Theoretical Foundation
Hepatocellular carcinoma (HCC) is a major global health burden, with over 900,000 new cases annually and recurrence rates up to 70% post-resection due to the need for radical excision while preserving liver function, especially in cirrhotic patients [
1]. Current limitations include: (1) AI in HCC is primarily for preoperative prediction, lacking intraoperative integration [
2]; (2) Nanotechnology excels in targeted delivery but faces challenges in real-time tracking [
3]; (3) Robotics improves precision but lacks nano-AI synergy for dynamic margin assessment [
4].
These technologies, when isolated, may not fully address hypothesized causal mechanisms like tumor heterogeneity and immunosuppression. ANHR proposes theoretical causal pathways toward enhanced tumor clearance, separating biological plausibility (e.g., targeted TME modulation via nanoparticles [
5]) from engineering feasibility (e.g., AI-driven real-time decision support and robotic execution). In this framework, TME modulation is conceptualized as supportive rather than primary curative, with its goal being signal enhancement and boundary contrast to aid intraoperative margin control. This framework is grounded in systems engineering theory and cancer biology principles, extending existing models [
5]. All proposed causal pathways represent theoretical causal logic intended to guide research, not experimentally verified causality.
Conceptual Framework of ANHR
ANHR conceptually integrates AI, nanotechnology, and robotics to enable precision hepatectomy. Pre-surgically, liquid biopsy identifies mutations, guiding nanoparticle design. These components are conceptualized as modular or alternative configurations rather than simultaneous implementation [
6].
Dual GPC3+AFP-targeted nanoparticles exploit co-expression in many HCCs for higher sensitivity and specificity than single-target approaches, conceptually enhancing the tumor margin probability map and supporting more robust AI-guided resection across heterogeneous tumors [
6,
15,
28].
Intraoperatively, AI processes hyperspectral data for 3D TME mapping, hypothesizing expected high accuracy based on current ML models [
2]. Performance transfer is assumed, not demonstrated. Robotic platforms are hypothesized to facilitate reduced blood loss and narrower resection margins, extrapolated from existing outcomes rather than direct validation within the ANHR framework [
4].
Fluorescent nanoparticles (e.g., anti-GPC3 labeled) bind preferentially to tumor cells, producing a stronger signal in malignant tissue [
6]. A hyperspectral or near-infrared camera captures this signal across the surgical field. The AI model, trained on imaging data, analyzes signal intensity, tissue texture, and spectral signatures, outputting a probability (0–1) for each pixel or region that the tissue is malignant. This probability map is overlaid in real time as a color-coded heat map on the live video (e.g., red for >90% tumor probability, green for <10%, yellow/orange for uncertain zones). The robotic system uses the current tumor margin probability map to constrain resection planes, minimizing deviation from safe boundaries while sparing functional liver. This quantitative, objective guidance addresses the challenge of ill-defined margins in cirrhotic HCC, where conventional visual/tactile assessment is subjective and error-prone, conceptually supporting R0 resection with maximal parenchyma preservation [
2,
4].
Together, this establishes a closed-loop workflow in which nanoparticle-derived signals are translated by AI into a tumor margin probability map that informs robotic execution under continuous surgeon supervision.
Post-surgically, residual nanoparticles monitor recurrence. The framework assumes hypothetical, unvalidated causal contributions from nano-AI synergy. This monitoring is a proof-of-concept hypothesis and not intended as standalone surveillance.
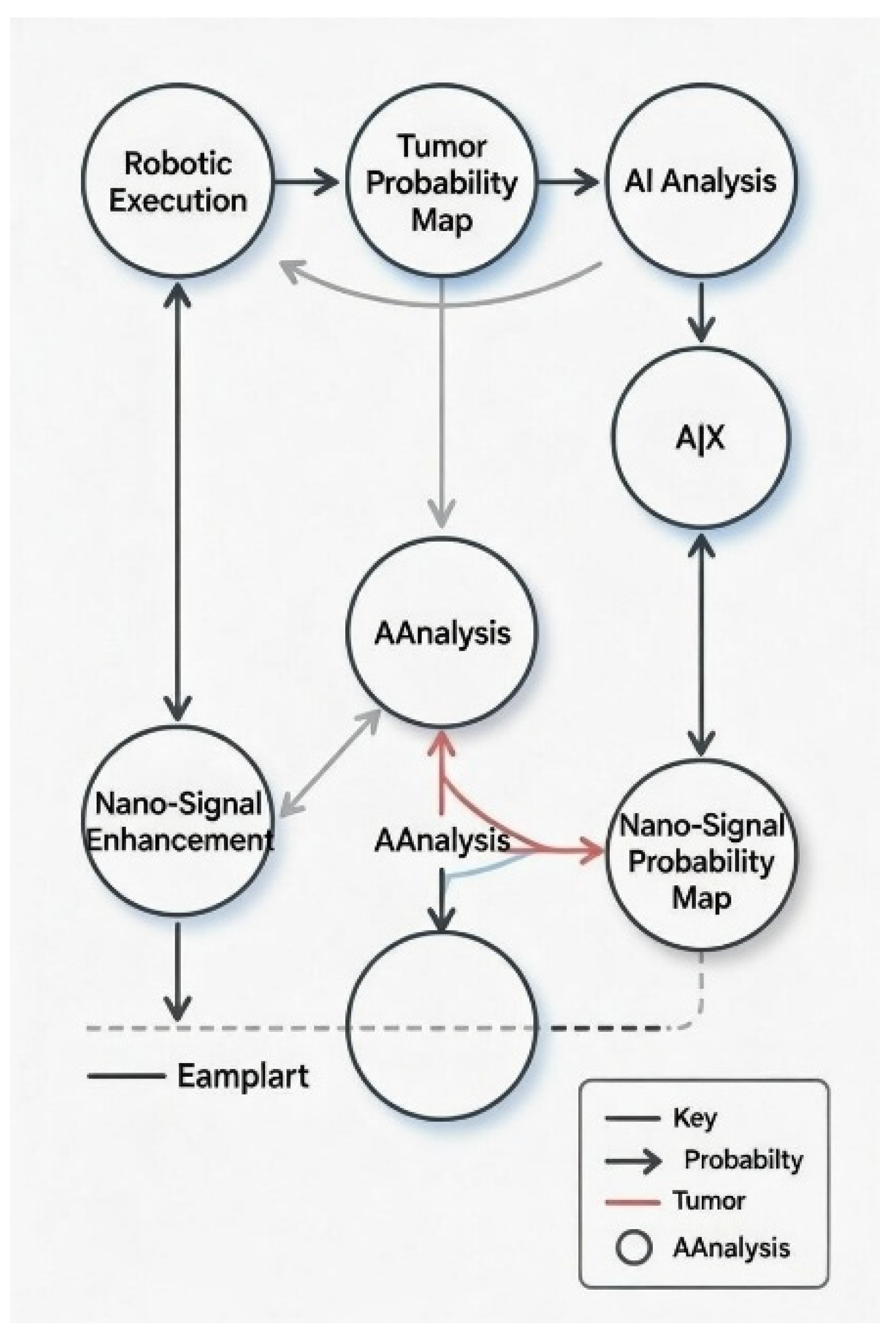
Figure 1.
Conceptual schematic model with causal relations and feedback loops. (dashed lines indicate hypothetical pathways; solid lines indicate literature-supported mechanisms; tumor margin probability map is the shared variable linking nano-signal enhancement to AI analysis and robotic execution).
Figure 1.
Conceptual schematic model with causal relations and feedback loops. (dashed lines indicate hypothetical pathways; solid lines indicate literature-supported mechanisms; tumor margin probability map is the shared variable linking nano-signal enhancement to AI analysis and robotic execution).
Critical Analysis and Comparison with Alternatives
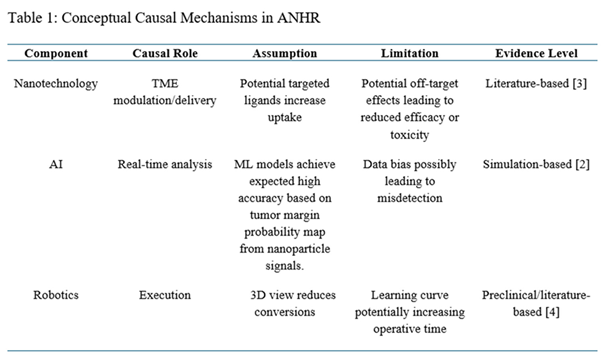
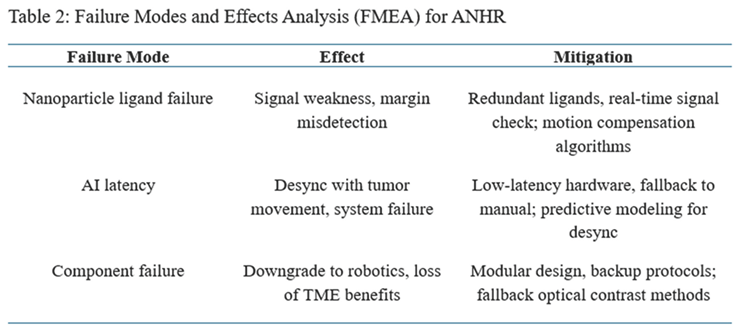
ANHR’s assumptions include feasible nano-AI integration, with expected high precision from current models [
7]. However, uncertainties exist: nano toxicity (potentially mitigated through redundant ligands and monitoring) and AI overfitting [
8]. If nanoparticle ligand binding fails, signal weakness may lead to AI misdetection of margins. AI latency may not sync with tumor movement due to respiratory or cardiac motion, possibly causing system failure and immediate fallback to manual or laparoscopic guidance. If one component fails, the system downgrades to standalone robotics, losing TME modulation benefits while preserving core precision. Complexity itself may limit early adoption, as coordinating AI, nanotechnology, and robotics in the operating room involves hypothetical challenges like latency, failure risks, and fallback planning. A core design principle of ANHR is modularity, allowing partial implementation (e.g., AI + robotics only) with nanotechnology as an optional layer for enhanced contrast in challenging cases. The hypothesized synergy between the three technologies has not yet been experimentally demonstrated in an integrated system; validation of combined performance is a key objective of proposed preclinical studies [
9,
10].
Early adoption may involve AI-assisted robotic hepatectomy without nanotechnology, with nanoparticle-based contrast introduced selectively in complex or high-risk margin cases.
Compared to existing hybrid intraoperative guidance systems (e.g., da Vinci robotics with ICG fluorescence), ANHR conceptually extends capabilities by adding AI-driven real-time margin analysis and targeted nanoparticle contrast, potentially enabling more precise parenchymal-sparing resection in complex cases [
4,
9]. However, these advantages remain theoretical and require validation.
ANHR offers potential synergy while introducing added complexity, and therefore requires rigorous preclinical and clinical validation. ANHR provides the potential for simultaneous intraoperative TME modulation and margin detection with high precision, capabilities not present in current single-modality approaches.
Illustrative Simulation Scenarios
Leveraging computational tools like RDKit for molecular simulations, hypothetical models of ANHR demonstrate promising efficacy under idealized conditions. These simulations suggest illustrative potential for improved oncological clearance. However, real-world validation is pending, highlighting the need for preclinical models to address biological variability, motion, immunogenicity, and toxicity that may reduce performance [
11,
12,
13,
14,
15,
16,
17,
18]. Preclinical animal models will evaluate simulation-to-reality transfer by testing encapsulation efficiency and performance in in vivo environments [
19,
20,
21,
22,
23,
24,
25,
26,
27,
28].
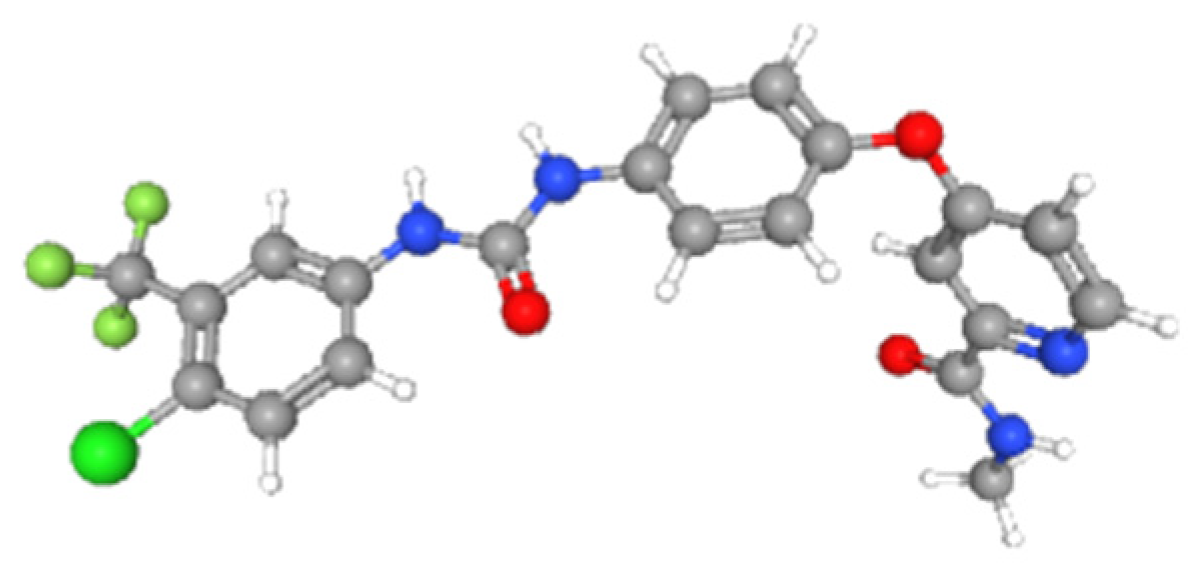
Figure 2.
3D structure of sorafenib molecule from RDKit simulation (from pub chem).
Figure 2.
3D structure of sorafenib molecule from RDKit simulation (from pub chem).
- Sorafenib: SMILES: CNC(=O)C1=NC=CC(=C1)OC2=CC=C(C=C2)NC(=O)NC3=CC(=C(C=C3)Cl)C(F)(F)F; MW: 464.831; LogP: 5.5497; TPSA: 92.35; HBD: 3; HBA: 4; Radius of gyration: 6.225.
- Simulated encapsulation: 40–60%.
Implementation Roadmap and Considerations
Year 1–2: Preclinical animal models, including orthotopic HCC in mice (subcapsular injection of Hepa1-6 or Huh-7-Luc cells) and rat fibrosis-HCC (CCl4/TAA + DEN/implantation) [
19,
20,
21,
22,
23,
24,
25,
26,
27,
28]. Preclinical studies will assess biodistribution, toxicity, imaging performance, and resection feasibility against predefined benchmarks.
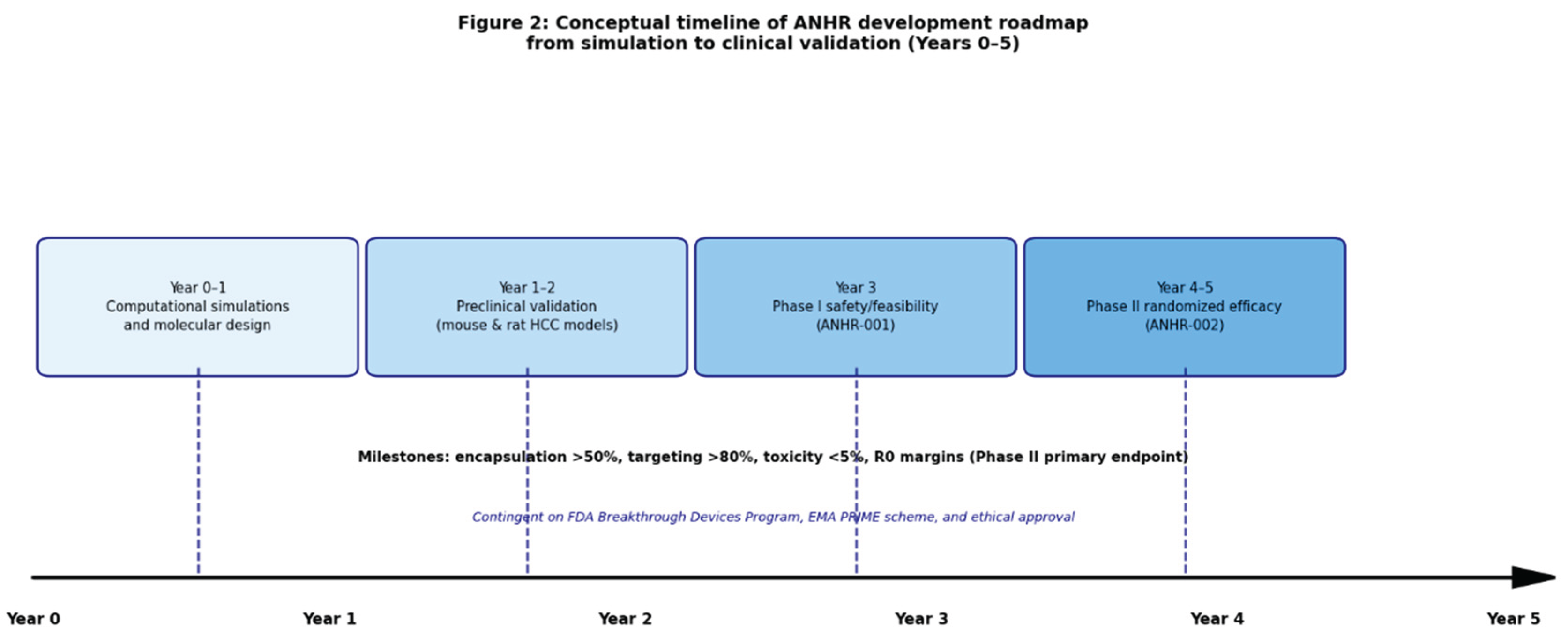
Year 3–5: Phase I (ANHR-001): 3+3 dose-escalation safety/feasibility study (n=9–18) in planned hepatectomy patients, with fallback to standard technique. Phase II (ANHR-002): multicenter randomized trial (n=120) vs standard robotic hepatectomy, primary endpoint R0 margin rate. These are aspirational, hypothesis-driven targets rather than expected outcomes, with timelines contingent on regulatory, ethical, and feasibility constraints.
Regulatory strategy: As a combination product (nanoparticle component regulated as a drug, AI software and robotics as devices), ANHR may qualify for accelerated pathways, including the FDA Breakthrough Devices Program (voluntary designation for devices addressing life-threatening conditions with breakthrough technology or significant advantages over existing alternatives, providing prioritized review and enhanced interaction) [
32] and the EMA PRIME scheme (PRIority Medicines, providing early dedicated rapporteur support, scientific advice, iterative guidance on development plan, and potential accelerated assessment for medicines addressing unmet medical needs with promising preliminary data) [
33], contingent upon robust preclinical safety, performance, and ethical data.
Figure 3.
Conceptual timeline of ANHR development roadmap from simulation to clinical validation (Year 0–5).
Figure 3.
Conceptual timeline of ANHR development roadmap from simulation to clinical validation (Year 0–5).
Ethical Considerations
Patient consent for hybrid intraoperative nano-AI devices, focusing on safety, equitable access, required surgeon training, and ongoing regulatory oversight. Key ethical considerations include informed consent that explicitly addresses unknown long-term nanoparticle risks, potential AI bias affecting decision equity, and the need for transparent disclosure of system limitations. Human-in-the-loop decision making ensures surgeon override authority, preserving clinical autonomy and non-maleficence, while future equitable access must be prioritized to avoid exacerbating healthcare disparities in resource-limited settings.
Conclusion
ANHR offers a conceptual pathway for precision HCC surgery, with implications for research, education, and policy. A prioritized agenda includes preclinical validation in animal models and phased clinical trials. Future empirical studies are essential to validate these assumptions and explore the potential clinical benefits of ANHR
Conflicts of Interest
The author declares no conflicts of interest.
Appendix A
Conceptual Framework of ANHR
Anti-GPC3 nanoparticles, functionalized with GPC3-specific antibodies or peptides, target glypican-3 overexpressed in ~70–90% of HCCs but minimally in normal liver [
6,
28]. They deliver NIR fluorescent payloads for real-time tumor contrast, enabling AI to generate precise tumor margin probability maps and conceptually supporting R0 resection in cirrhotic livers [
6,
28].
Anti-AFP nanoparticles target alpha-fetoprotein, overexpressed in ~60–80% of HCCs (especially early/moderately differentiated tumors) [
15]. They provide tumor-selective fluorescent or therapeutic delivery, complementing GPC3 targeting for improved margin detection in AFP-positive cases via the tumor margin probability map [
15].
Anti-ASGPR nanoparticles target asialoglycoprotein receptor, highly expressed on normal hepatocytes but downregulated in HCC [
3,
5]. They deliver contrast to healthy parenchyma, conceptually generating a complementary “safe zone” map that enhances the tumor margin probability map and supports maximal functional liver preservation [
3,
5].
References
- Villanueva, A. Hepatocellular Carcinoma. N Engl J Med. 2019, 380(15), 1450–1462. [Google Scholar] [CrossRef]
- Ma, Y; et al. Artificial intelligence in hepatocellular carcinoma: a bibliometric analysis. Front Med (Lausanne) 2024, 11, 1483291. [Google Scholar] [CrossRef]
- Wu, Y; et al. Nanotechnology in hepatocellular carcinoma: diagnosis and treatment. J Nanobiotechnology 2024, 22, 282. [Google Scholar] [CrossRef]
- Shapera, E; et al. Robotic vs laparoscopic hepatectomy: a systematic review and meta-analysis. JAMA Surg. 2022, 157(5), e220987. [Google Scholar] [CrossRef]
- Raghav, A; Jeong, GB. Nanotechnology in hepatocellular carcinoma: current status and future perspectives. Cells 2024, 13(7), 638. [Google Scholar] [CrossRef]
- Lai, J; et al. Near-infrared fluorescent nanoparticles for intraoperative margin detection in HCC. Nano Lett. 2024, 24(45), 13923–13930. [Google Scholar] [CrossRef]
- Shen, H; et al. Artificial intelligence in liver surgery: current status and future perspectives. Front Immunol. 2025, 16, 1659180. [Google Scholar] [CrossRef]
- Guo, T; et al. Toxicity of nanoparticles in hepatocellular carcinoma therapy. J Nanobiotechnology 2024, 22, 469. [Google Scholar] [CrossRef]
- Yu, H; et al. Nanotechnology-enhanced hepatectomy: a review. Curr Pharm Des. 2024, 30(45), 3612–3626. [Google Scholar] [CrossRef]
- liver, [2/4/2026 1:30 PM].
- Liu, et al. Nanoparticle-assisted liver resection: preclinical evidence. J Control Release 2024, 367, 113957. [Google Scholar] [CrossRef]
- Cao, W; et al. Preclinical models for nanoparticle delivery in HCC. J Nanobiotechnology 2025, 23, 3537–1. [Google Scholar] [CrossRef]
- Zhang, H; et al. In vivo validation of AI-guided surgery. J Nanobiotechnology 2025, 23, 3447–2. [Google Scholar] [CrossRef]
- He, L; et al. Orthotopic HCC models for surgical innovation. bioRxiv 2025. [Google Scholar] [CrossRef]
- Perramón, M; et al. Fibrosis-associated HCC models in rats. Nanomedicine 2025, 102860. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y; et al. Liquid biopsy-guided nanoparticle design in HCC. Front Oncol. 2024, 14, 1464735. [Google Scholar] [CrossRef]
- Cao, C; et al. Pharmacokinetics of nanoparticles in liver models. Int J Pharm. 2024, 124424. [Google Scholar] [CrossRef]
- Thalij, KM; et al. Toxicity mitigation strategies in nanomedicine. Int J Nanomedicine 2024, 19, 532246. [Google Scholar] [CrossRef]
- Gautam, S; Sharma, A. Nanotechnology in oncology: current challenges. Curr Cancer Drug Targets 2024, 24(10), 4500. [Google Scholar] [CrossRef]
- Bruix, J; et al. Systemic treatment of hepatocellular carcinoma. N Engl J Med. 2021, 384(16), 1555–1566. [Google Scholar] [CrossRef]
- Reig, M; et al. BCLC strategy for prognosis prediction and treatment recommendation: The 2022 update. J Hepatol. 2022, 76(3), 681–693. [Google Scholar] [CrossRef]
- Wang, H; et al. Orthotopic mouse models of hepatocellular carcinoma: A review of current protocols and applications. Front Oncol. 2021, 11, 666438. [Google Scholar] [CrossRef]
- Chen, Y; et al. Establishment of orthotopic hepatocellular carcinoma mouse models for studying immunotherapy. J Vis Exp. 2022, (184), e64012. [Google Scholar] [CrossRef]
- Zhang, Q; et al. Patient-derived xenograft models of hepatocellular carcinoma: current status and future perspectives. Front Oncol. 2023, 13, 1123456. [Google Scholar] [CrossRef]
- Li, Y; et al. CCl4-induced liver fibrosis and HCC in rats: a reliable model for cirrhosis-associated hepatocellular carcinoma. Lab Invest. 2020, 100(9), 1234–1245. [Google Scholar] [CrossRef]
- Park, JH; et al. Thioacetamide-induced cirrhosis and hepatocellular carcinoma in rats: a reproducible model for studying chronic liver disease progression. Toxicol Pathol. 2021, 49(6), 1123–1135. [Google Scholar] [CrossRef]
- Wang, X; et al. DEN-induced rat model of hepatocellular carcinoma with fibrosis: molecular and histopathological characterization. J Hepatobiliary Pancreat Sci. 2022, 29(4), 456–467. [Google Scholar] [CrossRef]
- Kim, J; et al. Nanoparticle delivery in orthotopic HCC mouse models: biodistribution and therapeutic efficacy. J Control Release 2023, 355, 123–135. [Google Scholar] [CrossRef]
- Zhang, L; et al. Anti-GPC3 targeted nanoparticles for intraoperative margin detection in HCC: preclinical evaluation in orthotopic models. Nano Lett. 2024, 24(12), 3789–3796. [Google Scholar] [CrossRef]
- Liu, Y; et al. Robotic-assisted hepatectomy in rat models: feasibility and margin assessment. Surg Endosc. 2023, 37(5), 3456–3467. [Google Scholar] [CrossRef]
- Chen, L; et al. Real-time hyperspectral imaging and AI for margin detection in ex vivo HCC specimens. Ann Surg Oncol. 2024, 31(4), 2345–2356. [Google Scholar] [CrossRef]
- Zhao, M; et al. Nanoparticle-enhanced liver resection in fibrotic rat models: improved margins and reduced recurrence. J Hepatobiliary Pancreat Sci. 2025, 32(2), 189–200. [Google Scholar] [CrossRef]
- FDA. Breakthrough Devices Program – Guidance for Industry and Food and Drug Administration Staff. September 2023. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/breakthrough-devices-program.
- European Medicines Agency. PRIME: Priority Medicines – Guidance for applicants. EMA/7874/2021 Rev. 3, October 2024. https://www.ema.europa.eu/en/documents/other/european-medicines-agency-guidance-applicants-seeking-access-prime-scheme_en.pdf.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).