1. Introduction
Total knee arthroplasty is one of the most effective orthopaedic procedures for advanced osteoarthritis[
1]; however, periprosthetic joint infection (PJI) remains one of its most serious and difficult-to-treat complications [
2]. Although single-stage revision may be appropriate in selected cases, two-stage replacement arthroplasty remains the most widely accepted treatment strategy worldwide for chronic knee PJI [
3,
4].
Regardless of the revision protocol, the condition of the periarticular soft tissues plays a pivotal role in infection management. The infection itself and repeated surgical interventions can compromise the soft tissue envelope, resulting in fistulas, retracted scarring, necrosis, wound dehiscence, or established coverage defects that prevent primary closure [
5]. Such alterations may jeopardise infection eradication, promote implant failure, and ultimately lead to knee arthrodesis or above-the-knee amputation [
6].
Several reconstructive options are available, ranging from pedicled to free microvascular flaps [
7]. The medial gastrocnemius flap is the most commonly employed pedicled option owing to its simplicity, anatomical proximity, reliable vascularity, and adequate tissue volume [
8]. Nevertheless, extensive defects, proximal exposure, or severe soft-tissue compromise may require free microvascular reconstruction, allowing tailored coverage according to defect size and tissue requirements [
7,
8,
9].
Despite the clinical relevance of soft-tissue reconstruction in knee PJI, comparative evidence evaluating infection outcomes in patients managed with and without formal flap coverage remains scarce. Furthermore, few studies have analysed outcomes according to the complexity of the reconstructive strategy employed. Therefore, this study was designed to test two hypotheses: first, that patients requiring formal soft-tissue reconstruction would not demonstrate inferior infection control compared with those managed without flap coverage; and second, that reconstructive complexity has increased over time, reflected by a growing need for microvascular free flaps. Accordingly, the primary aim was to evaluate infection control in chronic knee PJI patients treated with and without soft-tissue reconstruction, and, within the reconstruction group, to compare outcomes between pedicled medial gastrocnemius flaps and free microvascular flaps. A secondary objective was to analyse temporal trends in reconstructive complexity.
2. Materials and Methods
2.1. Study Design and Patient Selection
Following institutional review board approval (IRB-PR(AT)504/2025), a retrospective observational study was conducted including all consecutive patients treated for chronic knee periprosthetic joint infection using a two-stage exchange protocol at a 1,000-bed tertiary university hospital between 2010 and 2023. All procedures were performed within a dedicated multidisciplinary Septic Unit at a nationally recognised referral centre for musculoskeletal infection.
Inclusion criteria were: (1) a confirmed diagnosis of chronic PJI according to the European Bone and Joint Infection Society (EBJIS) definition [
10] ; (2) treatment with a two-stage arthroplasty replacement strategy; and (3) a minimum follow-up of 24 months after reimplantation. Patients with shorter follow-up or who did not meet all inclusion criteria were excluded. Demographic, clinical, surgical, and microbiological data were collected retrospectively from electronic medical records. When necessary, missing information was obtained through structured telephone interviews with patients or their relatives.
2.2. Study Groups
Patients were stratified into two groups according to the requirement for soft-tissue reconstruction:
No Soft-Tissue Reconstruction (N-STR) group: patients treated with a standard two-stage revision procedure without formal flap coverage.
Soft-Tissue Reconstruction (STR) group: patients requiring additional reconstructive procedures to address soft-tissue deficiencies, including medial gastrocnemius pedicled flaps or free microvascular flaps.
2.3. Outcome Measures and Definitions
The primary endpoint was infection control. Host-related variables included diabetes mellitus, malignancy, immunosuppression, American Society of Anesthesiologists (ASA) score, and Charlson comorbidity index. Surgical variables included the dates of surgery, type of spacer (articulated or static), isolated microorganisms, and characteristics of the final prosthesis.
Treatment success was defined according to the Delphi consensus criteria proposed by Diaz-Ledezma et al.[
11], which required: (a) a healed wound without sinus tract, persistent drainage, or pain and without recurrence caused by the same microorganism; (b) no reoperation for infection after reimplantation; and (c) no PJI-related mortality. Failure was further defined as the need for lifelong suppressive antibiotic treatment or the development of a new PJI caused by a different microorganism.
2.4. Operative Technique Description
All procedures were performed by one of four senior surgeons from the dedicated Infection Unit following a standardised two-stage protocol. Differences between groups were limited to the indication, technique, and timing of soft-tissue reconstruction.
During the first stage, a medial parapatellar approach was used and extended through a tibial tubercle osteotomy when safe mobilisation of the extensor mechanism or adequate exposure could not be achieved. The prosthesis and cement were removed, followed by aggressive debridement. At least six tissue samples were obtained for microbiological culture before antibiotic administration. The joint was irrigated with saline without additives using a low-pressure system, and an antibiotic-loaded cement spacer (Vancogenx®, Tecres, Sommacampagna, Italy) was implanted using vancomycin–gentamicin-loaded acrylic bone cement (Vancogenx®, Tecres SpA, Italy), supplemented with an additional 4 g of vancomycin and 4 g of tobramycin per 40-g cement bag. Static spacers were preferred in cases of major soft-tissue deficiency, extensor mechanism compromise, or extensive bone loss.
Postoperative antimicrobial therapy was guided by infectious disease specialists, members or our dedicated unit. Reimplantation was performed only in the absence of clinical signs of infection, after nutritional optimisation, and when a decreasing trend in C-reactive protein was observed. During the second stage, the spacer was removed, repeat debridement and sampling were performed, and definitive reconstruction was undertaken. Implant selection was based on bone loss, ligamentous stability, and bone quality; in most cases, a cemented modular rotating-hinge prosthesis was used (Endo-Model
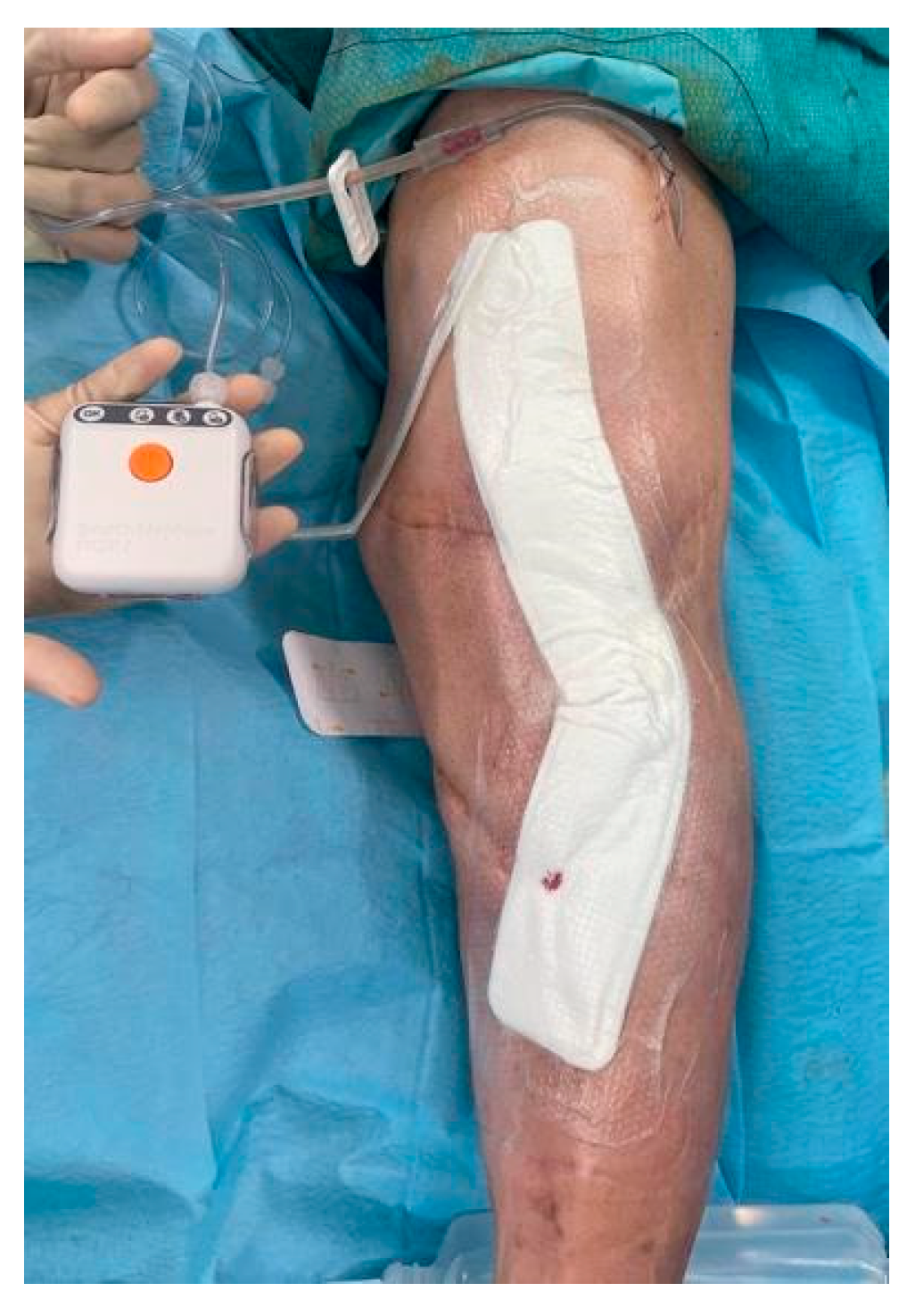
®-M; Waldemar Link, Germany). Prophylactic incisional negative pressure wound therapy (PICO
®, Smith & Nephew; Memphis, USA) was frequently applied for a minimum of 14 days in the vast majority of patients in both stages of treatment (
Figure 1).
2.5. Soft-Tissue Reconstruction Strategies
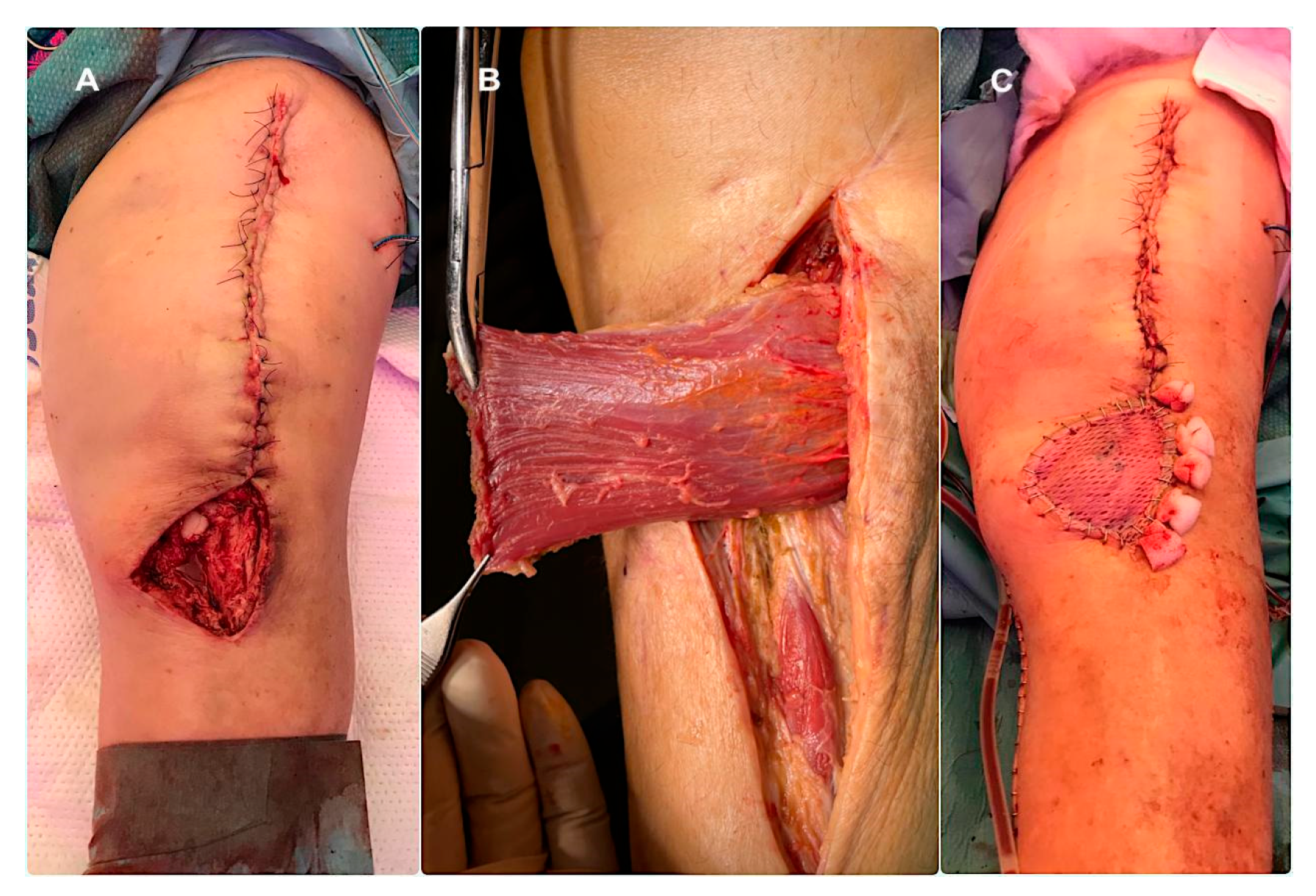
Soft-tissue reconstruction was planned and executed through a multidisciplinary orthopaedic–plastic surgery approach within the dedicated Infection Unit. When feasible, medial gastrocnemius pedicled flaps were preferentially performed during the second stage of the two-stage revision protocol (
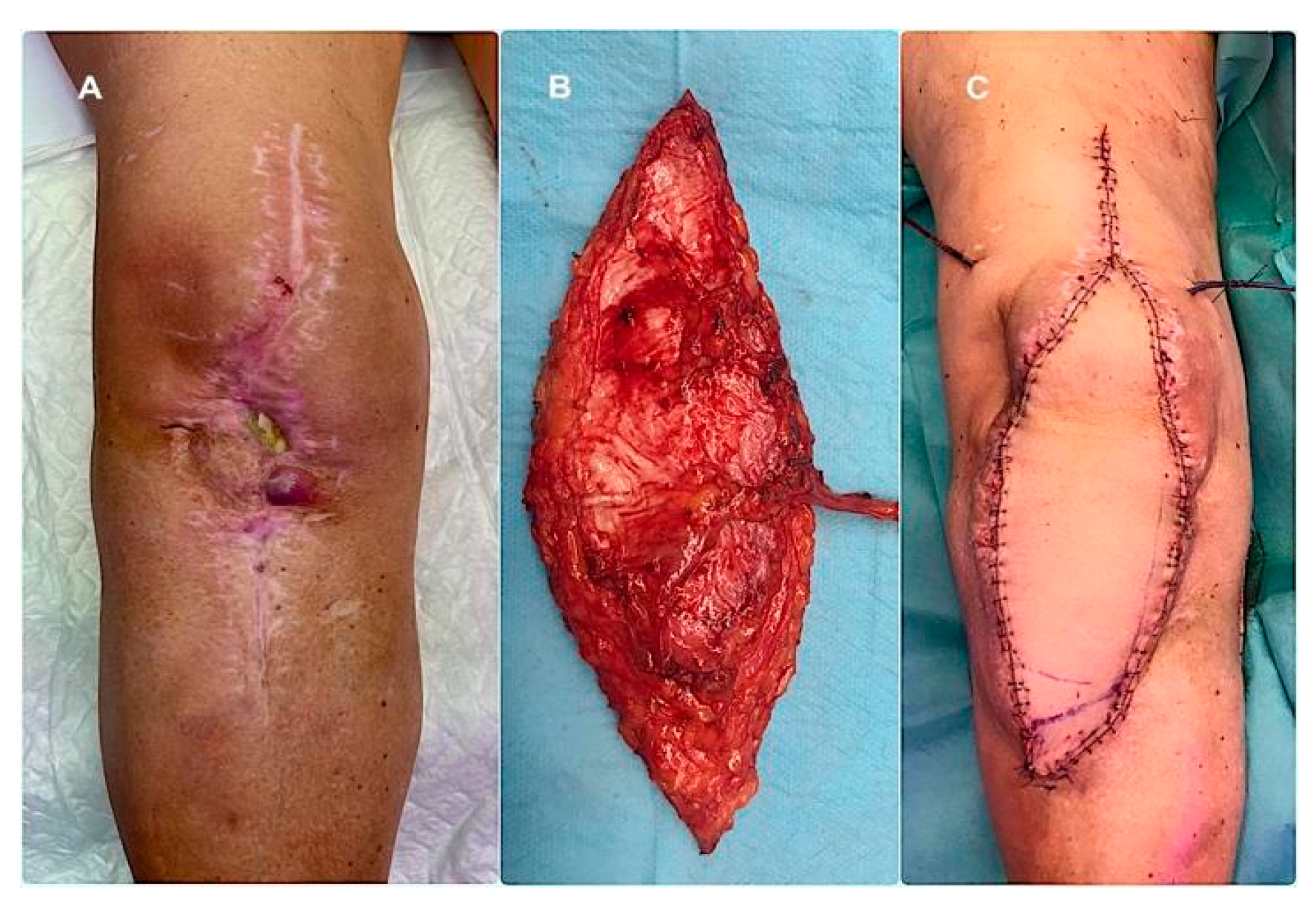
Figure 2). A comprehensive three-stage protocol was developed by our unit [
12] and adopted for patients requiring microsurgical reconstruction, with the anterolateral thigh (ALT) flap used as the preferred free flap (
Figure 3). The rationale for selecting a fasciocutaneous flap, particularly for anterior knee defects, was based on several considerations. First, fasciocutaneous tissue provides a more like-to-like reconstruction in an area subjected to repetitive flexion–extension movements and increased risk of friction, erosion, and superficial trauma. Second, fasciocutaneous flaps facilitate safer and easier re-elevation during subsequent surgical stages, particularly at the time of definitive joint reconstruction, compared with musculocutaneous alternatives. Under this three-stage protocol, surgical management is temporally divided in three discrete treatment phases; the first phase consisted of standard prosthesis removal, aggressive debridement, static spacer implantation, and temporary wound management, most commonly using negative pressure wound therapy. Definitive soft-tissue reconstruction was performed during the second phase, ideally within five days, following preoperative CT angiography to assess vascular anatomy and plan microsurgical anastomoses. Definitive joint reconstruction phase was subsequently undertaken once infection control had been confirmed, the flap demonstrated stable integration, and the patient had achieved adequate physiological optimisation, following the principles of a conventional second-stage reimplantation.
Immediate reconstruction was selectively performed in two specific scenarios: during the second stage when a planned medial gastrocnemius flap was indicated, following prosthesis implantation; or during the first stage in cases of acute soft-tissue loss with spacer exposure, provided that coverage with a medial gastrocnemius flap was feasible. In these cases, the flap was re-elevated during the reimplantation procedure to allow spacer removal and definitive prosthetic reconstruction.
2.6. Statistical Analysis
Statistical analyses were performed using GraphPad Prism® version 9.0. Categorical variables were compared using Fisher's exact test, and continuous variables were compared using Student's t-test or Mann-Whitney U test, depending on the distribution of the data. Normality was assessed using the Shapiro-Wilk test. All tests were two-tailed, and a p-value < 0.05 was considered statistically significant. Results are presented with the corresponding 95% confidence intervals.
3. Results
A total of 118 patients treated for periprosthetic knee joint infection (PJI) were included in the study. The mean age of the cohort was 69.7 ± 10.2 years, and comorbidities were common, as was a history of multiple previous surgical interventions. Baseline demographic and clinical characteristics are detailed in
Table 1.
Of the 118 patients, 78 (66.1%) were treated without formal soft tissue reconstruction (N-STR group), while 40 (33.9%) required additional soft tissue coverage (STR group). The gender distribution was similar between the groups (N-STR: 44.9% men; STR: 46.5% men), and patients in the STR group were slightly younger on average (68.0 ± 1.97 vs. 70.5 ± 1.05 years), although this difference was not statistically significant (p = 0.4206).
In terms of comorbidities and infection-related risk factors, no statistically significant differences were observed between groups. Diabetes mellitus (39.5% vs. 28.2%, p = 0.2887), the presence of a sinus tract (44.8% vs. 26.9%, p = 0.1022), and malignancy (14.1% vs. 2.6%, p = 0.1002) showed numerical differences without reaching statistical significance. Similarly, no significant differences were found in smoking status, obesity, Charlson comorbidity index, alcohol consumption, inflammatory diseases, cirrhosis, anticoagulant treatment, intra-articular pus, or suppressive antibiotic therapy.
Microbiological findings were comparable between groups, with no statistically significant differences in pathogen distribution (
Table 2). Methicillin-sensitive Staphylococcus aureus (MSSA) was isolated in 12.5% of patients in the STR group and 3.85% in the N-STR group (p = 0.1903), and
Enterococcus faecalis was identified in 12.5% and 3.85% of cases, respectively (p = 0.1903).
Enterobacter spp. was isolated more frequently among patients who required soft tissue reconstruction (9.38% vs. 1.28%), which was close to statistical significance (p = 0.0732), while the other microorganisms were detected in low and comparable proportions.
Patients in the STR group had undergone significantly more prior surgical procedures than those in the N-STR group (3.03 ± 0.31 vs. 2.08 ± 0.15; p = 0.0057), whereas the distribution of ASA scores did not differ significantly between the two groups (p = 0.8896).
With a minimum follow-up of 24 months (range: 24-120 months), the overall infection control rate after two-stage replacement arthroplasty was 94.23%. The absence of infection at final follow-up was achieved in 88.46% of patients treated without soft tissue reconstruction and in 100% of those requiring flap coverage, representing a statistically significant difference (p = 0.0294).
Within the soft tissue reconstruction cohort, 25 patients underwent medial gastrocnemius (MG) flap reconstruction and 15 patients received an anterolateral thigh (ALT) free flap. The distribution by age and sex was comparable between the groups (
Table 3). Diabetes mellitus was significantly more prevalent among patients with GM (60.87% vs. 6.67%; p = 0.0017), while inflammatory diseases (26.67% vs. 0%; p = 0.0185) and immunodeficiency (53.33% vs. 0%; p = 0.0001) were significantly more common in the ALT group. Intra-articular pus at the time of surgery was also observed more frequently in ALT patients (71.43% vs. 8.33%; p = 0.0095), while no significant differences were found in the Charlson comorbidity index, smoking, or obesity. Although the number of previous surgical interventions tended to be higher in the GM group (3.3 ± 0.33 vs. 2.67 ± 0.58; p = 0.1335), ALT patients required a significantly higher number of subsequent surgical procedures (2.5 ± 0.54 vs. 0.75 ± 0.31; p = 0.0022).
Microbiological profiles did not differ significantly between GM and ALT reconstructions (
Table 4). The rate of positive cultures, defined as those obtained intraoperatively during the coverage surgery, was 32% in the GM group and 40% in the ALT group, (p = 0.74). Regarding polymicrobial infections, these were observed in 25% of patients in the GM group and 16.7% in the ALT group (p = 0.999). The distribution of pathogen types was comparable between groups.
Postoperative complications were significantly more frequent in the ALT group (p = 0.0049). While 94.1% of patients treated with a gastrocnemius flap experienced no complications, only 46.7% of ALT flap patients were complication-free, with partial flap necrosis being the most common complication (26.7%). Nevertheless, the rate of complete flap loss was low, with total flap necrosis occurring in only 13.3% of cases (
Table 5). Despite these differences, infection control was achieved at final follow-up in 100% of patients in both reconstruction subgroups.
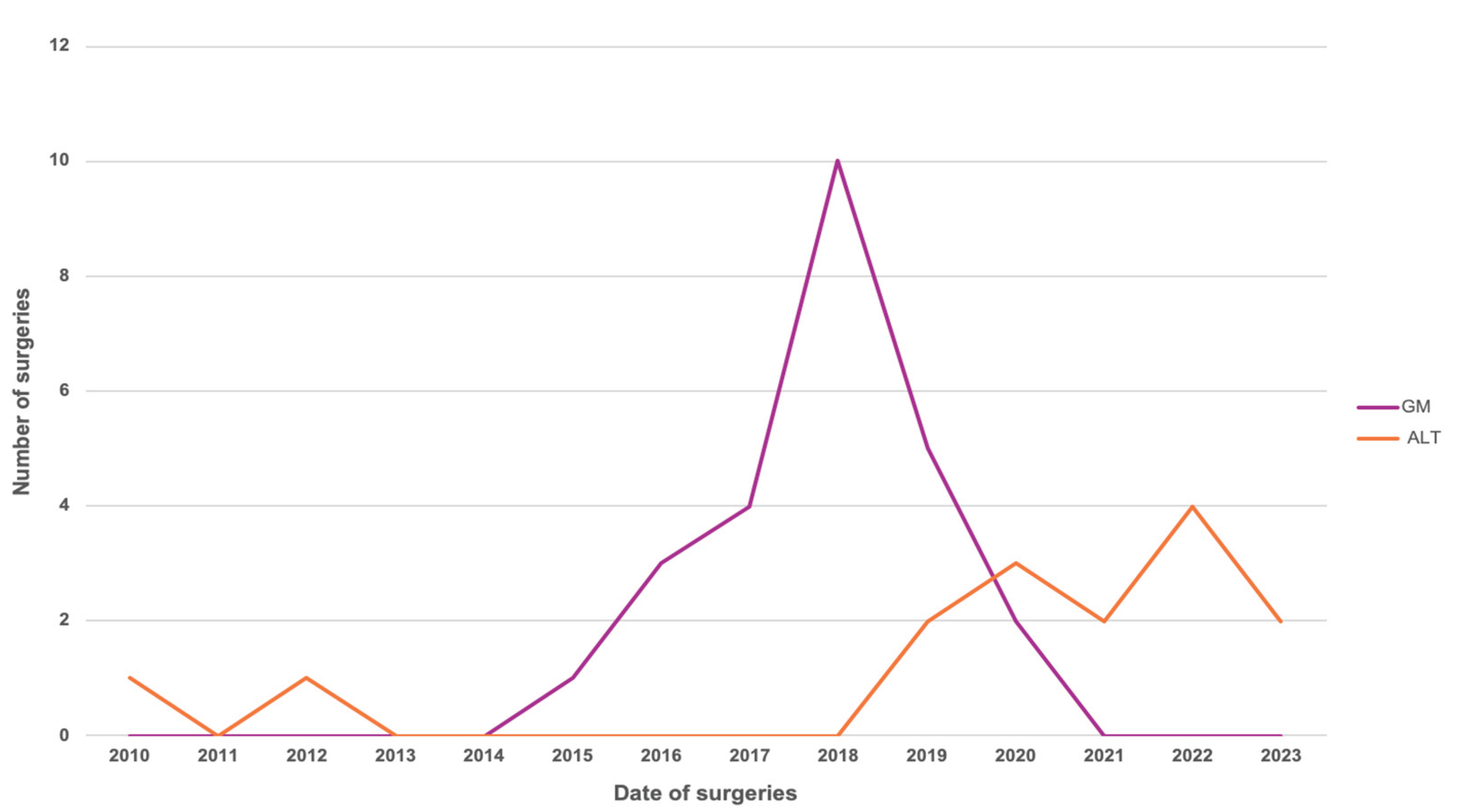
Finally, analysis of temporal trends showed a progressive change in the distribution and complexity of soft tissue reconstruction strategies over the last 13 years of the study period (
Figure 4).
During the early years, pedicled medial gastrocnemius flaps accounted for the majority of reconstructive procedures, while free microvascular flaps were used sporadically. Starting in 2018, there was a sustained annual increase in the number and proportion of anterolateral thigh free flaps, which equalled and subsequently surpassed medial gastrocnemius flaps in frequency starting in 2020.
4. Discussion
In this cohort of chronic knee periprosthetic joint infections treated with a standardised two-stage exchange protocol, approximately one-third of patients required formal soft-tissue reconstruction, reflecting the high clinical and biological complexity of contemporary PJI management. Consistent with our primary hypothesis, the need for soft-tissue reconstruction was not associated with inferior infection control. On the contrary, despite a significantly higher burden of previous surgeries and more advanced infection severity, patients requiring flap coverage achieved superior infection control compared with those managed without reconstruction (100 % vs 88.5 %, p = 0.0294). These findings indicate that, when appropriately integrated into a structured orthoplastic treatment pathway, soft-tissue reconstruction may counterbalance—rather than exacerbate—the negative prognostic impact of severe local compromise.
This observation contrasts with the findings reported by Baltzer et al., who analysed a cohort of 218 knee periprosthetic joint infections, 83.5% of which were managed using a two-stage strategy [
13]. In that study, the requirement for soft tissue reconstruction—together with increased surgical complexity—was identified as a factor associated with a higher risk of infectious failure in a population treated with mixed one- and two-stage revision strategies. Specifically, patients requiring flap coverage demonstrated a significantly lower likelihood of complete healing (30.8% vs 66.2%, p = 0.001) and a higher need for suppressive antibiotic therapy (42.3% vs 15.6%, p = 0.003) compared with those not requiring a flap. In contrast, our study did not demonstrate a negative impact of soft tissue reconstruction on infection control. Similarly, Moog et al. reported in a retrospective cohort of 78 patients with knee PJI that infection-free survival was achieved in 83% of patients undergoing soft tissue reconstruction compared with 57% of those managed without reconstruction (p = 0.0376) [
14]. These findings support the concept that restoration of a stable, well-vascularized soft tissue envelope facilitates infection eradication by improving local perfusion and antibiotic delivery while reducing dead space [
15].
Early and fully integrated collaboration between orthopaedic and plastic surgery teams represents a cornerstone in the management of complex knee periprosthetic joint infection and is a defining feature of our institutional model. In this context, Colen et al. [
16], showed that patients managed within a coordinated multidisciplinary framework, with early plastic surgery involvement, achieved higher prosthesis salvage rates and a lower risk of amputation compared with those referred after multiple prior interventions, reporting an overall infection control rate of 61.6%. Importantly, they identified the number of previous knee operations prior to definitive soft-tissue reconstruction as an independent predictor of reduced prosthesis salvage and increased limb loss. Consistent with these observations, patients requiring soft-tissue reconstruction in our series exhibited a markedly more compromised clinical profile. They had undergone a significantly higher number of prior surgical procedures (3.03 vs. 2.08 ± 0.15; p = 0.0057) and showed higher prevalence of sinus tract formation (44.8% vs. 26.9%; p=0.102) and diabetes mellitus (39.5% vs. 28.2%; p=0.288), factors classically associated with impaired wound healing and reduced soft-tissue resilience, but without reaching statistical significance. Similarly, Christiner et al. [
17] reported that two or more major prior operations significantly increase failure rates following two-stage revision, highlighting the cumulative detrimental effect of repeated surgical trauma. Taken together, these findings indicate that patients ultimately requiring soft-tissue reconstruction represent a subgroup in whom adverse biological and surgical risk factors accumulate, reflecting progressive soft-tissue deterioration, compromised local vascularity, and diminished healing capacity. This is further supported by the findings of Song et al. [
18] who reported inferior infection-free survival (80% vs. 94%, p = 0.006) and lower microorganism identification rates (69% vs. 88%, p = 0.006) in patients undergoing surgical intervention prior to referral to a specialised PJI centre. Collectively, these data underscore the importance of early referral and centralisation of complex PJI management in specialised, high-volume centres, in order to minimise unnecessary prior procedures and prevent progressive compromise of the soft-tissue envelope [
19].
Within the reconstruction subgroup, pedicled medial gastrocnemius (MG) flaps and free anterolateral thigh (ALT) flaps reflected fundamentally different clinical scenarios rather than interchangeable reconstructive options. Pedicled gastrocnemius flaps provided reliable coverage for small to moderate anterior and infrapatellar defects, as supported by Houdek et al., who reported 10-year revision-free and amputation-free survival rates of 68% and 79%, respectively. Importantly, wound size ≥50 cm² was identified as a significant predictor of reconstructive failure, supporting the limitation of this technique to moderate-sized defects [
20]. In contrast, free microvascular flaps were predominantly required in patients with extensive soft-tissue loss and advanced biological compromise, including intra-articular purulence, immunodeficiency, and inflammatory comorbidities. These patients required more surgical procedures and experienced higher complication rates, reflecting a more complex host and local environment. Consistent with our findings, Lee et al. reported limb salvage in 82% of patients treated with free flaps, although 18% ultimately required amputation and functional outcomes remained limited, while Hamrouni et al. achieved limb salvage in all cases at the cost of a substantial early complication rate [
21,
22]. Taken together, these data indicate that while free tissue transfer is superior for the management of large or complex soft-tissue defects, it identifies a distinct and more compromised patient population and is associated with greater surgical burden and potential functional limitations. Moreover, its implementation requires specialised expertise and is not universally available. Therefore, pedicled flaps should be reserved for localised, moderate defects, whereas microvascular reconstruction should be considered for extensive soft-tissue loss within specialised, multidisciplinary centres.
An interesting finding regarding microbiological profiles between reconstructive groups was that the use of temporary negative pressure wound therapy (NPWT) in the anterolateral thigh (ALT) free flap group was not associated with a higher rate of polymicrobial positive cultures. Previous studies have linked temporary NPWT to an increased incidence of polymicrobial infection and a higher risk of multidrug-resistant organisms in orthopaedic and trauma populations. Valenzuela et al. [
23] reported that open wound management, including NPWT, can convert monomicrobial to polymicrobial periprosthetic joint infection in 34% of cases, with only 45% of patients who converted to polymicrobial infection remaining infection-free at follow-up, compared with 70% of those who remained monomicrobial. In contrast, no such difference was observed in our cohort, as we observed polymicrobial cultures in only 16.7% of patients in the ALT group, with a subsequent infection eradication rate of 100%. This finding may be explained by the implementation of a strict and standardized wound management protocol. In cases managed using a three-stage reconstruction strategy, NPWT was applied for a maximum of five days, during which the wound remained sealed and isolated from the external environment. This limited exposure, combined with the local antibacterial effect of the antibiotic-loaded spacer, may mitigate the increased microbiological risk associated with prolonged NPWT use. Based on these observations, NPWT should be restricted to short, predefined intervals and employed only when definitive flap reconstruction is already scheduled.
A particularly relevant finding of this study is the progressive and sustained increase in the use of microvascular free flaps throughout the study period, a trend that became especially pronounced in recent years. Contemporary literature from high-volume centres consistently describes a growing reliance on microvascular reconstruction as a limb salvage strategy in infected prosthetic knees with complex soft-tissue defects, particularly in scenarios in which pedicled options are insufficient to provide stable and durable coverage [
24]. This reconstructive transition appears to reflect a substantial transformation in the clinical and biological profile of patients referred to tertiary centres, characterized by greater cumulative surgical burden, more advanced host compromise, and more extensive and complex soft-tissue deficiencies, rather than a simple shift in reconstructive team preference or technical approach. This evolving landscape is likely to define the future of limb salvage in complex periprosthetic knee infections, underscoring the need for reconstructive teams to be prepared for an increasing reliance on advanced microvascular techniques and multidisciplinary, protocol-driven care.
We acknowledge the limitations of our study. The first lies in the study’s retrospective nature. Retrospective studies rely on chart notes from which important data may be lacking, potentially increasing bias incidence. Second, the study was conducted in a national referral centre for musculoskeletal infection and complex reconstruction, which introduces a significant selection bias. Patients referred to our institution often represent the most severe and complex cases, characterised by advanced infection, multiple prior surgical procedures, compromised soft tissues, and unfavourable host factors. Consequently, the proportion of patients requiring soft-tissue reconstruction—particularly microsurgical free flaps—may be higher than that observed in non-referral settings. Third, four different surgeons carried out the study’s procedures, inevitably adding some variability of results. To balance this fact to some degree, all four surgeons were familiar with the protocol described and followed it faithfully. Other limitations to consider are the sample size and the follow-up period; even so, both of these factors were comparable or superior to those of previously published studies. Therefore, while our results demonstrate what can be achieved within a specialised setting, they should not be interpreted as evidence that similar outcomes can be expected universally.
Rather, they support the concept that concentration of complex PJI care within experienced centres may be necessary to reproduce such results.
5. Conclusions
In chronic knee periprosthetic joint infection treated with a two-stage exchange protocol, the need for soft-tissue reconstruction reflects increased clinical and biological complexity rather than an unfavourable prognostic factor. When embedded within a structured multidisciplinary orthoplastic approach, soft-tissue reconstruction—including microsurgical free flaps—enables high rates of infection eradication even in severely compromised cases. Differences between pedicled and free flap reconstruction primarily relate to defect extent and host factors, underscoring the importance of early referral and centralisation of complex PJI care in specialised centres.
Author Contributions
All authors contributed to the study conception and design, material preparation, data collection and analysis. The first draft of the manuscript was written by PC and CM, and all authors commented on the versions of the manuscript. All authors read and approved the final manuscript.
Funding
This work received no outside financial support. The study was conducted as part of the routine work of our institution.
Institutional Review Board Statement
The study was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki. The Ethics Committee of our center approved this study (Institutional Review Board -IRB- approval (PR(ATR)504/2025).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
We wish to thank our entire multidisciplinary committee, including Danilo Rivas member of the Plastic Surgery Department, Dr. Juan Carlos Juarez, member of our center’s Pharmacy Department, Dr. Carles Pigrau, Dr. Julia Sellarès and Dr. Dolors Rodriguez-Pardo, members of our infectious diseases department; Dra. Paula Salmeron and Dra. Mayli Lung, members of our microbiology team and Dr. Esther Marques, member of the anesthesiology department of our center. I would also like to acknowledge the inestimable help of Rosa Fraile Soriano, operating room nurse specialized in septic pathology. Without her help, it would be far more difficult to operate on patients of this type.
Conflicts of Interest
All Authors: None of the authors received payments or services, either directly or indirectly (i.e., via his or her institution), from a third party in support of any aspect of this work. One of the authors (PC) has done paid speaking engagements at scientific conferences during the thirty-six months prior to submission of this work, to an entity in the biomedical arena (Tecres SpA; Italy, Waldemar Link GmbH & Co.; Germany and Smith & Nephew; USA) whose products have been mentioned in the article. The author does not have any other conflict of interest with said companies. No author has had any other relationships or has engaged in any other activities that could be perceived to influence or have the potential to influence what is written in this work.
Abbreviations
The following abbreviations are used in this manuscript:
| PJI |
Periprosthetic joint infection |
| EBJIS |
European Bone and Joint Infection Society |
| N-STR |
No soft tissue reconstruction |
| STR |
Soft tissue reconstruction |
| ASA score |
American Society of Anesthesiologists score |
| GM |
Medial Gastrocnemius Muscle Flap |
| ALT |
Anterolateral Thigh Flap |
References
- Zhang, R.; Shen, X.; Yan, K.; Zhang, X.; Zhu, C. Comparative Efficacy and Safety of Bicompartmental versus Total Knee Arthroplasty: A Systematic Review and Update Meta-Analysis. J. Orthop. Surg. 2025, 20, 237. [Google Scholar] [CrossRef] [PubMed]
- Bozic, K.J.; Kurtz, S.M.; Lau, E.; Ong, K.; Chiu, V.; Vail, T.P.; Rubash, H.E.; Berry, D.J. The Epidemiology of Revision Total Knee Arthroplasty in the United States. Clin. Orthop. 2010, 468(1), 45–51. [Google Scholar] [CrossRef] [PubMed]
- Pérez, M.; Vicente, M.; Amat, C.; Lahoz, B.; Carrera, L.; Corona, P.S. Outcomes of a Cemented Modular Rotational-Hinge Design as the Final Implant in a Two-Stage Replacement Due to Chronic Knee Periprosthetic Joint Infection. Arch. Orthop. Trauma Surg. 2024, 144(12), 5239–5250. [Google Scholar] [CrossRef] [PubMed]
- Nelson, S.B.; Pinkney, J.A.; Chen, A.F.; Tande, A.J. Periprosthetic Joint Infection: Current Clinical Challenges. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2023, 77(7), e34–e45. [Google Scholar] [CrossRef]
- Raymond, A.C.; Liddle, A.D.; Alvand, A.; Donaldson, J.R.; Carrington, R.W.J.; Miles, J. Clinical Outcome of Free Latissimus Dorsi Flaps for Coverage of Soft Tissue Defects in Multiply Revised Total Knee Arthroplasties. J. Arthroplasty 2021, 36(2), 664–669. [Google Scholar] [CrossRef] [PubMed]
- Galat, D.D.; McGovern, S.C.; Larson, D.R.; Harrington, J.R.; Hanssen, A.D.; Clarke, H.D. Surgical Treatment of Early Wound Complications Following Primary Total Knee Arthroplasty. J. Bone Joint Surg. Am. 2009, 91(1), 48–54. [Google Scholar] [CrossRef] [PubMed]
- Gravvanis, A.; Kyriakopoulos, A.; Kateros, K.; Tsoutsos, D. Flap Reconstruction of the Knee: A Review of Current Concepts and a Proposed Algorithm. World J. Orthop. 2014, 5(5), 603–613. [Google Scholar] [CrossRef]
- Mayoly, A.; Mattei, J.-C.; Moullot, P.; Jaloux, C.; Rochwerger, A.; Casanova, D.; Bertrand, B.; Philandrianos, C. Gastrocnemius Myocutaneous Flaps for Knee Joint Coverage. Ann. Plast. Surg. 2018, 81(2), 208–214. [Google Scholar] [CrossRef] [PubMed]
- Cetrulo, CL, Jr.; Shiba, T.; Friel, MT.; Davis, B.; Buntic, RF.; Buncke, GM.; Brooks, D. Management of exposed total knee prostheses with microvascular tissue transfer. Microsurgery 2008, 28(8), 617–622. [Google Scholar] [CrossRef] [PubMed]
- McNally, M.; Sousa, R.; Wouthuyzen-Bakker, M.; Chen, A.F.; Soriano, A.; Vogely, H.C.; Clauss, M.; Higuera, C.A.; Trebše, R. The EBJIS Definition of Periprosthetic Joint Infection. Bone Jt. J. 2021, 103-B(1), 18–25. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Ledezma, C.; Higuera, C.A.; Parvizi, J. Success after Treatment of Periprosthetic Joint Infection: A Delphi-Based International Multidisciplinary Consensus. Clin. Orthop. 2013, 471(7), 2374–2382. [Google Scholar] [CrossRef]
- Corona, P.S.; Carbonell-Rosell, C.; Vicente, M.; Serracanta, J.; Tetsworth, K.; Glatt, V. Three-stage limb salvage in tibial fracture related infection with composite bone and soft-tissue defect. Arch. Orthop. Trauma Surg. 2022, 142(12), 3877–3887. [Google Scholar] [CrossRef]
- Baltzer, J.; Lording, T.; Pineda, T.; Ferry, T.; Servien, E.; Lustig, S.; Batailler, C. Satisfactory and Similar Outcomes After Knee Arthroplasty Revisions in One or Two Stages for Infection, Following a Surgical Strategy Based on Robust Guidelines. J. Arthroplasty 2025, S0883-5403((25)00882-4). [Google Scholar] [CrossRef] [PubMed]
- Moog, P.; Tinwald, I.; Aitzetmueller, M.; Bauer, A.-T.; Megerle, K.; Machens, H.-G.; Laszlo Kovacs, L.; Kuekrek, H. The Usage of Pedicled or Free Muscle Flaps Represents a Beneficial Approach for Periprosthetic Infection After Knee Arthroplasty. Ann. Plast. Surg. 2020, 85(5), 539–545. [Google Scholar] [CrossRef]
- Rovere, G.; Smakaj, A.; Calori, S.; Barbaliscia, M.; Ziranu, A.; Pataia, E.; Maccauro, G.; De Mauro, D.; Liuzza, F. Use of Muscular Flaps for the Treatment of Knee Prosthetic Joint Infection: A Systematic Review. Orthop. Rev. 2022, 14(5), 33943. [Google Scholar] [CrossRef] [PubMed]
- Colen, D.L.; Carney, M.J.; Shubinets, V.; Lanni, M.A.; Liu, T.; Levin, L.S.; Lee, G.-C.; Kovach, S.J. Soft-Tissue Reconstruction of the Complicated Knee Arthroplasty: Principles and Predictors of Salvage. Plast. Reconstr. Surg. 2018, 141(4), 1040–1048. [Google Scholar] [CrossRef]
- Christiner, T.; Sulcs, M.; Yates, P.; Prosser, G. Obesity, Comorbidities, and Prior Operations Additively Increase Failure in 2-Stage Revision Total Knee Arthroplasty for Prosthetic Joint Infection. J. Arthroplasty 2022, 37(2), 353–358. [Google Scholar] [CrossRef] [PubMed]
- Song, S.Y.; Goodman, S.B.; Suh, G.; Finlay, A.K.; Huddleston, J.I.; Maloney, W.J.; Amanatullah, D.F. Surgery Before Subspecialty Referral for Periprosthetic Knee Infection Reduces the Likelihood of Infection Control. Clin. Orthop. 2018, 476(10), 1995–2002. [Google Scholar] [CrossRef]
- Dombrowsky, A.R.; Jolissaint, J.E.; Posey, S.L.; Burger, J.M.; Metcalf, R.; Rowe, T.M.; Hietpas, K.T.; Fehring, T.K. Regional Periprosthetic Joint Infection Centers: The Time Has Come for a Paradigm Change in the Treatment of Periprosthetic Joint Infection. J. Arthroplasty 2024, 39(12), 2887–2892. [Google Scholar] [CrossRef] [PubMed]
- Houdek, M.T.; Wagner, E.R.; Wyles, C.C.; Harmsen, W.S.; Hanssen, A.D.; Taunton, M.J.; Moran, S.L. Long-Term Outcomes of Pedicled Gastrocnemius Flaps in Total Knee Arthroplasty. J. Bone Joint Surg. Am. 2018, 100(10), 850–856. [Google Scholar] [CrossRef]
- Lee, G.-C.; Colen, D.L.; Levin, L.S.; Kovach, S.J. Microvascular Free Flap Coverage for Salvage of the Infected Total Knee Arthroplasty. Bone Jt. J. 2020, 102-B(6_Supple_A), 176–180. [Google Scholar] [CrossRef] [PubMed]
- Hamrouni, N.; Højvig, J.H.; Knudsen, U.K.; Skovgaard, K.K.; Jensen, L.T.; Bonde, C.T.; Odgaard, A. Microvascular Free Flap Coverage of Complex Soft Tissue Defects after Revision Total Knee Arthroplasty: A Cross-Sectional Observation Study. Acta Orthop. 2024, 95, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, M.M.; Averkamp, B.J.; Odum, S.M.; Rowe, T.M.; Fehring, T.K. Polymicrobial Colonization of Prosthetic Joint Infections Treated With Open Wound Management. J. Arthroplasty 2022, 37(7S), S653–S656. [Google Scholar] [CrossRef] [PubMed]
- Kahramangil, B.; Pires, G.; Montorfano, L.; Riesgo, A.M.; Ghaznavi, A.M. The Utility of Flap Coverage for Limb Salvage in Patients With Severe Prosthetic Joint Infections of the Knee Requiring Implant Exchange. Ann. Plast. Surg. 2023, 91(1), 129–136. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |