; Holvoet, T.; Duysburgh, I. Nasogastric tube insertion length measurement and tip verification in adults: A narrative review. Crit. Care 2023, 27, 317. [Google Scholar] [CrossRef]
- Powers, J.; et al. Development of a competency model for placement and verification of nasogastric and nasoenteric feeding tubes for adult hospitalized patients. Nutr. Clin. Pract. 2021, 36, 517–533. [Google Scholar] [CrossRef] [PubMed]
- Rigobello, M.C.G.; et al. Accuracy and costs of bedside methods for confirming nasoenteral feeding tube position: A diagnostic accuracy study. J. Ultrasound 2024. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Li, Q.; Wu, X.; Wang, X.; Liu, D. Nurse-performed ultrasound: A new weapon against COVID-19. Crit. Care 2020, 24, 430. [Google Scholar] [CrossRef]
- Wang, L.; et al. Association of gastric antrum echodensity and acute gastrointestinal injury in critically ill patients. Nutrients 2022, 14, 566. [Google Scholar] [CrossRef] [PubMed]
- Bloom, L.; Seckel, M.A. Placement of nasogastric feeding tube and postinsertion care review. AACN Adv. Crit. Care 2022, 33, 68–84. [Google Scholar] [CrossRef] [PubMed]
- Melender, H.-L.; Maijala, V. Tactics for teaching evidence-based practice: Video as a support for learning international database searching in nursing education. Worldviews Evid. Based Nurs. 2018, 15, 326–328. [Google Scholar] [CrossRef] [PubMed]
- Jones, E.P. How to conduct a review of the literature. J. Dermatol. Physician Assist. 2023, 17. Available online: https://journals.lww.com/jdpa/fulltext/2023/17020/how_to_conduct_a_review_of_the_literature.9.aspx (accessed on 30 Jan 2026). [CrossRef]
- Page, M.J.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Blaivas, M. A new point of care ultrasound journal. Crit. Ultrasound J. 2009, 1, 1–2. [Google Scholar] [CrossRef]
- Boullata, J.I.; et al. ASPEN safe practices for enteral nutrition therapy. JPEN J. Parenter. Enteral Nutr. 2017, 41, 15–103. [Google Scholar] [CrossRef]
- Bahner, D.P.; Hughes, D.; Royall, N.A. I-AIM: A novel model for teaching and performing point-of-care ultrasound. J. Ultrasound Med. 2012, 31, 295–300. [Google Scholar] [CrossRef]
- Mak, M.Y.; Tam, G. Ultrasonography for nasogastric tube placement verification: An additional reference. Br. J. Community Nurs. 2020, 25, 328–334. [Google Scholar] [CrossRef]
- Tai, P.; et al. Nurse-performed ultrasonography in confirming the position of nasogastric tube in the emergency department. Hong Kong J. Emerg. Med. 2016, 23, 340–349. [Google Scholar] [CrossRef]
- Wenhong, G.M.; Shunji, G.M.; Qun-An, H.M.; Huijuan, X.M. Bedside ultrasonography-guided nasogastric tube placement in COVID-19 patients. Adv. Ultrasound Diagn. Ther. 2020, 4, 131. [Google Scholar] [CrossRef]
- Zhu, X.-J.; Liu, S.-X.; Li, Q.-T.; Jiang, Y.-J. Bedside ultrasonic localization of the nasogastric tube in a patient with severe COVID-19: A case report. World J. Clin. Cases 2022, 10, 4911–4916. [Google Scholar] [CrossRef]
- De Souza, R.; et al. Ultrasound as a method for confirming the positioning of enteral catheters in critically ill patients. Med. Ultrason. 2024, 26, 242. [Google Scholar] [CrossRef]
- Ferraboli, S.F.; Beghetto, M.G. Bedside ultrasonography for the confirmation of nasogastric tube placement: Agreement between nurse and physician. Rev. Gaucha Enferm. 2022, 43, e20220211. [Google Scholar] [CrossRef]
- Brun, P.-M.; et al. Two-point ultrasonography to confirm correct position of the gastric tube in prehospital setting. Mil. Med. 2014, 179, 959–963. [Google Scholar] [CrossRef]
- Chenaitia, H.; et al. Ultrasound to confirm gastric tube placement in prehospital management. Resuscitation 2012, 83, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Piton, G.; Parel, R.; Delabrousse, E.; Capellier, G. Echography for nasogastric tube placement verification. Eur. J. Clin. Nutr. 2017, 71, 669–670. [Google Scholar] [CrossRef]
- Vigneau, C.; Baudel, J.-L.; Guidet, B.; Offenstadt, G.; Maury, E. Sonography as an alternative to radiography for nasogastric feeding tube location. Intensive Care Med. 2005, 31, 1570–1572. [Google Scholar] [CrossRef] [PubMed]
- Mumoli, N.; et al. Bedside abdominal ultrasound in evaluating nasogastric tube placement. Chest 2021, 159, 2366–2372. [Google Scholar] [CrossRef] [PubMed]
- Chiesa, A.F.; et al. Accuracy of ultrasonography in confirming correct positioning of nasogastric tube in the intensive care setting. Chest Crit. Care 2025, 3, 100181. [Google Scholar] [CrossRef]
- Ong, M.Y.; et al. Accuracy of colour Doppler ultrasound for nasogastric tube placement in the emergency department. Emerg. Med. Australas. 2025, 37. [Google Scholar] [CrossRef] [PubMed]
- Hell, J.; Schelker, G.; Schumann, S.; Schmutz, A. Gastric insufflation with and without an inserted gastric tube in second-generation laryngeal mask airways. J. Clin. Anesth. 2024, 99, 111653. [Google Scholar] [CrossRef]
- Ragunathan, T.; et al. Performance of point-of-care ultrasonography in confirming feeding tube placement in mechanically ventilated patients. Diagnostics 2023, 13, 2679. [Google Scholar] [CrossRef]
- Güllüpınar, B.; et al. Nasogastric tube placement verification with ultrasound by emergency nurses. Ann. Clin. Anal. Med. 2022, 13. [Google Scholar] [CrossRef]
- McMullen, C.D.; Anstey, C.; Garrett, P.; Moore, J. Nasogastric tube placement under sonographic observation. Aust. Crit. Care 2022, 35, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Tsolaki, V.; et al. Ultrasonographic confirmation of nasogastric tube placement in the COVID-19 era. J. Pers. Med. 2022, 12, 337. [Google Scholar] [CrossRef] [PubMed]
- Yaseen, M.; et al. Point-of-care ultrasonography-assisted nasogastric tube placement in the emergency department. Eur. J. Emerg. Med. 2022, 29, 431–436. [Google Scholar] [CrossRef]
- Brotfain, E.; et al. Ultrasound assessment of gastric content and nasogastric tube placement in general ICU patients. Crit. Care 2019, 23, 235. [Google Scholar] [CrossRef]
- Ebert, E. Ultrasound for the confirmation of nasogastric tube placement in the emergency department. Brown Emergency Medicine Blog. Available online: https://brownemblog.com (accessed on 25 May 2025).
- Yıldırım, Ç.; et al. Verifying the placement of nasogastric tubes at an emergency center. Emerg. Med. Int. 2018, 2018, 2370426. [Google Scholar] [CrossRef]
- Nedel, W.L.; Jost, M.N.F.; Filho, J.W.F. A simple and fast ultrasonographic method of detecting enteral feeding tube placement. J. Intensive Care 2017, 5, 55. [Google Scholar] [CrossRef]
- Wong, K.W.; et al. Using color flow detection of air insufflation to improve accuracy in verifying nasogastric tube position. Am. J. Emerg. Med. 2017, 35, 333–336. [Google Scholar] [CrossRef]
- Zatelli, M.; Vezzali, N. Four-point ultrasonography to confirm correct position of nasogastric tube. J. Ultrasound 2017, 20, 53–58. [Google Scholar] [CrossRef]
- Kim, H.M.; et al. Effectiveness of ultrasonography in verifying nasogastric tube placement. Scand. J. Trauma Resusc. Emerg. Med. 2012, 20, 38. [Google Scholar] [CrossRef]
- Nguyen, L.; Lewiss, R.E.; Drew, J.; Saul, T. A novel approach to confirming nasogastric tube placement in the ED. Am. J. Emerg. Med. 2012, 30, 1662.e5–1662.e7. [Google Scholar] [CrossRef] [PubMed]
- Brotfain, E.; et al. Nurse-performed ultrasound assessment of gastric residual volume and nasogastric tube placement. Intensive Crit. Care Nurs. 2022, 69, 103183. [Google Scholar] [CrossRef]
- Metheny, N.A. Insufficient evidence to support ultrasonography as a method to rule out improperly positioned nasogastric tubes. Evid. Based Nurs. 2018, 21, 79. [Google Scholar] [CrossRef] [PubMed]
- Boling, B. Expanding the horizon: The case for point-of-care ultrasound in nurse practitioner practice. J. Am. Assoc. Nurse Pract. 2025, 37, 430–432. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; et al. Experiences of critical care nurses implementing point-of-care ultrasound. Nurs. Crit. Care 2025, 30, e70104. [Google Scholar] [CrossRef]
- Alfoti, B.O.O.; et al. Utilization of point-of-care ultrasound in emergency and critical care. Egypt. J. Chem. 2024, 67, 705–716. [Google Scholar] [CrossRef]
- Ventura, S.; et al. Implementation of point-of-care ultrasound training into nurse practitioner education. J. Nurse Pract. 2023, 19. [Google Scholar] [CrossRef]
- Pérez-Calatayud, Á.A.; Carillo-Esper, R. Role of gastric ultrasound to guide enteral nutrition in the critically ill. Curr. Opin. Crit. Care 2023, 29, 123–129. [Google Scholar] [CrossRef] [PubMed]
- askiran, N.; Sari, D. Effectiveness of auscultatory, capnometry, and pH methods to confirm nasogastric tube placement. Int. J. Nurs. Pract. 2022, 28, e13049. [Google Scholar] [CrossRef] [PubMed]
- Tsujimoto, H.; et al. Ultrasonography for confirmation of gastric tube placement. Cochrane Database Syst. Rev. 2017, 4, CD012083. [Google Scholar] [CrossRef] [PubMed]
| Refs. | Study Aim | Design | Setting | Sample |
| [4] | To prospectively evaluate the effectiveness of ultrasound-guided nasogastric tube placement and procedure-related parameters in ICU patients | Observacional study | ICU | 56 patients |
| [12] | To evaluate the diagnostic accuracy and direct costs of three bedside methods—ultrasound, epigastric auscultation, and pH measurement—for confirming nasoenteral tube placement compared with radiography | Observacional study | Clinical wards | 76 patients, 87 tube insertions |
| [22] | To investigate the effectiveness of point-of-care ultrasonography for verifying NGT placement in community-dwelling adults, and to evaluate its feasibility as a first-line reference in comparison with the gastric aspirate pH test | Observacional study | Other | 68 patients |
| [23] | To evaluate the diagnostic accuracy of nurse-performed ultrasonography in confirming the position of nasogastric tubes in the emergency department, compared with conventional methods and radiography | Observacional study | Emergency | 72 patients |
| [24] | To describe the feasibility and effectiveness of bedside ultrasonography to guide nasogastric tube placement in COVID-19 patients in isolation wards | Case report | Other | 2 elderly patients |
| [25] | To describe the use of bedside ultrasound for confirming nasogastric tube placement in a patient with severe COVID-19, when conventional methods were inconclusive or infeasible | Case report | Other | 1 elderly patient |
| [26] | To evaluate the agreement between the BUS and the plain radiography to confirm the positioning of the EC in critically ill patients, as well as to analyze the potential impact of this exam on the time to start the enteral nutrition | Observacional study | ICU | 83 patients |
| [27] | To evaluate the concordance between nurse and physician in determining the location of the nasoenteral tube using bedside ultrasonography, and to describe the main difficulties encountered by the nurse when performing the technique | Observacional study | ICU | 30 patients |
| [28] | To estimate the diagnostic accuracy of 2-point ultrasonography (esophagus + stomach) for confirming gastric tube placement in the prehospital setting | Observacional study | Emergency | 32 patients |
| [29] | To estimate the diagnostic accuracy of ultrasound in confirming gastric tube placement in the prehospital setting | Observacional study | Other | 130 patients |
| [30] | To describe and illustrate the use of abdominal ultrasonography for confirming the correct position of a nasogastric tube in the stomach, using a new dynamic turbulence test | Case report | ICU | 1 patient |
| [31] | To evaluate the accuracy and feasibility of bedside sonography performed by ICU physicians to confirm the position of weighted-tip nasogastric feeding tubes, compared with radiography | Observacional study | ICU | 33 patients; 35 procedures |
| [32] | To assess the diagnostic accuracy of bedside abdominal ultrasound (BAU) in confirming correct nasogastric tube placement compared with chest radiography | Observacional study | Other | 526 inpatients |
| [33] | To provide precise estimates and assess the accuracy of dynamic fogging detection by abdominal US in patient with a positive US (visible NGT) in oesophageal position to confirm correct tube placement | Observacional study | ICU | 182 patients |
| [34] | To investigated the accuracy of collor doppler ultrasonography for NGT placement confirmation in the ED setting | Observacional study | Emergency | 144 patients |
| [35] | To evaluate the risk of gastric insufflation with gastric ultrasound during increasing peak airway pressures in two different second-generation LMAs with and without an inserted gastric tube | Randomized controlled trial | ICU | 152 patients |
| [36] | To evaluate the diagnostic accuracy of point-of-care ultrasonography in confirming feeding tube placement in mechanically ventilated ICU patients compared to chest radiography. | Observacional study | ICU | 80 patients |
| [37] | To evaluate the ability of emergency nurses, after structured training, to confirm correct nasogastric tube placement using ultrasound compared with chest radiography. | Observacional study | ICU | 84 patients |
| [38] | To evaluate the diagnostic accuracy of ultrasound performed by nonradiologists for verifying nasogastric tube placement in mechanically ventilated ICU patients, compared with chest radiography | Observacional study | ICU | 25 patients |
| [39] | To evaluate the feasibility and diagnostic accuracy of ultrasonography for confirming NGT placement in COVID-19 patients, especially after frequent posture changes between prone and supine | Observacional study | ICU | 276 patients |
| [40] | To compare the accuracy and safety of real-time point-of-care ultrasonography-guided nasogastric tube insertion with the conventional blind insertion technique in the emergency department | Randomized controlled trial | Emergency | 118 patients |
| [41] | To validate a new ultrasound method of assessing gastric residual volume and nasogastric tube positioning performed by trained ICU nurses, compared with the standard protocol | Observacional study | ICU | 90 patients; 360 assessments |
| [42] | Not applicable | Not applicable | Emergency | Not applicable |
| [43] | To compare the accuracy of neck and subxiphoid ultrasound with chest radiography for verifying nasogastric tube placement in emergency department patients | Observacional study | Emergency | 49 patients |
| [44] | To prospectively evaluate the effectiveness of ultrasound in confirming correct enteral feeding tube placement in mechanically ventilated ICU patients and compare time to diagnosis with abdominal X-ray | Observacional study | ICU | 41 patients; 41 tube insertions |
| [45] | To evaluate the diagnostic accuracy of color flow Doppler ultrasonography in verifying nasogastric tube position compared to conventional two-dimensional ultrasound and chest radiography | Observacional study | Emergency | 100 patients |
| [46] | To estimate the diagnostic accuracy of a novel 4-point ultrasonography protocol to confirm nasogastric tube placement in critically ill ICU patients | Observacional study | ICU | 114 patients |
| [47] | To compare the effectiveness of auscultation, pH measurement, and ultrasonography in verifying nasogastric tube placement among patients with low consciousness in the emergency center | Observacional study | Emergency | 47 patients |
| [48] | To describe a novel approach using bedside two-dimensional ultrasound to confirm nasogastric tube placement in the emergency department | Case report | Emergency | 1 male patient |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




