Submitted:
08 February 2026
Posted:
10 February 2026
You are already at the latest version
Abstract
Keywords:
Background
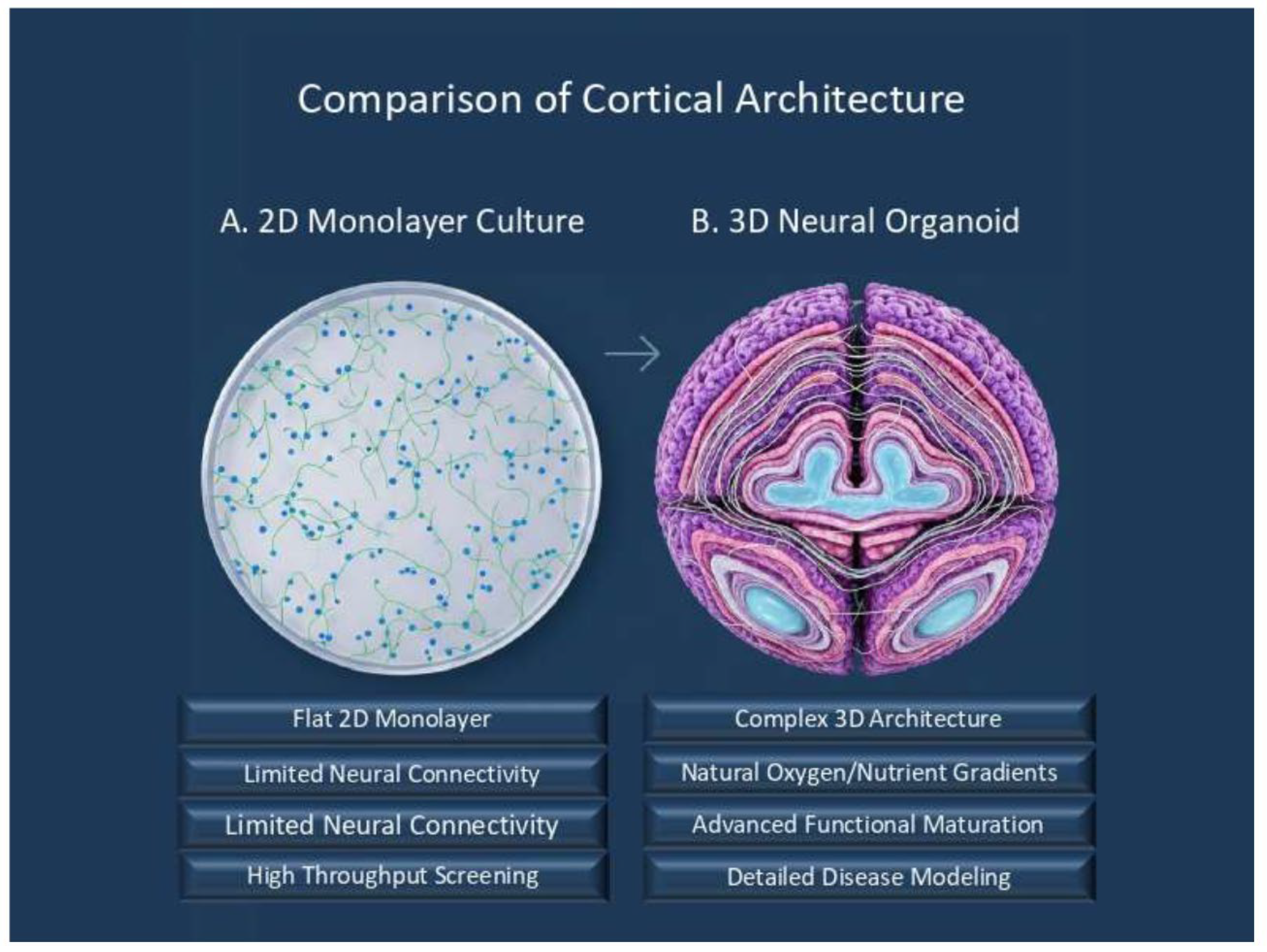
1. Architectural Superiority of 3D Systems
1.1. Scaffold-Free: Brain Organoids
1.2. Scaffold-Based: Hydrogels and 3D Bioprinting
2. Organ-on-Chip (OoC) and the Neurovascular Unit
2.1. The Blood-Brain Barrier (BBB) on a Chip
2.2. Microfluidic Modeling of Axonal Transport
3. Precision Medicine and Patient-Specific Modeling
3.1. High-Fidelity Disease Modeling (AD, PD, ALS)
3.2. Personalized Drug Screening and Toxicity
- Reduced Clinical Failure: By providing more predictive data during the preclinical phase, 3D models can filter out ineffective or toxic compounds before they reach expensive human trials.
- Patient-Centric Care: The use of patient-derived iPSCs in 3D systems allows for the development of “patient avatars,” enabling clinicians to test treatments in vitro before treating the patient.
- Ethical Advancement: These models offer a robust alternative to animal testing, particularly for studying human-specific neurological conditions that do not have accurate animal counterparts.
Authors’ Contributions
Ethics Approval and Consent to Participate
Consent for Publication
Competing Interests
Funding
Acknowledgments
References
- Jensen, C; Teng, Y. Is it time to start transitioning from 2D to 3D cell culture? Front Mol Biosci. 2020, 7, 33. [Google Scholar] [CrossRef]
- Abbott, A. Biology’s new dimension. Nature 2003, 424(6951), 870–2. [Google Scholar] [CrossRef]
- Cummings, JL; Morstorf, T; Zhong, K. Alzheimer’s disease drug-development pipeline: few candidates, frequent failures. Alzheimers Dement. 2014, 10(4), 432–8. [Google Scholar] [CrossRef]
- Gribkoff, VK; Kaczmarek, LK. The need for new models in antiepileptic drug discovery. Neuropharmacology 2017, 120, 1–2. [Google Scholar]
- Lancaster, MA; Knoblich, JA. Organogenesis in a dish: modeling development and disease using pluripotent stem cells. Science 2014, 345(6194), 1247125. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y; Inoue, H; Wu, JC; Yamanaka, S. Induced pluripotent stem cell technology: a decade of progress. Nat Rev Drug Discov. 2017, 16(2), 115–30. [Google Scholar] [CrossRef] [PubMed]
- Baker, BM; Chen, CS. Deconstructing the third dimension: how 3D culture microenvironments alter cellular cues. J Cell Sci. 2012, 125(13), 3015–24. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, AM; DeLaporte, L; Vunjak-Novakovic, G; et al. Nerve in a dish: materials for 3D in vitro neurons. Soft Matter 2015, 11(37), 7254–67. [Google Scholar]
- Tang-Schomer, MD; White, JD; Tien, LW; et al. Bioengineered functional 3D brain-like tissue. PNAS 2014, 111(38), 13811–6. [Google Scholar] [CrossRef]
- Lancaster, MA; Renner, M; Martin, CA; et al. Cerebral organoids model human brain development and microcephaly. Nature 2013, 501(7467), 373–9. [Google Scholar] [CrossRef]
- Cugola, FR; Fernandes, IR; Russo, FB; et al. The Brazilian Zika virus strain causes birth defects in experimental models. Nature 2016, 534(7606), 267–71. [Google Scholar] [CrossRef] [PubMed]
- Quadrato, G; Nguyen, T; Macosko, EZ; et al. Cell diversity and network dynamics in photosensitive human brain organoids. Nature 2017, 545(7652), 48–53. [Google Scholar] [CrossRef] [PubMed]
- Lozano, R; Stevens, L; Thompson, BC; et al. 3D printing of novel gellan gum-based scaffolds for neural tissue engineering. Biomaterials 2015, 67, 264–73. [Google Scholar] [CrossRef] [PubMed]
- Gu, Q; Tomaskovic-Crook, E; Wallace, GG; et al. 3D bioprinting of human neural progenitors and their activity in 3D-printed structures. Adv Healthc Mater. 2016, 5(12), 1429–38. [Google Scholar] [CrossRef]
- Murphy, SV; Atala, A. 3D bioprinting of tissues and organs. Nat Biotechnol. 2014, 32(8), 773–85. [Google Scholar] [CrossRef]
- Bhatia, SN; Ingber, DE. Microfluidic organs-on-chips. Nat Biotechnol. 2014, 32(8), 760–72. [Google Scholar] [CrossRef]
- Booth, R; Kim, H. Characterization of a microfluidic blood-brain barrier model. Lab Chip 2012, 12(10), 1784–92. [Google Scholar] [CrossRef]
- Herland, A; van der Meer, AD; FitzGerald, EA; et al. Distinct contributions of astrocytes and pericytes to neuroinflammation in a 3D microfluidic human blood-brain barrier model. PLoS One 2016, 11(3), e0150360. [Google Scholar] [CrossRef]
- Vatine, GD; Barrile, R; Kiernan, MJ; et al. Human iPSC-derived blood-brain barrier chips enable disease modeling and personalized medicine applications. Cell Stem Cell. 2019, 24(6), 995–1005. [Google Scholar] [CrossRef]
- Osaki, T; Sivathanu, V; Kamm, RD. Engineered 3D vascular and neural tissue models. Sci Adv. 2018, 4(10), eaat5847. [Google Scholar] [CrossRef]
- Taylor, AM; Blurton-Jones, M; Rhee, SW; et al. A microfluidic culture platform for CNS axonal injury, regeneration and transport. Nat Methods 2005, 2(8), 599–605. [Google Scholar] [CrossRef]
- Park, J; Wetzel, I; Marriott, I; et al. A 3D human triculture system modeling neurodegeneration and neuroinflammation in Alzheimer’s disease. Lab Chip 2014, 14(24), 4740–50. [Google Scholar] [CrossRef]
- Choi, SH; Kim, YH; Hebisch, M; et al. A three-dimensional human neural cell culture model of Alzheimer’s disease. Nature 2014, 515(7526), 274–8. [Google Scholar] [CrossRef] [PubMed]
- Lin, YT; Seo, J; Gao, F; et al. APOE4 causes widespread molecular and cellular alterations associated with Alzheimer’s disease phenotypes in human iPSC-derived brain cell types. Neuron 2018, 98(6), 1144–54. [Google Scholar] [CrossRef]
- Jo, J; Xiao, Y; Sun, AX; et al. Midbrain-like organoids from human pluripotent stem cells contain dopaminergic neurons. Cell Stem Cell. 2016, 19(2), 248–57. [Google Scholar] [CrossRef]
- Di Domenico, A; Carola, G; Calatayud, C; et al. Patient-specific iPSC-derived astrocytes contribute to non-cell autonomous neurodegeneration in Parkinson’s disease. Stem Cell Reports 2019, 12(2), 213–29. [Google Scholar] [CrossRef]
- Hubert, CG; Rivera, M; Spangler, LC; et al. A three-dimensional organoid culture system derived from human glioblastomas. Cancer Res. 2016, 76(8), 2468–77. [Google Scholar] [CrossRef]
- Linkous, A; Balamatsli, D; Snuderl, M; et al. Modeling patient-derived glioblastoma with cerebral organoids. Cell Rep. 2019, 26(12), 3203–11. [Google Scholar] [CrossRef] [PubMed]
- Pașca, AM; Sloan, SA; Clarke, LE; et al. Functional cortical neurons and astrocytes from human pluripotent stem cells in 3D culture. Nat Methods 2015, 12(7), 671–8. [Google Scholar] [CrossRef]
- Truong, DD; Fan, Y; Jureczek, J; et al. Engineering a neurovascular unit on a chip. Biomaterials 2019, 198, 63–77. [Google Scholar] [CrossRef] [PubMed]
| Feature | 2D Culture | 3D Organoids | Organ-on-Chip (3D) |
|---|---|---|---|
| Mechanical Environment | Non-physiological (Rigid) | Physiological (Soft) | Physiological (Soft + Flow) |
| Cell Morphology | Flattened, unnatural | Natural, 3D branching | Controlled, polarized |
| Nutrient Diffusion | Uniform | Gradient-based | Controlled/Dynamic |
| Predictive Power | Low (30-40%) [3,4] | High (60-80%) [10,23] | Very High (>80%) [19,30] |
| Translational Use | Basic signaling | Disease mechanisms | Drug screening/PK-PD |
| Disease | 3D Model Type | Key Molecular Finding | Ref |
|---|---|---|---|
| Alzheimer’s | iPSC 3D Hydrogel | Aβ plaque and Tau tangle formation | [23] |
| Parkinson’s | Midbrain Organoids | Selective dopaminergic loss | [25] |
| ALS | Motor Neuron Chip | Defective neuromuscular junction (NMJ) | [20] |
| Zika Virus | Cerebral Organoids | Viral-induced apoptosis of NPC cells | [11] |
| Glioblastoma | Patient-derived Spheroids | Personalized drug resistance profiling | [28] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




