Submitted:
06 February 2026
Posted:
09 February 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Epidemiology and Socio-Economic Impact
Predictors of Returning to Work
Psychological Factors
Clinical and Echocardiographic Considerations
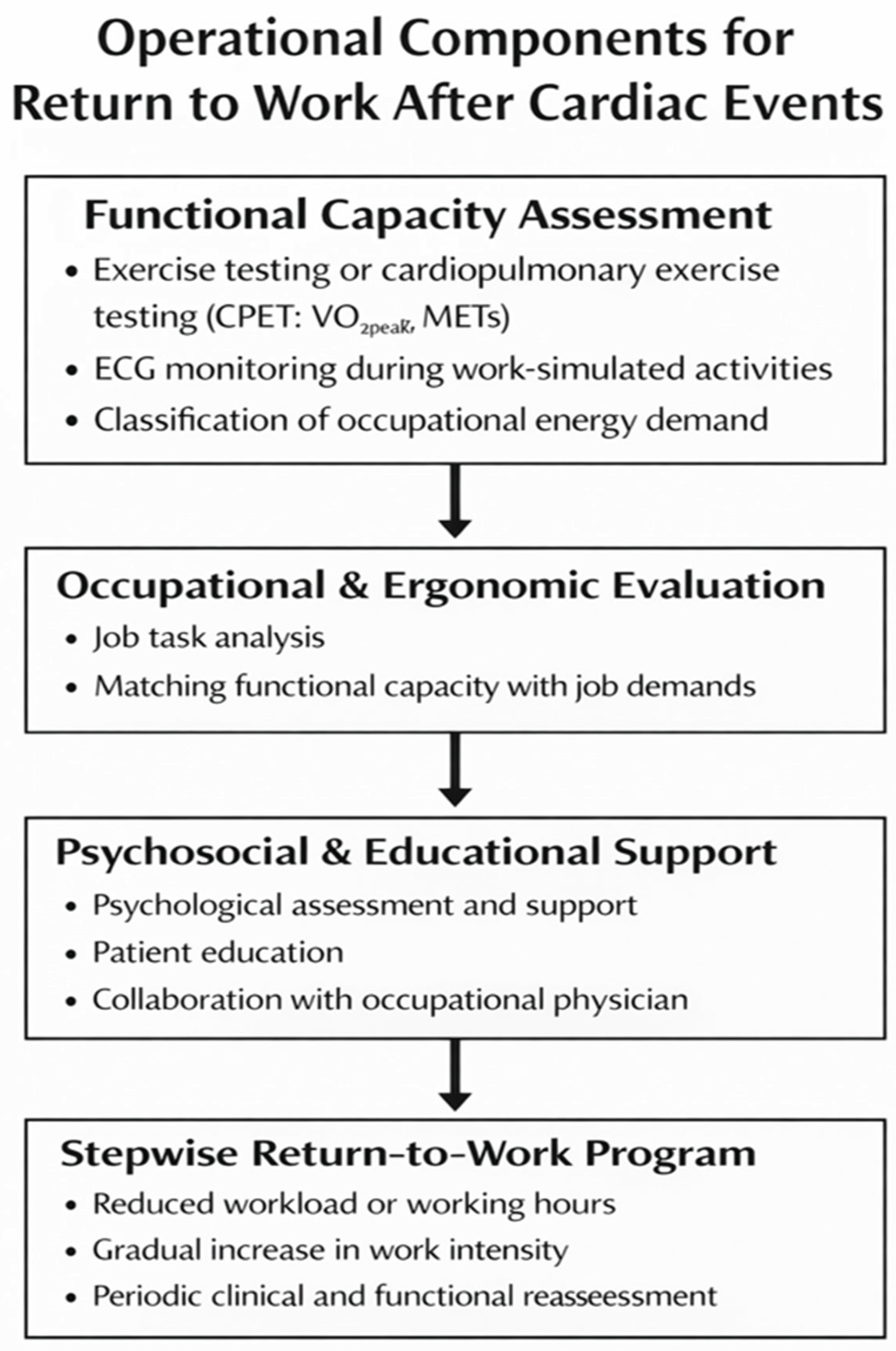
Functional Capacity and MET-Based Job Matching
The Role of Cardiac Rehabilitation
Measuring Occupational Outcomes: Work Performance Scale (WPS)
From the Acute Phase to Telerehabilitation
Gender Disparities and Socio-Occupational Barriers
Pathophysiological and Behavioral Linkages Between Work Stress and Cardiac Risk
Medication Optimization and Adherence in the Context of RTW
Legal, Policy, and Employer Engagement
Implementation Toolkit for Clinicians and Employers
Metrics, Quality, and Accreditation
Limitations and Research Gaps
Discussion
Conclusions
Abbreviations
| ACS | Acute Coronary Syndrome |
| AMI/MI | Acute Myocardial Infarction / Myocardial Infarction |
| CPET | Cardiopulmonary Exercise Testing |
| CR | Cardiac Rehabilitation |
| CS | Cardiogenic Shock |
| GLS | Global Longitudinal Strain |
| HF | Heart Failure |
| ICD | Implantable Cardioverter-Defibrillator |
| LVEF | Left Ventricular Ejection Fraction |
| MET | Metabolic Equivalent of Task |
| QoL | Quality of Life |
| RTW | Return to Work |
| WPS | Work Performance Scale |
References
- Smedegaard L, Numé AK, Charlot M, Kragholm K, Gislason G, Hansen PR. Return to Work and Risk of Subsequent Detachment From Employment After Myocardial Infarction: Insights From Danish Nationwide Registries. J Am Heart Assoc. 2017;6(10):e006486. [CrossRef]
- Reibis R, Salzwedel A, Abreu A, Corrà U, Davos C, Doehner W, et al. The importance of return to work: How to achieve optimal reintegration in ACS patients. Eur J Prev Cardiol. 2019;26(13):1358–69. [CrossRef]
- O’Brien L, Wallace S, Romero L. Psychosocial/vocational interventions and RTW post-MI. J Cardiopulm Rehabil Prev. 2018;38(4):215–23.
- Ambrosetti M, Abreu A, Corrà U, et al. Secondary prevention through comprehensive CR: 2020 update. Eur J Prev Cardiol. 2021;28(5):460–95.
- Kai SHY, Ferrières J, Rossignol M, Bouisset F, Herry J, Esquirol Y. Prevalence and determinants of return to work after various coronary events: meta-analysis of prospective studies. Sci Rep. 2022;12(1):15348. [CrossRef]
- Andersen EB, Kristiansen M, Jørgensen SMB. Barriers and facilitators to return to work following cardiovascular disease: a systematic review and meta-synthesis. BMJ Open. 2023;13(1):e069091. [CrossRef]
- Sun W, Gholizadeh L, Perry L, Kang K. Predicting return to work following myocardial infarction. Int J Environ Res Public Health. 2022;19(13):8032. [CrossRef]
- Perelshtein Brezinov O, Klempfner R, Zekry SB, et al. Prognostic value of ejection fraction in ACS. Medicine (Baltimore). 2017;96(9):e6226.
- D’Andrea A, Sperlongano S, Pacileo M, et al. New ultrasound technologies in CR. J Clin Med. 2020;9(10):3131.
- D’Andrea A, Radmilovic J, Mele D, et al. Speckle tracking in ICU. Echocardiography. 2018;35(4):506–19.
- Woo JS, Kim WS, Yu TK, et al. Prognostic value of serial GLS after STEMI. Am J Cardiol. 2011;108(3):340–7.
- Nucifora G, Delgado V, Bertini M, et al. LV muscle and fluid mechanics in AMI. Am J Cardiol. 2010;106(10):1404–9.
- Son JW, Park WJ, Choi JH, et al. Vortex flow and apical thrombus. Circ J. 2012;76(11):2640–6.
- McBurney CR, Eagle KA, Kline-Rogers EM, et al. Work-related outcomes after MI. Pharmacotherapy. 2004;24(11):1515–23.
- Cauter JV, De Bacquer D, Clays E, et al. RTW and psychosocial well-being in CHD: EUROASPIRE IV. Eur J Prev Cardiol. 2019;26(13):1386–95.
- Salzwedel A, Koran I, Langheim E, et al. PROs predict RTW/QoL after CR (OutCaRe). PLoS One. 2020;15(5):e0232752.
- Salzwedel A, Reibis R, Hadzic M, et al. Expectations of RTW and work capacity at CR discharge. Vasc Health Risk Manag. 2019;15:301–8.
- Byrne RA, Rossello X, Coughlan JJ, et al. 2023 ESC Guidelines for ACS. Eur Heart J. 2023;44(38):3720–26.
- Abreu A, Frederix I, Dendale P, et al. Standardization and quality improvement of CR in Europe. Eur J Prev Cardiol. 2021;28(5):496–509.
- Barresi F, Foster-Witassek F, Rickli H, Pedrazzini G, Roffi M, Puhan M, et al. Predictors of work inability after acute myocardial infarction in Switzerland. Sci Rep. 2024;14(1):13429. [CrossRef]
- Stendardo M, Bonci M, Casillo V, Miglio R, Giovannini G, Nardini M, et al. Predicting return to work after acute myocardial infarction: Socio-occupational factors overcome clinical conditions. PLoS One. 2018;13(12):e0208842. [CrossRef]
- Timóteo AT, Gouveia M, Soares C, Ferreira RC. Indirect costs of myocardial infarction in Portugal. Rev Port Cardiol (Engl Ed). 2020;39(5):245–51.
- O’Neil A, Sanderson K, Oldenburg B. Depression as a predictor of work resumption following myocardial infarction. Health Qual Life Outcomes. 2010;8:95. [CrossRef]
- Hu Y, Zhou T, Li X, et al. Factors influencing return to work 3 months after PCI in young and middle-aged patients. PLoS One. 2023;18(4):e0284100.
- Ruile S, Meisinger C, Burkhardt K, et al. Effort–reward imbalance and return to work 6 months after acute myocardial infarction. J Occup Rehabil. 2021;31(3):532–42.
- Lammerts L, Schaafsma FG, Eikelenboom M, et al. Longitudinal associations between biopsychosocial factors and sustainable return to work. J Occup Rehabil. 2016;26:70–9. [CrossRef]
- Hellström L, Christensen TN, Bojesen AB, Eplov LF. Predictors of return to work for people with anxiety or depression in supported employment trials. J Occup Rehabil. 2023;33:61–70.
- El-Menyar A, Zubaid M, AlMahmeed W, et al. Killip class in ACS. Am J Emerg Med. 2012;30(1):97–103.
- Lauridsen MD, Rørth R, Butt JH, et al. RTW after AMI with CS. Eur Heart J Acute Cardiovasc Care. 2022;11(5):397–406.
- Jetté M, Sidney K, Blümchen G. METs in exercise testing. Clin Cardiol. 1990;13(8):555–65.
- Vogel B, Jahed J, Bengel J, et al. Psycho-diagnostic programme in rehabilitation: pilot. Rehabilitation (Stuttg). 2009;48(6):361–8.
- Jette AM, Davies AR, Cleary PD, et al. Functional Status Questionnaire validation. J Gen Intern Med. 1986;1(3):143–9.
- Lamberti M, Ratti G, Gerardi D, et al. Work-related outcome after acute coronary syndrome: implications of complex cardiac rehabilitation. Int J Occup Med Environ Health. 2016;29(4):649–57. [CrossRef]
- Frederix I, Vanhees L, Dendale P, et al. Review of cardiac telerehabilitation. J Telemed Telecare. 2015;21(1):45–53.
- Frederix I, Caiani EG, Dendale P, et al. ESC e-Cardiology Position Paper on digital implementation. Eur J Prev Cardiol. 2019;26(11):1166–77.
- Avila A, Claes J, Buys R, et al. Home-based exercise with telemonitoring. Eur J Prev Cardiol. 2020;27(4):367–77. [CrossRef]
- Claes J, Cornelissen V, McDermott C, et al. Technology-enabled CR (PATHway) RCT. J Med Internet Res. 2020;22(2):e14221.
- Dreyer RP, Xu X, Zhang W, et al. RTW after AMI: young women vs men. Circ Cardiovasc Qual Outcomes. 2016;9(2 Suppl 1):S45–52.
- Jiang Z, Dreyer RP, Spertus JA, et al. Factors associated with RTW after AMI in China. JAMA Netw Open. 2018;1(7):e184831.
- Drory Y, Kravetz S, Koren-Morag N, Goldbourt U. Employment after first MI. Cardiology. 2005;103(1):37–43.
- Rauch B, Riemer T, Schwaab B, et al. Short-term comprehensive CR after AMI reduces 1-year mortality: OMEGA. Eur J Prev Cardiol. 2014;21(9):1060–9.
- Slebus FG, Jorstad HT, Peters RJ, et al. Patients’ perspective on RTW after ACS. Saf Health Work. 2012;3(2):117–22.
- Isaaz K, Coudrot M, Sabry MH, et al. Return to work after acute STEMI in the modern era. Arch Cardiovasc Dis. 2010;103(5):310–6.
- Hua Y, Wang Y, Fan F, et al. Predictors of job maintenance after percutaneous coronary intervention. Medicine (Baltimore). 2025;104(36):e44318.
- Karoff M, Held K, Bjarnason-Wehrens B. CR in Germany. Eur J Cardiovasc Prev Rehabil. 2007;14(1):18–27.
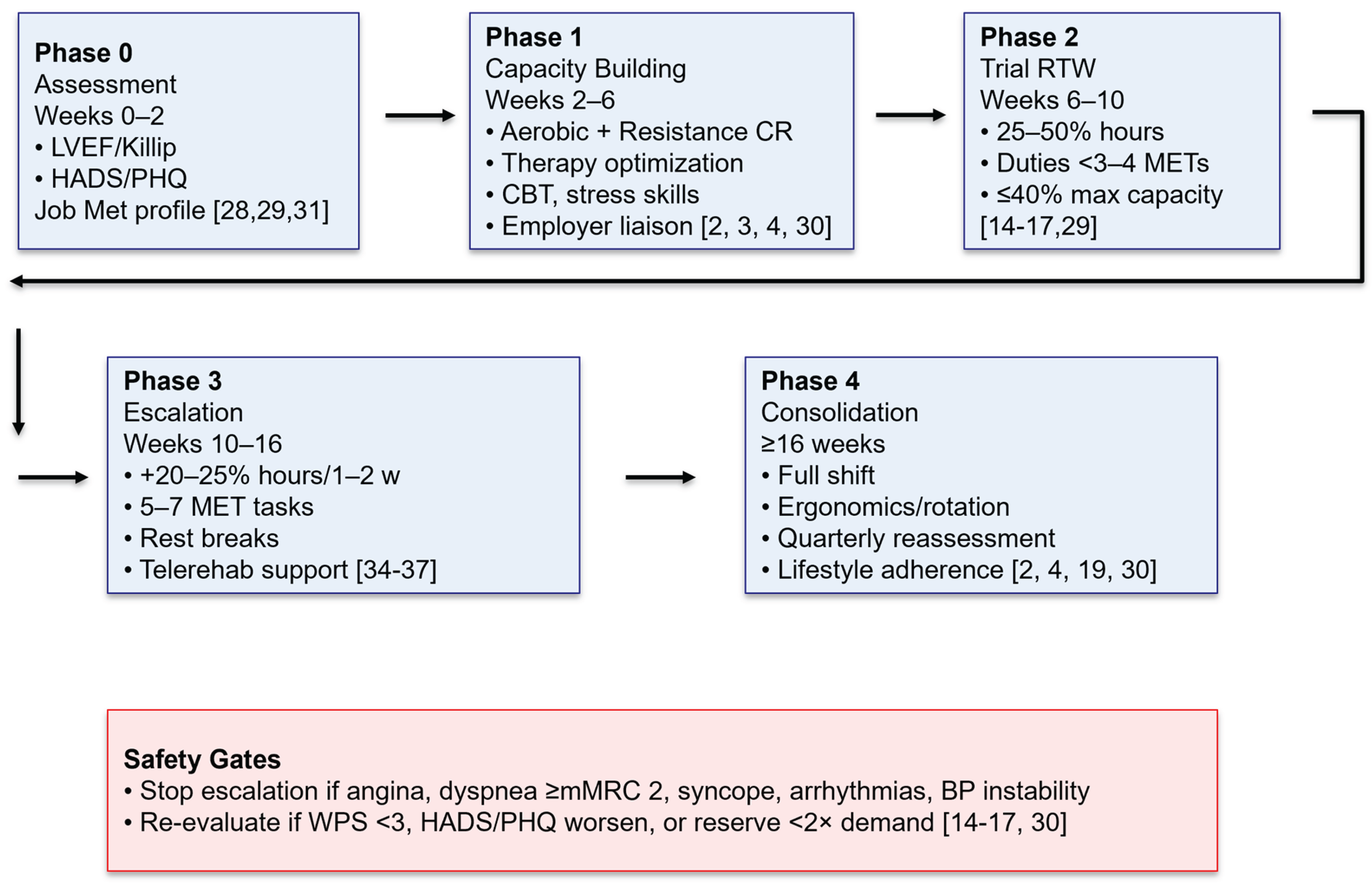
 |
| Risk stratum | Comorbidity profile | Suggested RTW start (weeks post-event) |
Duty intensity (avg METs) |
Min peak METs target (≥2× avg demand) |
Special conditions / notes |
|---|---|---|---|---|---|
| LVEF ≥50%; Killip I–II | Low (no diabetes/CKD; controlled BP) | 6–10 | ≤3–4 (light) | ≥6–8 | Stepwise hours; monitor WPS weekly; CBT for residual anxiety. |
| LVEF 40–49%; Killip II | Moderate (one stable comorbidity) | 8–12 | ≤3–4 → 5 (light→moderate) | ≥8–10 | Rotation of tasks; rest breaks; telerehab check-ins. |
| LVEF 30–39%; Killip II–III | High (≥2 comorbidities or CKD stage ≥3) | 10–16 | ≤3–4 (light) initially | ≥10–12 | Prolonged supervised CR; defer heavy/manual tasks; safety gates strict. |
| LVEF <30%; Killip III–IV | Very high (HF symptoms; frequent exacerbations) | >16 (individualized) | Very light only (≤2.5) | ≥5 (if RTW) | Extended rehabilitation; avoid safety-critical roles until stable. |
| Post–cardiogenic shock (CS) survivors | Variable; assess neurocognitive status | Individualized (often >16) | Very light → light | ≥6–8 | Neurocognitive screening; longer observation; gradual cognitive/physical load. |
| ICD carriers (post-AMI/HF) | Device-dependent risks | Per clinical stability | Role-dependent | Role-dependent | Avoid high-EMF tasks; device checks; emergency plan on site. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




