Main text
With advances in medical technology, complex chronic diseases have progressively supplanted infectious diseases as the predominant clinical challenge[
1]. IgA nephropathy (IgAN) is the most common primary glomerulonephritis worldwide and a leading cause of chronic kidney disease (CKD) and kidney failure[
2]. Nephrotic syndrome (NS) is a common clinical syndrome in renal disease, characterized primarily by massive proteinuria, hyperlipidemia, edema, and hypoalbuminemia. The precise etiologies of both conditions remain incompletely understood to date, and they are prone to recurrence, requiring patients to undergo regular follow-up. Herein, we report a case of IgAN with NS and prolonged hypercholesterolemia that was poorly responsive to immunosuppressive therapy but ultimately achieved remission following precise etiological interventions.
1. Case Presentation
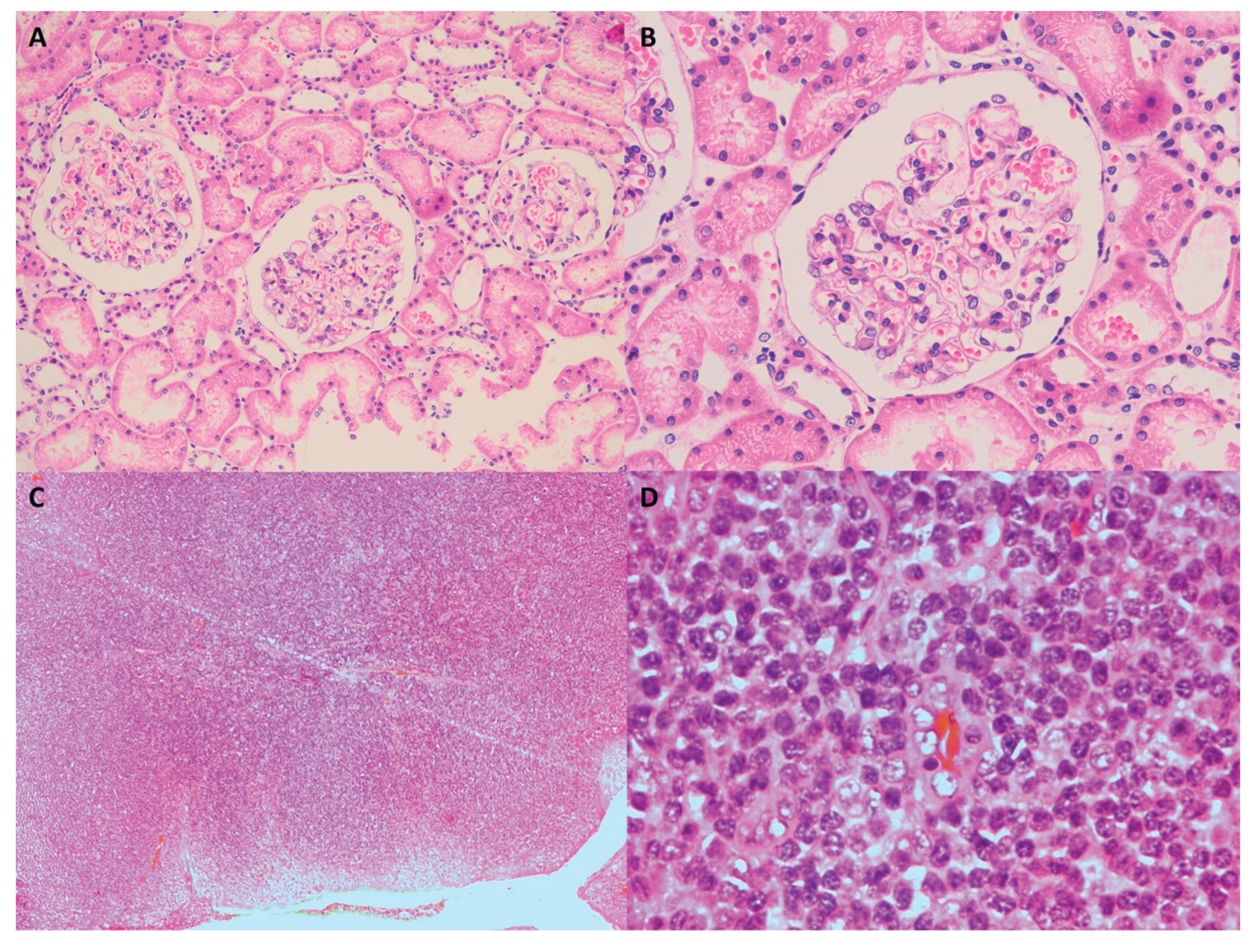
A 28.5-year-old female physician was admitted to the Nephrology Department on January 9, 2019, due to "bilateral lower limb edema for one month." Physical examination revealed mild pitting edema in both lower limbs. Urinalysis showed urine protein +3g/L and occult blood +1 Cell/uL. Serum albumin was 20.1 g/L. Lipid profile: total cholesterol (TC) 8.68 mmol/L, low-density lipoprotein cholesterol (LDL-C) 5.24 mmol/L. The 24-hour urinary protein excretion was 4.592 g/24h. Abdominal ultrasound indicated enhanced echogenicity of both renal parenchyma, with no other significant abnormalities. A renal biopsy performed on January 10, 2019, revealed minor lesion IgA nephropathy (
Figure 1, A and B). After excluding toxicant exposure at an occupational disease hospital, immunosuppressive therapy along with supportive lipid-lowering treatment was initiated on February 15, 2019. The treatment regimen was adjusted several times thereafter (
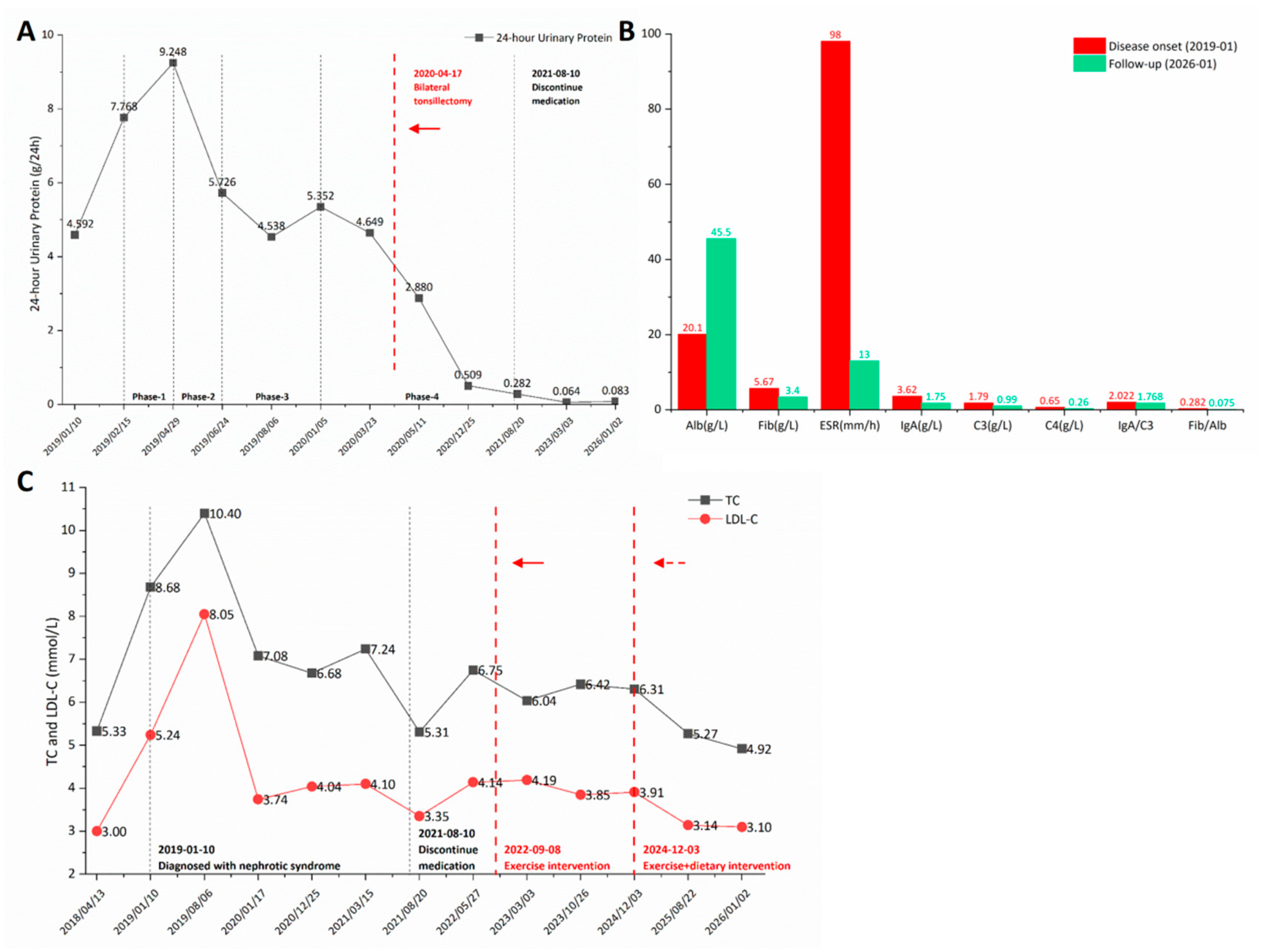
Table 1). The dynamic changes in 24-hour urinary protein are shown in
Figure 2A.
The patient reported a history of acute pharyngitis in early December 2018 during her initial hospitalization. However, this was not considered significant as she was asymptomatic (no sore throat, cough, or sputum) at admission, and the internal medicine examination did not reveal obviously enlarged tonsils. Subsequently, the patient sought consultations at several leading nephrology outpatient clinics, repeatedly emphasizing her history of tonsillar infection following night shifts, which consistently failed to garner adequate attention. Due to the persistent poor response to pharmacological therapy and based on the understanding that chronic tonsillar infection may drive IgAN via the mucosal immune pathway[
3,
4], the patient self-referred to the Department of Otolaryngology & Head and Neck. ENT examination revealed chronic congestion of the pharyngeal mucosa, Grade I enlargement with chronic congestion of both tonsils, enlarged crypt openings without purulent discharge, and lymphoid follicular hyperplasia of the posterior pharyngeal wall. She was admitted for chronic tonsillitis and underwent bilateral tonsillectomy on April 17, 2020. Postoperative tonsillar pathology showed lymphoid hyperplasia with bacterial colonies (
Figure 1, C and D). On postoperative day 24, the 24-hour urinary protein had decreased to below 3.5g/24h, after which immunosuppressants and corticosteroids were gradually tapered and completely discontinued by August 10, 2021, including lipid-lowering agents. Changes in inflammatory and immunological markers from disease onset to follow-up are shown in
Figure 2B.
Post-discontinuation laboratory tests revealed a rebound in blood lipids (
Figure 2C), with subsequent follow-ups consistently showing hypercholesterolemia (normal triglycerides). The patient’s hypoalbuminemia had resolved with NS remission, but the persistence of hypercholesterolemia suggested the presence of an independent metabolic factor. Reviewing past medical and body composition reports revealed mildly elevated TC as early as April 2018. Although her body mass index (BMI) was within the normal range, she exhibited a body composition phenotype marked by a high body fat percentage, elevated subcutaneous fat mass, and a low muscle mass percentage, consistent with concealed adiposity. Declining further drug intervention, the patient underwent exercise risk screening at a fitness center, received a personalized exercise prescription, and began a regimen of structured aerobic and strength training on September 8, 2022. However, annual check-ups showed her TC remained above 6 mmol/L. Subsequently, a semi-quantitative food frequency questionnaire (FFQ) revealed that, despite generally avoiding high-fat foods (e.g., fried foods, processed meats), she occasionally tended to consume foods high in cholesterol (e.g., organ meats, egg yolks). Notably, her prior adherence to a low-carbohydrate diet may have also contributed to her lipid profile, as such diets are associated with increased LDL-C in some individuals[
5,
6]. She implemented a targeted dietary modification plan on December 3, 2024, restricting high-cholesterol foods, increasing the intake of complex carbohydrates while increasing aerobic exercise volume[
7]. Follow-up tests subsequently showed a gradual normalization of her lipid profile.
2. Discussion
2.1. Etiological Mechanisms and Intervention Analysis in This Case
Current research suggests that a minority of IgAN patients can present with nephrotic syndrome[
2], and chronic tonsillitis may disrupt mucosal immune homeostasis, leading to aberrant IgA1 deposition in glomeruli, complement activation, and subsequent proteinuria[
3,
4]. Comparison of inflammatory and immunological markers between disease onset and follow-up (
Figure 2B) shows that ESR, serum IgA, and the Fib/Alb ratio decreased from 98 mm/h, 3.62 g/L, and 0.282, respectively, to within normal ranges (13 mm/h, 1.75 g/L, 0.075), indicating resolution of both the inflammatory state and immune hyperactivation. Further detailed history-taking revealed that the patient’s work environment harbored multidrug-resistant bacteria. Additionally, she was subject to chronic high-intensity work stress, irregular sleep-wake cycles due to night shifts, and physical inactivity—factors that may have collectively contributed to immune dysregulation and increased her susceptibility to infection. Immunosuppressive therapy likely failed to eliminate the persistent antigenic stimulus. In contrast, tonsillectomy, by radically removing the potential chronic infectious focus, led to the subsequent rapid remission of proteinuria. In summary, against a background of immune dysregulation fostered by long-term adverse psychosocial and behavioral factors, chronic tonsillar infection likely served as the key immunogenic trigger for the renal disease in this case.
2.2. Insights into the Gaps in Contemporary Disease Management: Lessons from This Case
In this case, a series of potentially modifiable etiological factors—from tonsillar infection to unhealthy lifestyle habits—were overlooked during routine clinical management, highlighting a structural deficiency within the current healthcare system regarding the integration of psychosocial and behavioral factors. Ample evidence indicates that adverse psychosocial and behavioral factors are closely linked to the development of many complex chronic diseases[
8,
9]; yet, their integration into routine clinical workflows remains inadequate: (1) Psychological Assessment: Psychology and mental health services currently face marginalization and underdevelopment within the biomedical healthcare systems both domestically and internationally. Medical training emphasizes biomedical knowledge and clinical skills, with insufficient attention to psychosocial factors[
10,
11,
12]. (2) Nutritional Assessment: Standardized nutritional risk screening is lacking in routine physical examinations and hospitalization processes, leading to significant underdiagnosis of malnutrition (including both undernutrition and overnutrition). Nutritional advice often depends on physician-initiated consultation, which is frequently overlooked due to inadequate training or low prioritization[
13,
14,
15,
16]. (3) Exercise Assessment: Exercise medicine content is sparse in medical school curricula, and clinicians lack systematic training in exercise prescription. Although substantial research supports the benefits of exercise for chronic and mental health conditions, these findings have not been adequately translated into clinical practice guidelines, resulting in a lag in application[
17,
18,
19,
20]. (4) Sleep Assessment: Sleep medicine is a quintessential interdisciplinary field. However, within a predominantly specialty-based healthcare system, effective integration pathways are lacking. Medical students receive minimal exposure to sleep medicine, leading to a general lack of awareness and competency among practicing physicians in recognizing and managing sleep disorders[
21,
22,
23].
2.3. Implications and Recommendations for Clinical Practice
While this case focuses on IgAN, the diagnostic challenges and therapeutic principles illustrated herein are reflective of broader issues encountered in the management of many complex chronic diseases. Currently, the diagnosis of many internal medicine diseases (including psychiatric disorders) remains primarily based on clinical symptom complexes and laboratory findings rather than etiology. In this case, the diagnosis of nephrotic syndrome was initially made based on the clinical syndrome, while IgAN was confirmed by renal immunopathology. This phenotype-based diagnostic approach often leads to diagnostic ambiguity or overlap. For instance, the diagnostic criteria for nephrotic syndrome in this case included hypercholesterolemia, complicating the clinical attribution and description of the disease. Following her illness, the patient sought a position transfer and engaged in lifestyle modifications including sleep regulation, dietary optimization, structured exercise, and the study of psychology and sociology to enhance self-awareness and regulate her behavior. As of the last follow-up in January 2026 (approximately seven years post-onset), her physical examination parameters were nearly normal. The resolution of hypercholesterolemia through targeted exercise and dietary adjustments further validates the importance of identifying concealed adiposity in solving complex metabolic problems, as well as the significant value of precision lifestyle interventions in chronic disease management. This case suggests that for patients with refractory chronic diseases, systematically assessing their psychosocial status, dietary, exercise, and sleep habits during the diagnostic and treatment process may help identify potential modifiable etiological factors. The phenomenon of disease remission following precision etiological intervention in this case, alongside the persistent difficulty in elucidating the precise causes of many complex chronic diseases in modern clinical practice, warrants greater attention from internists.
3. Conclusion and Future Perspectives
In modern medical practice, the unclear etiology of numerous complex chronic diseases profoundly reflects the complexity of living systems and the boundaries of current medical understanding. Contemporary clinical practice remains predominantly biomedically oriented, and the pervasive challenge of unknown etiology significantly impacts medical practice, urging a shift toward a more integrated biopsychosocial model. Etiological diagnosis is the cornerstone of clinical medicine, linking disease prevention, diagnosis, treatment, and prognosis. The relentless pursuit of etiological clarity remains the core driving force for medical advancement. It will propel medicine from experience-based to science-based practice, from symptomatic to etiological treatment, ultimately enabling more effective, precise, and patient-centered care.
Ethics Statement
This case report was reviewed and approved by the Ethics Committee of The First Affiliated Hospital of Guangxi Medical University (Approval No.: 2026-E0017).
Conflict of Interest Statement
This research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Vollset, S.E.; Ababneh, H.S.; Abate, Y.H.; Abbafati, C.; Abbasgholizadeh, R.; Abbasian, M.; Abbastabar, H.; Magied, A.H.A.A.A.; ElHafeez, S.A.; Abdelkader, A.; et al. Burden of Disease Scenarios for 204 Countries and Territories, 2022–2050: A Forecasting Analysis for the Global Burden of Disease Study 2021. The Lancet 2024, 403, 2204–2256. [CrossRef]
- Rovin, B.H.; Barratt, J.; Cook, H.T.; Noronha, I.L.; Reich, H.N.; Suzuki, Y.S.K.; Tang, S.C.W.; Trimarch, H.; Floege, J. KDIGO 2025 Clinical Practice Guideline for the Management of Immunoglobulin A Nephropathy (IgAN) and Immunoglobulin A Vasculitis (IgAV). Kidney International 2025, 108. [CrossRef]
- Tangon, N.; Kumfu, S.; Chattipakorn, N.; Chattipakorn, S.C. Links between Oropharyngeal Microbiota and IgA Nephropathy: A Paradigm Shift from Isolated Microbe to Microbiome. Microbiological Research 2025, 292, 128005. [CrossRef]
- Gesualdo, L.; Leo, V.D.; Coppo, R. The Mucosal Immune System and IgA Nephropathy. Seminars in Immunopathology 2021, 43, 657–668. [CrossRef]
- Burén, J.; Ericsson, M.D.L.N.; Damasceno, N.; Sjödin, A.N. A Ketogenic Low-Carbohydrate High-Fat Diet Increases LDL Cholesterol in Healthy, Young, Normal-Weight Women: A Randomized Controlled Feeding Trial. Nutrients 2021, 13, 814. [CrossRef]
- Schmidt, T.; Harmon, D.M.; Kludtke, E.; Mickow, A.; Simha, V.; Kopecky, S. Dramatic Elevation of LDL Cholesterol from Ketogenic-Dieting: A Case Series. American Journal of Preventive Cardiology 2023, 14, 100495. [CrossRef]
- Mach, F.; Koskinas, K.C.; Lennep, J.E.R. van; Tokgözoğlu, L.L.; Badimon, L.N.; Baigent, C.; Benn, M.R.A.N.; Binder, C.J.; Catapano, A.L.; Backer, G.G.D.; et al. 2025 Focused Update of the 2019 ESC/EAS Guidelines for the Management of Dyslipidaemias. European Heart Journal 2025, 46, 4359–4378. [CrossRef]
- Giuseppe, M.D.; Spatola, C.; Merlo, E.M.; Silvestro, O.; Giorgianni, C.M.; Juli, G.; Catalano, A.; Martino, G. RESEARCH ADVANCES IN CLINICAL PSYCHOLOGY OF CHRONIC DISEASES. Psychiatria Danubina 2025, 37, 56–62.
- Airhihenbuwa, C.O.; Tseng, T.-S.; Sutton, V.D.; Price, L. Global Perspectives on Improving Chronic Disease Prevention and Management in Diverse Settings. Preventing Chronic Disease 2021, 18. [CrossRef]
- Shalev, D.; Ekwebelem, M.I.; Brody, L.A.; Callahan, M.E.; Singh, N.; Reid, M.C. Hospice and Palliative Medicine Fellowship Training in Mental Health: A Survey of Program Directors. Journal of Pain and Symptom Management 2023, 66, 310–319. [CrossRef]
- Heetderks-Fong, E.; Bobb, A.N. Community Mental Health Workers: Their Workplaces, Roles, and Impact. Community mental health journal 2024, 60, 1547–1556. [CrossRef]
- Vaccarino, V.; Bremner, J.D. Stress and Cardiovascular Disease: An Update. Nature reviews. Cardiology 2024, 21, 603–616. [CrossRef]
- Serón-Arbeloa, C.; Labarta-Monzón, L.; Puzo-Foncillas, J.; Mallor-Bonet, T.; Lafita-López, A.; Bueno-Vidales, N.; Montoro-Huguet, M. Malnutrition Screening and Assessment. Nutrients 2022, 14, 2392. [CrossRef]
- Santos, L. The Impact of Nutrition and Lifestyle Modification on Health. European Journal of Internal Medicine 2022, 97, 18–25. [CrossRef]
- Hachey, S.M.; Hamilton, C.; Goins, B.; Underwood, P.; Chao, A.M.; Dolin, C.D. Nutrition Education and Nutrition Knowledge Among Obstetrics and Gynecology Residents. Journal of Women’s Health 2024, 33, 741–748. [CrossRef]
- Tang, H.Y.; Yang, M. Nutritional Assessment in Patients with Chronic Diseases: Tools, Challenges, and Future Directions. Nutrients 2023, 15, 4794. [CrossRef]
- Wood, L.N.; Ash, G. Exercise Prescription and Patient Outcomes. BMC Primary Care 2025, 26. [CrossRef]
- Wang, Y.F.; McCarthy, A.L.; Hayes, S.C.; Gordon, L.G.; Chiu, V.; Bailey, T.G.; Stewart, E.; Tuffaha, H. Economic Evaluation of Exercise Interventions for Individuals with Cancer: A Systematic Review. Preventive Medicine 2023, 172, 107491. [CrossRef]
- Tao, D.; Awan-Scully, R.; Cole, A.; Gao, Y.; Ash, G.I.; Gu, Y.D.; Dutheil, F.; Sun, Y.; Baker, J.S. Integration of Exercise Prescription into Medical Provision as a Treatment for Non-Communicable Diseases: A Scoping Review. Frontiers in Public Health 2023, 11, 1126244. [CrossRef]
- Greenslade, K.; Nelson, J.; Murray, A.; McCrea-Routray, R.; Hall, A.J. Is Medical Training Adequate to Promote Health and Give Patients What They Need? The Role of Sport and Exercise Medicine in 21st Century Healthcare. British Journal of Sports Medicine 2023, 57, 558–559. [CrossRef]
- BaHammam, A.S.; Al-Abri, M.A.; Rashid, R.A.; Amra, B.; Oweidat, K.A.; Chan, J.W.Y.; Chen, N.-H.; Chirakalwasan, N.R.C.; Dizon, R.V.; Gupta, R.; et al. Mapping the Landscape of Sleep Medicine Training across Asia. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine 2024, 20, 1647–1656. [CrossRef]
- BaHammam, A.S.; Han, F.; Inoue, Y. The Asian Sleep Medicine Fellowship Training Curriculum: A Milestone in Sleep Medicine Education. Annals of Thoracic Medicine 2025. [CrossRef]
- Behkar, A.; Amirifard, H.; Samadi, S.; Alemohammad, Z.B.; Golkhandan, A.R.; Heidari, R.; Akbarpour, S.; Sadeghniiat-Haghighi, K.; Jalilolghadr, S.; Najafi, A. Global Practice of Sleep Medicine: Iran. Journal of Clinical Sleep Medicine 2024, 20, 1009–1015. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).