Submitted:
09 February 2026
Posted:
10 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
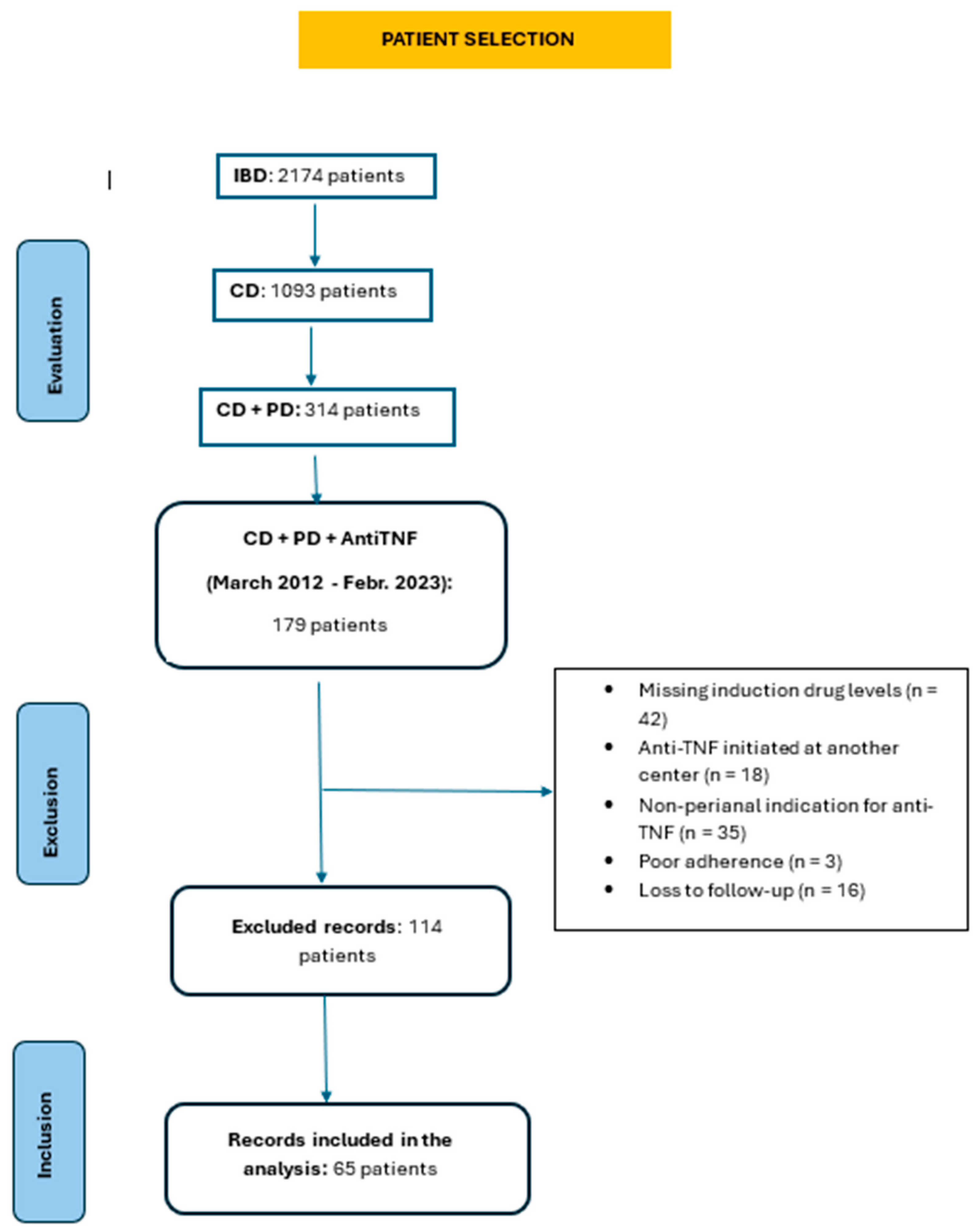
2. Materials and Methods
3. Results
3.1. Study Population
3.2. Clinical and Radiological Outcomes
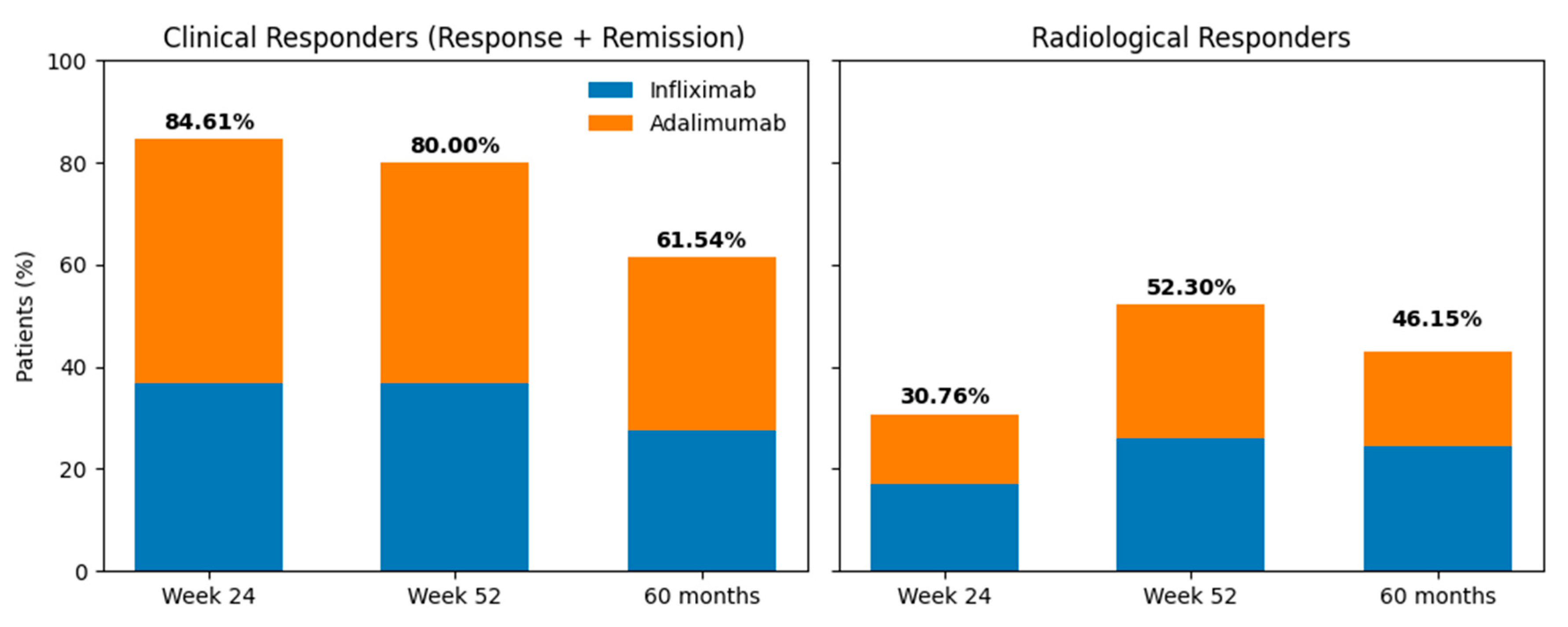
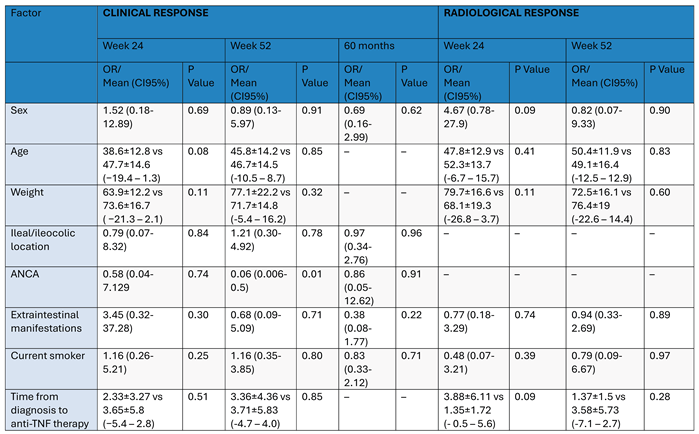
3.3. Predictors of Clinical and Radiological Outcomes
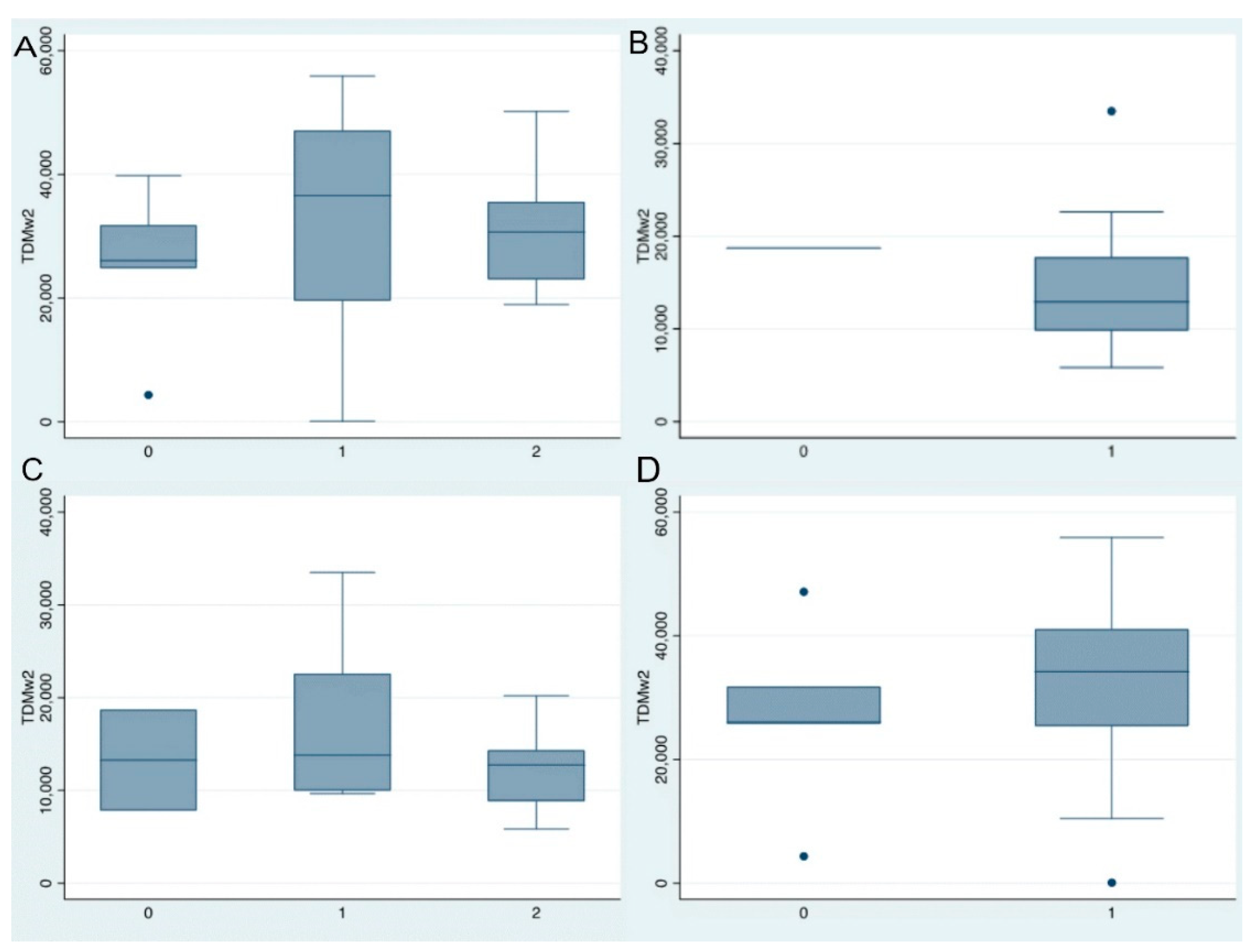
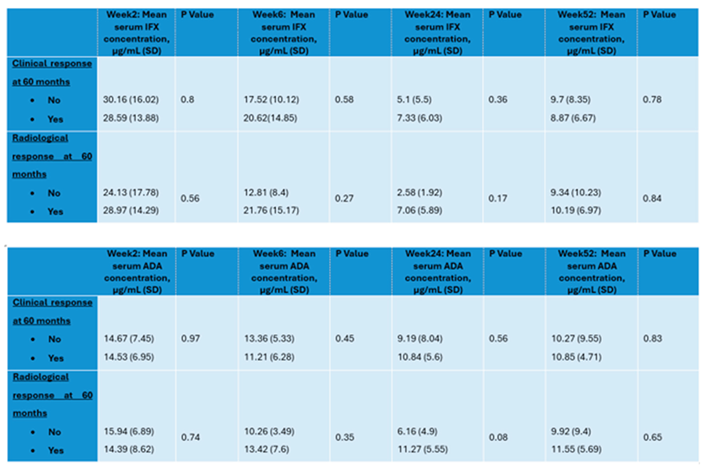
3.4. Anti-TNF Levels and Clinical and Radiological Outcomes
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADA | Adalimumab |
| ANCA | Antineutrophil Cytoplasmic Antibodies |
| ASCA | Anti-Saccharomyces cerevisiae Antibodies |
| AUC | Area Under the Curve |
| CD | Crohn´s Disease |
| CI | Confidence Interval |
| EFO | External Fistula Opening |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| EUA | Examination Under Anesthesia |
| IBD | Inflammatory Bowel Disease |
| IFX | Infliximab |
| MRI | Magnetic Resonance Imaging |
| OR | Odds Ratio |
| PD | Perianal Disease |
| ROC | Receiver Operating Characteristic |
| SD | Standard Deviation |
| TNF | Tumor Necrosis Factor |
References
- Magro, F; Gionchetti, P; Eliakim, R; Ardizzone, S; Armuzzi, A; Barreiro-de Acosta, M; et al. Third European Evidence-based Consensus on Diagnosis and Management of Ulcerative Colitis. Part 1: Definitions, Diagnosis, Extra-intestinal Manifestations, Pregnancy, Cancer Surveillance, Surgery, and Ileo-anal Pouch Disorders. J Crohns Colitis 2017, 11(6), 649–70. [Google Scholar] [CrossRef] [PubMed]
- Gomollón, F; Dignass, A; Annese, V; Tilg, H; Van Assche, G; Lindsay, JO; et al. European evidence-based consensus on the diagnosis and management of Crohn’s disease 2016: Part 1: Diagnosis and medical management. J Crohn’s Colitis 2017, 11(1), 3–25. [Google Scholar] [CrossRef]
- Feuerstein, JD; Ho, EY; Shmidt, E; Singh, H; Falck-Ytter, Y; Sultan, S; et al. AGA Clinical Practice Guidelines on the Medical Management of Moderate to Severe Luminal and Perianal Fistulizing Crohn’s Disease. Gastroenterology 2021, 160(7), 2496–508. [Google Scholar]
- Panaccione, R; Steinhart, AH; Bressler, B; Khanna, R; Marshall, JK; Targownik, L; et al. Canadian Association of Gastroenterology Clinical Practice Guideline for the Management of Luminal Crohn’s Disease. Clin Gastroenterol Hepatol 2019, 17(9), 1680–713. [Google Scholar] [CrossRef] [PubMed]
- Parks, AG; Gordon, PH; Hardcastle, JD. A classification of fistula-in-ano. Br J Surg 1976, 63(1), 1–12. [Google Scholar]
- Sandborn, WJ; Fazio, VW; Feagan, BG; Hanauer, SB. AGA technical review on perianal Crohn’s disease. Gastroenterology 2003, 125(5), 1508–30. [Google Scholar] [CrossRef] [PubMed]
- Geldof, J; Iqbal, N; LeBlanc, JF; Anandabaskaran, S; Sawyer, R; Buskens, C; et al. Classifying perianal fistulising Crohn’s disease: an expert consensus to guide decision-making in daily practice and clinical trials. Lancet Gastroenterol Hepatol. 2022, 7(6), 576–84. [Google Scholar] [CrossRef]
- Rimola, J; Capozzi, N. Magnetic Resonance in Crohn Disease: Imaging Biomarkers in Assessing Response to Therapy. Magn Reson Imaging Clin N Am [Internet] 2020, 28(1), 45–53. [Google Scholar] [CrossRef]
- Present, DH; Rutgeerts, P; Targan, S; Hanauer, SB; Mayer, L; van Hogezand, RA; et al. Infliximab for the treatment of fistulas in patients with Crohn’s disease. N Engl J Med 1999, 340(18), 1398–405. [Google Scholar]
- Sands, BE; Anderson, FH; Bernstein, CN; Chey, WY; Feagan, BG; Fedorak, RN; et al. Infliximab maintenance therapy for fistulizing Crohn’s disease. N Engl J Med. 2004, 350(9), 876–85. [Google Scholar] [CrossRef]
- Colombel, JF; Schwartz, DA; Sandborn, WJ; Kamm, MA; D’Haens, G; Rutgeerts, P; et al. Adalimumab for the treatment of fistulas in patients with Crohn’s disease. Gut 2009, 58(7), 940–8. [Google Scholar] [CrossRef]
- Fu, Y-M; Chen, M; Liao, A-J. A Meta-Analysis of Adalimumab for Fistula in Crohn’s Disease. Gastroenterol Res Pract. 2017, 2017, 1745692. [Google Scholar]
- Bouguen, G; Siproudhis, L; Gizard, E; Wallenhorst, T; Billioud, V; Bretagne, JF; et al. Long-term outcome of perianal fistulizing crohn’s disease treated with infliximab. Clin Gastroenterol Hepatol [Internet] 2013, 11(8), 975–981.e4. [Google Scholar] [CrossRef] [PubMed]
- Karmiris, K; Bielen, D; Vanbeckevoort, D; Vermeire, S; Coremans, G; Rutgeerts, P; et al. Long-term monitoring of infliximab therapy for perianal fistulizing Crohn’s disease by using magnetic resonance imaging. Clin Gastroenterol Hepatol Off Clin Pract J Am Gastroenterol Assoc. 2011, 9(2), 130–6. [Google Scholar] [CrossRef]
- Lichtiger, S; Binion, DG; Wolf, DC; Present, DH; Bensimon, AG; Wu, E; et al. The CHOICE trial: adalimumab demonstrates safety, fistula healing, improved quality of life and increased work productivity in patients with Crohn’s disease who failed prior infliximab therapy. Aliment Pharmacol Ther. 2010, 32(10), 1228–39. [Google Scholar] [CrossRef]
- Castaño-Milla, C; Chaparro, M; Saro, C; De Acosta, MB; García-Albert, AM; Bujanda, L; et al. Effectiveness of adalimumab in perianal fistulas in Crohn’s disease patients naive to Anti-TNF therapy. J Clin Gastroenterol. 2015, 49(1), 34–40. [Google Scholar] [CrossRef] [PubMed]
- Sirmai, L; Pelletier, AL; Gault, N; Zallot, C; Bouguen, G; Bouchard, D; et al. Relationship between clinical remission of perianal fistulas in Crohn’s disease and serum adalimumab concentrations: A multicenter cross-sectional study. World J Gastroenterol. 2022, 28(9), 961–72. [Google Scholar] [CrossRef]
- Gordon, H; Minozzi, S; Kopylov, U; Verstockt, B; Chaparro, M; Buskens, C; et al. ECCO Guidelines on Therapeutics in Crohn’ s Disease: Medical Treatment 2024, (June), 1531–55.
- Panes, J; Reinisch, W; Rupniewska, E; Khan, S; Forns, J; Khalid, JM; et al. Burden and outcomes for complex perianal fistulas in Crohn’s disease: Systematic review. World J Gastroenterol 2018, 24(42), 4821–34. [Google Scholar] [CrossRef] [PubMed]
- Friedberg, S; Choi, D; Hunold, T; Choi, NK; Garcia, NM; Picker, EA; et al. Upadacitinib Is Effective and Safe in Both Ulcerative Colitis and Crohn’s Disease: Prospective Real-World Experience. Clin Gastroenterol Hepatol. 2023, 21(7), 1913–1923.e2. [Google Scholar] [CrossRef]
- Elford, AT; Bishara, M; Plevris, N; Gros, B; Constantine-Cooke, N; Goodhand, J; et al. Real-world effectiveness of upadacitinib in Crohn’s disease: a UK multicentre retrospective cohort study. Frontline Gastroenterol 2024, 15(4), 297–304. [Google Scholar] [CrossRef] [PubMed]
- Zare, B; Gros, B; Lal, N; Dawson, P; Sharma, E; Dart, RJ; et al. Effectiveness of risankizumab induction and maintenance therapy for refractory Crohn’ s disease: a real- world experience from a preapproval access programme and early access to medicines scheme 2024, 1–8.
- Makowiec, F; Jehle, EC; Starlinger, M. Clinical course of perianal fistulas in Crohn’s disease. Gut 1995, 37(5), 696–701. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Value |
|---|---|
| Age (years), mean (SD) | 46.28 (14.62) |
| Sex, male/female (% male) | 30/35 (46,2%) |
| Body mass index (kg/m²) mean (SD) | 25.57 (5.09) |
|
Current smoker, N (%) Former smoker, N (%) Never smoker, N (%) |
21 (32.3%) 15 (23.1%) 29 (44.6%) |
| Extraintestinal manifestations,N (%) | 24 (36.9%) |
| Disease Location (L1/L2/L3), n (%) | 10 (15.4%) / 32 (49.2%) / 23 (35.4%) |
| Disease behavior (B1/B2/B3), n (%) | 35 (53.8%) / 12 (18.5%) / 18 (27.7%) |
|
Previous surgery before Anti-TNF No, N (%) Yes, N (%)
|
21 (32.3%) 44 (67.7%) 9 2 39 |
Treatment prior to Anti-TNF therapy
|
61 (93.8%) 29 (44.6%) 23 (35.4%) |
Anti-TNF therapy
|
10 (15.4%) 22 (33.8%) 26 (40%) 7 (10.8%) |
Time from CD diagnosis to onset of PD
|
28 (43.1%) 14 (21.5%) 23 (35.4%) |
| Fistula type | |
|
12 (18.5) |
|
53 (81.5) |
| Parks classification | |
|
6 (9.38) |
|
9 (14.06) |
|
43 (66.15) |
|
5 (7.81) |
|
2 (3.12) |
Number of fistulous tracts
|
|
| 32 (48.65) | |
| 12 (18.75) | |
| 12 (18.75) | |
| 9 (13.85) | |
| Rectovaginal fistulas | |
|
9 (13.85) |
|
56 (86.15) |
| Associated abscess | |
|
58 (89.23) |
|
7 (10.77) |
 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




