Submitted:
06 February 2026
Posted:
09 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Scope and Non-Goals
3. Prior Hypotheses and Partial Models
4. Core Proposal
5. Model Formalism
5.1. State Vector and Coupling Terms
| Component | Meaning (model) | Example measurable proxies (illustrative) |
|---|---|---|
| M | Mechanical compliance/strain state | Elastography/strain; stiffness; ROM-linked stiffness |
| E | Electrical/impedance state (ionic conductance proxies) | Skin impedance/admittance; galvanic response; bioimpedance |
| Θ | Temperature | Thermometry; thermography; local skin temperature gradients |
| Φ | Biochemical/metabolic state proxy | Metabolic surrogates (context-dependent); oxygenation/lactate proxies (if available) |
| I | Interstitial/ionic milieu (ECF composition/hydration-related proxies) | Hydration proxies; edema tendency; interstitial surrogates |
| F | Fascial glide/viscoelastic friction proxy | Ultrasound fascial sliding; shear-wave measures; glide tests |
| N | Neural excitability proxy | Pressure sensitivity thresholds; reflex/excitability surrogates; EMG tone (if available) |
| P | Interstitial/compartment pressure proxy (incl. lymphatic congestion proxies) | Circumference/edema; tissue turgor; lymph-related surrogates |
| V | Perfusion/flow proxy | Capillary refill; Doppler microflow; near-infrared perfusion surrogates |
| C | Context-dependent coupling strength across layers (not an energy variable) | Cross-modal lag/phase relations; coupling indices from multi-proxy time series |
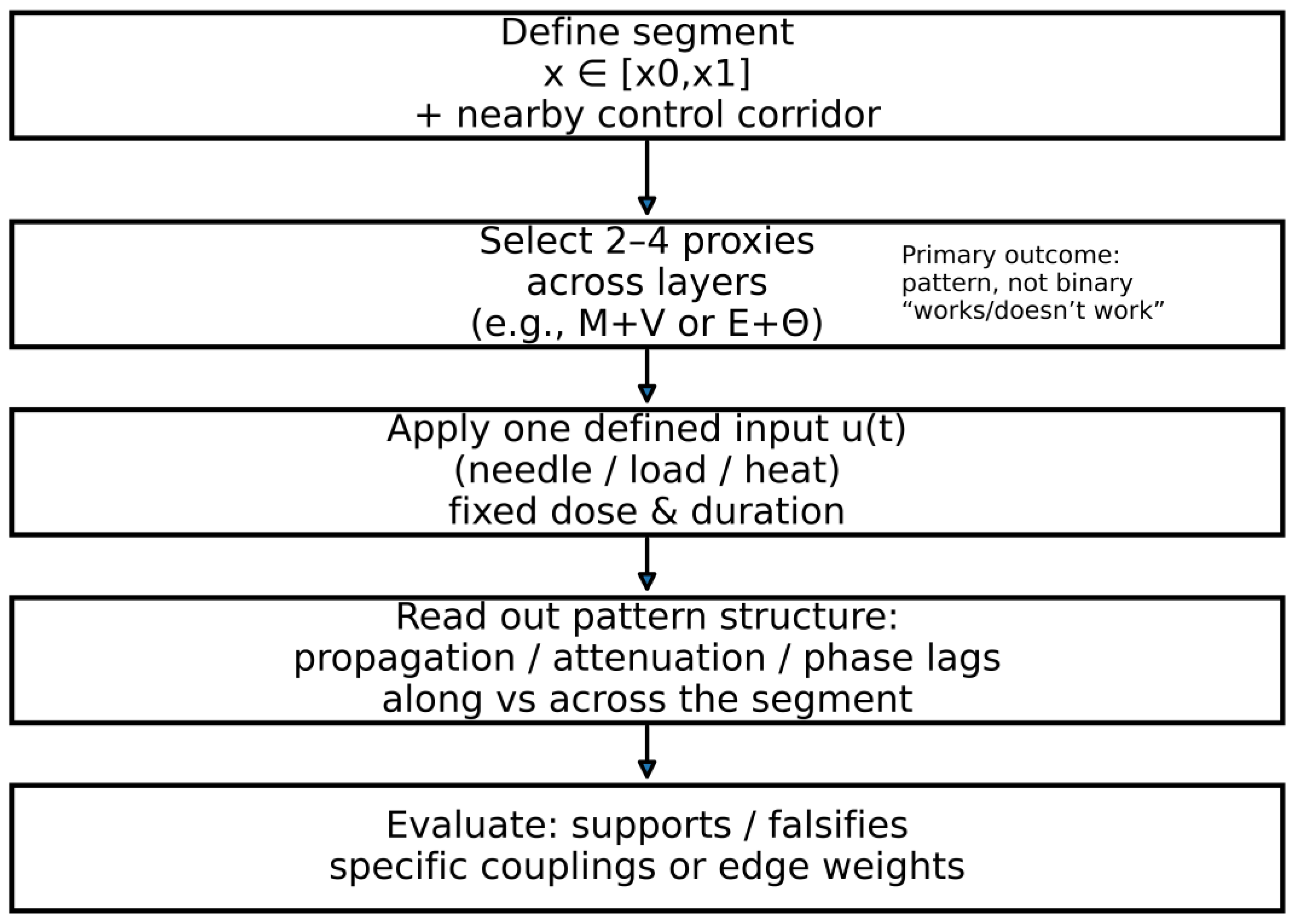
5.1.1. Operationalization: Minimal Workflow for Testing Predictions

5.2. Minimal Segment Formalism (Optional)
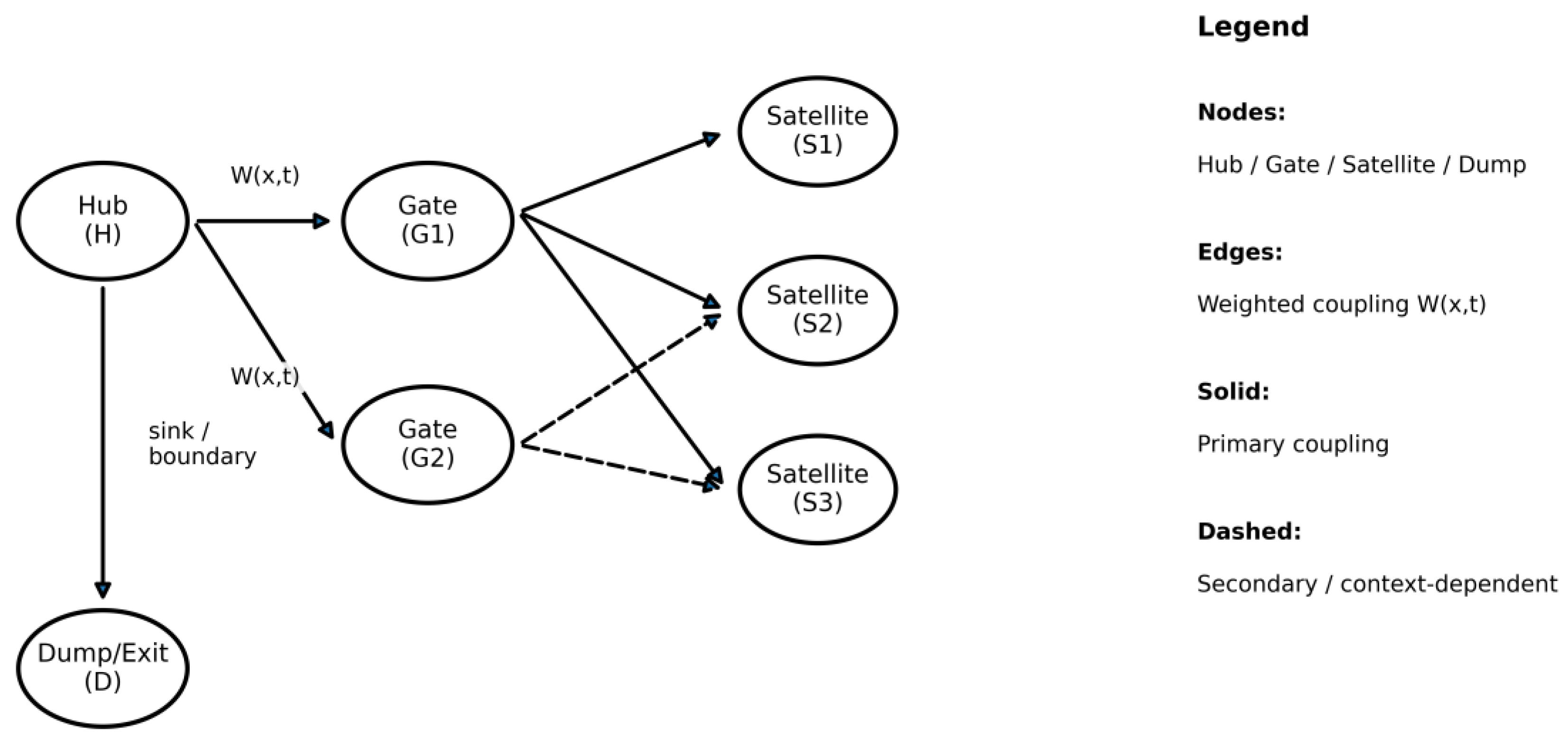
5.3. Network Representation: Meridians as Graph Waveguides
5.3.1. Practical Estimation of Edge Weights (Data-Driven, Non-Circular)
5.4. Why a “Waveguide” Analogy Is Useful
5.5. Interventions as State Perturbations
6. Testable Predictions and Minimal Validation Roadmap
7. Practice-Oriented Translation for Acupuncture Practice
7.1. What an Acupuncturist Can Use Immediately
7.2. A Minimal Mapping and Reassessment Loop
7.3. How This Model Informs Point Strategy Without Prescribing Points
7.4. Lay Explanation (Optional)
7.5. Example Practice Use Cases (Schematic, Non-Prescriptive)
8. Scope Boundaries and Limitations
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Schleip, R.; Jäger, H.; Klingler, W. What is ‘fascia’? A review of different nomenclatures. J. Bodyw. Mov. Ther. 2012, 16, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Kumka, M.; Bonar, J. Fascia: a morphological description and classification system based on a literature review. J Can Chiropr Assoc. 2012, 56, 179–191. [Google Scholar] [PubMed]
- Langevin, H.M.; Yandow, J.A. Relationship of acupuncture points and meridians to connective tissue planes. Anat. Rec. 2002, 269, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Langevin, H.M.; Churchill, D.L.; Wu, J.; Badger, G.J.; Yandow, J.A.; Fox, J.R.; Krag, M.H. Evidence of Connective Tissue Involvement in Acupuncture. FASEB J. 2002, 15, 2275–2282. [Google Scholar] [CrossRef] [PubMed]
- Benias, P.C.; Wells, R.G.; Sackey-Aboagye, B.; Klavan, H.; Reidy, J.; Buonocore, D.; Miranda, M.; Kornacki, S.; Wayne, M.; Carr-Locke, D.L.; et al. Structure and Distribution of an Unrecognized Interstitium in Human Tissues. Sci. Rep. 2018, 8, 4947. [Google Scholar] [CrossRef] [PubMed]
- Tomov, N.; Atanasova, D.; Dimitrov, N. Is the Newly Described Interstitial Network the Anatomical Basis of Acupuncture Meridians? A Commentary. Anat. Rec. 2019, 303, 2169–2170. [Google Scholar] [CrossRef] [PubMed]
- Levin, M. Bioelectric signaling: Reprogrammable circuits underlying embryogenesis, regeneration, and cancer. Cell 2021, 184, 1971–1989. [Google Scholar] [CrossRef] [PubMed]
- Young, M.; Lewis, A.H.; Grandl, J. Physics of mechanotransduction by Piezo ion channels. J. Gen. Physiol. 2022, 154, e202113044. [Google Scholar] [CrossRef] [PubMed]
- Ahn, A.C.; Park, M.; Shaw, J.R.; McManus, C.A.; Kaptchuk, T.J.; Langevin, H.M. Electrical Impedance of Acupuncture Meridians: The Relevance of Subcutaneous Collagenous Bands. PLOS ONE 2010, 5, e11907. [Google Scholar] [CrossRef]
- Soh, K.-S. Bonghan Circulatory System as an Extension of Acupuncture Meridians. J. Acupunct. Meridian Stud. 2009, 2, 93–106. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Rong, P.J.; Ben, H.; et al. Meridian studies in China: a systematic review. J Acupunct Meridian Stud. 2010, 3, 1–9. [Google Scholar]
- Elahee, S.F.; Pardhan, S.; Behara, D.; et al. Meridian system and mechanism of acupuncture action: A scientific evaluation. World J Acupunct Moxibustion. 2020, 30, 233–238. [Google Scholar] [CrossRef]
- Kovich, A. A new definition of an acupuncture meridian. J Acupunct Meridian Stud. 2019, 12, 25–28. [Google Scholar] [CrossRef] [PubMed]
- Brizhik, L.; Eremko, A.; Pietak, A.; et al. Modeling meridians within the quantum field theory. J Acupunct Meridian Stud. 2019, 12, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Ren, Y.-L.; Chen, Y.-H.; Chen, J.; Tang, X.-J.; Zhang, Z.-M. A “4D” systemic view on meridian essence: Substantial, functional, chronological and cultural attributes. J. Integr. Med. 2022, 20, 96–103. [Google Scholar] [CrossRef] [PubMed]
- LIANG, Y.; ZHOU, J.; DU, J.-Y.; SHAO, X.-M.; FANG, J.-Q. Prospects for the development of acupuncture analgesia from an international perspective. World J. Acupunct. - Moxibustion 2023, 33, 6–8. [Google Scholar] [CrossRef]
- Liu, W.H. Brief discussion on the broad-spectrum effect and specific effect of acupuncture-moxibustion. World J Acupunct Moxibustion. 2023, 33, 294–295. [Google Scholar] [CrossRef]
- Xie, Y.-X.; Xiong, Z.-Y.; Liu, C.-Z.; Yan, S.-Y. Placebo effect of acupuncture in clinical study. World J. Acupunct. - Moxibustion 2023, 33, 309–313. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




