Submitted:
05 February 2026
Posted:
09 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Clinical Trials Data Used for Model Development and Validation
2.2. PBPK Model Development
2.1.1. Drug Physicochemical Properties and Blood Proteins Binding
2.1.2. Dissolution and Absorption
2.1.3. Volume of Distribution and Tissue-to-Plasma Partitioning
2.1.4. Metabolism and Excretion
2.1.5. Enzyme- and Transporter-Mediated Drug–Drug Interactions
2.2. QSP Model Development
2.2.1. QSP Model Parametrisation
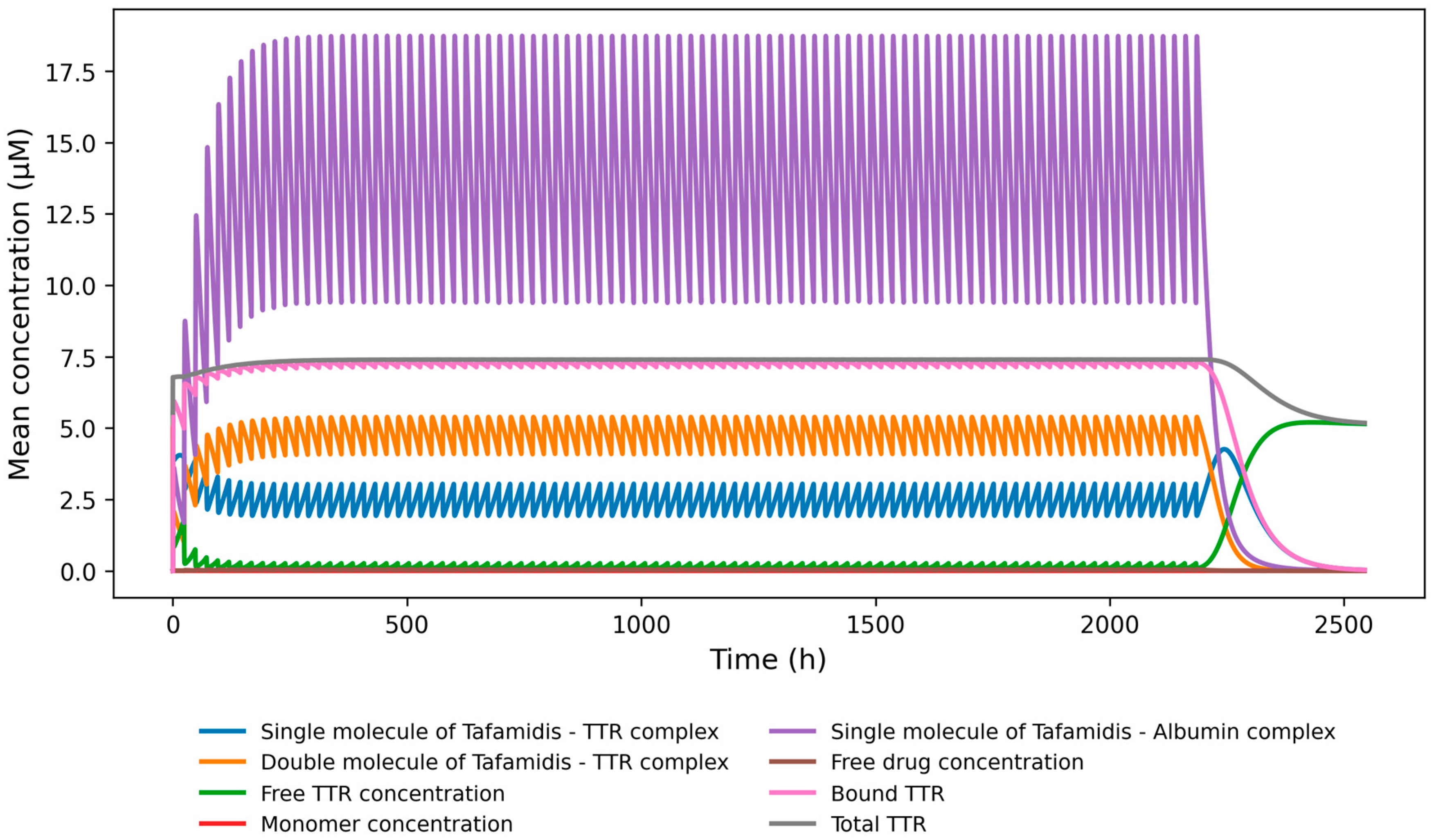
2.2.2. Equations Describing Tafamidis-TTR Kinetics
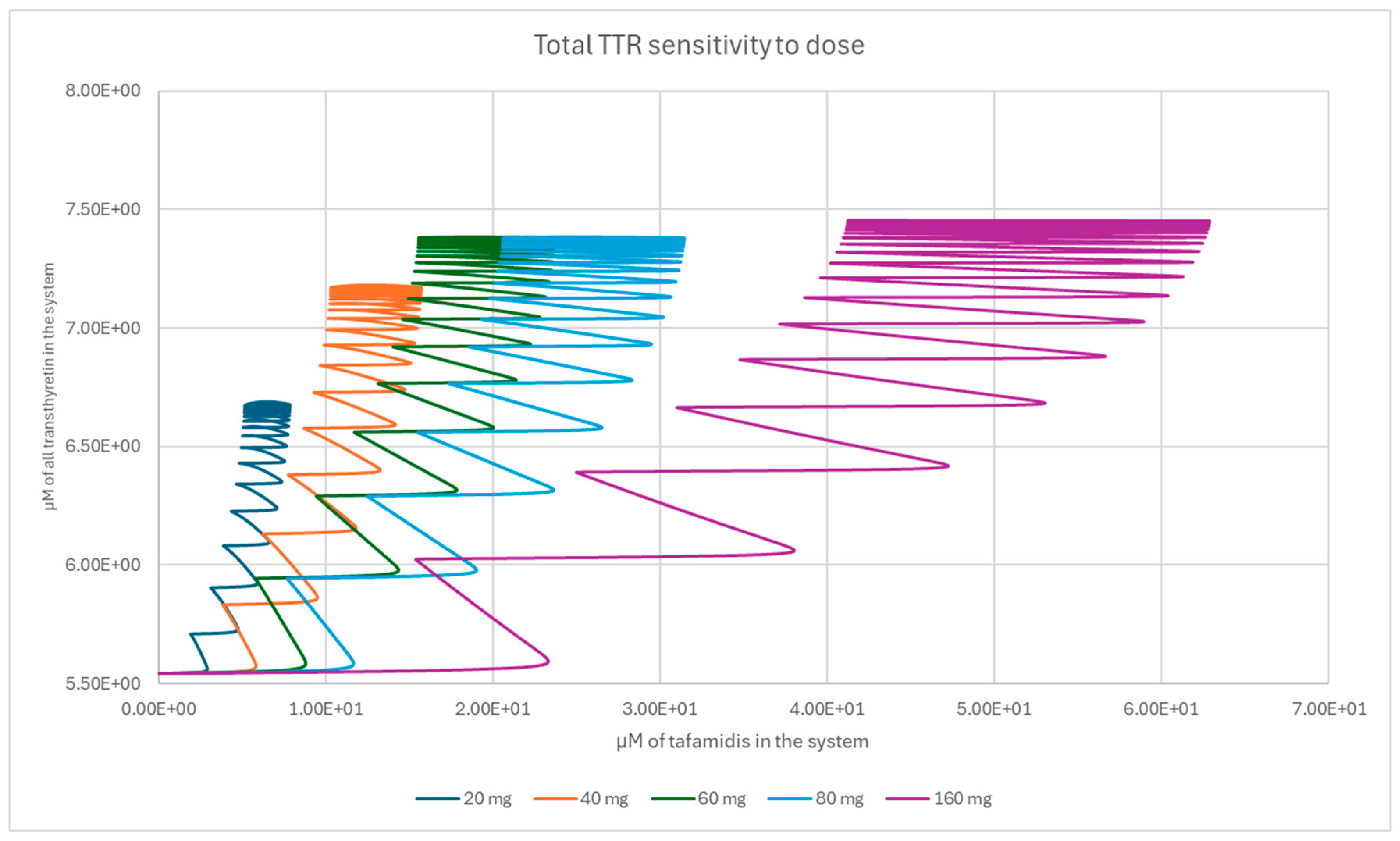
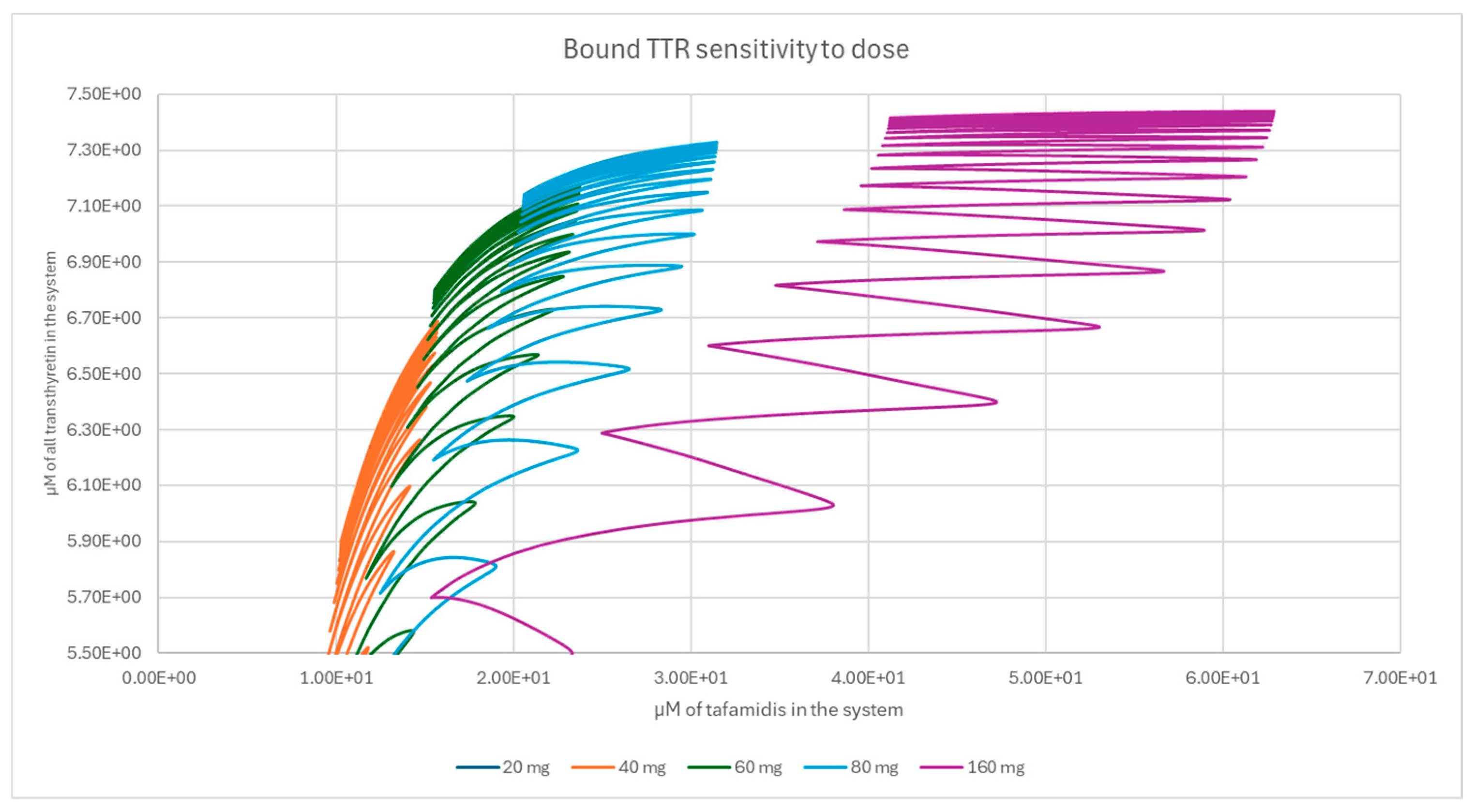
2.3. Population-Level Sampling of TTR Concentrations
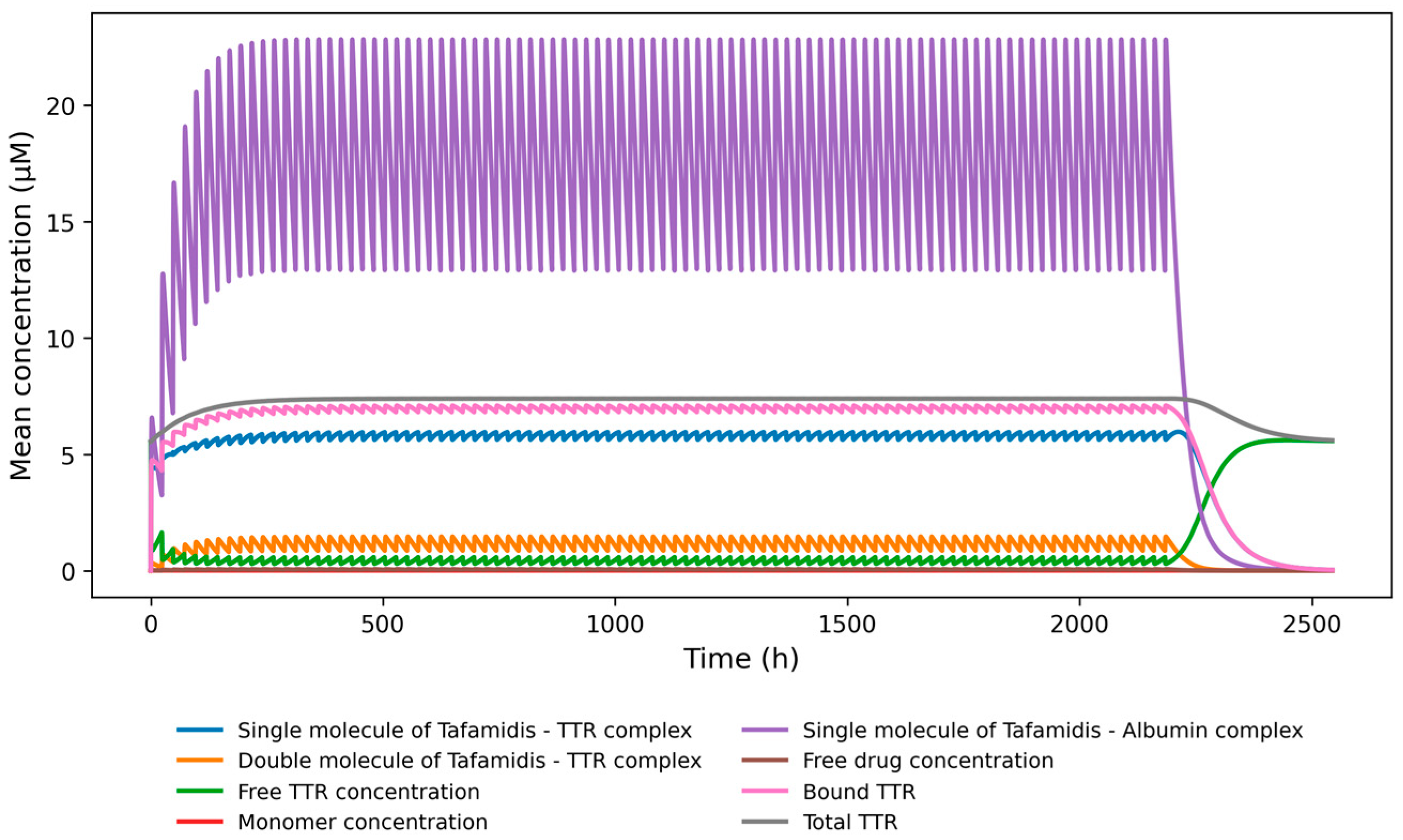
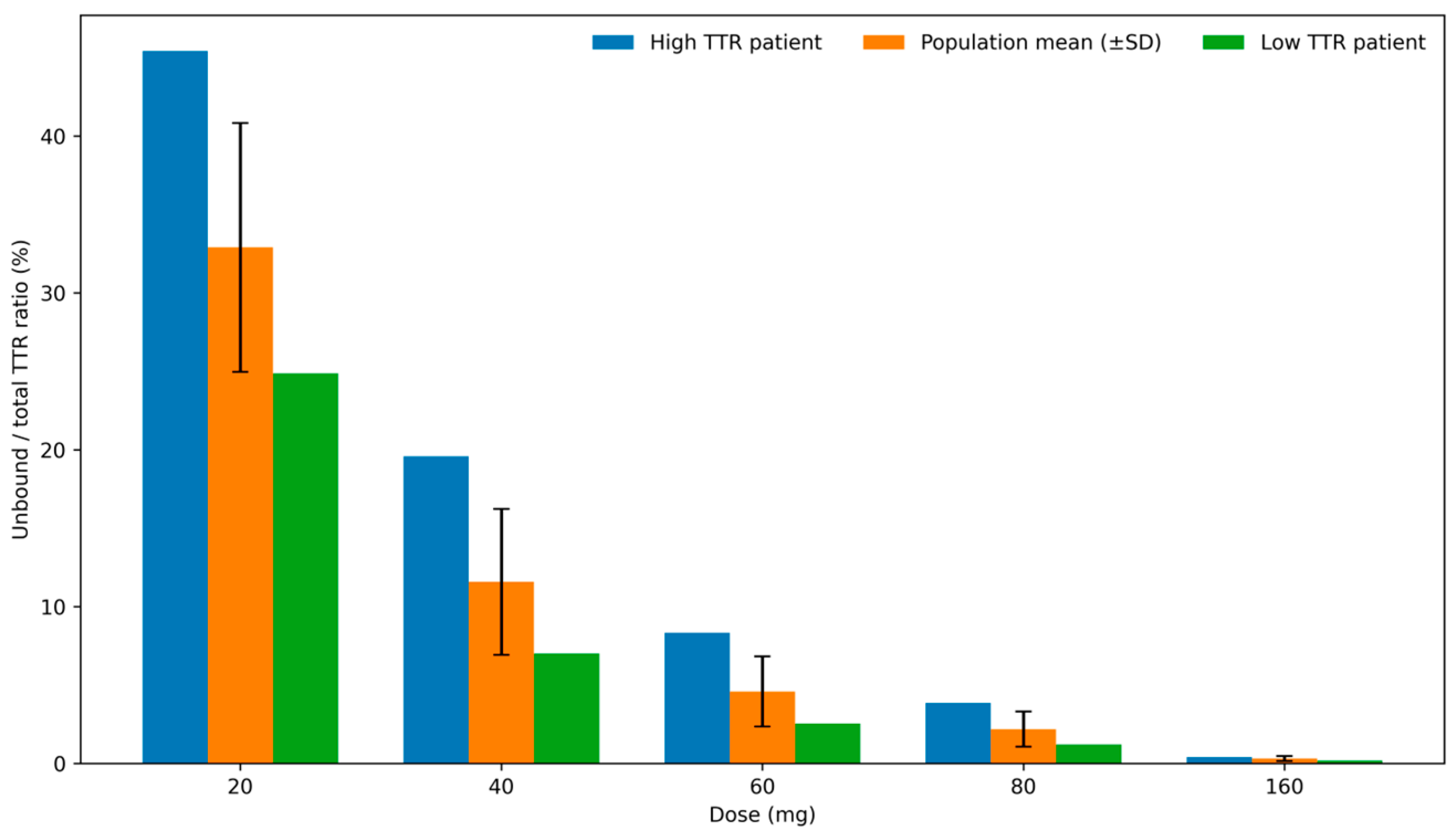
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AAFE | Absolute Average Fold Error |
| ADAM | Advanced Dissolution, Absorption, and Metabolism |
| AFE | Average Fold Error |
| AUC | Area Under the Curve |
| ATTR | Transthyretin Amyloidosis |
| BCRP | Breast Cancer Resistance Protein |
| BCS | Biopharmaceutics Classification System |
| Cmax | Maximum Plasma Concentration |
| FDA | Food and Drug Administration |
| GIT | Gastrointestinal Tract |
| ka | Association Rate Constant |
| kdiss | Dissociation Rate Constant |
| kon | Association Rate Constant |
| koff | Dissociation Rate Constant |
| Mdeg | Monomer Degradation Rate Constant |
| PBPK | Physiologically Based Pharmacokinetics |
| PD | Pharmacodynamics |
| Peff | Effective Permeability |
| Papp | Apparent Permeability |
| PK | Pharmacokinetics |
| QSP | Quantitative Systems Pharmacology |
| SD | Standard Deviation |
| So | Intrinsic Solubility |
| Tdeg | TTR Degradation Rate Constant |
| Tmax | Time to Maximum Concentration |
| TTR | Transthyretin |
| UGT | Uridine 5′-Diphospho-Glucuronosyltransferase |
| Vss | Volume of Distribution at Steady State |
Appendix A
| Parameter | Value | Origin of data | Reference |
|---|---|---|---|
| Physicochemical properties and blood binding | |||
| Type of compound | Acid | Based on structure | - |
| MW, free acid (g/mol) | 308.12 | Based on structure | - |
| LogP | 3.91 | Predicted with ALOGPS | [43] |
| pKa | 3.7 | - | [21] |
| PSA (Å2) | 63.3 | Predicted | - |
| Blood and plasma binding | |||
| Blood to plasma ratio (B:P) | 0.55 | Methodological details unknown. Derived from regulatory documents. | [18] |
| Fraction unbound in plasma (fup) | 0.008 | Experimental | [18] |
|
Absorption (solubility and permeability) Advanced Dissolution, Absorption and Metabolism model (ADAM) | |||
| fugut | 0.67393 | Predicted | [33] |
| Caco-2 apparent permeability (Papp) (10-6 cm/s) | 30.9 | Experimental | [24] |
| Metoprolol permeability as a reference for Caco-2 experiment (Papp) (10-6 cm/s) | 15.5 | Experimental | [24] |
| Effective permeability in human intestine (10-4 cm/s) | 6.9706 | Predicted | [25] |
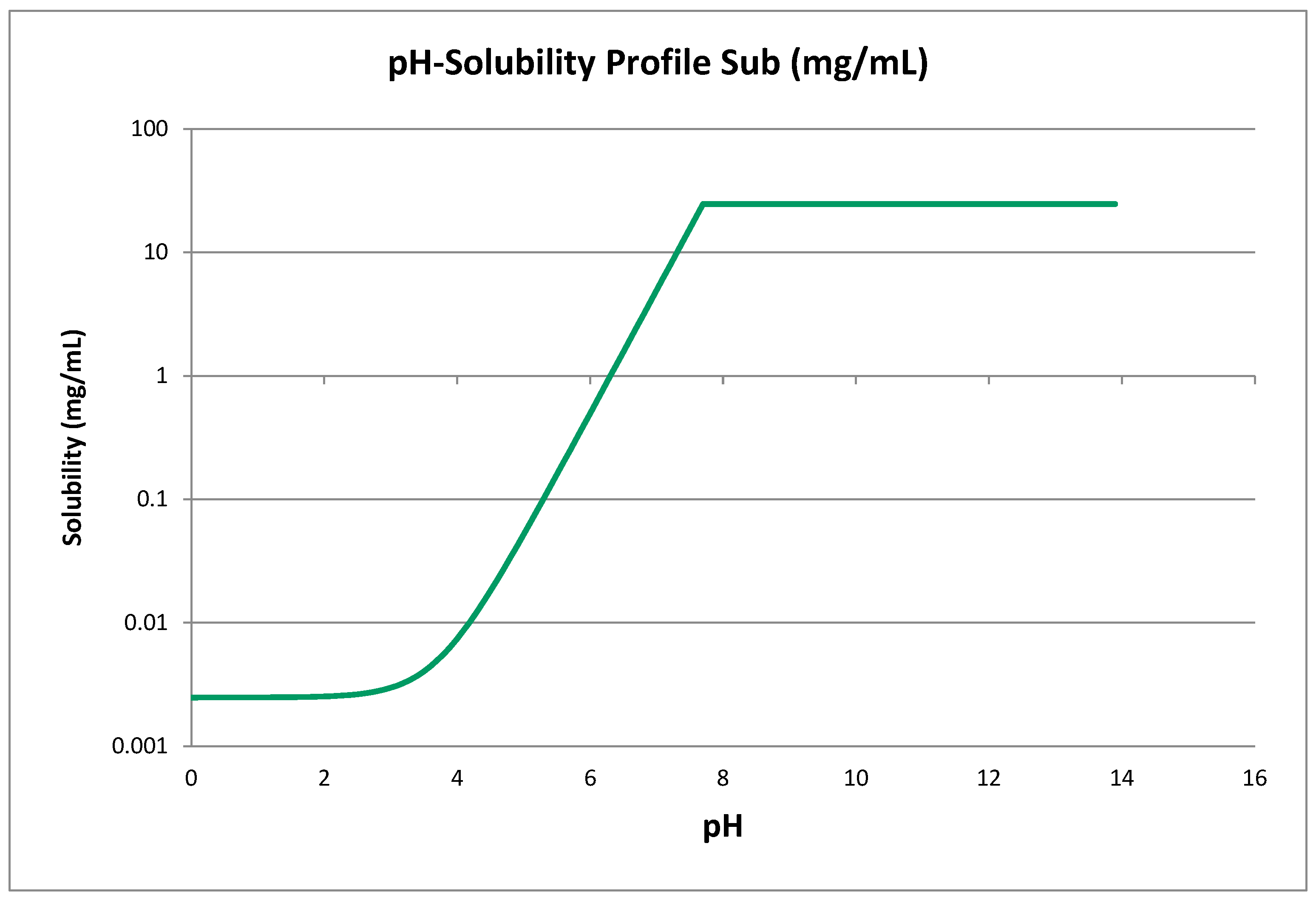
| Solubility at pH 6.8 (mg/mL) | 3.121 | Experimental | [26] |
| Intrinsic Solubility S0 (mg/mL) | 0.0024771 | Predicted with Henderson-Hasselbach equation using solubility at pH 6.8 as an input. | [44] |
| Distribution | |||
| Volume of distribution at steady state | 0.16909 L/kg | Predicted with Simcyp population data, B:P and tissue-to-plasma partition coefficients. | [31,45] |
| Tissue to plasma partition coefficients (Kp) | |||
| Adipose | 0.027 | Obtained from rats. Rats distribution of small molecules is expected to be similar to that of humans. | [24] |
| Brain | 0.022 | ||
| Gut | 0.254 | ||
| Heart | 0.113 | ||
| Kidney | 0.59 | ||
| Liver | 4.01 | ||
| Lung | 0.107 | ||
| Spleen | 0.063 | ||
| Bone | 0.10203 | Predicted using the Rodgers and Rowland method. | [45] |
| Pancreas | 0.06316 | ||
| Muscle | 0.036922 | ||
| Skin | 0.28047 | ||
| Additional Organ | 0.039244 | ||
| Elimination | |||
| Described as a total clearance = 0.262 L/hr. Tafamidis is eliminated mainly through glucuronidation in the liver, unfortunately there is not enough experimental data shared for bottom-up clearance prediction. | |||
| Parameter Name | Value | Unit | Source |
|---|---|---|---|
| TTR Synthesis rate | Patient-specific | Based on Ingenbleek et al. dataset, as explained in “Population-level sampling of TTR concentrations” section [9,14,41]. | |
| TTR degradation rate | Patient-specific | Based on Ingenbleek et al. dataset, as explained in “Population-level sampling of TTR concentrations” section [9,14,41]. | |
| Dissociation rate of unbound TTR | 0.0024 | [8] | |
| Association rate of unbound TTR | 360000 | [46] | |
| Monomer degradation rate | 0.016 | There is no data on this parameter available in the literature. We assume that for healthy volunteers it is the same as average degradation rate of TTR [14]. | |
| Association rate constant for first tafamidis-TTR binding | 16200 | [37] | |
| Dissociation rate constant for first tafamidis-TTR binding | 50.2 OR 28.8 |
Two alternative values presented: one fitted using KD from Nelson et al. (2020) combined with kon from Corazza et al. (2019); the other directly using koff from Corazza et al. (2019) [37,40]. | |
| Association rate constant for second tafamidis-TTR binding | 12600 | [37] | |
| Dissociation rate constant for second tafamidis-TTR binding | 2998.8 OR 216 |
Two alternative values presented: one fitted using KD from Nelson et al. (2020) combined with kon from Corazza et al. (2019); the other directly using koff from Corazza et al. (2019) [37,40]. | |
| Association rate constant for tafamidis-albumin binding | 1000 | [40]. | |
| Dissociation rate constant for tafamidis-albumin binding | 1800 | Exact value unknown. Determined by simultaneous fitting with the albumin-tafamidis complex association rate to achieve KD = kdiss/ka [40]. | |
| Individual albumin concentration | Patient-specific | Albumin concentration calculated using a baseline (50.34 g/L) adjusted for age (coefficient = -0.0575) and BMI (coefficient = -0.0738), with an inter-individual coefficient of variation (CV) of 10%. | |
| Irreversible elimination rate of bound TTR-tafamidis complexes | Tdeg * 0.75 | Analytically solved for ~ 33% increase of total TTR concentration |
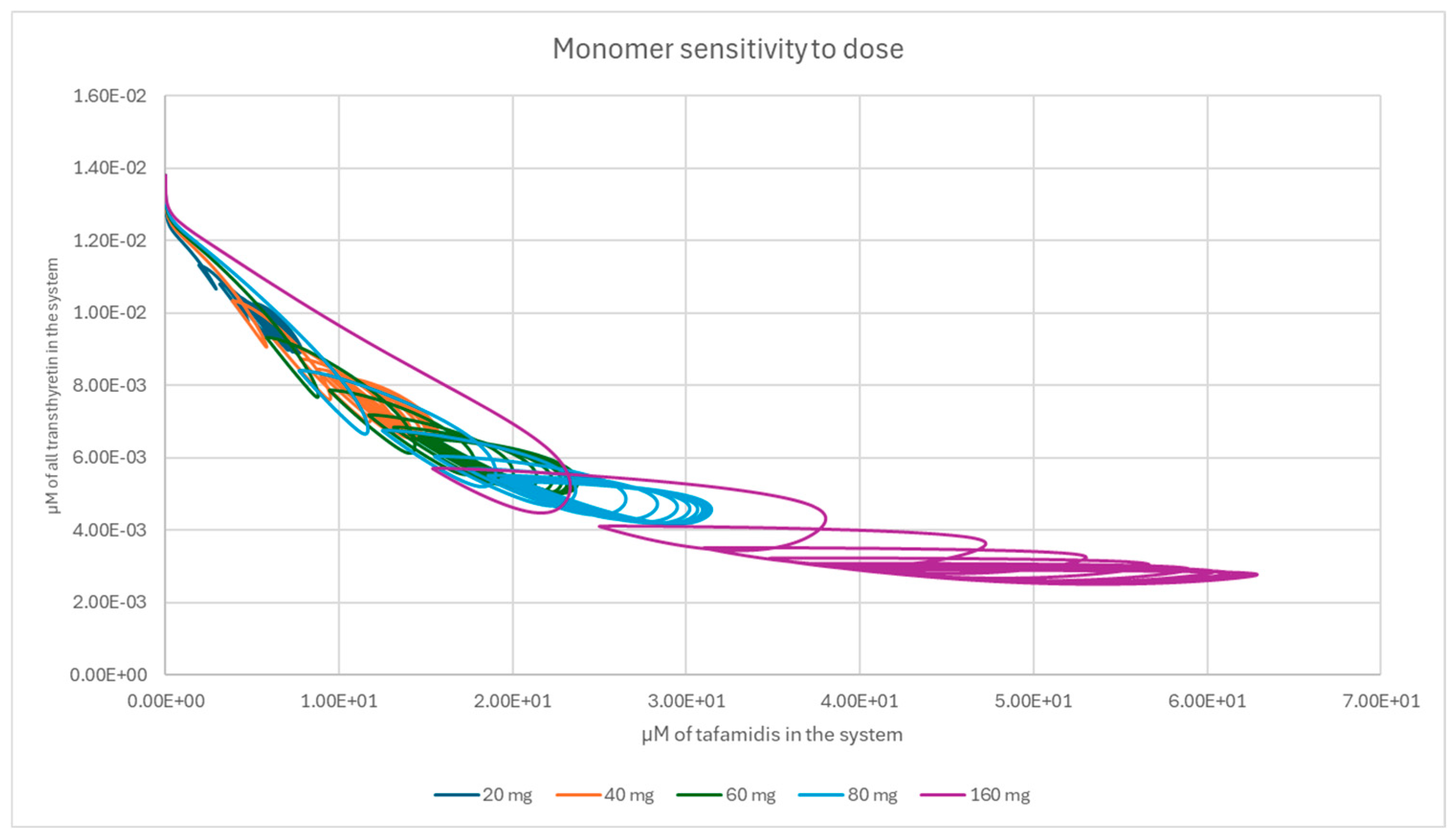
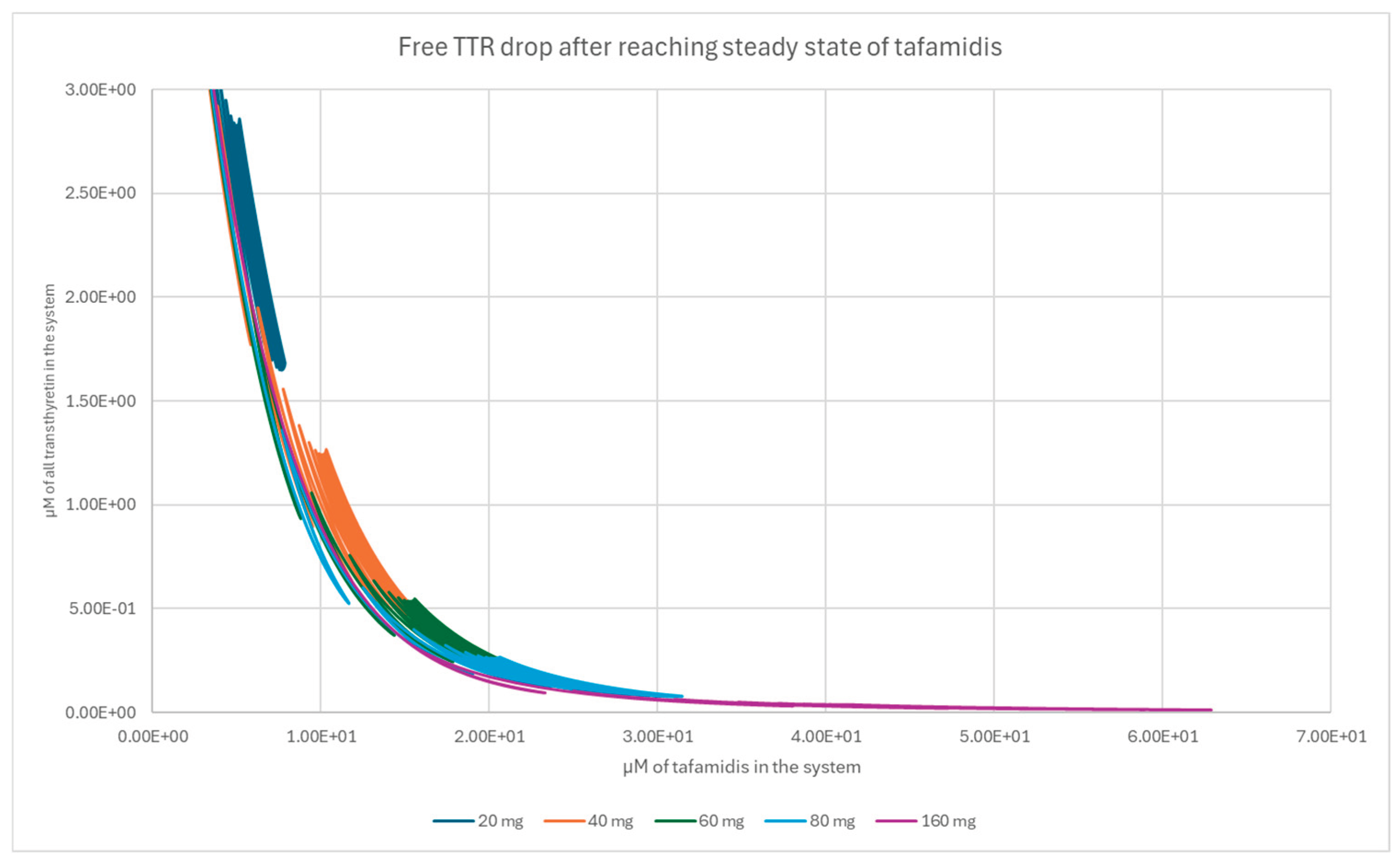
References
- Ruberg, F.L.; Berk, J.L. Transthyretin (TTR) Cardiac Amyloidosis. Circulation 2012, 126, 1286–1300. [Google Scholar] [CrossRef]
- Ueda, M. Transthyretin: Its function and amyloid formation. Neurochem. Int. 2022, 155, 105313. [Google Scholar] [CrossRef]
- Powers, E.T.; Amass, L.; Baylor, L.; Fernández-Arias, I.; Riley, S.; Kelly, J.W. Transthyretin Kinetic Stabilizers for ATTR Amyloidosis: A Narrative Review of Mechanisms and Therapeutic Benefits. Cardiol. Ther. 2025, 14, 333–350. [Google Scholar] [CrossRef]
- FDA approves new treatments for heart disease caused by a serious rare disease, transthyretin mediated amyloidosis. Jun 2019. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-new-treatments-heart-disease-caused-serious-rare-disease-transthyretin-mediated#:∼:text=On%20May%203%2C%20the%20U.S.,approved%20treatments%20for%20ATTR%2DCM.
- Maurer, M.S.; et al. Tafamidis Treatment for Patients with Transthyretin Amyloid Cardiomyopathy. N. Engl. J. Med. 2018, 379, 1007–1016. [Google Scholar] [CrossRef] [PubMed]
- Falk, R.H.; Haddad, M.; Walker, C.R.; Dorbala, S.; Cuddy, S.A.M. Effect of Tafamidis on Serum Transthyretin Levels in Non-Trial Patients With Transthyretin Amyloid Cardiomyopathy. JACC CardioOncology 2021, 3, 580–586. [Google Scholar] [CrossRef]
- Gamino, D.; Teruya, S.; De Los Santos, J.; Helmke, S.; Guadalupe, S.; Maurer, M. Tafamidis Increases Serum TTR (Prealbumin) Levels in both ATTRh and ATTRwt Cardiac Amyloidosis. J. Card. Fail. 2019, 25, S21. [Google Scholar] [CrossRef]
- Rappley, I.; et al. Quantification of Transthyretin Kinetic Stability in Human Plasma Using Subunit Exchange. Biochemistry 2014, 53, 1993–2006. [Google Scholar] [CrossRef] [PubMed]
- Ulaszek, S.; Wiśniowska, B.; Lisowski, B. No body fits in the test tube – the case of transthyretin. Amyloid 2024, 31, 347–349. [Google Scholar] [CrossRef] [PubMed]
- Lisowski, B.; Ulaszek, S.; Wiśniowska, B.; Bernhauerová, V.; Polak, S. Phenomenological model of transthyretin stabilization. Sci. Rep. 2026. [Google Scholar] [CrossRef]
- Tess, D.A.; Maurer, T.S.; Li, Z.; Bulawa, C.; Fleming, J.; Moody, A.T. Relationship of binding-site occupancy, transthyretin stabilisation and disease modification in patients with tafamidis-treated transthyretin amyloid cardiomyopathy. Amyloid 2023, 30, 208–219. [Google Scholar] [CrossRef]
- Rowland, M.; Peck, C.; Tucker, G. Physiologically-Based Pharmacokinetics in Drug Development and Regulatory Science. Annu. Rev. Pharmacol. Toxicol. 2011, 51, 45–73. [Google Scholar] [CrossRef] [PubMed]
- Danhof, M.; de Jongh, J.; De Lange, E.C.M.; Della Pasqua, O.; Et, A. Mechanism-Based Pharmacokinetic-Pharmacodynamic Modeling: Biophase Distribution, Receptor Theory, and Dynamical Systems Analysis. Annu. Rev. Pharmacol. Toxicol. 2007. [Google Scholar] [CrossRef] [PubMed]
- Oppenheimer, J.H.; Surks, M.I.; Bernstein, G.; Smith, J.C. Metabolism of Iodine-131-Labeled Thyroxine-Binding Prealbumin in Man. Science 1965, 149, 748–751. [Google Scholar] [CrossRef]
- Socolow, E.L.; Woeber, K.A.; Purdy, R.H.; Holloway, M.T.; Ingbar, S.H. Preparation of I-131-labeled human serum prealbumin and its metabolism in normal and sick patients. J. Clin. Invest. 1965, 44, 1600–1609. [Google Scholar] [CrossRef]
- Klamerus, K.J.; Watsky, E.; Moller, R.; Wang, R.; Riley, S. The effect of tafamidis on the QTc interval in healthy subjects. Br. J. Clin. Pharmacol. 2015, 79, 918–925. [Google Scholar] [CrossRef]
- Pfizer. “A Phase 1, Open-label, Randomized, Four-period, Four-sequence, Single-dose, Crossover Study In Healthy Volunteers, To Determine The Relative Bioavailability Of Pf-06291826 61 Mga Tafamidis Free Acid Soft Gelatin Capsules Compared To Commercial 4 × 20 Mg Tafamidis Meglumine Soft Gelatin Capsules Administered Under Fasted And Fed Conditions And The Effect Of Food On The Oral Bioavailability Of Pf-06291826 61 Mga Tafamidis Free Acid Soft Gelatin Capsules,” clinicaltrials.gov, Clinical trial registration NCT03280173, Apr. 2018. Available online: https://clinicaltrials.gov/study/NCT03280173 (accessed on Jan. 16, 2026).
- Center for drug evaluation and research. “Application number 211996Orig1s000 212161Orig1s000.” FDA Office of Clinical Pharmacology, Nov. 02, 2018. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/211996Orig1s000,%20212161Orig1s000ClinPharmR.pdf.
- Lockwood, P.A.; et al. The Bioequivalence of Tafamidis 61-mg Free Acid Capsules and Tafamidis Meglumine 4 × 20-mg Capsules in Healthy Volunteers. Clin. Pharmacol. Drug Dev. 2020, 9, 849–854. [Google Scholar] [CrossRef]
- Jamei, M.; et al. Population-Based Mechanistic Prediction of Oral Drug Absorption. AAPS J. 2009, 11, 225–237. [Google Scholar] [CrossRef]
- Sinha, U.; Rao, S. Methods of treating ttr amyloidosis using ag10. Available online: https://patents.google.com/patent/CA3094711A1/.
- Tetko, I.V.; et al. Virtual Computational Chemistry Laboratory – Design and Description. J. Comput. Aided Mol. Des. 2005, 19, 453–463. [Google Scholar] [CrossRef]
- Tafamidis - NDA 212161. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/211996Orig1s000,%20212161Orig1s000PharmR.pdf (accessed on Oct. 15, 2025).
- Lee, K.-R.; et al. Pharmacokinetics of tafamidis, a transthyretin amyloidosis drug, in rats. Xenobiotica 2018, 48, 831–838. [Google Scholar] [CrossRef]
- Sun, D.; et al. Comparison of Human Duodenum and Caco-2 Gene Expression Profiles for 12,000 Gene Sequences Tags and Correlation with Permeability of 26 Drugs. Pharm. Res. 2002, 19, 1400–1416. [Google Scholar] [CrossRef] [PubMed]
- “VYNDAQEL PRODUCT MONOGRAPH,” Pfizer, Oct. 2022. Available online: https://pdf.hres.ca/dpd_pm/00067760.PDF.
- Hansen, N.T.; Kouskoumvekaki, I.; Jørgensen, F.S.; Brunak, S.; Jónsdóttir, S.Ó. Prediction of pH-Dependent Aqueous Solubility of Druglike Molecules. J. Chem. Inf. Model. 2006, 46, 2601–2609. [Google Scholar] [CrossRef]
- Tchaparian, E.; Xu, G.; Huang, T.; Jin, L. Cell Based Experimental Models as Tools for the Prediction of Human Intestinal Absorption. Poster Presentation, 15th North American Regional International Society for the Study of Xenobiotics Meeting, 2008. [Google Scholar]
- Hens, B.; et al. Gastrointestinal transfer: In vivo evaluation and implementation in in vitro and in silico predictive tools. Eur. J. Pharm. Sci. 2014, 63, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Australian Public Assessment Report for Tafamidis and Tafamidis meglumine. Therapeutic Goods Administration. Sep 2020. Available online: https://www.tga.gov.au/sites/default/files/auspar-tafamidis-200903.pdf.
- Sawada, Y.; Hanano, M.; Sugiyama, Y.; Harashima, H.; Iga, T. Prediction of the volumes of distribution of basic drugs in humans based on data from animals. J. Pharmacokinet. Biopharm. 1984, 12, 587–596. [Google Scholar] [CrossRef] [PubMed]
- Mayumi, K.; Tachibana, M.; Yoshida, M.; Ohnishi, S.; Kanazu, T.; Hasegawa, H. The Novel In Vitro Method to Calculate Tissue-to-Plasma Partition Coefficient in Humans for Predicting Pharmacokinetic Profiles by Physiologically-Based Pharmacokinetic Model With High Predictability. J. Pharm. Sci. 2020, 109, 2345–2355. [Google Scholar] [CrossRef]
- Rodgers, T.; Rowland, M. Physiologically based pharmacokinetic modelling 2: Predicting the tissue distribution of acids, very weak bases, neutrals and zwitterions. J. Pharm. Sci. 2006, 95, 1238–1257. [Google Scholar] [CrossRef]
- Committee for Medicinal Products for Human Use (CHMP), “EMA assessment report - Vyndaqel,” Assessment report - Vyndaqel. Available online: https://www.ema.europa.eu/en/documents/variation-report/vyndaqel-h-c-2294-x-0049-g-epar-assessment-report_en.pdf (accessed on Dec. 15, 2025).
- Vyndaqel and Vyndamax full prescribing information. Available online: https://www.fda.gov/media/126283/download.
- Ji, A.X.; Betz, A.; Sinha, U. Differential Binding Affinities and Kinetics of Transthyretin Stabilizers. J. Cardiovasc. Pharmacol. 2025, 86, 204. [Google Scholar] [CrossRef]
- Corazza, A.; et al. Binding of Monovalent and Bivalent Ligands by Transthyretin Causes Different Short- and Long-Distance Conformational Changes. J. Med. Chem. 2019, 62, 8274–8283. [Google Scholar] [CrossRef]
- Sousa, M.M.; Saraiva, M.J. Internalization of Transthyretin. J. Biol. Chem. 2001, 276, 14420–14425. [Google Scholar] [CrossRef] [PubMed]
- Romine, C.; Wiseman, R.L. Starting at the beginning: endoplasmic reticulum proteostasis and systemic amyloid disease. Biochem. J. 2020, 477, 1721–1732. [Google Scholar] [CrossRef]
- Nelson, L.T.; Paxman, R.J.; Xu, J.; Webb, B.; Powers, E.T.; Kelly, J.W. Blinded potency comparison of transthyretin kinetic stabilisers by subunit exchange in human plasma. Amyloid 2021, 28, 24–29. [Google Scholar] [CrossRef]
- Ingenbleek, Y. Plasma Transthyretin Reflects the Fluctuations of Lean Body Mass in Health and Disease. In Recent Advances in Transthyretin Evolution, Structure and Biological Functions; Richardson, S. J., Cody, V., Eds.; Springer: Berlin, Heidelberg, 2009; pp. 329–357. [Google Scholar] [CrossRef]
- Sekijima, Y.; Tokuda, T.; Kametani, F.; Tanaka, K.; Maruyama, K.; Ikeda, S.-I. Serum transthyretin monomer in patients with familial amyloid polyneuropathy. Amyloid 2001, 8, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Drug bank website. Available online: https://go.drugbank.com/drugs/DB11644.
- Avdeef, A. Solubility of sparingly-soluble ionizable drugs. Adv. Drug Deliv. Rev. 2007, 59, 568–590. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, T.; Leahy, D.; Rowland, M. Physiologically Based Pharmacokinetic Modeling 1: Predicting the Tissue Distribution of Moderate-to-Strong Bases. J. Pharm. Sci. 2005, 94, 1259–1276. [Google Scholar] [CrossRef]
- Wiseman, R.L.; Johnson, S.M.; Kelker, M.S.; Foss, T.; Wilson, I.A.; Kelly, J.W. Kinetic Stabilization of an Oligomeric Protein by a Single Ligand Binding Event. J. Am. Chem. Soc. 2005, 127, 5540–5551. [Google Scholar] [CrossRef] [PubMed]
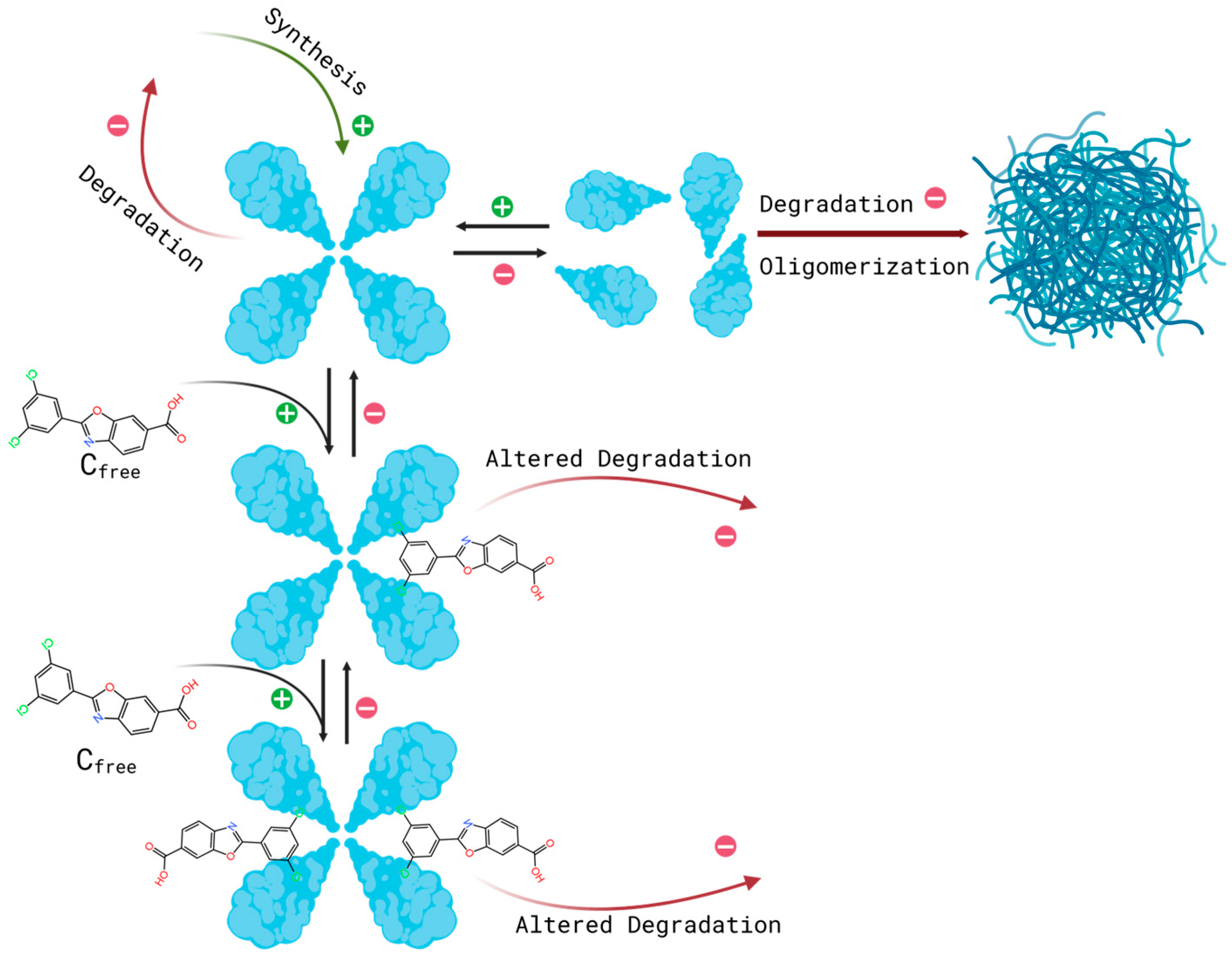
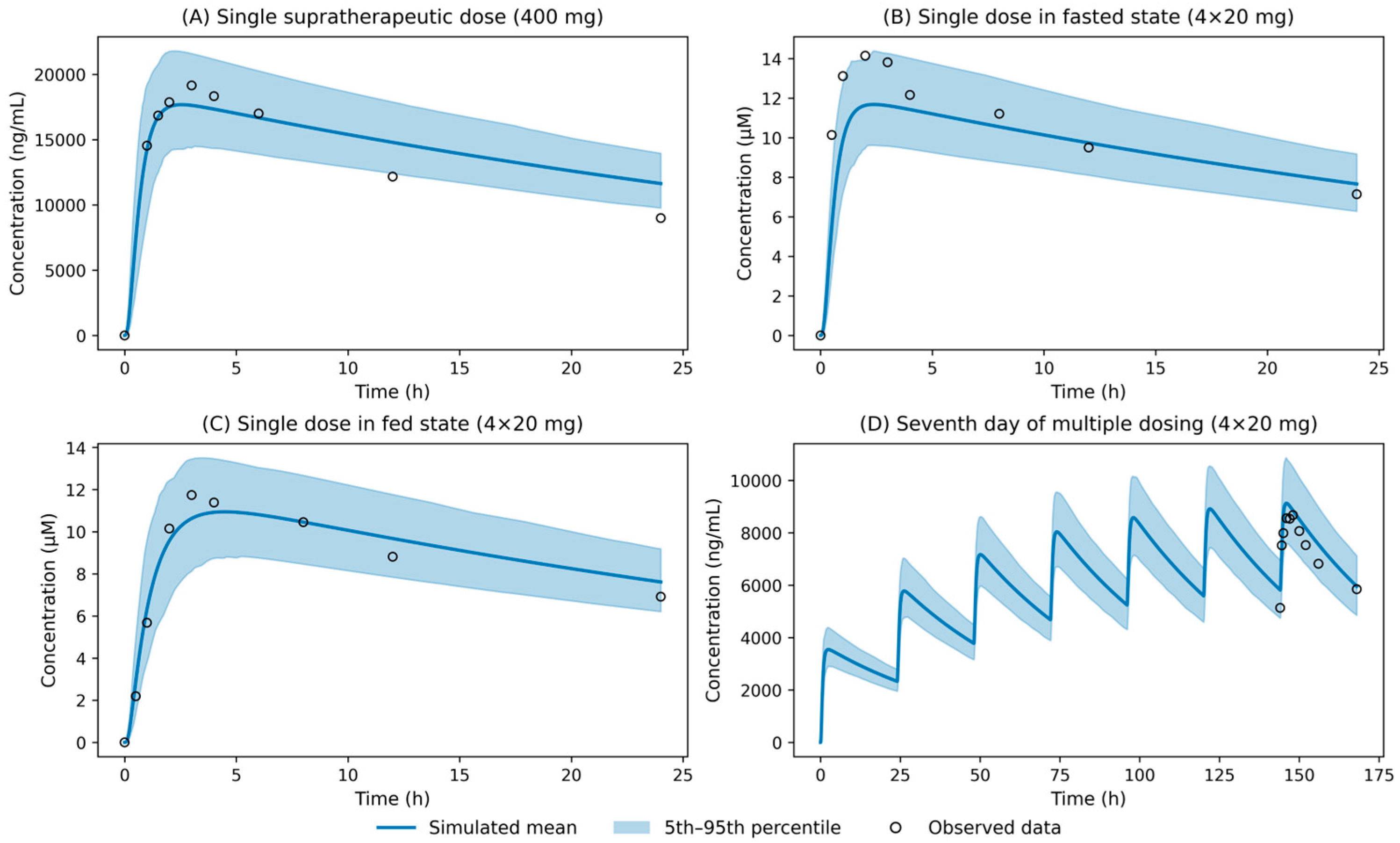
| Study / Trial identifier | Study purpose and design | Dose regimen and PK data used in this work |
|---|---|---|
| Thorough QT study (NCT01775761) [16]. | Phase I; randomized crossover; healthy volunteers (n = 42); fasted; QTc evaluation with PK as secondary endpoint. | Single oral supratherapeutic dose of tafamidis meglumine (400 mg); observed plasma PK (0–24 h); mean concentration–time data used to evaluate high-exposure behavior (Figure 2A). |
| Single-dose relative bioavailability and food-effect study (B3461054) [17,18]. | Phase I; open-label; randomized 4-period, 4-sequence crossover; healthy volunteers (n = 16); comparison of free acid and meglumine formulation; comparison of fasted and fed conditions. | Single oral dose of tafamidis meglumine 80 mg (4×20 mg); observed plasma PK under fasted and fed conditions; mean concentration–time profiles used to evaluate food effect (Figure 2B,C). |
| Phase Ib bioequivalence study (B3461056 - Lockwood et al., 2020) [19]. |
Phase I; open-label; randomized crossover; healthy male volunteers (n = 30); fasted; bioequivalence study. | Tafamidis meglumine 80 mg (4×20 mg) administered once daily for 7 days; observed steady-state plasma PK; mean concentration–time profiles on day 7 used to evaluate multiple-dose behavior (Figure 2D). |
| Study / scenario | Dosing regimen | State | PK parameter | Statistic | Observed | Predicted | Fold error (Pred/Obs) |
|---|---|---|---|---|---|---|---|
| B3461056 | Multiple dose, 4 × 20 mg, Day 7 | Fasted | AUC144–168h (ng x h/mL) | Mean (SD) | 169600 (35637) | 180557 (20998) | 1.06 |
| AUC144–168h (ng x h/mL) | Median (range) | 171000 (125000–258000) | 179721 (127744–254519) | 1.05 | |||
| AUC144–168h (ng x h/mL) | Geometric mean (%CV) | 166200 (20) | 179362 (12) | 1.08 | |||
| Cmax (ng/mL) | Mean (SD) | 9241 (1796) | 9160 (1039.26) | 0.99 | |||
| Cmax (ng/mL) | Median (range) | 8950 (6580–14600) | 9085 (6819.28–12840.15) | 1.02 | |||
| Cmax (ng/mL) | Geometric mean (%CV) | 9087 (18) | 9102 (11) | 1.00 | |||
| Tmax (h) | Median (range) | 2.0 (0.5–6.0) | 1.90 (1.18–3.84) | 0.95 | |||
| B3461054 | Single dose, 80 mg (4 × 20 mg) | Fasted | AUC0–inf (ng x h/mL) | Geometric mean (%CV) | 203400 (18) | 188274 (12) | 0.93 |
| Fasted | Cmax (ng/mL) | Geometric mean (%CV) | 4835 (20) | 3586 (12) | 0.74 | ||
| Fasted | Tmax (h) | Median (range) | 1.5 (0.5–4.05) | 2.26 (1.33–4.53) | 1.51 | ||
| Fed | AUC0–inf (ng x h/mL) | Geometric mean (%CV) | 208100 (23) | 188505 (12) | 0.91 | ||
| Fed | Cmax (ng/mL) | Geometric mean (%CV) | 4132 (15) | 3446.93 (12) | 0.83 | ||
| NCT01775761 | Single dose, 400 mg (supratherapeutic dose study) | Fasted | Cmax (ng/mL) | Geometric mean (%CV) | 20360 (18) | 17659 (13) | 0.87 |
| Fasted | AUC0–24h (ng x h/mL) | Geometric mean (%CV) | 305400 (15) | 342313 (13) | 1.12 | ||
| Fasted | Tmax (h) | Median (range) | 2.0 (1–6) | 2.3 (1.37–5.07) | 1.15 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




